[1] HO TT, WARR MR, ADELMAN ER, et al. Autophagy maintains the metabolism and function of young and old stem cells. Nature. 2017; 543(7644):205-210.
[2] MARCHETTI B, TIROLO C, L’EPISCOPO F, et al. Parkinson’s disease, aging and adult neurogenesis: Wnt/β-catenin signalling as the key to unlock the mystery of endogenous brain repair. Aging Cell. 2020;19(3):e13101.
[3] WU J, LIU S, MENG H, et al. Neuropeptide Y enhances proliferation and prevents apoptosis in rat bone marrow stromal cells in association with activation of the Wnt/β-catenin pathway in vitro. Stem Cell Res. 2017;21:74-84.
[4] KOLOŠA K, MOTALN H, HEROLD-MENDE C, et al. Paracrine effects of mesenchymal stem cells induce senescence and differentiation of glioblastoma stem-like cells. Cell Transplant. 2015;24(4):631-644.
[5] 李渊,周玥,王亚平,等.人参皂苷Rg1基于SIRT6/NF-κB信号通路对辐射致造血干/祖细胞衰老的保护作用[J].中草药,2017,48(21): 4497-4501.
[6] YANG M, TENG S, MA C, et al. Ascorbic acid inhibits senescence in mesenchymal stem cells through ROS and AKT/mTOR signaling. Cytotechnology. 2018;70(5):1301-1313.
[7] CIESLIK KA, TRIAL J, CRAWFORD JR, et al. Adverse fibrosis in the aging heart depends on signaling between myeloid and mesenchymal cells; role of inflammatory fibroblasts. J Mol Cell Cardiol. 2014;70:56-63.
[8] ZENG Y, HU W, JING P, et al. The regulation of ginsenoside Rg1 upon aging of bone marrow stromal cell contribute to delaying senescence of bone marrow mononuclear cells (BMNCs). Life Sci. 2018;209:63-68.
[9] SCHOFIELD R. The relationship between the spleen colony-forming cell and the haemopoietic stem cell. Blood Cells. 1978;4(1-2):7-25.
[10] CHACÓN-MARTÍNEZ CA, KOESTER J, WICKSTRÖM SA. Signaling in the stem cell niche: regulating cell fate, function and plasticity. Development. 2018;145(15):dev165399.
[11] ROSSI L, SALVETTI A. Planarian stem cell niche, the challenge for understanding tissue regeneration. Semin Cell Dev Biol. 2019;87:30-36.
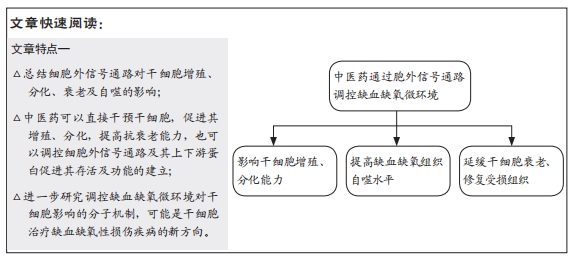
[12] 王诗琦,张金生.中医药调控缺血缺氧微环境对骨髓间充质干细胞增殖、分化及衰老的影响[J].中国组织工程研究,2021,25(7):1129-1134.
[13] ZHANG Y, ZHANG L, ZHANG Y, et al. YiQiFuMai Powder Injection Attenuates Coronary Artery Ligation-Induced Heart Failure Through Improving Mitochondrial Function via Regulating ROS Generation and CaMKII Signaling Pathways. Front Pharmacol. 2019;10:381.
[14] 赵慧杰.加味丹参饮含药血清对骨髓间充质干细胞增殖及分泌bFGF、VEGF 的影响[J].亚太传统医药,2019,15(8):41-43.
[15] 涂献坤,杨滨,涂德文,等.PI3K/Akt通路在丙泊酚减轻大鼠脑缺血性损伤中的作用[J].中国药理学通报,2019,35(5):630-633.
[16] LIU B, LUO C, ZHENG Z, et al. Shengui Sansheng San extraction is an angiogenic switch via regulations of AKT/mTOR, ERK1/2 and Notch1 signal pathways after ischemic stroke. Phytomedicine. 2018;44:20-31.
[17] JEONG JE, PARK JH, KIM CS, et al. Neuroprotective effects of erythropoietin against hypoxic injury via modulation of the mitogen-activated protein kinase pathway and apoptosis. Korean J Pediatr. 2017;60(6):181-188.
[18] MA Y, QI M, AN Y, et al. Autophagy controls mesenchymal stem cell properties and senescence during bone aging. Aging Cell. 2018;17(1): e12709.
