中国组织工程研究 ›› 2021, Vol. 25 ›› Issue (29): 4672-4679.doi: 10.12307/2021.167
• 组织构建细胞学实验 cytology experiments in tissue construction • 上一篇 下一篇
肠道微生态制剂联合改良低碳饮食靶向调控肥胖症患者体脂代谢及肠屏障功能的效果分析
刘建国1,潘 勇2, 3,李翰宇4,刘 燕1,张圳甜1,林秀萍1,张 苑1,刘亚静1,张 帆4,张雷钧1,肖烈辉1,徐爱民2, 3,朱翠凤1, 2, 3
- 1南方医科大学深圳医院健康管理中心(营养科),广东省深圳市 518000;2香港大学李嘉诚医学院内分泌科,中国香港特别行政区;3生物医药技术国家重点实验室香港大学伙伴实验室,中国香港特别行政区;4北京大学深圳医院内分泌科,广东省深圳市 518000
-
收稿日期:2020-07-20修回日期:2020-07-28接受日期:2020-11-19出版日期:2021-10-18发布日期:2021-07-22 -
通讯作者:朱翠凤,博士,主任医师,南方医科大学深圳医院营养科,广东省深圳市 518000;香港大学李嘉诚医学院内分泌科,生物医药技术国家重点实验室香港大学伙伴实验室,中国香港特别行政区 -
作者简介:刘建国,男,1961年生,湖南省人,汉族,1984年南华大学毕业,副主任医师。南方医科大学深圳医院健康管理中心,广东省深圳市 518000 -
基金资助:深圳市科技创新委基础研究基金立项资助课题(JCYJ20170307162004873),项目负责人:朱翠凤;深圳市宝安区科创局基础研究基金立项资助课题(2017JD017),项目负责人:刘建国;香港研究资助局合作研究基金(C7055-14G,C7037-17W),项目负责人:徐爱民;香港大学博士后启动基金(201507176257),项目负责人:朱翠凤
Effect of microecological preparation combined with an improved low-carbon diet on fat metabolism and intestinal barrier function in obese patients
Liu Jianguo1, Pan Yong2, 3, Li Hanyu4, Liu Yan1, Zhang Zhentian1, Lin Xiuping1, Zhang Yuan1, #br# Liu Yajin1, Zhang Fan4, Zhang Leijun1, Xiao Liehui1, Xu Aimin2, 3, Zhu Cuifeng1, 2, 3, *#br#
- 1Health Management Center (Department of Nutrition), Shenzhen Hospital of Southern Medical University, Shenzhen 518000, Guangdong Province, China; 2Department of Endocrinology, Li Ka Shing School of Medicine, University of Hong Kong, Hong Kong Island West, Kong Kong Special Administration Region, China; 3State Key Laboratory of Biomedical Technology, University of Hong Kong Partner Laboratory, Hong Kong Island West, Kong Kong Special Administration Region, China; 4Department of Endocrinology, Shenzhen Hospital, Peking University, Shenzhen 518000, Guangdong Province, China
-
Received:2020-07-20Revised:2020-07-28Accepted:2020-11-19Online:2021-10-18Published:2021-07-22 -
Contact:Zhu Cuifeng, MD, Chief physician, Health Management Center (Department of Nutrition), Shenzhen Hospital of Southern Medical University, Shenzhen 518000, Guangdong Province, China; Department of Endocrinology, Li Ka Shing School of Medicine, University of Hong Kong, Hong Kong Island West, Kong Kong Special Administration Region, China; State Key Laboratory of Biomedical Technology, University of Hong Kong Partner Laboratory, Hong Kong Island West, Kong Kong Special Administration Region, China -
About author:Liu Jianguo, Associate chief physician, Health Management Center (Department of Nutrition), Shenzhen Hospital of Southern Medical University, Shenzhen 518000, Guangdong Province, China -
Supported by:This work was supported by the Basic Research Fund of the Shenzhen Science and Technology Innovation Commission, No. JCYJ20170307162004873 (to ZCF), the Basic Research Fund of the Science and Technology Innovation Bureau of Shenzhen Baoan District, No. 2017JD017 (to LJG), Cooperative Research Fund of the Hong Kong Research Grants Council, No. C7055-14G and C7037-17W (to XAM), Post-doctoral Foundation of the University of Hong Kong, No. 201507176257 (to ZCF)
