中国组织工程研究 ›› 2021, Vol. 25 ›› Issue (32): 5171-5177.doi: 10.12307/2021.220
• 组织构建综述 tissue construction review • 上一篇 下一篇
骨关节炎的单克隆抗体治疗
张新潮1,王炳南2,葛 喆2
- 1复旦大学附属金山医院骨科,上海市 200540;2复旦大学附属金山医院,上海市 200540
-
收稿日期:2020-07-20修回日期:2020-07-22接受日期:2020-11-13出版日期:2021-11-18发布日期:2021-07-26 -
作者简介:张新潮,男,1967年生,河南省洛阳市人,汉族,1992年上海医科大学毕业,主任医师,教授,硕士生导师,主要从事创伤骨科、关节外科的研究
Monoclonal antibody therapy for osteoarthritis
Zhang Xinchao1, Wang Bingnan2, Ge Zhe2
- 1Department of Orthopedics, Jinshan Hospital, Fudan University, Shanghai 200540, China; 2Jinshan Hospital, Fudan University, Shanghai 200540, China
-
Received:2020-07-20Revised:2020-07-22Accepted:2020-11-13Online:2021-11-18Published:2021-07-26 -
About author:Zhang Xinchao, Chief physician, Professor, Master’s supervisor, Department of Orthopedics, Jinshan Hospital, Fudan University, Shanghai 200540, China
摘要:
文题释义:
单克隆抗体:针对单一抗原决定基,由一个识别一种抗原决定基的B细胞克隆合成并分泌的抗体,称为单克隆抗体。作为一种新型的生物制剂药物,单克隆抗体近年来被大量开发并逐渐投入临床。单克隆抗体的优点是靶向性强、疗效好、药效持续时间长、不良反应小以及研发速度快、成本低等,因此受到国内外学者的重视。
背景:在骨关节炎的发病过程中有大量炎症因子产生,针对炎症因子的抗体最近成为了研究热点。
目的:概述治疗骨关节炎单克隆抗体的最新研究进展,为临床治疗骨关节炎提供参考。
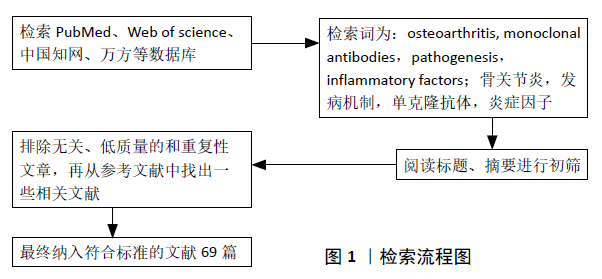
方法:利用计算机检索CNKI、万方、PubMed及Web of Science数据库2006年1月至2020年5月的相关文章,检索词为“骨关节炎,单克隆抗体,炎症因子,osteoarthritis,monoclonal antibodies,inflammatory factors”。查阅相关文章,最终共纳入69篇文献进行结果分析。
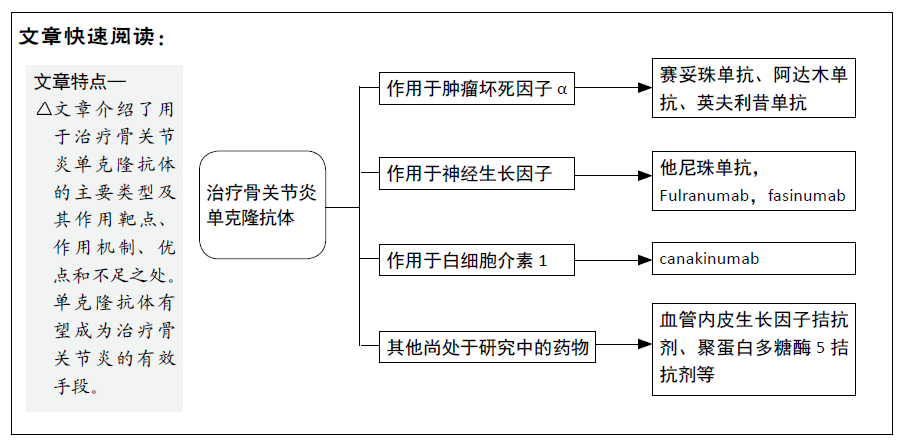
结果与结论:①骨关节炎是一种炎症性、退行性疾病,与传统非类固醇类抗炎药相比,单克隆抗体因其靶向性好、不良反应小的特点,在治疗骨关节炎疼痛方面已显示出良好效果;②部分单克隆抗体会加大患者关节置换的风险,因此药物选择应当因人而异;③随着研究的不断深入,会有更多针对不同靶点的单克隆抗体产生,为临床治疗骨关节炎提供更多选择。
中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程
中图分类号:
引用本文
张新潮, 王炳南, 葛 喆. 骨关节炎的单克隆抗体治疗[J]. 中国组织工程研究, 2021, 25(32): 5171-5177.
Zhang Xinchao, Wang Bingnan, Ge Zhe. Monoclonal antibody therapy for osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(32): 5171-5177.
