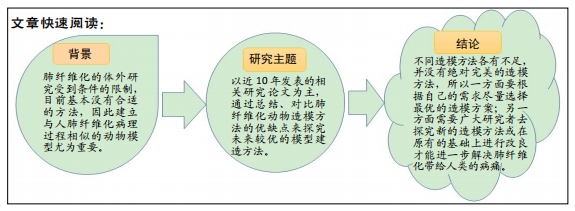
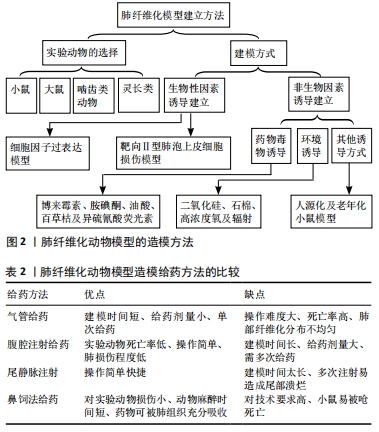
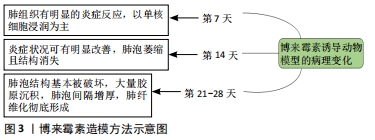
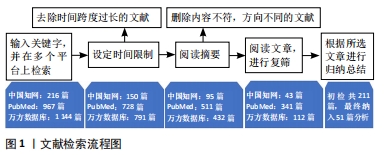
[1] RAGHU G, GHEN SY, YEH WS, et al. Idiopathic pulmonary fibrosis in US medicare beneficiaries aged 65 years and older: incidence, prevalence and survival. Lancet Reapir Med. 2014;2(7):566-572.
[2] RAGHU G, COLLARD HR, EGAN JJ, et al. An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med. 2011;183(6):788-824.
[3] COTTIN V, BERGOT E, BOURDIN A, et al. Adherence to guidelines in idiopathic pulmonary fibrosis: a follow-up national survey. ERJ Open Res. 2015;1(2):1-8.
[4] 韩永禧,杨军,黄芪对博来霉素肺纤维化大鼠血清NO水平的影响[J].中国社区医师(医学专业),2012,14(302):8-9.
[5] WOLLIN L, WEX E, PAUTSCH A, et al. Mode of action of nintedanib in the treatment of idiopathic pulmonary fibrosis. Eur Respir J. 2015;45(5):1434-1445.
[6] 陈孟毅,孟爱民.肺纤维化动模型及研究进展[J].中国比较医学杂志,2016, 26(6):88-92.
[7] 庄文越,马周义,赵娜,等.肺纤维化细胞因子参与机制及中药治疗研究进展[J].北华大学学报(自然科学版),2017,18(5):615-618.
[8] SQUIRES MS, HUDSON EA, HOWELLS L, et al. Relevance of mitogen activated protein kinase (MAPK) and phosphotidy linositol-3-kinase/protein kinase B (PI3K/PKB) pathways to induction of apoptosis by curcumin in breast cells. Biochem Pharmacol. 2003;65(3):361-376.
[9] SUN L, LOUIE MC, VANNELLA KM, et al. New concepts of IL-10-induced lung fibrosis: fibrocyte recruitment and M2 activation in a CCL2/CCR2 axis. Am J Physiol Lung Cell Mol Physiol. 2011;300(3):L341-L353.
[10] 肖莉.白细胞介素13(IL-13)致肺纤维化作用及干预机制的研究[D].沈阳:中国医科大学,2004.
[11] RAFII R, JUAREZ MM, ALBERTSON TE, et al. A review of current and novel therapies for idiopathic pulmonary fibrosis. J Thorac Dis. 2013;5(1):48-73.
[12] SISSON TH, MENDEZ M, CHOI K, et al. Targeted injury of type II alveolar epithelial cells induces pulmonary fibrosis. Am J Respir Crit Care Med. 2010;181(3):254-263.
