中国组织工程研究 ›› 2022, Vol. 26 ›› Issue (22): 3602-3608.doi: 10.12307/2022.292
• 生物材料综述 biomaterial review • 上一篇
骨生物支架材料诱导的血管生成
李杜晨晖,田 艾,唐正龙
- 贵州医科大学口腔医学院/附属口腔医院,贵州省贵阳市 550004
-
收稿日期:2020-12-03修回日期:2021-01-27接受日期:2021-05-20出版日期:2022-08-08发布日期:2022-01-13 -
通讯作者:唐正龙,博士,教授,主任医师,博士生导师,贵州医科大学附属口腔医院口腔颌面外科,贵州省贵阳市 550004 -
作者简介:李杜晨晖,男,1993年生,贵州省贵阳市人,汉族,贵州医科大学在读硕士,医师,主要从事骨组织再生与骨免疫方面的研究。 -
基金资助:国家自然科学基金地区科学基金(81760192),项目名称:巨噬细胞影响骨替代品修复骨缺损的分子机制研究初探,项目负责人:田艾;贵阳市科技局联合基金([2018]1-82),项目名称:甲磺酸去铁铵改善骨质疏松下种植体骨结合的实验研究,项目负责人:田艾
Angiogenesis induced by bone bioscaffold materials
Li Duchenhui, Tian Ai, Tang Zhenglong
- School/Hospital of Stomatology of Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Received:2020-12-03Revised:2021-01-27Accepted:2021-05-20Online:2022-08-08Published:2022-01-13 -
Contact:Tang Zhenglong, MD, Professor, Chief physician, Doctoral supervisor, School/Hospital of Stomatology of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Li Duchenhui, Master candidate, Physician, School/Hospital of Stomatology of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
Supported by:National Natural Science Foundation of China, No. 81760192 (to TA); Joint Fund of Guiyang Science and Technology Bureau, No. [2018]1-82 (to TA)
摘要:
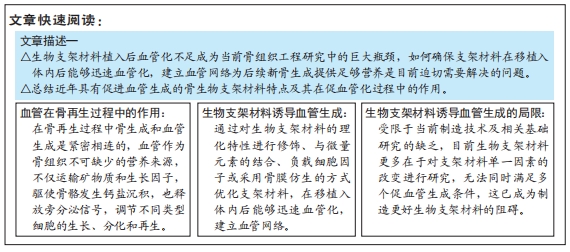
文题释义:
骨稳态:健康的骨骼会持续不断的更新和重塑,包括破坏和形成细胞外骨骼基质等,使骨组织能够适应机械和生物刺激。由于体内平衡的控制,骨组织的吸收和形成过程是平衡的,并且骨骼的数量保持恒定。
骨免疫:骨形成的过程不是单一骨骼系统的参与调节,而是血液循环系统、免疫系统等多系统同时参与、相互串联相互影响的过程。成骨、破骨细胞和免疫细胞会通过两者间共有的调节通路相互影响。
背景:生物支架材料诱导建立的血管网络对于细胞存活和新骨形成是至关重要的。近年来,骨组织工程的学者们更多地关注了骨再生过程中不同支架材料对血管生成的影响。
目的:阐述能促进血管生成骨生物支架材料的特点及其在促血管化过程中的作用。
方法:由第一作者以“Osteogenesis、Biomaterials、bone defect、Angiogenesis、vascularization”为英文检索词,以“成骨分化、生物材料、骨缺损、血管生成、血管化”为中文检索词,检索 PubMed、知网数据库中 2016 至 2020 年已发表的相关文献,并进行筛选、归纳与总结,最终纳入 82篇相关文献进行综述。
结果与结论:支架材料植入后建立的早期血管网络能够提供足够的营养并运输代谢物质,如果局部血管网络形成缓慢造成血液供应缺乏,将导致成骨过程的延迟甚至无法形成新骨。研究人员通过改变支架材料理化性质、负载生长因子缓释系统、结合微量元素或模拟骨膜结构等方式对支架材料进行修饰,使其在诱导骨再生过程中能促进早期血管生成,有利于整个骨再生过程。但受限于当前制造技术及缺乏相关基础研究,目前生物支架材料更多在于对支架材料单一因素的改变进行研究。未来随着制造技术的提升及更多新型生物材料的研发,将制作出集多尺寸孔隙结构、微/纳米级多层级表面形貌为一体的生物支架材料,同时随着对体内骨再生过程中各生长因子分泌释放的精确性研究,以及对缓释、控释系统的精确把握,也将进一步提升生物支架材料的促血管生成作用。
https://orcid.org/0000-0002-7058-2101 (李杜晨晖)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料; 口腔生物材料; 纳米材料; 缓释材料; 材料相容性;组织工程
中图分类号:
引用本文
李杜晨晖, 田 艾, 唐正龙. 骨生物支架材料诱导的血管生成[J]. 中国组织工程研究, 2022, 26(22): 3602-3608.
Li Duchenhui, Tian Ai, Tang Zhenglong. Angiogenesis induced by bone bioscaffold materials[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(22): 3602-3608.