2.1.1 赛妥珠单抗 赛妥珠单抗是一种聚乙二醇化Fab片段的抗肿瘤坏死因子α、且对肿瘤坏死因子α具有高亲和力的人源化单克隆抗体。赛妥珠单抗缺乏FC片段,这导致它的作用机制与其他肿瘤坏死因子α拮抗剂不同,该药物缺乏诱导调节型巨噬细胞形成、抗体依赖性细胞毒性、补体依赖性细胞毒性和通过反向信号诱导细胞凋亡的能力。然而,赛妥珠单抗可以像英夫利昔单抗和阿达木单抗一样有效抑制炎症递质和增加调节性T细胞的活性[11]。有学者将99Tcm标记的赛妥珠单抗通过静脉注射入患者体内[12],用于观察肿瘤坏死因子α在侵蚀性骨关节炎患者体内的分布,以及关节对于赛妥珠单抗的吸收情况。在注射单克隆抗体24 h之后,观察发现,在肿胀的关节中示踪剂显著吸收的比例达到14/23(61.0%),而正常关节中,仅有10/67(14.9%),二者差异显著(P < 0.001);有压痛的关节与无压痛的相比,存在吸收更多的倾向。因此该实验表明,赛妥珠具有较好的靶向性。但该文章并未说明患者病变关节的炎症是否好转。目前赛妥珠单抗已广泛应用于治疗类风湿性关节炎、活动性银屑病关节炎、强直性脊柱炎等疾病,但是关于骨关节炎的治疗报道较少,其疗效和安全性还需要更多的临床实验来证明。
2.1.2 阿达木单抗 阿达木单抗的商品名为修美乐,是首个成功研发的重组全人源化免疫球蛋白 (IgG) 单克隆抗体,目前已广泛应用于风湿性关节炎、强直性脊柱炎、银屑病、银屑病关节炎和克罗恩病等的治疗[14-16]。阿达木单抗对可溶性肿瘤坏死因子α具有很高的亲和力,通过阻断肿瘤坏死因子α与其受体p55和p75的相互作用可有效抵消肿瘤坏死因子α生物学功能,且免疫原性低,半衰期长[13]。德国学者OSSENDORFF等[17]通过体外实验来探究肿瘤坏死因子α与软骨细胞生长的关系,以及阿达木单抗的作用,该学者从牛的球关节处分离获得软骨细胞分组培养:①对照组不额外添加药品;②添加20 μg/L的肿瘤坏死因子α;③添加10 g/L
阿达木单抗;④添加20 μg/L肿瘤坏死因子α+10 g/L 阿达木单抗。在培养2周之后通过测量发现,软骨细胞DNA总量在肿瘤坏死因子α组最低,且氨基葡萄糖(构成软骨的成分)的含量也显著低于对照组(P=0.029),这表明肿瘤坏死因子α会抑制软骨细胞的增殖;肿瘤坏死因子α的抑制作用可被阿达木单抗所拮抗:在肿瘤坏死因子α+阿达木组中,氨基葡萄糖的量是肿瘤坏死因子α组的5倍(P < 0.01),软骨细胞基因的表达产物软骨寡聚物蛋白、胶原1和胶原2的含量要显著高于肿瘤坏死因子α组(P < 0.01)。MA等[18]在骨关节炎的体内模型中证明了阿达木单抗不仅可以减少软骨和软骨下骨的降解,而且在mRNA水平上降低了基质金属蛋白酶3的活性。MAKSYMOWYCH等[19]的实验表明,阿达木单抗可以有效改善膝关节炎的疼痛指数。然而根据CHEVALIER等[20]和AITKEN等[21]的报道,与使用安慰剂的对照组相比,阿达木单抗在治疗手关节炎时镇痛作用并不显著,这说明阿达木单抗可能对于膝关节炎有一定效果,但是对手关节炎的疗效不佳。
2.1.3 英夫利昔单抗 英夫利昔单抗是具有人IgG1和k恒定区以及鼠可变区的嵌合二价抗体[16]。动物实验表明,英夫利昔单抗可以通过减缓软骨变性、减少滑液中的肿瘤坏死因子α和一氧化氮来预防实验性诱导骨关节炎[22]。有学者为了探究英夫利昔单抗对于治疗手部侵蚀性关节炎的疗效和安全性,设计了一个临床试验[23]。该学者招募了20例符合条件的患者,将其随机分为2组,治疗组和对照组各10例,经12个月治疗后,治疗组的目测类比(VAS)疼痛评分有了显著降低,且未发现严重的不良反应,但是该试验参与人数较少,需要更多的临床试验来阐明英夫利昔单抗对骨关节炎的治疗效果。
2.2 神经生长因子拮抗剂 神经生长因子在胚胎形成期也被称为神经营养因子,对神经元的存活和生长发育都起到了重要作用。神经生长因子可与两种受体结合:一种是高亲和力的酪氨酸激酶受体(tyrosine receptor kinases,trks),另一种是低亲和力的肿瘤坏死因子P75受体。现已证实,神经生长因子不仅起到营养神经的作用,而且与炎症的发生、疼痛信号的传递及疼痛的超敏反应都有关[24-25]。神经生长因子在外周损伤后由免疫细胞释放,参与局部的炎症反应。在炎症环境下,神经生长因子与痛觉感受器上的TrkA受体结合,激活感受器产生关节疼痛[26]。TrkA还与疼痛的超敏反应密切相关。神经生长因子与炎症细胞上的TrkA受体结合,刺激多种炎症递质的释放,如组胺、血清素、5-羟色胺、前列腺素E2和神经生长因子本身[27-28]。这些介质可与痛觉感受器外周终端的受体结合,并启动引起痛觉感受器敏感化的信号。目前针对靶点神经生长因子的药物主要有3种:他尼珠单抗(Tanezumab)、Fulranumab和fasinumab。
2.2.1 他尼珠单抗 他尼珠单抗是一种针对神经生长因子的人源化单克隆抗体,可以抑制神经生长因子与其受体的结合,有效减轻膝、髋部骨关节炎引发的疼痛及腰背痛[29-31]。SCHNITZER等[32]学者为了研究他尼珠单抗对膝骨关节炎的效果,设计了一个临床试验,该试验包含了2个实验组:试验第1天和第8周时关节内注射他尼珠2.5 mg(低剂量组);试验第1天注射他尼珠2.5 mg,第8周时注射他尼珠5 mg(高剂量组);以及一个对照组。在第16周时对疗效进行评估,结果发现低剂量组的WOMAC疼痛指数降低了3.5,与对照组(降低2.7)相比差异显著(P=0.01);高剂量组的WOMAC疼痛指数降低了3.7,与对照组相比差异显著(P=0.002);且治疗组WOMAC功能指数也有显著改善。KATZ[33]的研究也显示了相似结果。虽然皮下注射他尼珠单抗是一种有效的治疗骨关节炎的方法,但是有报道称他尼珠单抗会导致病情进展加速,关节置换的风险加大。且他尼珠联合非类固醇类抗炎药时,这种加速作用更显著[34]。这种加速作用主要表现在两个方面:①导致关节间隙缩窄的速度加快;②导致骨的丢失和破坏[35]。美国学者为了研究关节炎进展、关节置换与使用他尼珠单抗治疗之间的关系,设计了一个随机对照试 验[36],该试验将符合条件的患者分为3组,分别使用高剂量他尼珠、低剂量他尼珠和安慰剂进行治疗。结果发现,他尼珠与安慰剂相比确实有更强的镇痛效果,但是在治疗40周时高剂量组有6.9%的患者接受了关节置换,低剂量组的比例为3.5%,安慰剂组为1.7%,差异有显著性意义,且随着治疗剂量的增加关节置换的风险也加大。所以患者接受他尼珠单抗治疗,就要接受关节炎进展加速、关节置换风险增加的可能性,对于那些极有可能出现全关节置换术相关并发症以及希望避免接受全关节置换术的患者来说,他尼珠单抗不是一个好的选择。美国西北大学的SCHNITZER教授[32]通过临床试验发现,他尼珠单抗还存在使用后感觉异常和停药后疼痛加剧等不良反应。
2.2.2 Fulranumab Fulranumab是一种重组免疫球蛋白G2单克隆抗体,具有特异性中和人类神经生长因子生物学作用的能力[37]。美国学者SANGA等[38]通过长期的研究发现,在每4周3 mg或每8周10 mg的剂量下(关节腔注射Fulranumab),与使用安慰剂的对照组相比,骨关节炎患者的WOMAC功能评分和疼痛评分在第4周时已有显著改善。根据MAYORGA
等[39]的报道,患者在接受每4周3 mg或每4周9 mg的
Fulranumab关节腔注射治疗后,在12周时出现了显著的疼痛减轻和关节功能的改善。Fulranumab对骨关节炎的镇痛效果甚至优于羟考酮。严重的不良事件中最常见的是骨关节炎进展加速、关节间隙缩窄、关节置换的需求加大。SANGA等[38]的报道称,在使用Fulranumab治疗后,膝关节炎和髋关节炎的患者分别有10%和7%接受了关节置换术,且在总的接受关节置换的71例患者中,有63例(89%)接受了Fulranumab治疗。与他尼珠类似的是,Fulranumab与非类固醇类抗炎药物联合使用比单一用药导致的骨关节炎病变速度更快。此外,在治疗组中有28%出现了神经系统相关的不良反应,如手指感觉异常、双臂刺痛感等(13%),腕管综合征(8%),感觉迟钝(7%),而对照组出现神经相关不良反应的比例为14%,而此类不良反应在停药之后都消失了;随着治疗剂量的增加,也未见到患者出现剂量相关的严重不良反应,这说明Fulranumab(富拉尼单抗)是一种相对安全的药物。