[13] MOURATIS MA, AIDINIS V. Modeling pulmonary fibrosis with bleomycin. Curr Opin Pulm Med. 2011;17(5):355-361.
[14] 邱雨雨,张港,王鸣尚,等.抗肿瘤药物博来霉素诱导小鼠肺纤维化模型[J].山东化工,2021,50(5):55.
[15] CARRINGTON R, JORDAN S, PITCHFORD SC, et al. Use of animal models in IPF research. Pulm Pharmacol Ther. 2018;51:73-78.
[16] SHEPPARD D. Transforming growth factor beta: a central modulator of pulmonary and airway inflammation and fibrosis. Proc Am Thorac Soc. 2006;3(5):413-417.
[17] 卢锦辉,张丽,刘子豪,等.博来霉素诱导小鼠肺纤维化模型的建立及评价[J].兰州大学学报(医学版),2019,45(6):41.
[18] DEGRYSE AL, TANJORE H, XU XC, et al. Repetitive intratracheal bleomycin models several features of idiopathic pulmonary fibrosis. Am J Physiol: 2010;299(4): L442-L452.
[19] 王萍,李铁刚.百草枯中毒致小鼠肺纤维化模型的建立[J].实用药物与临床, 2020,23(10):886-889.
[20] CHUA F, GAULDIE J, LAURENT GJ. Pulmonary fibrosis: searching for model answers. Am J Respir Cell Mol Biol. 2005;33(1):9-13.
[21] 周平,王磊,何春香,等.博来霉素和油酸致大鼠肺纤维化病理模型的比较[J].世界最新医学信息文摘,2015,15(87):48.
[22] 夏洪颖,任晓明.胺碘酮肺毒性的研究进展[J].医学先驱报,2010,29(6): 758.
[23] DHOKARH R, LIG, SCHMICKL CN, et al. Drug associated acute lung injury: a population based cohort study. Chest. 2012;142(4):845-850.
[24] 王和枚,瞿文生,余寿忠,等.胺碘酮所致大鼠肺毒性[J].中国药理学与毒理学杂志,1999,13(4):301-305.
[25] GONCALVES-DE-ALBUQUERQUE CF, SILVA AR, BURTH P, et al. Acute respiratory distress syndrome: role of oleic acid-triggered lung injury and inflammation. Mediators Inflamm. 2015;2015:1-9.
[26] BALTAZAR T, DINIS-OLIVEIRA RJ, DUARTE JA, et al. Paraquat research: do recent advances in limiting its toxicity make its use safer. Br J Pharmacol. 2013;168(1):44-45.
[27] 李学军,崔社怀.肺纤维化治疗研究进展[J].中国综合临床,2001,17(10):725.
[28] 杨珊珊,贾晓民,赵杰,等.三个品系小鼠百草枯肺纤维化模型的比较[J].山西医科大学学报,2014,45(6):456-459.
[29] 杨晓燕,沈杰.肺纤维化动物模型的研究进展[J].生物技术通讯,2015,26(6):869.
[30] ISHIDA Y, TAKAYASU T, KIMURA A, et al. Gene expression of cytokines and gro with factors in the lungs after paraquat adminstration in mice. Legal Med. 2006;8(2): 102-109.
[31] MOORE BB, HOGABOAM CM. Murine Animal models of pulmonary fibrosis. Am J Physiol Lung Cell Mol Physiol. 2008;294(2):L154-L155.
[32] CHRISTENSEN PJ, GOODMAN RE, PASTORIZA L, et al. Induction of lung fibrosis in the mouse by intratracheal instillation of fluorescein isothiocyanate is not T-cell-dependent. Am J Pathol. 1999;155(5):1775-1776.
[33] O’ DWYER DN, MOORE BB. Animal models of pulmonary fibrosis. lung innate immunity and inflammation: methods and protocols. Methods Mol Biol. 2018; 1809:373-374.