随着微米和纳米制造技术的进步,使得在生物支架材料表面形成范围宽广、定义明确的微米或纳米形貌成为可能,而不同的表面形貌似乎具有不同免疫调节作用,进而影响血管生成[7-10]。早前已有文献报道微观尺度的凹凸表面显著影响巨噬细胞的形状和大小,但未发现与细胞极化状态相关[7]。PATEL等[11]用碳纳米管(25 nm)修饰生物聚合物纳米纤维(500 nm)的表面,形成碳纳米管包覆的纳米纤维材料,在其表面形成独特的双峰纳米形貌;体内研究结果显示,在植入材料的第2周和第4周可见碳纳米管包覆的纳米纤维材料周围有大量成纤维细胞聚集和新生血管形成,同时检测到血管性血友病因子阳性细胞明显增加。其原因可能是由于碳纳米管与生物蛋白(例如血液和黏附蛋白)的相互作用导致其表面特性的改变,影响了巨噬细胞的募集和融合以减少黏附介导的异物反应,同时通过增强内皮细胞黏附而刺激内皮细胞功能,最终缩短血管生成时间。尽管生物支架材料的微米或纳米形貌修饰策略对增强其组织重塑功能具有一定的价值,但是最近研究显示相较于单一因素的改变,微/纳米组合分层结构表现出协同刺激干细胞生物活性的作用[12-13]。YANG等[14]通过光刻和水热技术相结合在羟基磷灰石生物陶瓷上制备了定义明确的微/纳米层级结构,发现微/纳米组合层级结构具有协同刺激作用,可使干细胞具有更强的生物活性,并通过直接刺激内皮细胞和间接刺激活化巨噬细胞的多途径机制调节组织再生期间的血管生成,显著增强了血管生成因子如内皮型一氧化氮合酶、血管内皮生长因子和碱性成纤维细胞生长因子的表达。这意味着不同的支架材料表面形貌可能会影响血管生成作用,而微观/纳米层级结构具有通过形成良好的骨免疫微环境来促进血管生成、加强骨再生的潜力。
支架材料上的孔隙结构能够允许细胞通过,同时输送血管中的氧气和营养物质,并清除代谢产物[15-16]。孔隙结构会影响细胞的接种效率、细胞活力、迁移增殖能力、排列形态、分化能力及血管生成[17-24]。孔隙结构的几个重要参数,包括孔径、几何形状、方向均匀性、互连性和孔隙率[25-27],这些参数是相互关联的,影响支架材料的血管生成能力。通常有效血管生成的孔隙尺寸阈值是100 μm[20,28-29],当孔径小于100 μm时,整个支架中的细胞分布和血管生成会受到限制[20, 28-29],导致细胞活力降低,细胞增殖和分化被延迟,最终形成纤维组织而不是骨骼[24]。HAYASHI等[30]制造了具有不同孔隙尺寸(100,200和300 μm)的蜂窝状支架,这种蜂窝状结构内的孔隙尺寸均匀且有序排列,具有极高的互连性,蜂窝状支架由AB型碳酸盐磷灰石和约8.5%的碳酸根离子组成,即与人骨矿物质组成相同;在植入兔股骨缺损4周后发现3个孔隙尺寸的蜂窝状支架均形成了新的骨骼和血管,其中300 μm的孔隙内被新骨组织广泛占据。表明支架材料孔隙结构的高度互连性和均匀性可以有效提高孔隙尺寸的阈值,更利于血管生成,促进骨骼再生,同时保证了支架材料的强度。LIU 等[31]使用3-羟基丁酸酯-co-3-羟基己酸酯作为聚合物基质,将其浸入含有氯化钠颗粒的成孔剂中使支架材料上形成大孔结构,再使用硅质介孔泡沫形成纳米级中孔结构,最后通过1,4-二恶烷冷冻干燥在大孔孔壁表面形成微孔,制造了同时具有大孔、中孔和微孔的多尺寸孔隙结构支架,该支架的多尺寸孔隙结构有利于细胞的黏附与浸润,并能促进更多硅离子的释放,进而刺激血管生成特异性基因的表达,如血管内皮生长因子、成纤维细胞生长因子2、血管内皮细胞生长因子受体2。其中血管内皮生长因子和成纤维细胞生长因子2作为启动血管生成所必需的生长因子,是上皮细胞的趋化因子[32-33]。血管内皮生长因子还被认为是内皮细胞增殖、迁移的有效刺激剂,并且通过与它的两个主要受体血管内皮细胞生长因子受体1和血管内皮细胞生长因子受体2的相互作用在体内诱导血管生成。表明骨生物支架材料的多尺寸孔隙结构可能有利于血管生成。LI等[34]通过糖模板-浸出技术将羟磷灰石制作成两种分级多孔支架:①在中心具有1 100-1 250 μm的大孔结构,在外围具有500-650 μm小孔结构的HALS;②在中心具有500-650 μm的小孔结构,而外围有1 100-1 250 μm大孔结构的HASL,体内研究显示不同的孔径分布对血管形成有显著影响,支架材料植入1个月后新生血管成功渗入整个支架,但渗入HASL支架外周的血管直径明显大于中心,而HALS支架的中心则很少形成血管,表现出血管异质化。表明分级支架中的孔隙尺寸分布会影响早期血管形成,而分级支架周围的较大孔径结构可能增强血管生成。
近年来,三维(3D)打印技术已成为一种通用的平台技术,可准确地将生物材料、生物分子甚至活细胞定位在特定区域内,从而显著提高制作具有特定结构、机械强度、物理/化学和生物特性的复杂组织工程支架的能力[35-37]。其中低温3D打印技术已引起越来越多的关注,并被用于开发具有合适机械性能、仿生分层多孔结构和生物分子原位递送能力的骨组织工程支架,以增强细胞反应、促进血管生成和骨形成[38-40]。成骨肽和血管生成肽是功能上分别等同于骨形态发生蛋白2和血管内皮生长因子的合成肽,其具有低成本、较细胞因子更稳定的优点。WANG等[41]在低温条件下将含有水/聚(乳酸-乙醇酸)/二氯甲烷的成骨肽乳液油墨和β-磷酸三钙进行3D打印,制作出具有骨传导性和骨诱导性平衡的定制骨组织工程支架,该支架材料具有分级的多孔结构,并且在机械强度上与人的松质骨相当;再将含血管生成肽的胶原水凝胶包被在支架表面,使该支架材料具有快速释放血管生成肽和缓慢持续释放成骨肽特性;体外研究发现大鼠内皮细胞和骨髓间充质干细胞在支架上均显示出高生存力;同时,由于成骨肽与血管生成肽递送的协同作用可改善大鼠内皮细胞的体外迁移能力,并加速诱导支架内血管化,使新生血管明显增多。
此外还有研究表明,磁响应生物材料能够对外部施加的磁场做出响应,并产生刺激以促进组织再生[42],这种刺激被认为是细胞的机械信号[43]。在掺入了磁性纳米颗粒的聚合物纳米纤维中,纳米粒子在基质中形成聚集体,当支架被外部磁场磁化时,纳米粒子的磁矩运动可能会在聚合物纤维中产生力,这些力会使纤维材料发生微小的形变作用,使附着在支架上的细胞产生拉伸、弯曲或收缩[43-44]。超顺磁性支架是带有meso-2,3-二巯基琥珀酸涂层的磁性g-Fe2O3纳米颗粒。HAO等[45]利用磁化后的超顺磁性支架作为机械刺激平台,向生长在支架上的巨噬细胞施加刺激,发现超顺磁性支架与巨噬细胞的相互作用能抑制TLR2/4的激活并增强血管内皮细胞生长因子受体2的活性,从而抑制促炎性细胞因子白细胞介素1β、肿瘤坏死因子α、单核细胞趋化蛋白1,促破骨细胞分化的细胞因子基质金属蛋白酶9和抗酒石酸磷酸酶的表达;同时上调血管生成因子血管内皮生长因子和血小板源生长因子表达,最终增强了内皮细胞的血管生成作用。
2.2 骨生物支架材料微量元素对血管生成的影响 近年来有研究发现,微量元素对血管生成具有一定的影响。硅作为人体中的主要微量元素能促进骨骼稳态和再生[46]。原硅酸、生物二氧化硅和二氧化硅纳米粒子等在体外实验中能刺激人成骨样细胞中的Ⅰ型胶原合成和成骨细胞分化[47-48]。SUN等[49]通过将纤维内非结晶二氧化硅渗透进胶原蛋白基质中制作出的硅化胶原蛋白支架,被证明具有促进成骨过程中血管生成的潜力;在植入硅化胶原蛋白支架的小鼠颅骨缺损模型中发现,其显著增加了血管连通性和血管管壁厚度,并可从新骨形成区域内鉴定出抗酒石酸酸性磷酸酶阳性单核细胞、巢蛋白阳性骨髓间充质干细胞及血小板-内皮细胞黏附分子阳性和内黏蛋白阳性的新血管数量增加。已有研究表明,血小板-内皮细胞黏附分子(CD31)和内黏蛋白(CD31hiEmcnhi)强阳性的特定血管亚型,偶联血管生成和成骨[50-51];而破骨细胞分泌的血小板源生长因子BB在骨重塑过程中诱导内黏蛋白(CD31hiEmcnhi)阳性血管形成。此外,硅化胶原蛋白支架能够持续释放硅酸,刺激血液中单核细胞及其衍生的巨噬细胞分化为抗酒石酸酸性磷酸酶阳性细胞,并增加基质细胞衍生因子1α、转化生长因子β、血管内皮生长因子a和血小板源生长因子BB的表达,这些细胞因子进一步促进了骨髓间充质干细胞和内皮细胞的归巢以及新血管形成。表明硅化胶原蛋白支架具有通过刺激单核细胞及其衍生巨噬细胞进行免疫调节来增强骨缺损修复过程中血管生成的能力。SONG等[52]将硅酸锌加入支架材料,通过3D打印技术制作的硅酸锌/纳米羟基磷灰石/胶原蛋白支架显著增加了抗酒石酸酸性磷酸酶阳性细胞、巢蛋白阳性骨髓间充质干细胞及血小板-内皮细胞黏附分子(CD31)阳性新生血管的数量,并上调了血管生成相关基因(血管内皮生长因子a和血小板-内皮细胞黏附分子)在新生骨骼中的表达水平;此外,负载10%硅酸锌的硅酸锌/纳米羟基磷灰石/胶原蛋白支架还能激活单核细胞中的p38信号通路,使单核细胞分化为抗酒石酸酸性磷酸酶阳性细胞,并表达高水平的细胞因子基质细胞衍生因子1、转化生长因子β、血管内皮生长因子a和血小板源生长因子BB,将骨髓间充质干细胞和内皮细胞募集到缺损区域,创建一个良好的成骨微环境,促进骨髓间充质干细胞的迁移、分化及血管形成。