2.2.3 Fasinumab Fasinumab是一种完全人源化的IgG4型抗神经生长因子的单克隆抗体,可靶向作用于神经生长因子。Tiseo等[41]设计了一项为期24周的临床试验,受试者全部为膝或髋部骨关节炎患者,分别接受0.03,0.1,0.3 mg/kg的Fasinumab或安慰剂治疗(第1天和第57天行关节腔内注射),在第24周行主要疗效分析时发现,与安慰剂相比使用Fasinumab治疗患者疼痛减轻,运动能力也有显著改善。美国学者Dakin等[42]设计了一项试验(随机、双盲、安慰剂对照),旨在确定Fasinumab的作用、安全性和耐受性,受试者为膝或髋部骨性关节炎慢性疼痛患者,分别接受每4周1次Fasinumab 1 mg、 Fasinumab 3 mg,Fasinumab 6 mg,Fasinumab 9 mg或安慰剂治疗,治疗时间为16周。结果发现在第16周时4个治疗组的WOMAC疼痛分数与对照组相比都有了显著的降低;与基线水平相比,4个治疗组的WOMAC疼痛分数(分值为0-10)降低值分别达到3.5,3.4, 3.1和3.8,患者的整体评价也有显著改善;Fasinumab的耐受性较好,在16周的治疗中实验组和对照组与治疗相关的不良事件发生率分别为62%和55%,但大多数都比较轻微,包括呼吸道感染、神经系统、皮肤、皮下组织疾病,因严重的不良事件而退出的患者,治疗组中共有14例(4%),对照组有1例(1%)。研究者通过常规的影像学监测发现,Fasinumab可导致关节病(包括关节间隙缩窄,骨质的改变,软骨下成分的缺损等)的发生,并且与使用剂量呈正相关[40]。Fasinumab是一种相对安全的药物,而且与安慰剂相比可以显著减轻关节疼痛,改善关节功能。
总体来说,针对神经生长因子的药物都有镇痛、改善关节功能的效果,但是也普遍存在导致骨关节炎进展加速的问题,因此对该类药物的选择应该因人而异。
2.3 白细胞介素1拮抗剂 白细胞介素1在骨关节炎患者的软骨、滑膜、滑膜液中广泛表达,在骨关节炎的发病过程中发挥着关键作用。白细胞介素1α和白细胞介素1β具有协同作用,当白细胞介素1β与其受体白细胞介素1R1结合后,引起髓样分化蛋白88(myeloid differentiation factor 88,MyD88)和白细胞介素1R相关激酶1(interleukin-1 receptor-associated kinase 1,IRAK1)的募集。而IRAK会激活肿瘤坏死因子受体相关因子6。肿瘤坏死因子受体相关因子6随后作用于下游靶点,导致TAK1[转化生长因子激酶1,是丝裂源激活蛋白激酶激酶激酶(MAP3K)家族成员之一]、TAB1、TAB2等蛋白的募集。TAK1可促进IκB激酶的磷酸化,引起转录因子核周因子κB的激活。同时,MAPK家族中的P38以及c-Jun N-terminal kinase(JNK)等转录因子也会被TAK1激活,引起多种基因的迅速表达:包括其他细胞因子、趋化因子、黏附分子、炎症递质和酶[43-46]。核因子κB和MAPKs是关节炎发病机制中两条重要的信号通路[47]。白细胞介素1可诱导表达基质金属蛋白酶、自由基、一氧化氮和前列腺素,抑制胶原蛋白和蛋白多糖的合成,因此,抑制白细胞介素1的释放可能会缓解骨关节炎的进展。
目前报道可用于骨关节炎的针对白细胞介素1的单克隆抗体如下:Canakinumab (ACZ885)是一种人IgGk型单克隆抗体,可以阻断白细胞介素1β受体。最近,CHELESCHI等[48]设计了一个体外实验,来探究canakinumab对人软骨细胞的作用,研究者通过透射电镜观察发现,与使用安慰剂的对照组相比,canakinumab可显著增加肿瘤坏死因子α培养细胞的蛋白聚糖水平,并降低了一氧化氮水平。但是,由于缺乏相关的临床试验,关节内注射canakinumab的安全性、耐受性、药代动力学和对疼痛的影响,仍有待进一步研究。
2.4 其他类型尚处于研究中的药物
2.4.1 低密度脂蛋白受体相关蛋白1(low-density lipoprotein receptor–related protein-1,LRP-1)脱落酶抑制剂 骨关节炎发病的一个重要原因是基质金属蛋白酶和聚蛋白多糖酶(ADAMTS)升高导致的软骨降解破坏。研究发现,基质金属蛋白酶和聚蛋白多糖酶5本质上都是由正常的软骨组织产生的,但是它们会迅速的通过软骨细胞表面的LRP-1介导后通过胞饮途径被摄取,然后在胞内降解[49-51]。聚蛋白多糖酶4和组织金属蛋白酶抑制剂3也通过LRP-1介导入胞[52- 53],这表明LRP-1是软骨基质降解系统中的关键调节器。由于骨关节炎患者软骨细胞中LRP-1蛋白水平的降低,使得这种内吞通路在软骨中受损,导致基质金属蛋白酶和聚蛋白多糖酶5的浓度在胞外升高。有学者猜想LRP-1蛋白浓度的降低是由于脱落酶的作用使得LRP-1裂解,使用脱落酶抑制剂可以降低基质金属蛋白酶和聚蛋白多糖酶5的胞外浓度,从而阻止骨关节炎患者关节软骨的退化(脱落酶是膜结合型的ADAM或基质金属蛋白酶)[54]。YAMAMOTO等[49]通过蛋白质印记(Western
blotting)法检测发现,与正常人相比骨关节炎患者软骨中LRP-1的α链减少了48%,β链减少了65%,且聚蛋白多糖酶5半衰期是正常人的2.8倍(骨关节炎患者210 min;正常人
75 min)。脱落酶不仅使LRP-1的胞外域部分降解,并且会使聚蛋白多糖酶5的活性增加3倍。根据YAMAMOTO等[54]的报道,在骨关节炎患者的软骨细胞中,单独使用抗ADAM-17的抗体(D1A12)或抗基质金属蛋白酶14的抗体(E2C6)都会使LRP-1的α链和β链在细胞水平上增加,且两者联合使用效果更加显著。两种抗体联合使用还会使聚蛋白多糖酶5的清除速率增加2.4倍(使用抗体时聚蛋白多糖酶5的半衰期为95 min,未使用时半衰期为210 min),蛋白多糖和胶原的降解也有显著减少。虽然LPR-1脱落酶抑制剂在细胞水平上显示出了治疗骨关节炎的效果,但是仍缺乏相关动物实验、临床试验来证明它在体内的疗效。
2.4.2 聚蛋白多糖酶5抑制剂 完整的聚蛋白多糖包含N端球状结构域,该区域将糖蛋白锚定在透明质酸上,并被蛋白水解敏感的球状结构域(IGD)和富含糖胺聚糖(GAG)侧链的C端区域分隔保护[4],这对于维持软骨弹性、软骨内水含量和结构的完整性非常重要。研究表明,聚蛋白多糖酶家族,尤其是聚蛋白多糖酶5所导致的蛋白多糖水解被认为是引起骨关节炎早期软骨缺损的关键[55]。聚蛋白多糖酶5是一种多域的金属蛋白酶,包括一个大型的前肽结构域,一个催化域,一个解聚素域;两个血小板反应蛋白域(TS-1,TS-2),一个富含半胱氨酸域,一个间隔域[50]。不同亚型的单克隆抗体针对聚蛋白多糖酶5的位点也不同。SANTAMARIA等[50]通过细胞实验发现,2D3和2D11型的抗体分别作用于聚蛋白多糖酶5的催化区和解聚素区,2D5作用于TS-1区,2B9作用于间隔区。虽然作用位点不同,但是这些抗体都表现出了对聚蛋白多糖酶5的抑制作用。实验证明,聚蛋白多糖酶5缺乏的小鼠经DMM法(通过手术的方式人为破坏小鼠内侧半月板的稳定性,destabilization of the medial meniscus,DMM法)造模后,仍可长期免受骨关节炎的侵袭[56]。LARKIN等[4]学者为了验证GSK2394002型抗体(靶点横跨催化区和解聚素区)的作用,设计了一个动物实验,首先取一定量的小鼠通过DMM法来构建骨关节炎模型,术后6周单次给药(用IR-800标记,16 mg/kg),定期摄片。发现在腹腔给药30 min后,小鼠腹腔内就出现了高信号;4 d后取切片观察,IR-800的信号在全身都有分布,且在膝关节区域的浅层软骨、半月板、软骨下骨的绿色荧光信号较强,这表明该抗体具有较好的穿透性、靶向性;在术后4周开始给药[10 mg/(kg?