摘要:
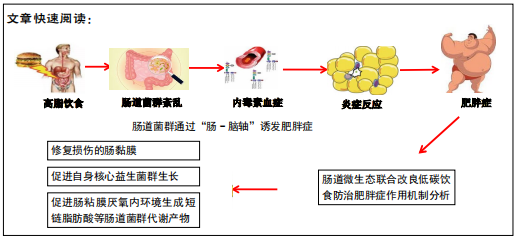
文题释义:
肠-脑轴与肥胖症的关系:肠道微生物和大脑的相互作用可能是肥胖的后果或发病因素,肠-脑轴是它们相互作用的联络枢纽。
肠-脑轴调控肥胖症发生的可能机制:肠道微生物可以通过肠道激素以及迷走神经连接(影响能量消耗和中枢神经系统中与饮食行为相关的区域)来影响肠-脑轴,从而改变宿主行为。同时,肠道微生物代谢物和其产物还可以充当信号分子并调节肠内分泌细胞的激素分泌,从而调节食欲、肠道运动、能量吸收和储存以及能量消耗等摄食相关行为,进而影响肥胖的发展。
背景:研究表明改善肠道菌群微生态或进食低碳饮食能够缓解肥胖及其并发症的发生率,但单独给予菌群治疗肥胖效果并不显著,而单纯低碳饮食安全性也颇有争议。因此,寻求更安全有效的减肥方法成为临床营养等相关学科的研究热点。
目的:探讨肠道微生态制剂联合改良低碳饮食靶向调控单纯性肥胖症患者体脂代谢及肠屏障功能的效果。
方法:试验纳入2017年8月至2019年5月在南方医科大学深圳医院营养科、健康管理中心门诊就诊的75例成人单纯性肥胖患者,随机分为3组,分别为接受肠道微生态制剂+改良低碳饮食减肥法干预组,接受单纯改良低碳饮食减肥法干预组及均衡饮食对照组,每组25例,同时与25例均衡饮食正常体质量人群进行对照比较。观察各组患者体质量指数、腰臀比、体脂量、血浆血脂指标、脂肪肝发病率及其严重程度变化,同时监测各组营养干预前后D-乳酸、细菌脂多糖、二胺氧化酶等肠屏障功能指标的变化并比较其差异性。
结果与结论:给予不同营养方式干预3个月后,①肠道微生态制剂联合改良低碳饮食的肥胖患者的体质量指数、体脂量、血脂指标、脂肪肝分级和肠屏障功能各项指标的改善更明显(P < 0.05或P < 0.01),比单纯低碳饮食的临床效果好;②联合肠道微生态制剂的低碳饮食能更显著地降低肥胖症患者血清肠道损伤指标D-乳酸、细菌脂多糖和二胺氧化酶水平
(P < 0.05或P < 0.01);③上述数据证实,肠道微生态制剂联合强化补充维生素矿物质的改良低碳饮食减肥法能够有效降低患者血清肠屏障损伤指标水平,而且相比于单独低碳饮食治疗,这种新的饮食治疗方案可更显著降低此类患者的体脂和血脂水平,减轻脂肪肝的严重程度,改善肠屏障功能。
https://orcid.org/0000-0003-0707-6772(刘建国);https://orcid.org/0000-0001-7111-7087(朱翠凤)
中图分类号:
引用本文
刘建国, 潘 勇, 李翰宇, 刘 燕, 张圳甜, 林秀萍, 张 苑, 刘亚静, 张 帆, 张雷钧, 肖烈辉, 徐爱民, 朱翠凤. 肠道微生态制剂联合改良低碳饮食靶向调控肥胖症患者体脂代谢及肠屏障功能的效果分析[J]. 中国组织工程研究, 2021, 25(29): 4672-4679.
Liu Jianguo, Pan Yong, Li Hanyu, Liu Yan, Zhang Zhentian, Lin Xiuping, Zhang Yuan, Liu Yajin, Zhang Fan, Zhang Leijun, Xiao Liehui, Xu Aimin, Zhu Cuifeng. Effect of microecological preparation combined with an improved low-carbon diet on fat metabolism and intestinal barrier function in obese patients[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(29): 4672-4679.
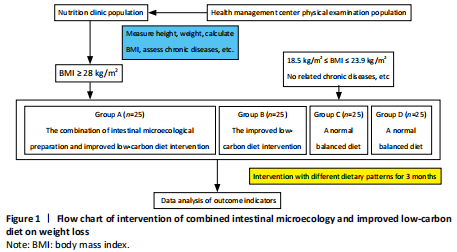
The trial followed the principle of intention analysis, 100 participants were involved in the result analysis, and there were no shedding cases, as shown in Figure 1.
Comparison of general data and a 24-hour retrospective dietary survey, a lifestyle survey, a medical history, and family history between study groups before the nutritional intervention
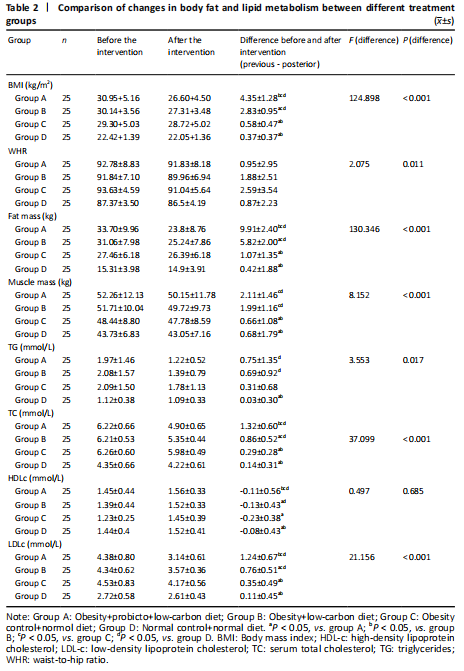
There were no significant difference in the general data among four groups, including case number, age, sex, smoking, alcohol usage, and high blood pressure (P > 0.05), but the weekly physical activity and protein energy ratio were lower in obese subjects than healthy controls (P < 0.01). Daily total energy intake, fat energy ratio, abnormal glucose metabolism, and the prevalence of carotid atherosclerosis were significantly increased in three obese patients (groups A, B, C) than normal control group (group D) (P < 0.05 or
P < 0.01). These results indicate that obesity is a high-risk factor for abnormal glucose metabolism and atherosclerosis (Table 1).