近期文献报道,功能化的膜用锌能增强成骨和血管生成反应,并增加组织矿化和类骨质新骨的形成[53]。也有研究表明,在静态体外实验中锌离子的掺杂能增强成骨细胞的黏附与增殖能力[54]。二氧化硅纳米颗粒掺杂(MMA)1-co-(HEMA)1和(MA)3-co-(HEA)2的电纺复合膜,由于负载二氧化硅纳米颗粒,可通过刺激支架的生物活性增强骨传导性[55],以及修饰磷酸钙沉积物的表面来促进其与人体组织的整合;同时具有驱动祖细胞的成骨分化作用。TOLEDAN等[56]用锌离子与该电纺复合膜进行结合制作得到Zn-HOOC-Si-Membrane复合膜,在体内研究中发现该复合膜降低了M1/M2巨噬细胞的比例,促进了促愈合表型M2型巨噬细胞数量的增加,从而诱导血管化,并表现出明显的脉管系统重塑迹象;同时发现,成骨细胞在缺损周围的数量增加,其分泌的血管内皮生长因子能有效促进血管生成,并在血管生长阶段有效抑制金属蛋白酶释放介导的支架降解。
已有研究表明,Sr离子可以刺激骨形成并降低骨吸收[57]。Sr离子可以通过激活膜结合钙敏感受体和Wnt/β-catenin信号通路来激活成骨细胞,促进骨祖细胞向成骨细胞的增殖和分化[58-59]。此外,Sr离子更具有促进血管生成的功能。有研究显示,组织工程骨中Sr离子的释放可以刺激血管内皮生长因子、碱性成纤维细胞生长因子和基质金属蛋白酶2等促血管生成因子的表达[60]。Sr离子还可以通过促进成骨细胞分泌血管生成相关细胞因子来促进血管生成[61-64]。但由于植入部位的炎症反应,成骨细胞对早期血管生成的作用极其有限。ZHAO等[65]发现单分散的含锶生物活性玻璃微球可通过调节巨噬细胞表型控制局部炎症反应,显著促进早期血管生成。使用含锶生物活性玻璃微球原料提取物的条件培养基培养巨噬细胞,巨噬细胞表现出向M2型极化的趋势,并表达高水平的血小板源生长因子BB;同样条件培养基下,当巨噬细胞与脐静脉内皮细胞共培养时,能上调抗炎因子白细胞介素10及精氨酸酶1的表达,显著下调促炎因子如肿瘤坏死因子α、白细胞介素1、白细胞介素6的表达水平,并能检测出大量血小板-内皮细胞黏附分子(CD31)阳性细胞,以及血管生成素和基质细胞衍生因子表达的显著上调;此外,还观察到一些被血小板-内皮细胞黏附分子(CD31)阳性蛋白包围的圆形区域,意味着有相对成熟的血管腔形成;体内研究也在含锶生物活性玻璃微球的支架中心观察到大量新血管及血小板-内皮细胞黏附分子(CD31)阳性蛋白,表明其促进早期的血管生成。
2.3 负载细胞因子的骨生物支架材料对血管生成的影响 过去基于血管内皮生长因子或micro-RNA(miR-26a)的呈递策 略[66-67],将具有骨传导性的磷酸钙水泥与负载血管内皮生长因子的凝胶支架相结合,发现其能够提供成血管和成骨的复合作用[68]。但骨再生作为一个动态而全面的过程,需要在特定的阶段精确释放一定剂量的生长因子和信号分子[69],因此体内模拟不仅仅只是生长因子从给药系统或呈递系统中释放,由于药物自身的快速或持续释放不能确保与体内动态环境精准协调,所以在骨组织再生修复策略中时间、剂量等因素的多维协调非常重要。
早期研究表明在免疫调节中,M1型巨噬细胞可以分泌血管内皮生长因子以促进早期血管芽的形成,而M2型巨噬细胞可以分泌血小板源生长因子BB和其他参与血管重塑的因子一起作用,使血管稳定和成熟[70]。与传统β-磷酸三钙支架相比,5%硅酸钙-β-磷酸三钙支架可增强血管形成和骨诱导作用,并刺激血小板源生长因子BB和基质细胞衍生因子1向周围环境的分泌[71]。基于这一研究基础,LI等[72]将能够促进M1型巨噬细胞极化的干扰素γ负载在5%硅酸钙-β-磷酸三钙支架上,使干扰素γ和Si离子从支架中顺序释放,以调控M1和M2巨噬细胞的极化顺序;体内模型研究显示,在材料植入早期(1-3 d),支架释放干扰素γ,促进巨噬细胞向M1型极化并在缺损处募集更多的巨噬细胞;然后支架释放Si离子,诱导巨噬细胞向M2型极化,与此同时生物支架材料开始降解,在这一过程中能检测出大量血小板-内皮细胞黏附分子(CD31)阳性的血管内皮细胞,以及血管内皮生长因子、基质细胞衍生因子1和血小板源生长因子表达水平的升高,以促进造血干细胞的募集、稳定及血管生成作用。
在负载生长因子的骨生物支架材料研发过程中,如何保持生长因子的生物活性并模仿体内供应模式是极具挑战性的。TANG等[73]制造的2-N,6-O-硫酸壳聚糖功能化的双模块支架,由分级多孔结构的生物活性玻璃(模块Ⅰ)和丙烯酰化明胶水凝胶柱(模块Ⅱ)组成,该支架材料能够实现成骨因子重组人骨形态发生蛋白2和血管内皮生长因子的差异化递送,以协调骨骼再生过程中的成骨作用和血管生成作用。体外和体内研究均表明,该支架材料在初期能释放高浓度的血管内皮生长因子,后随着时间推移血管内皮生长因子的释放浓度逐渐降低,而重组人骨形态发生蛋白2的释放则表现为缓慢、可持续,这种差异性释放模式类似于生长因子在体内供应的模式。在早期血管内皮生长因子释放过程中能检测出内皮细胞基因标志物(血小板-内皮细胞黏附分子CD31)、血管生成素、血管性假血友病因子的表达水平升高,促进毛细管形成和小血管增生。研究者还对2-N,6-O-硫酸壳聚糖功能化的双模块支架进行化学修饰,形成仿生细胞外基质环境,从而增强生长因子的生物活性,降低头蛋白的拮抗作用。
脱矿骨基质支架具有良好的生物相容性、低抗原性、天然的多孔结构与无细胞毒性,是骨骼再生的理想材料之一。但是在制备过程中,常常会因去除骨诱导性生长因子而破坏脱矿骨基质支架的骨诱导能力。LIANG等[74]开发的人骨形态发生蛋白2基因激活脱矿骨基质支架,能增强脱矿骨基质的骨诱导性并改善骨修复,通过逐层自组装将聚乙烯亚胺和hBMP2质粒(phBMP2)依次包被在骨髓间充质干细胞衍生的微泡上形成微泡-聚乙烯亚胺/phBMP2非病毒基因载体,最后通过将微泡-聚乙烯亚胺/phBMP2加载到脱矿骨基质支架上制造出基因激活的支架(脱矿骨基质/微泡-聚乙烯亚胺/phBMP2)。研究表明该支架材料能促进内皮细胞向缺损部位的迁移,并表现出Ⅰ型跨膜糖蛋白(CD34)阳性细胞的增加;同时,由于骨髓间充质干细胞衍生的微泡的存在,使该基因激活支架可以加速诱导损伤部位的内皮细胞重建新血管。
2.4 仿生人工骨膜生物支架材料对血管生成的影响 骨膜是高度血管化的组织,并深入参与骨愈合过程,在骨骼的发育和损伤愈合过程中都起着至关重要的作用。但是很少有研究集中在人造骨膜上,人造骨膜的构造也很复杂,且临床上存在生物学风险,因此受到了限制。受骨膜的分阶段发育过程及其结构特征的启发,WU等[75]将微溶胶电纺技术与胶原纳米纤维自组装技术相结合,构建出能持久释放血管内皮生长因子的分层微/纳米纤维仿生骨膜,其可以单独使用或与现有的骨移植材料结合使用,同时作用于骨膜和骨缺损两个方面,以降低临床骨缺损患者发生骨不连的可能性。将血管内皮生长因子包裹在透明质酸水溶胶中,然后将透明质酸水溶胶分散在纺丝溶液中形成乳液,最终获得乳液中的微滴,即透明质酸微溶胶颗粒,该工艺方法被称为微溶胶静电纺丝法。在静电纺丝过程中,透明质酸微溶胶颗粒被封装到左旋聚乳段纤维中,形成用于血运重建的芯鞘结构;再将Ⅰ型胶原蛋白纳米纤维装载到电纺膜的孔中,形成微/纳米多层膜结构支架,能够增强纤维膜的生物相容性和物理性能,最终形成适合间充质干细胞存活、黏附、增殖和成骨分化的微环境,实现骨膜仿生结构及促进骨组织再生。透明质酸溶胶颗粒不仅可以形成一个隔离系统来保护血管内皮生长因子在有机溶剂中的生物活性,还可以作为血管内皮生长因子自由扩散释放的储存库。该支架材料能在4周左右的时间内缓慢稳定释放血管内皮生长因子,促进细胞的迁移,诱导由细胞聚集体形成散射状的管样结构组成初期血管样网络,从而加速血管的生成。