周)],在第8周时使用甲苯胺蓝对骨关节炎小鼠的膝关节软骨组织进行染色观察,发现实验组小鼠与对照组软骨破坏程度明显减轻。与小分子的广谱金属蛋白酶抑制剂相比,该型单克隆抗体对聚蛋白多糖酶5的亲和力更高,靶向性更好,且药效可达广谱金属蛋白酶抑制剂的30倍[4]。CHIUSAROLI等[57]的报道称,通过向患有骨关节炎的小鼠关节腔内注射CRB0017型的抗体(靶点为间隔区),不仅靶向性好,而且起到了较好的软骨保护作用。MILLER等[58]的研究发现,在经DMM法构建的骨关节炎小鼠模型中,接受12F4.1H7型单克隆单体治疗的小鼠与接受安慰剂及普通igG2抗体治疗的小鼠相比,其软骨的损失量、骨赘的宽度都有显著减小。总体来说,针对聚蛋白多糖酶5的单克隆抗体在动物体内已取得良好效果,但是该药物对人体的疗效还有待进一步研究。需要注意的是,蛋白聚糖还存在于心血管等其他系统内,那么抑制多功能蛋白聚糖的周转,是否会损害相关器官?可以考虑给药剂量或给药方法,使其对软骨的影响最大,但不影响其他组织。一些可能的措施包括关节内给药、间歇性给药和低剂量抗聚蛋白多糖酶5抗体联合其他靶向于骨关节炎通路的药物[59]。
2.4.3 血管内皮生长因子抑制剂 血管内皮生长因子可作用于人体内的多种细胞,具有广泛而重要的生理作用。血管内皮生长因子可维持软骨细胞的存活,促进关节内软骨的骨化和骨的生长发育,促进损伤骨组织的修复[60]。然而,过量血管内皮生长因子所介导的血管新生在骨关节炎的发展中起着重要作用。软骨细胞血管内皮生长因子表达增强会导致新生血管侵蚀关节软骨,诱导基质金属蛋白酶1、基质金属蛋白酶3、基质金属蛋白酶13等炎症因子生成增加[61,67]。高剂量的血管内皮生长因子是诱导骨关节炎起始和发展的重要因素[63]。Bevacizumab是一种抗血管内皮生长因子的单克隆抗体,通常被用作抗肿瘤的药物。研究发现,Bevacizumab不仅可以抑制肿瘤组织内血管生长,还具有抑制软骨退化的作用[60]。LI等[64]的研究表明,通过石膏制动法构建兔的膝关节炎模型后[65],向关节腔内注射4 mg的Bevacizumab可明显减少血管生成、抑制滑膜增生、降低血管内皮生长因子和基质金属蛋白酶1的表达。与关节内注射透明质酸和曲安奈德相比,Bevacizumab对兔原发性膝关节骨性关节炎有更好的软骨保护作用。NAGAI等[66]的研究也显示,在兔的骨关节炎模型中,使用Bevacizumab治疗可有效降低关节软骨损失、骨赘形成和滑膜炎。动物实验显示Bevacizumab具有较好的治疗骨关节炎的潜能,但是目前仍缺乏相关的临床试验,所以关于Bevacizumab对人体骨关节炎的治疗功效还需进一步探究。
2.4.4 粒细胞巨噬细胞集落刺激因子抑制剂 粒细胞巨噬细胞集落刺激因子与许多炎症性疾病,包括骨关节炎的发病机制有关。有报道称,通过对骨关节炎患者的软骨标本检查发现,其粒细胞巨噬细胞集落刺激因子相关mRNA和蛋白水平要显著高于正常人[68]。有学者通过向鼠关节腔内注射胶原酶来构建小鼠的骨关节炎(collagenase-induced osteoarthritis,CiOA)模型,并探究粒细胞巨噬细胞集落刺激因子及相关单克隆抗体的作用[68],结果发现,粒细胞巨噬细胞集落刺激因子基因敲除的小鼠与正常小鼠相比,在建模后第2周滑膜炎症状更轻,骨赘尺寸更小,软骨缺损也更小;而使用鼠型单克隆抗体(MP1-22E9)的治疗组小鼠,与使用普通抗体的小鼠相比,不仅镇痛效果好(在第3天就出现了明显差异),持续时间长(镇痛效果一直持续到第6周实验结束时),而且骨赘小、软骨缺损少。LEE等[69]也通过CiOA法来构建小鼠的骨关节炎模型,结果发现,早期的骨关节炎通过单次腹腔注射鼠型单克隆抗体(22E9,剂量为6.7 μg/g),可有效镇痛并缓解疾病进展;骨关节炎若进展至中晚期,仅单次给药疼痛会在短期内显著降低,但1周后又恢复到治疗前水平;若持续给药(每周2次),不仅可以有效镇痛,还能缓解骨关节炎进展。这些研究表明,抗粒细胞巨噬细胞集落刺激因子治疗可能具有缓解骨关节炎患者关节疼痛的作用,但仍需更多的临床试验对该类药物的功效进行探究。

| [1] 张希, 刘萍. 骨性关节炎所致慢性持续性疼痛的发病机制 [J]. 中国骨质疏松杂志,2017,23(2):248-255. [2] MARTEL-PELLETIER J, BARR A J, CICUTTINI FM, et al. Osteoarthritis. Nat Rev Dis Primers. 2016;2:16072. [3] 贺娟娟, 颜春鲁, 安方玉, 等. 炎症因子与炎症因子相关信号通路在膝骨关节炎中的调控机制研究进展[J].中国临床药理学杂志, 2019,35(12):1308-1311. [4] LARKIN J, LOHR TA, ELEFANTE L, et al.Translational development of an ADAMTS-5 antibody for osteoarthritis disease modification. Osteoarthritis Cartilage. 2015;23(8):1254-1266. [5] WANG T, HE C. Pro-inflammatory cytokines: The link between obesity and osteoarthritis. Cytokine Growth Factor Rev. 2018;44:38-50. [6] CHEN D, LU D, LIU H, et al. Pharmacological blockade of PCAF ameliorates osteoarthritis development via dual inhibition of TNF-α-driven inflammation and ER stress. EBioMedicine. 2019;50:395-407. [7] LóPEZ-ARMADA MJ, CARAMéS B, LIRES-DEáN M, et al. Cytokines, tumor necrosis factor-alpha and interleukin-1beta, differentially regulate apoptosis in osteoarthritis cultured human chondrocytes. Osteoarthritis Cartilage. 2006;14(7):660-669. [8] SHU S, ZHU J, LIU Z, et al. Endoplasmic reticulum stress is activated in post-ischemic kidneys to promote chronic kidney disease. EBioMedicine. 2018;37:269-280. [9] LIU-BRYAN R, TERKELTAUB R. Emerging regulators of the inflammatory process in osteoarthritis. Nat Rev Rheumatol. 2015:11(1):35-44. [10] ZHAO Y, LI Y, QU R, et al. Cortistatin binds to TNF-α receptors and protects against osteoarthritis. EBioMedicine. 2019:41:556-570. [11] YAMAZAKI H, SO R, MATSUOKA K, et al. Certolizumab pegol for induction of remission in Crohn’s disease. Cochrane Database Syst Rev. 2019;8(8):Cd012893. [12] WITTOEK R, CARRON P, LAMBERT B, et al. Immunoscintigraphic detection of tumour necrosis factor by radiolabelled certolizumab pegol in patients with erosive hand osteoarthritis: a proof-of-concept study. Ann Rheum Dis. 2018;77(2):310-311. [13] 安富荣,崔岚,王勤.