[34] MOORE B, LAWSON WE, OURY TD, et al. Animal models of fibrotic lung disease. Am J Respir Cell Mol Biol. 2013;49(2):167-179.
[35] 蒋荣,陈明霞.二氧化硅诱导的内皮间质转化参与矽肺纤维化的实验研究[J].现代预防医学,2020,47(9):1665-1669.
[36] 蔡婷峰,宋向荣,李宏玲,等.不同时间点大鼠矽肺模型病理改变观察[J].中国职业医学,2020,47(4):385-389.
[37] Heather F, Lakatos, Heather A, et al. Oropharyngeal aspiration of a silica suspension produces a superior model of silicosis in the mouse when compared to intratracheal instillation. Exp Lung Res. 2006;32:181-199.
[38] 张志斌,穆丹,李明,等.牛磺酸对大鼠肺纤维化的干预作用[J].工业卫生与职业病,2014,40(4):255-258.
[39] LAKATOS HF, BURGESS HA, THATCHER TH, et al. Oropharyngeal aspiration of a silica suspension produces a superior model of silicosis in the mouse when compared to intratracheal instillation. Exp Lung Res. 2006;32(5):181-199.
[40] CSAAEL SL, EISENBARTH SC, IYER SS, et al. The Nalp3 inflamma some is essential for the development of silicosis. Proc Natl Acad Sci. 2008;105(26):9035-9040.
[41] TASHIRO J, RUBIO GA, LIMPER AH, et al. Exploring animal models that resemble idiopathic pulmonary fibrosis. Front Med. 2017;4:118.
[42] 肖国兵.石棉致纤维化机制研究新进展[J].职业卫生与病伤,1997,12(1):51-53.
[43] RUBIN P, FINKELSTEIN J, SCHAPIRO D. Molecular biology mechanisms in the radiation induction of pulmonary injury syndromes: interrelationship between the alveolar macrophage and the septal fibroblast. Int Radiat Oncol Biol Phys. 1992;24(1):93-101.
[44] MCDONALD S, RUBIN P, CHANG AY, et al. Pulmonary changes induced by combined mouse beta-interferon (rMuIFN-beta) and irradiation in normal mice-toxic versus protective effects. Radiother Oncol. 1993;26(3):212-218.
[45] LIU H, XUE JX, LI X, et al. Quercetin liposomes protect against radiation-induced pulmonary injury in a murine model. Oncol Lett. 2013;6(2):453-459.
[46] VAN OOIJ PJ, HOLLMANN MW, VAN HULST RA, et al. Assessment of pulmonary oxygen toxicity: relevance to professional diving: a review. Respir Physiol Neurobiol. 2013;189(1):117-128.
[47] RUDLOFF I, CHO SX, BUI CB, et al. Refining anti-inflammatory therapy strategies for bronchpulmonary dysplasia. J Cell Mol Med. 2017;21(6):1128-1138.
[48] WARNER BB, STUART LA, PAPES RA, et al. Functional and pathological effects of prolonged hyperoxia in neonatal mice. Am J Physiol. 1998;275(1):L110-L117.
[49] 徐凤丹,王优,吴文燊,等.高氧暴露对新生小鼠肺部炎性反应及纤维化的影响[J].中国医药导报,2017,14(26):21-24.
[50] PIERCE EM, CARPENTER K, JAKUBZICK C, et al. Therapeutic targeting of CC ligand 21 or CC chemokine receptor 7 abrogates pulmonary fibrosis induced by the adoptive transfer of human pulmonary fibroblasts to immunodeficient mice. Am J pathol. 2007;170(4):1152-1164.
[51] SUEBLINVONG V, NEVEU WA, NEUJAHR DC, et al. Aging promotes pro-fibrotic matrix production and increases fibrocyte recruitment during acute lung injury. Adv Biosci Biotechnol. 2014;5(1):19-30.
|