已有研究证明,由骨膜基质组成的生物支架能进行自发的生物矿化作用,并在骨缺损修复中发挥积极作用[76-77]。传统的脱细胞支架由于其密集的胶原纤维网络,使得细胞通透性较差[78],导致其在骨骼损伤修复早期对炎症反应的调控较差。细胞外基质水凝胶是一种多孔隙、可降解的生物材料,在血管生成和免疫调节过程中诱导各种祖细胞和巨噬细胞的趋化性,促进M1型巨噬细胞向M2型巨噬细胞极化,调控炎症反应,促进组织再生[79-80]。QIU等[81]发现,源自骨膜的细胞外基质水凝胶能在没有其他生物活性剂的情况下调节不同的骨骼修复阶段,体外研究显示,源自骨膜的细胞外基质水凝胶能诱导巨噬细胞募集并向M2型极化,促进骨髓间充质干细胞分化为内皮样细胞,以诱导血管生成。随着骨骼修复过程的推进,源自骨膜的细胞外基质水凝胶促进了血管迁移、较大血管的发育以及功能性血管的形成。源自骨膜的细胞外基质水凝胶的促进修复机制可能归因于生物活性因子和脱细胞细胞外基质超微结构的综合优势。尽管水凝胶失去了其原有结构,但保留了天然的生化线索,提供生物分子并重现了细胞外基质结构。LIU等[82]通过乳液法制备磷酸钙纳米粒子,然后通过电纺纤维将磷酸钙纳米粒子与明胶-甲基丙烯酰基合并,构建出混合水凝胶纤维,这是一种有机-无机混合仿生骨膜纤维,具有良好的形态和机械性能,能够诱导局部矿化并控制离子的长期释放;使用混合水凝胶纤维与人脐静脉内皮细胞共培养后,可以发现新生血管样组织长度更长,并具有更多的分支连接,从而形成更加紧密的血管网络结构,这意味着其具有促进体外血管生成的能力。

| [1] BALAGANGADHARAN K, DHIVYA S, SELVAMURUGAN N. Chitosan based nanofibers in bone tissue engineering. Int J Biol Macromol. 2017;104(Pt B):1372-1382. [2] ALMUBARAK S, NETHERCOTT H, FREEBERG M, et al. Tissue engineering strategies for promoting vascularized bone regeneration. Bone. 2016; 83:197-209. [3] BATTISTON KG, CHEUNG JWC, JAIN D, et al. Biomaterials in co-culture systems: towards optimizing tissue integration and cell signaling within scaffolds. Biomaterials. 2014;35(15):4465-4476. [4] PARK M, KIM H, SONG KH, et al. Ultra-thin, aligned, free-standing nanofiber membranes to recapitulate multi-layered blood vessel/tissue interface for leukocyte infiltration study. Biomaterials. 2018;169:22-34. [5] CHEN Z, BACHHUKA A, HAN S, et al. Tuning Chemistry and Topography of Nanoengineered Surfaces to Manipulate Immune Response for Bone Regeneration Applications. ACS Nano. 2017;11(5):4494-4506. [6] ROH JD, SAWH-MARTINEZ R, BRENNAN MP, et al. Tissue-engineered vascular grafts transform into mature blood vessels via an inflammation-mediated process of vascular remodeling. Proc Natl Acad Sci U S A. 2010;107(10):4669-4674. [7] MALHEIRO V, LEHNER F, DINCA V, et al. Convex and concave micro-structured silicone controls the shape, but not the polarization state of human macrophages. Biomater Sci. 2016;4(11):1562-1573. [8] SINGH S, AWUAH D, ROSTAM HM, et al. Unbiased Analysis of the Impact of Micropatterned Biomaterials on Macrophage Behavior Provides Insights beyond Predefined Polarization States. ACS Biomater Sci Eng. 2017;3(6): 969-978. [9] WANG K, HOU WD, WANG X, et al. Overcoming foreign-body reaction through nanotopography: Biocompatibility and immunoisolation properties of a nanofibrous membrane. Biomaterials. 2016;102:249-258. [10] GULATI K, HAMLET SM, IVANOVSKI S. Tailoring the immuno-responsiveness of anodized nano-engineered titanium implants. J Mater Chem B. 2018;6(18):2677-2689. [11] PATEL KD, KIM TH, MANDAKHBAYAR N, et al. Coating biopolymer nanofibers with carbon nanotubes accelerates tissue healing and bone regeneration through orchestrated cell- and tissue-regulatory responses. Acta Biomater. 2020;108:97-110. [12] ZHAO C, WANG X, GAO L, et al. The role of the micro-pattern and nano-topography of hydroxyapatite bioceramics on stimulating osteogenic differentiation of mesenchymal stem cells. Acta Biomater. 2018;73: 509-521. [13] JAGGY M, ZHANG P, GREINER AM, et al. Hierarchical Micro-Nano Surface Topography Promotes Long-Term Maintenance of Undifferentiated Mouse Embryonic Stem Cells. Nano Lett. 2015;15(10): 7146-7154. [14] YANG C, ZHAO C, WANG X, et al. Stimulation of osteogenesis and angiogenesis by micro/nano hierarchical hydroxyapatite via macrophage immunomodulation. Nanoscale. 