自身免疫性疾病的生物治疗新药阿达木单抗[J].中国新药杂志,2010,19(14):1197-1200+1233. [14] 范晓蕾, 刘中华, 岳涛, 等. 血清TNF-α、IL-1β、IL-6和IL-17表达水平在阿达木单抗治疗活动性类风湿关节炎中的疗效预测作用[J]. 中国医科大学学报,2018,47(6):556-561. [15] HORNEFF G, SEYGER MMB, ARIKAN D, et al. Safety of Adalimumab in Pediatric Patients with Polyarticular Juvenile Idiopathic Arthritis, Enthesitis-Related Arthritis, Psoriasis, and Crohn’s Disease. J Pediatr. 2018;201:166-175.e163. [16] LIM H, LEE SH, LEE HT, et al. Structural Biology of the TNFα Antagonists Used in the Treatment of Rheumatoid Arthritis. Int J Mol Sci. 2018; 19(3):768. [17] OSSENDORFF R, GRAD S, STODDART MJ, et al. Autologous Chondrocyte Implantation in Osteoarthritic Surroundings: TNFalpha and Its Inhibition by Adalimumab in a Knee-Specific Bioreactor. Am J Sports Med. 2018; 46(2):431-440. [18] MA CH, LV Q, YU YX, et al. Protective effects of tumor necrosis factor-alpha blockade by adalimumab on articular cartilage and subchondral bone in a rat model of osteoarthritis. Braz J Med Biol Res. 2015;48(10):863-870. [19] MAKSYMOWYCH WP, RUSSELL AS, CHIU P, et al. Targeting tumour necrosis factor alleviates signs and symptoms of inflammatory osteoarthritis of the knee. Arthritis Res Ther. 2012;14(5):R206. [20] CHEVALIER X, RAVAUD P, MAHEU E, et al. Adalimumab in patients with hand osteoarthritis refractory to analgesics and NSAIDs: a randomised, multicentre, double-blind, placebo-controlled trial. Ann Rheum Dis. 2015;74(9):1697-1705. [21] AITKEN D, LASLETT L L, PAN F, et al. A randomised double-blind placebo-controlled crossover trial of HUMira (adalimumab) for erosive hand OsteoaRthritis - the HUMOR trial. Osteoarthritis Cartilage. 2018; 26(7):880-887. [22] ZHANG Q, LV H, CHEN A, et al. Efficacy of infliximab in a rabbit model of osteoarthritis. Connect Tissue Res. 2012;53(5):355-358. [23] FIORAVANTI A, FABBRONI M, CERASE A, et al. Treatment of erosive osteoarthritis of the hands by intra-articular infliximab injections: a pilot study. Rheumatol Int. 2009;29(8):961-965. [24] SEIDEL M F, WISE B L, LANE N E. Nerve growth factor: an update on the science and therapy. Osteoarthritis Cartilage. 2013;21(9):1223-1228. [25] DENK F, BENNETT D L, MCMAHON S B. Nerve Growth Factor and Pain Mechanisms. Annu Rev Neurosci. 2017;40:307-325. [26] SOUSA-VALENTE J, CALVO L, VACCA V, et al. Role of TrkA signalling and mast cells in the initiation of osteoarthritis pain in the monoiodoacetate model. Osteoarthritis Cartilage. 2018;26(1):84-94. [27] MANTYH PW, KOLTZENBURG M, MENDELL LM, et al. Antagonism of nerve growth factor-TrkA signaling and the relief of pain. Anesthesiology. 2011;115(1):189-204. [28] SCHMELZ M, MANTYH P, MALFAIT AM, et al.Nerve growth factor antibody for the treatment of osteoarthritis pain and chronic low-back pain: mechanism of action in the context of efficacy and safety. Pain. 2019;160(10):2210-2220. [29] KIVITZ AJ, GIMBEL JS, BRAMSON C, et al. Efficacy and safety of tanezumab versus naproxen in the treatment of chronic low back pain. Pain. 2013;154(7):1009-1021. [30] SPIERINGS EL, FIDELHOLTZ J, WOLFRAM G, et al. A phase III placebo- and oxycodone-controlled study of tanezumab in adults with osteoarthritis pain of the hip or knee. Pain. 2013;154(9):1603-1612. [31] LANE NE, SCHNITZER TJ, BIRBARA