Changes in body composition and lipid metabolism
Before intervention: BMI, waist-hip ratio, fat mass, TG, TC, and LDL-c were significantly higher in obese groups than in the healthy control group (P < 0.05), and there was no significant difference among the three intervention groups (P > 0.05; Table 2). After nutritional intervention, the parameters of BMI, waist-hip ratio and fat mass in obese subjects fed with low-carbon diet with or without microecological preparation were significantly improved (P < 0.05). Obese patients given microecological preparation combined with low-carbon diet exhibited better improvement in BMI and fat mass than those with low-carbon diet alone. These results suggest that microecological preparation combined with low-carbon diet is more effective to reduce body fat and BMI than a low-carbon diet alone (Table 2).
Microecological preparation combined with low-carbon diet or single low-carbon diet intervention significantly improved serum lipid profiles, including reduction of TG, TC, and LDLc (P < 0.05; Table 2). The levels of TC and LDLc were lower in obese patients given microecological preparation combined with low-carbon diet than those given single low-carbon diet (P < 0.01). These results reveal that microecological preparation combined with low-carbon diet is more effective to reduce serum hyperlipidemia than low-carbon diet alone.
Pathological changes in hepatic remodeling
After 3-month intervention, the incidence and severity (Table 3) of non-alcoholic fatty liver disease were decreased in obese patients given both microecological preparation combined with low-carbon diet and single low-carbon diet, and combined treatment of microecological preparation and low-carbon diet had better clinical outcomes. These results suggest that microecological preparation combined with low-carbon diet is more effective to reduce the severity of fatty liver disease than a low-carbon diet alone.
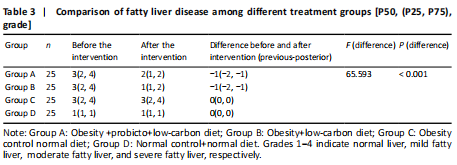
Changes in intestinal barrier function
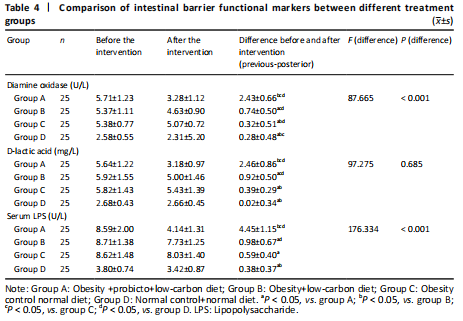
Before the intervention, serum levels of D-lactic acid, diamine oxidase and bacterial LPS were significantly higher in obese subjects, as compared with healthy subjects (P < 0.05). Whereas serum levels of D-lactic acid,diamine oxidase and LPS were significantly decreased in subjects fed with two intervention (P < 0.05). More importantly, the serum levels of D-lactic acid,diamine oxidase and LPS were lower in subjects treated with microecological preparation and low-carbon diet than with single low-carbon diet (P < 0.05). These results suggested that microecological preparation combined improved low-carbon diet had better effective than improved low-carbon diet alone in repairing intestinal mucosal damage, reducing intestinal permeability, and inhibiting LPS translocation (Table 4).
Adverse reaction events
No adverse reaction was found in each group during the trial.
| [1] NCD RISK FACTOR COLLABORATION (NCD-RISC). Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: a pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. Lancet. 2017;390(10113):2627-2642. [2] LEE EY, YOON KH. Epidemic obesity in children and adolescents: risk factors and prevention. Front Med. 2018;12(6):658-666. [3] ZHANG X, ZHANG M, ZHAO Z, et al. Geographic variation in prevalence of adult obesity in China: Results from the 2013-2014 National Chronic Disease and Risk Factor Surveillance. Ann Intern Med. 2020;172(4):291-293. [4] LUO NY. Establishment and Issue of Healthy China Action Committee (2019-2030). Zhongyiyao Guanli Zazhi. 2019;27(14):cover 2. [5] WILLIAMS R, AITHAL G, ALEXANDER GJ, et al. Unacceptable failures: the final report of the Lancet Commission into liver disease in the UK. Lancet. 2020;395(10219):226-239. [6] GBD 2015 OBESITY COLLABORATORS, AFSHIN A, FOROUZANFAR MH, et al. Health effects of overweight and obesity in 195 countries over 25 years. N Engl J Med. 2017;377(1):13-27. [7] MOORADIAN AD. In search for an alternative to sugar to reduce obesity. Int J Vitam Nutr Res. 2019;89(3-4):113-117. [8] HUI S, COWAN AJ, ZENG X, et al. Quantitative fluxomics of circulating metabolites. Cell Metab. 2020;32(4):676-688.e4. [9] GOMEZ-ARBELAEZ D, CRUJEIRAS AB, CASTRO AI, et al. Acid-base safety during the course of a very low-calorie-ketogenic diet. Endocrine. 2017;58(1):81-90. [10] GIANNOUDAKI E, HERNANDEZ-SANTANA YE, MULFAUL K, et al. Interleukin-36 cytokines alter the intestinal microbiome and can protect against obesity and metabolic dysfunction. Nat Commun. 