2019;11(38):17699-17708. [15] BOBBERT FSL, ZADPOOR AA. Effects of bone substitute architecture and surface properties on cell response, angiogenesis, and structure of new bone. J Mater Chem B. 2017;5(31):6175-6192. [16] KIM SS, UTSUNOMIYA H, KOSKI JA, et al. Survival and function of hepatocytes on a novel three-dimensional synthetic biodegradable polymer scaffold with an intrinsic network of channels. Ann Surg. 1998; 228(1):8-13. [17] AMINI AR, ADAMS DJ, LAURENCIN CT, et al. Optimally porous and biomechanically compatible scaffolds for large-area bone regeneration. Tissue Eng Part A. 2012;18(13-14):1376-1388. [18] MARCOS-CAMPOS I, MAROLT D, PETRIDIS P, et al. Bone scaffold architecture modulates the development of mineralized bone matrix by human embryonic stem cells. Biomaterials. 2012;33(33):8329-8342. [19] MURPHY CM, HAUGH MG, O’BRIEN FJ. The effect of mean pore size on cell attachment, proliferation and migration in collagen-glycosaminoglycan scaffolds for bone tissue engineering. Biomaterials. 2010;31(3):461-466. [20] REINWALD Y, JOHAL RK, GHAEMMAGHAMI AM, et al. Interconnectivity and permeability of supercritical fluid-foamed scaffolds and the effect of their structural properties on cell distribution. Polymer. 2014;55(1): 435-444. [21] LIU X, WU S, YEUNG KEK, et al. Relationship between osseointegration and superelastic biomechanics in porous NiTi scaffolds. Biomaterials. 2011;32(2):330-338. [22] KIM K, DEAN D, WALLACE J, et al. The influence of stereolithographic scaffold architecture and composition on osteogenic signal expression with rat bone marrow stromal cells. Biomaterials. 2011;32(15):3750-3763. [23] SICCHIERI LG, CRIPPA GE, OLIVEIRA PT, et al. Pore size regulates cell and tissue interactions with PLGA-CaP scaffolds used for bone engineering. J Tissue Eng Regen Med. 2012:6(2):155-162. [24] FENG B, JINKANG Z, ZHEN W, et al. The effect of pore size on tissue ingrowth and neovascularization in porous bioceramics of controlled architecture in vivo. Biomed Mater. 2011:6(1):015007. [25] DIAS MR, FERNANDES PR, GUEDES M, et al. Permeability analysis of scaffolds for bone tissue engineering. J Biomech. 2012;45(6):938-944. [26] ZADPOOR AA. Bone tissue regeneration: the role of scaffold geometry. Biomater Sci. 2015;3(2):231-245. [27] YILGOR P, YILMAZ G, ONAL M B, et al. An in vivo study on the effect of scaffold geometry and growth factor release on the healing of bone defects. J Tissue Eng Regen Med. 2013;7(9):687-696. [28] BOBBERT FSL, ZADPOOR AA. Effects of bone substitute architecture and surface properties on cell response, angiogenesis, and structure of new bone. J Mater Chem B. 2017;5(31):6175-6192. [29] ZHANG Y, FAN W, MA Z, et al. The effects of pore architecture in silk fibroin scaffolds on the growth and differentiation of mesenchymal stem cells expressing BMP7. ACTA BIOMATER. 2010;6(8):3021-3028. [30] HAYASHI K, MUNAR ML, ISHIKAWA K. Effects of macropore size in carbonate apatite honeycomb scaffolds on bone regeneration. Mater Sci Eng C. 2020;111:110848. [31] LIU Y, YANG S, CAO L, et al. Facilitated vascularization and enhanced bone regeneration by manipulation hierarchical pore structure of scaffolds. Mater Sci Eng C. 2020;110:110622. [32] DECKERS MML, MARCEL K, CHRIS VDB, et al. Expression of vascular endothelial growth factors and their receptors during osteoblast differentiation. Endocrinology. 2000;141(5):1667-1674. [33] FERRARA N, GERBER HP, LECOUTER J. The biology of VEGF and its receptors. Nat Med. 2003;9(6):669-676. [34] LI J, XU T, HOU W, et al. The response of host blood vessels to graded distribution of macro-pores size in the process of ectopic osteogenesis. Mater Sci Eng C. 2020;109:110641. [35] BRUNELLO G, SIVOLELLA S, MENEGHELLO R, et al. Powder-based 3D printing for bone tissue engineering. Biotechnol Adv. 2016;34(5):740-753. [36] WANG X, XU S, ZHOU S, et al. Topological design and additive manufacturing of porous metals for bone scaffolds and orthopaedic implants: A review. Biomaterials. 