CA, et al. Tanezumab for the treatment of pain from osteoarthritis of the knee. N Engl J Med. 2010; 363(16):1521-1531. [32] SCHNITZER TJ, EASTON R, PANG S, et al. Effect of Tanezumab on Joint Pain, Physical Function, and Patient Global Assessment of Osteoarthritis Among Patients With Osteoarthritis of the Hip or Knee: A Randomized Clinical Trial. JAMA. 2019;322(1):37-48. [33] KATZ JN. Tanezumab for Painful Osteoarthritis. JAMA. 2019;322(1): 30-32. [34] SCHNITZER TJ, EKMAN EF, SPIERINGS EL, et al. Efficacy and safety of tanezumab monotherapy or combined with non-steroidal anti-inflammatory drugs in the treatment of knee or hip osteoarthritis pain. Ann Rheum Dis. 2015;74(6):1202-1211. [35] NAGASHIMA H, SUZUKI M, ARAKI S, et al. Preliminary assessment of the safety and efficacy of tanezumab in Japanese patients with moderate to severe osteoarthritis of the knee: a randomized, double-blind, dose-escalation, placebo-controlled study. Osteoarthritis Cartilage. 2011;19(12):1405-1412. [36] CISTERNAS MG, MURPHY L, SACKS JJ, et al. Alternative Methods for Defining Osteoarthritis and the Impact on Estimating Prevalence in a US Population-Based Survey. Arthritis Care Res (Hoboken). 2016;68(5): 574-580. [37] KELLY KM, SANGA P, ZAKI N, et al. Safety and efficacy of fulranumab in osteoarthritis of the hip and knee: results from four early terminated phase III randomized studies. Curr Med Res Opin. 2019;35(12): 2117-2127. [38] SANGA P, KATZ N, POLVEREJAN E, et al. Long-Term Safety and Efficacy of Fulranumab in Patients With Moderate-to-Severe Osteoarthritis Pain: A Phase II Randomized, Double-Blind, Placebo-Controlled Extension Study. Arthritis Rheumatol. 2017;69(4):763-773. [39] MAYORGA AJ, WANG S, KELLY KM, et al. Efficacy and safety of fulranumab as monotherapy in patients with moderate to severe, chronic knee pain of primary osteoarthritis: a randomised, placebo- and active-controlled trial. Int J Clin Pract.2016;70(6): 493-505. [40] BERENBAUM F. Targeting nerve growth factor to relieve pain from osteoarthritis: What can we expect? Joint Bone Spine. 2019;86(2): 127-128. [41] TISEO PJ, KIVITZ AJ, ERVIN JE, et al. Fasinumab (REGN475), an antibody against nerve growth factor for the treatment of pain: results from a double-blind, placebo-controlled exploratory study in osteoarthritis of the knee. Pain. 2014;155(7):1245-1252. [42] DAKIN P, DIMARTINO SJ, GAO H, et al. The Efficacy, Tolerability, and Joint Safety of Fasinumab in Osteoarthritis Pain: A Phase IIb/III Double-Blind, Placebo-Controlled, Randomized Clinical Trial. Arthritis Rheumatol. 2019:71(11):1824-1834. [43] FENG Z, LI X, LIN J, et al. Oleuropein inhibits the IL-1β-induced expression of inflammatory mediators by suppressing the activation of NF-κB and MAPKs in human osteoarthritis chondrocytes. Food Funct. 2017;8(10):3737-3744. [44] WOJDASIEWICZ P, PONIATOWSKI ŁA, SZUKIEWICZ D. The role of inflammatory and anti-inflammatory cytokines in the pathogenesis of osteoarthritis. Mediators Inflamm. 2014;2014:561459. [45] CHEN J, GU YT, XIE JJ, et al. Gastrodin reduces IL-1β-induced apoptosis, inflammation, and matrix catabolism in osteoarthritis chondrocytes and attenuates rat cartilage degeneration in vivo. Biomed Pharmacother. 2018;97:642-651. [46] KAPOOR M, MARTEL-PELLETIER J, LAJEUNESSE D, et al. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat Rev Rheumatol. 