2019;10(1):4003. [11] FORTE N, FERNÁNDEZ-RILO AC, PALOMBA L, et al. Obesity affects the microbiota-gut-brain axis and the regulation thereof by endocannabinoids and related mediators. Int J Mol Sci. 2020;21(5):1554. [12] BLÜHER M. Obesity: global epidemiology and pathogenesis. Nat Rev Endocrinol. 2019; 15(5):288-298. [13] TODA G, SOEDA K, OKAZAKI Y, et al. Insulin- and lipopolysaccharide-mediated signaling in adipose tissue macrophages regulates postprandial glycemia through Akt-mTOR activation. Mol Cell. 2020;79(1):43-53.e4. [14] GUO ZP. Quantitative medical imaging evaluation of fatty liver. Zhonghua Jiankang Guanglixue Zazhi. 2020;14(6):583-586. [15] XIA MF, BIAN H, GAO X. Thoughts on the change of nomenclature from non-alcoholic fatty liver disease to metabolic associated fatty liver disease. Zhonghua Tangniaobing Zazhi. 2020;12(7):445-450. [16] SONG XG, HE YB. Advances in the application of magnetic resonance imaging in hepatic fat quantification in patients with nonalcoholic fatty liver disease. Chin Hepatol. 2020;25(5):536-538. [17] D’SOUZA MS, DONG TA, RAGAZZO G, et al. From fad to fact: evaluating the impact of emerging diets on the prevention of cardiovascular disease. Am J Med. 2020; 133(10):1126-1134. [18] SARTORIUS K, SARTORIUS B, MADIBA TE, et al. Does high-carbohydrate intake lead to increased risk of obesity? A systematic review and meta-analysis. BMJ Open. 2018; 8(2):e018449. [19] LYONS L, SCHOELER NE, LANGAN D, et al. Use of ketogenic diet therapy in infants with epilepsy: a systematic review and meta-analysis. Epilepsia. 2020;61(6):1261-1281. [20] GUO YL, RUAN LT, DANG Y, et al. Application value of liver and spleen stiffness measured by real-time shear wave elastography combined with hepatic steatosis index in diagnosis of hepatic steatosis. Journal of China Clinic Medical Imaging. 2019;30(6):416-420. [21] SHI HP, YU KY, ZHOU H. Low carbon diet, cancer metabolism and nutrition. Electronic J. 2019;6(1):1-6. [22] WAISE TMZ, RASTI M, DUCA FA, et al. Inhibition of upper small intestinal mTOR lowers plasma glucose levels by inhibiting glucose production. Nat Commun. 2019;10(1):714. [23] RASTELLI M, CANI PD, KNAUF C. The gut microbiome influences host endocrine functions. Endocr Rev. 2019;40(5):1271-1284. [24] VISCONTI A, LE ROY CI, ROSA F, et al. Interplay between the human gut microbiome and host metabolism. Nat Commun. 2019;10(1):4505. [25] GUAZZELLI MARQUES C, DE PIANO GANEN A, ZACCARO DE BARROS A, et al. Weight loss probiotic supplementation effect in overweight and obesity subjects: a review. Clin Nutr. 2020; 39(3):694-704. [26] LI S, QI C, ZHU H, et al. Lactobacillus reuteri improves gut barrier function and affects diurnal variation of the gut microbiota in mice fed a high-fat diet. Food Funct. 2019;10(8): 4705-4715. [27] ZUO K, LI J, XU Q, et al. Dysbiotic gut microbes may contribute to hypertension by limiting vitamin D production. Clin Cardiol. 2019;42(8): 710-719. [28] WU T, SUN M, LIU R, et al. Bifidobacterium longum subsp. longum remodeled roseburia and phosphatidylserine levels and ameliorated intestinal disorders and liver metabolic abnormalities induced by high-fat diet. J Agric Food Chem. 2020;68(16):4632-4640. [29] MARTENS EC, NEUMANN M, DESAI MS. Interactions of commensal and pathogenic microorganisms with the intestinal mucosal barrier. Nat Rev Microbiol. 2018;16(8):457-470. [30] VIRTUE AT, MCCRIGHT SJ, WRIGHT JM, et al. The gut microbiota regulates white adipose tissue inflammation and obesity via a family of microRNAs. Sci Transl Med. 2019; 11(496):eaav1892. [31] LIU JG, LIN XP, ZHANG Y, et al. Analysis of the effects and mechanism of compound intestinal microecological preparation combined with modified low-carbon diet on appetite and body fat metabolism in children with simple obesity. Chin Gen Prac. 2020;23(29):3675-3681. |
| [1] | 罗安玉, 刘瀚霖, 谢小飞, 黄 琛. 抗氧化合剂对膝骨关节炎模型大鼠结构性退变的作用[J]. 中国组织工程研究, 2021, 25(23): 3625-3629. |
| [2] | 方 雯, 李 泽, 柳昭明, 冯 红. 中等强度训练干预大鼠骨骼肌抗增殖蛋白PHB1表达及线粒体呼吸功能的变化[J]. 中国组织工程研究, 2020, 24(8): 1207-1212. |
| [3] | 冯程钦, 曾 平, 罗 钧, 刘金富, 黄佳兴. 维生素D受体基因多态性与骨关节炎关系的Meta分析[J]. 中国组织工程研究, 2020, 24(33): 5405-5412. |
| [4] | 温霜威, 武青梅. 有氧运动联合左旋甲状腺素与维生素D3改善亚临床甲减大鼠骨质疏松的作用[J]. 中国组织工程研究, 2020, 24(26): 4118-4124. |
| [5] | 吴鹏波, 宋 琪, 俞媛洁, 饶 倩, 张 国, 郭一天, 谭诗云. 姜黄素激活自噬干预非酒精性脂肪肝病模型大鼠氧化应激及炎症反应[J]. 中国组织工程研究, 2020, 24(11): 1720-1725. |
| [6] | 张文将, 易 健, 贾 平, 陈博威, 王 勇, 刘柏炎 . 载脂蛋白E基因敲除(APOE-/-)小鼠基因的鉴定方法及模型应用[J]. 中国组织工程研究, 2019, 23(7): 1103-1108. |