2016;83:127-141. [37] ZAMANI Y, AMOABEDINY G, MOHAMMADI J, et al. 3D-printed poly(E-caprolactone) scaffold with gradient mechanical properties according to force distribution in the mandible for mandibular bone tissue engineering. J Mech Behav Biomed Mater. 2020;104:103638. [38] LEE H, YANG GH, KIM M, et al. Fabrication of micro/nanoporous collagen/dECM/silk-fibroin biocomposite scaffolds using a low temperature 3D printing process for bone tissue regeneration. Mater Sci Eng C. 2018;84:140-147. [39] LUO C, XIE R, ZHANG J, et al. Low temperature 3D printing of tissue cartilage engineered with gelatin methacrylamide. Issue Eng Part C Methods. 2020;26(6):306-316. [40] WANG Z, FLORCZYK SJ. Freeze-FRESH: A 3D Printing Technique to Produce Biomaterial Scaffolds with Hierarchical Porosity. Materials (Basel). 2020;13(2):354. [41] WANG C, LAI J, LI K, et al. Cryogenic 3D printing of dual-delivery scaffolds for improved bone regeneration with enhanced vascularization. Bioact Mater. 2021;6(1):137-145. [42] YUN HM, AHN SJ, PARK KR, et al. Magnetic nanocomposite scaffolds combined with static magnetic field in the stimulation of osteoblastic differentiation and bone formation. Biomaterials. 2016;85:88-98. [43] SAPIR Y, COHEN S, FRIEDMAN G, et al. The promotion of in vitro vessel-like organization of endothelial cells in magnetically responsive alginate scaffolds. Biomaterials. 2012;33(16):4100-4109. [44] FUHRER R, HOFMANN S, HILD N, et al. Pressureless Mechanical Induction of Stem Cell Differentiation Is Dose and Frequency Dependent. PloS One. 2013;8(11):1362. [45] HAO S, MENG J, ZHANG Y, et al. Macrophage phenotypic mechanomodulation of enhancing bone regeneration by superparamagnetic scaffold upon magnetization. Biomaterials. 2017;140:16-25. [46] RODELLA LF, BONAZZA V, LABANCA M, et al. A review of the effects of dietary silicon intake on bone homeostasis and regeneration. J Nutr Health Aging. 2014;18(9):820-826. [47] REFFITT DM, OGSTON N, JUGDAOHSINGH R, et al. Orthosilicic acid stimulates collagen type 1 synthesis and osteoblastic differentiation in human osteoblast-like cells in vitro. Bone. 2003;32(2):127-135. [48] WIENS M, WANG X, SCHLOSSMACHER U, et al. Osteogenic potential of biosilica on human osteoblast-like (SaOS-2) cells. Calcif Tissue Int. 2010;87(6):513-524. [49] SUN JL, JIAO K, NIU LN, et al. Intrafibrillar silicified collagen scaffold modulates monocyte to promote cell homing, angiogenesis and bone regeneration. Biomaterials. 2017;113:203-216. [50] KUSUMBE AP, RAMASAMY SK, ADAMS RH. Coupling of angiogenesis and osteogenesis by a specific vessel subtype in bone. Nature. 2014; 507(7492):323-328. [51] RAMASAMY SK, KUSUMBE AP, WANG L, et al. Endothelial Notch activity promotes angiogenesis and osteogenesis in bone. Nature. 2014;507(7492):376-380. [52] SONG Y, WU H, GAO Y, et al. Zinc Silicate/Nano-Hydroxyapatite/Collagen Scaffolds Promote Angiogenesis and Bone Regeneration via the p38 MAPK Pathway in Activated Monocytes. ACS Appl Mater Interfaces. 2020;12(14):16058-16075. [53] MANUEL T, JOSÉ L GP, AIDA GC, et al. Novel non-resorbable polymeric-nanostructured scaffolds for guided bone regeneration. Clin Oral Investig. 2020;24(6):2037-2049. [54] OSORIO R, CARRASCO-CARMONA L, TOLEDANO M, et al. Ex vivo investigations on bioinspired electrospun membranes as potential biomaterials for bone regeneration. J Dent. 2020;98:103359. [55] CASTILLO-DALÍ G, CASTILLO-OYAGÜE R, TERRIZA A, et al. Pre-prosthetic use of poly (lactic-co-glycolic acid) membranes treated with oxygen plasma and TiO2 nanocomposite particles for guided bone regeneration processes. J Dent. 2016;47:71-79. [56] TOLEDANO M, TOLEDANO-OSORIO M, OSORIO R, et al. Doxycycline and Zinc Loaded Silica-Nanofibrous Polymers as Biomaterials for Bone Regeneration. Polymers. 