[19] JAKOVLJEVIC J, HARRELL CR, FELLABAUM C, et al. Modulation of autophagy as new approach in mesenchymal stem cell-based therapy. Biomed Pharmacother. 2018;104:404-410.
[20] WEICHHART T. mTOR as Regulator of Lifespan, Aging, and Cellular Senescence: A Mini-Review. Gerontology. 2018;64(2):127-134.
[21] RASTALDO R, VITALE E, GIACHINO C. Dual Role of Autophagy in Regulation of Mesenchymal Stem Cell Senescence. Front Cell Dev Biol. 2020;8:276.
[22] MRAKOVCIC M, FRÖHLICH LF. p53-Mediated Molecular Control of Autophagy in Tumor Cells. Biomolecules. 2018;8(2):14.
[23] YANG M, WEN T, CHEN H, et al. Knockdown of insulin-like growth factor 1 exerts a protective effect on hypoxic injury of aged BM-MSCs: role of autophagy. Stem Cell Res Ther. 2018;9(1):284.
[24] DALGAARD LT, ELIASSON L. An ‘alpha-beta’ of pancreatic islet microribonucleotides. Int J Biochem Cell Biol. 2017;88:208-219.
[25] CAPPELLETTI C, GALBARDI B, BRUTTINI M, et al. Aging-associated genes and let-7 microRNAs: a contribution to myogenic program dysregulation in oculopharyngeal muscular dystrophy. FASEB J. 2019; 33(6):7155-7167.
[26] KATZ S, CUSSIGH D, URBÁN N, et al. A Nuclear Role for miR-9 and Argonaute Proteins in Balancing Quiescent and Activated Neural Stem Cell States. Cell Rep. 2016;17(5):1383-1398.
[27] LYU G, GUAN Y, ZHANG C, et al. TGF-beta signaling alters H4K20me3 status via miR-29 and contributes to cellular senescence and cardiac aging. Nat Commun. 2018;9(1):2560.
[28] JAUHARI A, SINGH T, SINGH P, et al. Regulation of miR-34 Family in Neuronal Development. Mol Neurobiol. 2018;55(2):936-945.
[29] HADAR A, MILANESI E, WALCZAK M, et al. SIRT1, miR-132 and miR-212 link human longevity to Alzheimer’s Disease. Sci Rep. 2018;8(1):8465.
[30] OKADA M, KIM HW, MATSU-URA K, et al. Abrogation of Age-Induced MicroRNA-195 Rejuvenates the Senescent Mesenchymal Stem Cells by Reactivating Telomerase. Stem Cells. 2016;34(1):148-159.
[31] LI X, SONG Y, LIU D, et al. MiR-495 Promotes Senescence of Mesenchymal Stem Cells by Targeting Bmi-1. Cell Physiol Biochem. 2017;42(2):780-796.
[32] JEDARI B, RAHMANI A, NADERI M, et al. MicroRNA-7 promotes neural differentiation of trabecular meshwork mesenchymal stem cell on nanofibrous scaffold. J Cell Biochem. 2020;121(4):2818-2827.
[33] KIM T, MEHTA SL, MORRIS-BLANCO KC, et al. The microRNA miR-7a-5p ameliorates ischemic brain damage by repressing α-synuclein. Sci Signal. 2018;11(560):eaat4285.
[34] KONDO MA, MOHAN A, MATHER KA. Going around in circles: deciphering the role of circular RNAs in neurodegenerative disease. Curr Opin Psychiatry. 2020;33(2):141-147.
[35] JIANG L, LI H, FAN Z, et al. Circular RNA expression profiles in neonatal rats following hypoxic-ischemic brain damage. Int J Mol Med. 2019; 43(4):1699-1708.
[36] BACHMAYR-HEYDA A, REINER AT, AUER K, et al. Correlation of circular RNA abundance with proliferation--exemplified with colorectal and ovarian cancer, idiopathic lung fibrosis, and normal human tissues. Sci Rep. 2015; 5:8057.
[37] KUMAR L, SHAMSUZZAMA, HAQUE R, et al. Circular RNAs: the Emerging Class of Non-coding RNAs and Their Potential Role in Human Neurodegenerative Diseases. Mol Neurobiol. 2017;54(9):7224-7234.
[38] GUERRA BS, LIMA J, ARAUJO B, et al. Biogenesis of circular RNAs and their role in cellular and molecular phenotypes of neurological disorders. Semin Cell Dev Biol. 2020 Sep 3. doi: 10.1016/j.semcdb.2020.08.003. Online ahead of print.
[39] LI HM, MA XL, LI HG. Intriguing circles: Conflicts and controversies in circular RNA research. Wiley Interdiscip Rev RNA. 2019;10(5):e1538.