| [7] | 卢秋平,桂裕昌,许建文,覃海飚,饶远森. 维生素D受体基因多态性与胸腰段骨质疏松性椎体压缩骨折半定量分型的关联[J]. 中国组织工程研究, 2019, 23(35): 5577-5581. |
| [8] | 杨忠明,蒋满意,许思毛. 中强度耐力运动与大蒜素对高脂饮食致脂肪肝模型大鼠具有协同抵抗效应[J]. 中国组织工程研究, 2019, 23(31): 5030-5035. |
| [9] | 刘文静,孙养鹏,郑有华,张志光. 1,25-二羟维生素D3和地塞米松促进人颞下颌关节滑液间充质干细胞的体外成骨分化[J]. 中国组织工程研究, 2018, 22(9): 1443-1449. |
| [10] | 赵英鹏,李 立,陈 刚,白建华,刘其雨. 减体积肝移植在大鼠脂肪肝供肝肝移植模型中的应用[J]. 中国组织工程研究, 2018, 22(4): 582-586. |
| [11] | 丁 晨,江 艺,潘 凡,蔡秋程. 脂肪肝模型大鼠自体原位肝移植后的脂质代谢变化[J]. 中国组织工程研究, 2018, 22(36): 5821-5827. |
| [12] | 刘 锐1,何 嘉2,刘秀芳2,冯晓秋1,古展鑫2,康善平1 . 多烯磷脂酰胆碱干预大鼠非酒精性脂肪肝细胞模型基因芯片组学分析[J]. 中国组织工程研究, 2018, 22(36): 5833-5839. |
| [13] | 黄晓东,邓国英,王伟恒,徐立璋,马 俊,叶晓健. 生长因子对椎间盘终板软骨细胞抗氧化能力的影响[J]. 中国组织工程研究, 2017, 21(4): 520-526. |
| [14] | 陈尚雅,崔冠群,薄存香,张 玉,张恩国,杨 叶,杜忠君,邵 华. 大鼠肺泡Ⅱ型上皮细胞系RLE-6TN诱导脂肪间充质干细胞体外分化[J]. 中国组织工程研究, 2017, 21(33): 5280-5286. |
| [15] | 张良明,杨 阳,陈振翔,何天威,何 磊,刘 斌,戎利民. 中国南方地区中老年患者血清维生素D水平与骨密度的相关性[J]. 中国组织工程研究, 2017, 21(28): 4448-4453. |
In recent years, the restriction of carbohydrates intake has been recognized as an important way to overcome the challenges in weight loss. Several approaches can be used for weight loss, including fasting, low-carbon diet, and ketogenic diet[7-8]. Despite the benefits of these therapies, there is an international debate over adverse effects and safety[9]. Therefore, seeking safer and more effective weight-loss diets has become a hot topic in medical research.
Intestinal microecological imbalance in recent years has been considered to be a key factor leading to simple obesity and other metabolic diseases[10-11]. Numerous studies have shown that a high-fat diet can alter the abundance and structure of the intestinal microbiota in mice, leading to obesity and insulin resistance[12-13]. Our previous studies found that fecal transplantation from normal-weight mice to obese mice can significantly reduce body weight in the latter group. Furthermore, the administration of dietary fiber or prebiotics and probiotics reduced body weight and insulin resistance of obese mice. However, these studies mainly focus on animal experiments and there are no sufficient evidences from human. Therefore, their mechanisms remain explored. In view of this, this study was designed to evaluate the effect of a weight-loss therapy using microecological preparation combined with an improved low-carbon diet on fat metabolism and intestinal barrier function in overweight and obese patients and to analyze the possible mechanisms. 中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程
A prospective randomized controlled trial.
Time and settings
This study was completed in the Health Management Center and Nutrition Department of Shenzhen Hospital of Southern Medical University, China from August 2017 to October 2019.
Participants
Case collection and random grouping: 75 adults with obesity aged 18 to 60 were selected from the outpatient department of the Nutrition and Health Management Center of Shenzhen Hospital Southern Medical University. Study participation was voluntary, and the enrolled patients were randomly assigned to different groups using a table of random numbers (no blinding). Patients who had number 1 to 3 were assigned to group A (n=25) and were given microecological preparation + an improved low-carbon diet. Participants who had number 4 to 6 were assigned to group B (n=25) and received an improved low-carbon diet only. Patients who had number 7 to 9 were assigned to group C (n=25) (food intervention control group with obesity) and were given a balanced diet. If the random number was 0, a second draw would be performed. Another 25 people with normal physical examination (normal weight, no other chronic diseases, infectious diseases or other genetic diseases) were given a balanced diet and served as healthy controls (group D, n=25). This study was approved by the Ethics Committee of Shenzhen Hospital of Southern Medical University in 2017, with an approval No. NYSZYYEC20170026, and all the subjects signed an informed consent.
Inclusion criteria
Obesity was defined either according to body mass index (BMI) or waist circumference, and the definitions of metabolic abnormalities were derived from five different criteria. The diagnostic criteria for overweight and obesity according to BMI were established according to the Expert Consensus on Medical Nutrition Therapy for Overweight/Obesity in China (2016) using the following BMI (weight [kg]/height [m]2) ranges: 25 kg/m2 ≤ BMI < 28 kg/m2, overweight; and BMI ≥ 28 kg/m2, obese.