2020;12(5):1201. [57] PILMANE M, SALMA-ANCANE K, LOCA D, et al. Strontium and strontium ranelate: Historical review of some of their functions. Mater Sci Eng C. 2017;78:1222-1230. [58] YANG F, YANG D, TU J, et al. Strontium enhances osteogenic differentiation of mesenchymal stem cells and in vivo bone formation by activating Wnt/catenin signaling. Stem Cells. 2011;29(6):981-991. [59] FROMIGUE O, HAY E, BARBARA A, et al. Calcium sensing receptor-dependent and receptor-independent activation of osteoblast replication and survival by strontium ranelate. J Cell Mol Med. 2009; 3(8B):2189-2199. [60] WANG X, WANG Y, LI L, et al. Stimulations of strontium-doped calcium polyphosphate for bone tissue engineering to protein secretion and mRNA expression of the angiogenic growth factors from endothelial cells in vitro. Ceram Int. 2014;40(5):6999-7005. [61] ZHAO S, ZHANG J, ZHU M, et al. Three-dimensional printed strontium-containing mesoporous bioactive glass scaffolds for repairing rat critical-sized calvarial defects. Acta Biomater. 2015;12:270-280. [62] GU Z, XIE H, LI L, et al. Application of strontium-doped calcium polyphosphate scaffold on angiogenesis for bone tissue engineering. J Mater Sci Mater Med. 2013;24(5):1251-1260. [63] MAO L, XIA L, CHANG J, et al. The synergistic effects of Sr and Si bioactive ions on osteogenesis, osteoclastogenesis and angiogenesis for osteoporotic bone regeneration. Acta Biomater. 2017;61:217-232. [64] ZHANG W, CAO H, ZHANG X, et al. A strontium-incorporated nanoporous titanium implant surface for rapid osseointegration. Nanoscale.2016;8(9):5291-5301. [65] ZHAO F, LEI B, LI X, et al. Promoting in vivo early angiogenesis with sub-micrometer strontium-contained bioactive microspheres through modulating macrophage phenotypes. Biomaterials. 2018;178:36-47. [66] KAIGLER D, SILVA EA, MOONEY DJ. Guided Bone Regeneration Using Injectable Vascular Endothelial Growth Factor Delivery Gel. J Periodontol. 2013;84(2):230-238. [67] LI Y, FAN L, LIU S, et al. The promotion of bone regeneration through positive regulation of angiogenic–osteogenic coupling using microRNA-26a. Biomaterials. 2013;34(21):5048-5058. [68] AHLFELD T, AKKINENI AR, FORSTER Y, et al. Design and Fabrication of Complex Scaffolds for Bone Defect Healing: Combined 3D Plotting of a Calcium Phosphate Cement and a Growth Factor-Loaded Hydrogel. Ann Biomed Eng. 2017;45(1):224-236. [69] SPILLER K, NASSIRI S, WITHEREL CE, et al. Sequential delivery of immunomodulatory cytokines to facilitate the M1-to-M2 transition of macrophages and enhance vascularization of bone scaffolds. Biomaterials. 2015;37:194-207. [70] SPILLER KL, ANFANG RR, SPILLER KJ, et al. The role of macrophage phenotype in vascularization of tissue engineering scaffolds. Biomaterials. 2014;35(15):4477-4488. [71] DENG Y, JIANG C, LI C, et al. 3D printed scaffolds of calcium silicate-doped beta-TCP synergize with co-cultured endothelial and stromal cells to promote vascularization and bone formation. Sci Rep.2017;7(1):5588. [72] LI T, PENG M, YANG Z, et al. 3D-printed IFN-gamma-loading calcium silicate-beta-tricalcium phosphate scaffold sequentially activates M1 and M2 polarization of macrophages to promote vascularization of tissue engineering bone. Acta Biomater. 2018;71:96-107. [73] TANG W, YU Y, WANG J, et al. Enhancement and orchestration of osteogenesis and angiogenesis by a dual-modular design of growth factors delivery scaffolds and 26SCS decoration. Biomaterials. 