2011;7(1):33-42. [47] LEE HJ, LEE SM, MOON YG, et al. Membrane-Free Stem Cell Components Inhibit Interleukin-1alpha-Stimulated Inflammation and Cartilage Degradation in vitro and in vivo: A Rat Model of Osteoarthritis. Int J Mol Sci. 2019;20(19):4869. [48] CHELESCHI S, CANTARINI L, PASCARELLI N A, et al. Possible chondroprotective effect of canakinumab: an in vitro study on human osteoarthritic chondrocytes. Cytokine.2015;71(2):165-172. [49] YAMAMOTO K, TROEBERG L, SCILABRA SD, et al. LRP-1-mediated endocytosis regulates extracellular activity of ADAMTS-5 in articular cartilage. FASEB J. 2013;27(2):511-521. [50] SANTAMARIA S, YAMAMOTO K, BOTKJAER K, et al. Antibody-based exosite inhibitors of ADAMTS-5 (aggrecanase-2). Biochem J. 2015; 471(3):391-401. [51] YAMAMOTO K, OKANO H, MIYAGAWA W, et al. MMP-13 is constitutively produced in human chondrocytes and co-endocytosed with ADAMTS-5 and TIMP-3 by the endocytic receptor LRP1. Matrix Biol. 2016;56:57-73. [52] SCILABRA SD, TROEBERG L, YAMAMOTO K, et al. Differential regulation of extracellular tissue inhibitor of metalloproteinases-3 levels by cell membrane-bound and shed low density lipoprotein receptor-related protein 1. J Biol Chem. 2013;288(1):332-342. [53] YAMAMOTO K, OWEN K, PARKER AE, et al. Low density lipoprotein receptor-related protein 1 (LRP1)-mediated endocytic clearance of a disintegrin and metalloproteinase with thrombospondin motifs-4 (ADAMTS-4): functional differences of non-catalytic domains of ADAMTS-4 and ADAMTS-5 in LRP1 binding. J Biol Chem. 2014;289(10): 6462-6474. [54] YAMAMOTO K, SANTAMARIA S, BOTKJAER KA, et al. Inhibition of Shedding of Low-Density Lipoprotein Receptor-Related Protein 1 Reverses Cartilage Matrix Degradation in Osteoarthritis. Arthritis Rheumatol. 2017;69(6):1246-1256. [55] ZHENG S, HUNTER D J, XU J, et al. Monoclonal antibodies for the treatment of osteoarthritis. Expert Opin Biol Ther. 2016;16(12): 1529-1540. [56] MALFAIT AM, RITCHIE J, GIL AS, et al. ADAMTS-5 deficient mice do not develop mechanical allodynia associated with osteoarthritis following medial meniscal destabilization. Osteoarthritis Cartilage. 2010;18(4): 572-580. [57] CHIUSAROLI R, VISENTINI M, GALIMBERTI C, et al. Targeting of ADAMTS5’s ancillary domain with the recombinant mAb CRB0017 ameliorates disease progression in a spontaneous murine model of osteoarthritis. Osteoarthritis Cartilage. 2013;21(11):1807-1810. [58] MILLER RE, TRAN PB, ISHIHARA S, et al. Therapeutic effects of an anti-ADAMTS-5 antibody on joint damage and mechanical allodynia in a murine model of osteoarthritis. Osteoarthritis Cartilage. 2016;24(2): 299-306. [59] APTE SS. Anti-ADAMTS5 monoclonal antibodies: implications for aggrecanase inhibition in osteoarthritis. Biochem J. 2016;473(1):e1-4. [60] LEE S, NEMEñO JG, LEE J I. Repositioning Bevacizumab: A Promising Therapeutic Strategy for Cartilage Regeneration. Tissue Eng Part B Rev. 2016;22(5):341-357. [61] HAMILTON JL, NAGAO M, LEVINE B R, et al. Targeting VEGF and Its Receptors for the Treatment of Osteoarthritis and Associated Pain. J Bone Miner Res. 2016;31(5):911-924. [62] LI L, LIU F, HUANG W, et al. Ricolinostat (ACY-1215) inhibits VEGF expression via PI3K/AKT pathway and promotes apoptosis in osteoarthritic osteoblasts. Biomed Pharmacother. 2019;118:109357. [63] NAGAO M, HAMILTON JL, KC R, et al. Vascular Endothelial Growth Factor in Cartilage Development and Osteoarthritis. Sci Rep. 2017:7(1): 13027. [64] LI W, LIN J, WANG Z, et al. Bevacizumab tested for treatment of knee osteoarthritis via inhibition of synovial vascular hyperplasia in rabbits. J Orthop Translat. 