Exclusion criteria
(1) Endocrine diseases caused by obesity or drug use; (2) usage of other weight-loss methods, including surgery and therapy; (3) individuals who received antioxidant or functional food for 2 weeks or more in the past 3 months; (4) individuals who used probiotics, prebiotics, yogurt, and other food products that might affect the structure of intestinal microbiota for 1 week or more in the last month; (5) subjects treated with antibiotic drugs in the last 2 weeks that might affect the structure of intestinal microbiota; (6) fever, skin soft tissue infection, acute enteritis, and other diseases or symptoms in the past week that might affect the structure of intestinal microbiota; (7) patients with cancer who received chemotherapy or radiotherapy in the past 6 months; (8) non-compliance and loss of follow-up; pregnant and nursing women; (9) significant organ dysfunction or serious complications in the heart, brain, kidney, eyes, feet, and neuropathy; (10) patients with mental disorders; and (11) excluded patients with poor compliance.
Methods
Nutritional therapy and lifestyle intervention in the study groups
Group A (obesity intervention group 1) were given microecological preparation + improved low-carbon diet.
Microecological preparation is described as follows: 1) Enteric-coated capsules of glutamine (purchased from Chengdu Diao Pharmaceutical Group Co., Ltd., China), including L-glutamine, ginseng, licorice (honey broach), atractylodes, Poria cocos (250 mg per capsule), two tablets twice a day (at breakfast and dinner); 2) Bifidobacterium trifecta enteral solution capsules (compound preparation, purchased from Jincheng Hayes Pharmaceutical Co., Ltd., China), each gram contains Bifidobacterium longum ≥ 1.0 × 106 CFU, Lactobacillus acidophilus ≥ 4.0 × 106 CFU, and Enterococcus faecalis ≥ 1.0 × 106 CFU, two tablets twice a day (at breakfast and dinner); 3) Probiotics (ekeli) (purchased from Beijing Tongze Kang-Cheng Medical Technology Co., Ltd., China): galactose oligosaccharide, trisaccharide, and mannose oligosaccharide), 2 g once a day (at breakfast); 4) Compound metabolites of probiotics (purchased from Beijing Ketuo Heng-Tong Biotechnology Co., Ltd., China), comprised of the metabolites of five strains probiotics (Lactobacillus casei Zhang, Lactobacillus plantarum p-8, Bifidobacterium lactis V9, Lactobacillus rhamnosus M9, and Bifidobacterium lactis M8). The products are rich in metabolic factors such as probiotics, active peptides, polysaccharides, gamma-aminobutyric acid and natural antibacterial substances (phenyllactic acid). The amount of each strain is ≥ 50 CFU/g. 2 g packs, once a day, 1 pack each time, after breakfast. Improved low-carbon diet scheme is described as follows: Three meals a day, with a daily total energy intake (TEI) of 4282.49-4575.37 kJ, 93.18-105.88 g of protein (36.41%-38.73% of TEI), 48.29-60.09 g of carbohydrate (18.87%-21.98% of TEI), and 52.04-60.14 g of fat (45.76%-49.50% of TEI).
1) Staple food: Limit the consumption of fruits and vegetables containing more than 5% of sugar, and replace these products to nutrition bars (100 g per day) (purchased from Hangzhou Saineng Pharmaceutical Co., China) as staple food (25 g at breakfast, 50 g at lunch, and 25 g at dinner. The nutrition bars have the following composition per 100 g: total calories, 1612.9 kJ; protein, 28.8 g; fat, 14.6 g; carbohydrate, 34.7 g; dietary fiber, 13.0 g; sodium, 515 mg.
2) Non-staple food: Breakfast: 1 egg + 50 g of dried tofu + 200 g of vegetables (tomato or cucumber); lunch: 50 g of meat + 80 g of fish or shrimp + 200 g vegetables with < 5% sugar (green vegetable, melon, and eggplant vegetables, bacterial and algal food). Dinner:
50 g of meat + 80 g of fish or shrimp + 200 g vegetables with < 5% sugar (green vegetable, melon, and eggplant vegetables, bacterial and algal food).
3) Soda water: Dissolve 0.5 g of sodium bicarbonate in 2 000 mL warm water, intake 200 mL per hour once, 10 times a day.
4) Dietary supplements: i) calcium (Wyeth-Ayerst (China) Ltd., China): 300 mg (men) or 600 mg (women) once a day after lunch; ii) Centrum tablets (Wyeth-Ayerst (China) Ltd.), one tablet once a day after lunch; iii) ferrous succinate tablets (Jinling Pharmaceutical Company Limited Nanjing Jinling Pharmaceutical Factory, China): 0.1 g (men) or 0.2 g (women), twice a day (at breakfast and dinner).
Group B (obesity intervention group 2) should have the same improved low-carbon diet as group A, except for microecological preparation. Other foods and treatment plans were the same as group A.
Group C (obesity control group) should eat a balanced diet (10%-15% of protein, 55%-60% of carbohydrate, and 25%-30% of fat), corresponding to 104.6-117.2 kJ/kg per day.
Group D (healthy control group) should eat the same balanced diet as group C.
Exercise
Moderate intensity aerobic exercise, 30-60 minutes per day, corresponding to a daily energy expenditure of 1 255.2-2 092.0 kJ, 3-5 times a week, was conducted. All four groups had same exercise level. Moreover, other variables, such as water consumption and food types in four groups, should remain consistent, while energy supply ratios were different in each group as required.