2020; 232:119645. [74] LIANG Z, LUO Y, LV Y. Mesenchymal stem cell-derived microvesicles mediate BMP2 gene delivery and enhance bone regeneration. J Mater Chem B. 2020;8(30):6378-6389. [75] WU L, GU Y, LIU L, et al. Hierarchical micro/nanofibrous membranes of sustained releasing VEGF for periosteal regeneratio. Biomaterials. 2020;227:119555. [76] LIN X, ZHAO C, ZHU P, et al. Periosteum Extracellular‐Matrix‐Mediated Acellular Mineralization during Bone Formation. Adv Healthc Mater. 2018;7(4):1700660. [77] CHEN K, LIN X, ZHANG Q, et al. Decellularized periosteum as a potential biologic scaffold for bone tissue engineering. Acta Biomater. 2015;19:46-55. [78] LOVATI AB, MARTA B, MATTEO M. Decellularized and Engineered Tendons as Biological Substitutes: A Critical Review. Stem Cells Int. 2016;2016:7276150. [79] SALDIN LT, CRAMER MC, VELANKAR SS, et al. Extracellular matrix hydrogels from decellularized tissues: Structure and function. Acta Biomater. 2017;49:1-15. [80] SPANG MT, CHRISTMAN KL. Extracellular matrix hydrogel therapies: In vivo applications and development. Acta Biomater. 2018;68:1-14. [81] QIU P, LI M, CHEN K, et al. Periosteal matrix-derived hydrogel promotes bone repair through an early immune regulation coupled with enhanced angio- and osteogenesis. Biomaterials. 2020;227:119552. [82] LIU W, BI W, SUN Y, et al. Biomimetic organic-inorganic hybrid hydrogel electrospinning periosteum for accelerating bone regeneration. Mater Sci Eng C Mater Biol Appl. 2020;110:110670. |
| [1] | 薛亚东, 周新社, 裴立家, 孟繁宇, 李 键, 王金子. 自体髂骨块联合钛板重建Paprosky Ⅲ型髋臼骨缺损为假体提供坚强的初始固定#br#[J]. 中国组织工程研究, 2022, 26(9): 1424-1428. |
| [2] | 姚晓玲, 彭建城, 许岳荣, 杨志东, 张顺聪. 可变角度零切迹前路椎间融合内固定系统治疗脊髓型颈椎病:30个月随访[J]. 中国组织工程研究, 2022, 26(9): 1377-1382. |
| [3] | 王 景, 熊 山, 曹 金, 冯林伟, 王 信. 白细胞介素3在骨代谢中的作用及机制[J]. 中国组织工程研究, 2022, 26(8): 1260-1265. |
| [4] | 安维政, 何 萧, 任 帅, 刘建宇. 肌源干细胞在周围神经再生中的潜力[J]. 中国组织工程研究, 2022, 26(7): 1130-1136. |
| [5] | 张璟琳, 冷 敏, 朱博恒, 汪 虹. 干细胞源外泌体促进糖尿病创面愈合的机制及应用[J]. 中国组织工程研究, 2022, 26(7): 1113-1118. |
| [6] | 吴玮玥, 郭晓东, 包崇云. 工程化外泌体在骨修复再生中的应用[J]. 中国组织工程研究, 2022, 26(7): 1102-1106. |
| [7] | 周洪琴, 吴丹丹, 杨 琨, 刘 琪. 传递特定miRNA的外泌体可调控成骨并促进成血管[J]. 中国组织工程研究, 2022, 26(7): 1107-1112. |
| [8] | 杨 峰, 赵 骞, 张世轩, 赵铁男, 冯 博. 雷帕霉素联合CD133抗体支架预防血管再狭窄的有效性和安全性[J]. 中国组织工程研究, 2022, 26(4): 579-584. |
| [9] | 陈晓旭, 罗雅馨, 毕浩然, 杨 琨. 脱细胞支架制备及其在组织工程和再生医学中的应用[J]. 中国组织工程研究, 2022, 26(4): 591-596. |
| [10] | 康坤龙, 王新涛. 生物支架材料促进骨髓间充质干细胞成骨分化的研究热点[J]. 中国组织工程研究, 2022, 26(4): 597-603. |
| [11] | 沈佳华, 付 勇. 基于石墨烯的纳米材料可否在干细胞领域应用[J]. 中国组织工程研究, 2022, 26(4): 604-609. |
| [12] | 王阮彬, 程丽乾, 陈 凯. 高分子材料在3D打印生物骨骼及支架中的应用与价值[J]. 中国组织工程研究, 2022, 26(4): 610-616. |
| [13] | 张 通, 蔡金池, 袁志发, 赵海燕, 韩兴文, 王文己. 基于透明质酸的复合水凝胶修复骨关节炎软骨损伤:应用与机制[J]. 中国组织工程研究, 2022, 26(4): 617-625. |
| [14] | 李 辉, 陈良龙. 植骨材料在脊柱结核治疗中的应用与特征[J]. 中国组织工程研究, 2022, 26(4): 626-630. |
| [15] | 高仓健, 杨 振, 刘舒云, 李 浩, 付力伟, 赵天元, 陈 威, 廖志垚, 李品学, 眭 翔, 郭全义. 静电纺丝技术在肩袖损伤修复中的应用[J]. 中国组织工程研究, 2022, 26(4): 637-642. |
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料; 口腔生物材料; 纳米材料; 缓释材料; 材料相容性;组织工程
1.2 资料筛选
纳入标准:与研究骨生物支架材料相关的文章;与骨再生、血管生成相关的文章;同一领域选择近期发表或在权威杂志上发表的相关文章。
排除标准:重复性研究;内容陈旧、相关性差及无关文章。
1.3 资料提取与文献质量评价 根据检索词,计算机共检索到242篇相关文献,阅读标题和摘要,按照纳入及排除标准筛选后,最后共纳入82篇文献进行分析和整理,见图1。

中国组织工程研究杂志出版内容重点:生物材料;骨生物材料; 口腔生物材料; 纳米材料; 缓释材料; 材料相容性;组织工程

文题释义:
骨稳态:健康的骨骼会持续不断的更新和重塑,包括破坏和形成细胞外骨骼基质等,使骨组织能够适应机械和生物刺激。由于体内平衡的控制,骨组织的吸收和形成过程是平衡的,并且骨骼的数量保持恒定。
骨免疫:骨形成的过程不是单一骨骼系统的参与调节,而是血液循环系统、免疫系统等多系统同时参与、相互串联相互影响的过程。成骨、破骨细胞和免疫细胞会通过两者间共有的调节通路相互影响。
生物支架材料诱导建立的血管网络对于细胞存活和新骨形成是至关重要的,该文通过搜索近五年生物支架材料促进血管生成相关研究文献,总结归纳目前主流的生物支架材料优化策略包括改变支架材料理化性质、负载生长因子缓释系统、结合微量元素或模拟骨膜结构等方式;集中讨论各类策略的优缺点及其在血管生成中的作用。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||