2019;19:38-46. [65] LU W, WANG L, YAO J, et al. C5a aggravates dysfunction of the articular cartilage and synovial fluid in rats with knee joint immobilization. Mol Med Rep. 2018;18(2):2110-2116. [66] NAGAI T, SATO M, KOBAYASHI M, et al. Bevacizumab, an anti-vascular endothelial growth factor antibody, inhibits osteoarthritis. Arthritis Res Ther. 2014;16(5):427. [67] LI L, LIU F, HUANG W, et al. Ricolinostat (ACY-1215) inhibits VEGF expression via PI3K/AKT pathway and promotes apoptosis in osteoarthritic osteoblasts. Biomed Pharmacother. 2019;118:109357. [68] COOK AD, POBJOY J, STEIDL S, et al. Granulocyte-macrophage colony-stimulating factor is a key mediator in experimental osteoarthritis pain and disease development. Arthritis Res Ther. 2012;14(5):R199. [69] LEE KM, PRASAD V, ACHUTHAN A, et al. Targeting GM-CSF for collagenase-induced osteoarthritis pain and disease in mice. Osteoarthritis Cartilage. 2020;28(4):486-491. |
| [1] | 方兴艳, 田侦丽, 赵哲仪, 文平, 谢婷婷. 亚砷酸钠对人脐静脉内皮细胞损伤及鞘氨醇激酶1/1-磷酸鞘氨醇信号轴的影响[J]. 中国组织工程研究, 2023, 27(在线): 1-7. |
| [2] | 郭淑慧, 杨晔, 江杨洋, 许建文. 神经源性膀胱miRNA-mRNA调控网络的筛选与验证[J]. 中国组织工程研究, 2023, 27(在线): 1-8. |
| [3] | 李晓敏, 田向东 , 谭冶彤 , 朱光宇 , 王荣田 , 王 剑 , 薛志鹏, 马 晟, 胡元一, 黄 叶, 丁天送. 骨质疏松性内侧室膝骨关节炎胫骨高位截骨后下肢力线及膝关节功能的变化[J]. 中国组织工程研究, 2023, 27(9): 1325-1329. |
| [4] | 黄林科, 韦林华, 蒋 捷, 刘 倩, 陈蔚蔚. 雌激素与跑台运动干预卵巢切除模型小鼠骨量和关节软骨的变化[J]. 中国组织工程研究, 2023, 27(8): 1166-1171. |
| [5] | 高 煜, 韩佳慧, 葛 新. 脊髓缺血再灌注损伤后的免疫炎性微环境[J]. 中国组织工程研究, 2023, 27(8): 1300-1305. |
| [6] | 王 敏, 尹秀山, 王盈熹, 张 妍, 赵 龙, 夏书月. 吸入骨髓间充质干细胞来源外泌体减轻慢性阻塞性肺疾病的炎性损伤[J]. 中国组织工程研究, 2023, 27(6): 827-834. |
| [7] | 邵子晨, 李华南, 顾 兵, 章晓云, 孙伟康, 刘永钱, 甘 斌. 痛风过程中微小RNA、长链非编码RNA和环状RNA介导降尿酸、抗炎及调控骨代谢的协同调节作用[J]. 中国组织工程研究, 2023, 27(5): 765-771. |
| [8] | 袁长深, 官岩兵, 李 哲, 容伟明, 廖书宁, 陈乐伟, 梅其杰, 段 戡. 骨关节炎坏死性凋亡关键基因的筛选与验证[J]. 中国组织工程研究, 2023, 27(5): 695-700. |
| [9] | 李 龙, 李光第, 石 豪, 邓柯淇. 环状RNA作为内源性竞争RNA参与调控骨性关节炎的发生[J]. 中国组织工程研究, 2023, 27(5): 751-757. |
| [10] | 李志超, 谭国庆, 苏 辉, 徐展望, 薛海鹏. 非编码RNAs作为潜在治疗靶点在脊髓损伤中的调控效应[J]. 中国组织工程研究, 2023, 27(5): 758-764. |
| [11] | 李雅萍, 刘 红, 高 真, 陈晓琳, 黄武杰, 江 征. 太极拳练习伴膝关节疼痛者下肢生物力学表现的三维运动分析[J]. 中国组织工程研究, 2023, 27(4): 520-526. |
| [12] | 谷明西, 王 博, 田丰德, 安 宁, 郝瑞胡, 王常成, 郭 林. 同期和分期双侧全膝关节置换的早期疗效和安全性比较[J]. 中国组织工程研究, 2023, 27(4): 565-571. |
| [13] | 郭颖奇, 宫先旭, 张 岩, 肖 寒, 王 野, 谷文光. 半月板外突与髌股关节软骨损伤及骨髓病变:MRI半定量评分的评价[J]. 中国组织工程研究, 2023, 27(4): 600-605. |
| [14] | 张力宸, 陈 亮, 顾 勇. 无机离子仿生骨膜调控免疫微环境促进骨修复[J]. 中国组织工程研究, 2023, 27(3): 346-353. |
| [15] | 李晓敏, 田向东, 谭冶彤, 薛志鹏, 马 晟, 胡元一, 黄 叶, 丁天送. 胫骨结节远端单平面截骨患者髌股关节退变及髌骨高度的改变[J]. 中国组织工程研究, 2023, 27(27): 4367-4372. |
中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程
1.2 入选标准
1.2.1 纳入标准 ①论述骨关节炎病因及发病机制的文章;②与骨关节炎单克隆抗体治疗相关的文章;③与单克隆抗体作用机制相关需提取资料的文章。
1.2.2 排除标准 ①治疗其他疾病抗体的文章;②重复性文章;③来源于非核心期刊和其他低影响因子期刊。
1.3 质量评估 ①内容与主题高度相关;②以近10年为主;③文献质量高,中文文献来自核心期刊,英文文献均为影响因子3分以上的文献。
1.4 数据提取 通过关键词检索及被引文献检索,共检索到1 922篇文献,其中中文文献668篇,英文文献1 254篇;排除与主题相关性差的,以及陈旧的、重复的文献,纳入符合标准的69篇文献进行综述;见图1和表1。


文题释义:
单克隆抗体:针对单一抗原决定基,由一个识别一种抗原决定基的B细胞克隆合成并分泌的抗体,称为单克隆抗体。作为一种新型的生物制剂药物,单克隆抗体近年来被大量开发并逐渐投入临床。单克隆抗体的优点是靶向性强、疗效好、药效持续时间长、不良反应小以及研发速度快、成本低等,因此受到国内外学者的重视。
中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程骨关节炎是一种与年龄有关的疾病,其患病率会随着年龄增长而增加。骨关节炎的主要症状是关节的疼痛、活动受限。目前,有许多药物被用来治疗关节炎相关疼痛,包括非类固醇抗炎药(NSAIDs)、扑热息痛、阿片类药物、以及改变痛觉中枢敏感化的药物,如加巴喷丁和度洛西汀。这些药物都有一定的毒性或效力上的局限性。这促使人们加大力度研究治疗骨关节炎疼痛的新药物(尤其是非阿片类药物)。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||