Body composition assessment by bioelectrical impedance analysis (BIA)
Body composition was measured by Tanita MC-980 body composition analyzer (Tanita Corp., Tokyo, Japan). The instrument principle is described below:
For most clinical and health-related purposes, human body composition is the relative proportion of fat and lean tissue. Fat mass is the total fat mass, including nerve tissues and brain. Fat-free mass is body fat in other tissues, including body fluids and skeleton. Lean body mass is used interchangeably with fat-free mass.
BIA measures the impedance or resistance of fluids present in lean tissue with a low-level electric current through body. Impedance is low in lean tissue, where most intracellular and extracellular fluids and electrolytes are present, and high in fat tissue. Therefore, impedance is proportional to total body water. Lean body mass is calculated by using a hydration fraction of 73.2% for lean tissue, and this percentage is used in other methods that assess body composition. Fat mass is calculated as the difference between body weight and lean body mass.
Impedance has two components: resistance and reactance. Conductive characteristics are measured in body fluids, whereas frequency-dependent reactance is measured in cell membranes, which act as imperfect isolating transistors. Measuring impedance at different frequencies (e.g., 5 and 200 kHz) and applying predictive equations allows estimating both extracellular water and total body water, respectively, and intracellular water is determined by deduction. Extracellular water is related to extracellular mass and intracellular water is related to body cell mass.
Body composition was measured in overweight and obese or normal-weight people with standing position, barefoot, according to the manufacturer’s instructions. The device used multiple-frequency (5, 50, 250, and 500 kHz) BIA technology and had eight tactile electrodes. Two electrodes were placed in contact with the palm and thumb of each hand, and two remained in contact with the anterior and posterior aspect of the sole of each foot. Patients were instructed to stand with their soles in contact with the foot electrodes and grab the hand electrodes. Total and visceral adiposity was determined by a firmware embedded in the Tanita MC-980 body composition analyzer using a proprietary equation developed by the manufacturer.
The original prediction equations built into Tanita’s BIA software were derived by Professor Steven Heymsfield’s research team at St Luke’s/Roosevelt Hospital, College of Physicians and Surgeons, Columbia University, New York. Tanita BIA analyzers have been more extensively validated against alternative body composition techniques than other BIAs. A selection of studies published in abstract form can be found at: http://www.tanita.co.uk/professional_index.cfm?page=professional_resource04.
Measurement of blood lipids
Venous blood serum samples were collected from the antecubital vein into Vacutainer tubes after a 12-hour overnight fast. Before and after the intervention, fasting blood samples were drawn within 24 hours of admission. Serum total cholesterol (TC) (normal range,
2.16-5.20 mmol/L), triglycerides (TG; normal range, < 2.26 mmol/L),
high-density lipoprotein cholesterol (HDL-c; normal range,
> 1.68 mmol/L), and low-density lipoprotein cholesterol (LDL-c; normal range, < 3.34 mmol/L) were measured by an enzymatic colorimetric method in an automated analyzed model Roche Cobas c702 (Roche Diagnostics, Basel, Switzerland).
Ultrasonographical diagnosis of fatty liver
Fatty liver was evaluated by a Kelly S40 Plus ultrasound device. Fatty liver disease is a clinicopathological syndrome characterized by liver steatosis and can be divided into alcoholic and nonalcoholic. The disease spectrum includes fatty liver, fatty hepatitis and cirrhosis. In this study, color Doppler ultrasound was used to diagnose and grade fatty liver.
Ultrasonic examinations were conducted using Kelly S40 Plus color Doppler ultrasound, with a probe frequency of 3.5 Hz by two physicians certified in color Doppler ultrasonography (color Doppler flow imaging/doctor operation). After fasting for at least 8 hours, the patient lied on the left side, breathed normally, exhaled continuously over 5 seconds. The Doppler sample volume was placed in the center of the lumen to avoid contacting with the luminal wall which prevented effects of inferior vena cava. High-quality images were recorded[14].
Doppler spectrum characteristics: The waveform is classified into three types by the Doppler spectrum characteristics of the pulse in the right hepatic vein: (1) normal type (HV0 type): three-phase or four-phase waveform, with transient reverse blood flow, manifested as two negative waves and one or two positive waves; (2) attenuating type (HV1 type): the wave amplitude is reduced, and the short-term reverse blood flow disappears, manifested as two negative waves; (3) flat (HV2): one-way flow curve waveform is similar to the portal vein waveform[14].
Fatty liver is classified by the ultrasonographic characteristics: (1) mild fatty liver, in which the light spot is fine, near-field echo is slightly enhanced, far-field echo is attenuated by 1/3, and the liver tubular structure is visible; (2) moderate fatty liver, wherein liver margins are well-defined, near-field echo is enhanced, far-field echo is attenuated by 50%, and the tubular structure is poorly defined; (3) severe fatty liver, in which the liver spot is fine, near-field echo is significantly enhanced, far-field echo is attenuated by 75%, and the tubular structure cannot be identified. The sonographic findings without the above manifestations and other liver lesions are normal[15-16].
Analysis of intestinal barrier function
A portable biochemical analysis system (JY-DLT; Beijing Zhongsheng Jinyu Diagnostic Technology Co., Ltd., China) was used to quantify D-lactic acid (a marker of intestinal permeability changes), Diamine oxidase (a marker of intestinal mucosal damage), and bacterial endotoxin lipopolysaccharide (LPS, a marker of bacterial and endotoxin translocation).
Sample preparation: After blood collection with serum gel tubes, the tubes were gently inverted for four or five times and then vortexed. The samples were left to stand for 30 minutes, centrifuged at 3 500–4 000 r/min for 10 minutes, and analyzed.