[40] LI L, CHEN Y, NIE L, et al. MyoD-induced circular RNA CDR1as promotes myogenic differentiation of skeletal muscle satellite cells. Biochim Biophys Acta Gene Regul Mech. 2019;1862(8):807-821.
[41] LIU L, LIU FB, HUANG M, et al. Circular RNA ciRS-7 promotes the proliferation and metastasis of pancreatic cancer by regulating miR-7-mediated EGFR/STAT3 signaling pathway. Hepatobiliary Pancreat Dis Int. 2019;18(6):580-586.
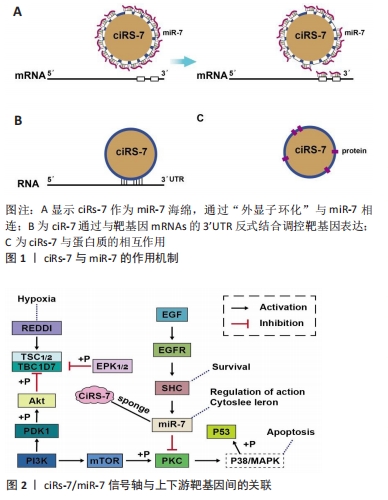
[42] HANSEN TB, JENSEN TI, CLAUSEN BH, et al. Natural RNA circles function as efficient microRNA sponges. Nature. 2013;495(7441):384-388.
[43] PENG L, YUAN XQ, LI GC. The emerging landscape of circular RNA ciRS-7 in cancer (Review). Oncol Rep. 2015;33(6):2669-2674.
[44] CAI W, ZHANG Y, SU Z. ciRS-7 targeting miR-135a-5p promotes neuropathic pain in CCI rats via inflammation and autophagy. Gene. 2020;736:144386.
[45] LIN SP, YE S, LONG Y, et al. Circular RNA expression alterations are involved in OGD/R-induced neuron injury. Biochem Biophys Res Commun. 2016;471(1):52-56.
[46] ZHOU X, JIANG L, FAN G, et al. Role of the ciRS-7/miR-7 axis in the regulation of proliferation, apoptosis and inflammation of chondrocytes induced by IL-1β. Int Immunopharmacol. 2019;71:233-240.
[47] GENG HH, LI R, SU YM, et al. The Circular RNA Cdr1as Promotes Myocardial Infarction by Mediating the Regulation of miR-7a on Its Target Genes Expression. PLoS One. 2016;11(3):e0151753.
[48] CHEN L, YAO H, CHEN X, et al. Ginsenoside Rg1 Decreases Oxidative Stress and Down-Regulates Akt/mTOR Signalling to Attenuate Cognitive Impairment in Mice and Senescence of Neural Stem Cells Induced by D-Galactose. Neurochem Res. 2018;43(2):430-440.
[49] LI YB, WANG Y, TANG JP, et al. Neuroprotective effects of ginsenoside Rg1-induced neural stem cell transplantation on hypoxic-ischemic encephalopathy. Neural Regen Res. 2015;10(5):753-759.
[50] TANG YL, ZHOU Y, WANG YP, et al. SIRT6/NF-κB signaling axis in ginsenoside Rg1-delayed hematopoietic stem/progenitor cell senescence. Int J Clin Exp Pathol. 2015;8(5):5591-5596.
[51] LIU D, YE Y, XU L, et al. Icariin and mesenchymal stem cells synergistically promote angiogenesis and neurogenesis after cerebral ischemia via PI3K and ERK1/2 pathways. Biomed Pharmacother. 2018;108:663-669.
[52] FU X, LI S, ZHOU S, et al. Stimulatory effect of icariin on the proliferation of neural stem cells from rat hippocampus. BMC Complement Altern Med. 2018;18(1):34.
[53] HE Q, LIU Q, CHEN Y, et al. Long-Zhi Decoction Medicated Serum Promotes Angiogenesis in Human Umbilical Vein Endothelial Cells Based on Autophagy. Evid Based Complement Alternat Med. 2018; 2018:6857398.
[54] 周胜强.补阳还五汤调控Cav1/mTOR/ULK1通路介导的自噬抗脑缺血损伤机制研究[D].长沙:湖南中医药大学,2017.
[55] 刘振伟,孙慧君,张金生.补肾化瘀生新方对大脑中动脉栓塞模型大鼠海马区神经干细胞增殖的影响[J].中医学报,2020,35(7):1485-1490.
[56] 张宝霞,张金生,惠小珊,等.补肾化瘀生新方改善缺血缺氧性微环境延缓骨髓间充质干细胞衰老的作用[J].中国实验方剂学杂志, 2020, 26(16):87-92.
|