Sample detection procedure: The serum was collected by a pipette and 20 μL serum was transferred to the corresponding number cartridge. (1) The cartridge was inserted into the 37℃ warm bath for 10 minutes; (2) the cartridge was inserted into the analyzer detection port after the warm bath time, and the detection results were read after 2 seconds.
The dry chemistry analyzer principle of detection methodology is the reflection spectrophotometry, chemical reaction between reagent and substance that measured in blood samples would produce a colored product, and the changes of absorbance are directly related to the concentration of the substance of interest.
Methodology and principles: dry chemistry method using following reagents:
1) Diamine oxidase: the oxidogenic substrate putrescine reacts with Diamine oxidase in the blood to form H2O2. H2O2 reacts with a chromogenic agent in the presence of POD and produces a red product, within the range of the standard curve, and color intensity is linearly correlated with diamine oxidase concentration.
2) D-lactic acid: D-lactic acid which presents in sera interacts with nicotinamide adenine dinucleotide (coenzyme I) then produces acetoacetic acid and reducibility coenzyme I under the action of d-lactate dehydrogenase. Reducibility coenzyme I reduces nitroblue tetrazolium in the presence of diaphorase, resulting in the formation of blue formazan. Color intensity was positively correlated with D-lactate concentration in the blood.
3) Bacterial endotoxin LPS: Bacterial endotoxins hydrolyze β-naphthylamine substrates, and the product reacts with a diazonium compound and assumes a reddish color. The color intensity is positively correlated with bacterial endotoxin activity.
Main outcome measures
Body composition analysis (height, weight, body fat rate, body fat volume, muscle mass), lipid metabolism markers (TG, TC, HDL-c, LDL-c), fatty liver, intestinal barrier function were observed.
Statistical analysis
A database was created, and statistical analysis was performed using SPSS software version 23.0. Descriptive analysis of count data was expressed as percentage (%), measurement data as descriptive analysis, normal data as the mean ± standard deviation (SD) and non-normal data as quartile [P50, (P25, P75)]. x2 test was used for inter-group comparisons, and Z test was used for inter-group comparisons (Bonferroni test corrected P values). Analysis of variance was used for the comparison of normality data, and SNK method was used for the pair-wise comparison between groups. The rank sum test was used for non-normal data comparison between groups, and K-W test was used for pair comparison between groups. A value of P < 0.05 was considered statistically significant.
The trial followed the principle of intention analysis, 100 participants were involved in the result analysis, and there were no shedding cases, as shown in Figure 1.
Comparison of general data and a 24-hour retrospective dietary survey, a lifestyle survey, a medical history, and family history between study groups before the nutritional intervention
There were no significant difference in the general data among four groups, including case number, age, sex, smoking, alcohol usage, and high blood pressure (P > 0.05), but the weekly physical activity and protein energy ratio were lower in obese subjects than healthy controls (P < 0.01). Daily total energy intake, fat energy ratio, abnormal glucose metabolism, and the prevalence of carotid atherosclerosis were significantly increased in three obese patients (groups A, B, C) than normal control group (group D) (P < 0.05 or
P < 0.01). These results indicate that obesity is a high-risk factor for abnormal glucose metabolism and atherosclerosis (Table 1).
Changes in body composition and lipid metabolism
Before intervention: BMI, waist-hip ratio, fat mass, TG, TC, and LDL-c were significantly higher in obese groups than in the healthy control group (P < 0.05), and there was no significant difference among the three intervention groups (P > 0.05; Table 2). After nutritional intervention, the parameters of BMI, waist-hip ratio and fat mass in obese subjects fed with low-carbon diet with or without microecological preparation were significantly improved (P < 0.05). Obese patients given microecological preparation combined with low-carbon diet exhibited better improvement in BMI and fat mass than those with low-carbon diet alone. These results suggest that microecological preparation combined with low-carbon diet is more effective to reduce body fat and BMI than a low-carbon diet alone (Table 2).
Microecological preparation combined with low-carbon diet or single low-carbon diet intervention significantly improved serum lipid profiles, including reduction of TG, TC, and LDLc (P < 0.05; Table 2). The levels of TC and LDLc were lower in obese patients given microecological preparation combined with low-carbon diet than those given single low-carbon diet (P < 0.01). These results reveal that microecological preparation combined with low-carbon diet is more effective to reduce serum hyperlipidemia than low-carbon diet alone.
Pathological changes in hepatic remodeling
After 3-month intervention, the incidence and severity (Table 3) of non-alcoholic fatty liver disease were decreased in obese patients given both microecological preparation combined with low-carbon diet and single low-carbon diet, and combined treatment of microecological preparation and low-carbon diet had better clinical outcomes. These results suggest that microecological preparation combined with low-carbon diet is more effective to reduce the severity of fatty liver disease than a low-carbon diet alone.
Changes in intestinal barrier function
Before the intervention, serum levels of D-lactic acid, diamine oxidase and bacterial LPS were significantly higher in obese subjects, as compared with healthy subjects (P < 0.05). Whereas serum levels of D-lactic acid,diamine oxidase and LPS were significantly decreased in subjects fed with two intervention (P < 0.05). More importantly, the serum levels of D-lactic acid,diamine oxidase and LPS were lower in subjects treated with microecological preparation and low-carbon diet than with single low-carbon diet (P < 0.05). These results suggested that microecological preparation combined improved low-carbon diet had better effective than improved low-carbon diet alone in repairing intestinal mucosal damage, reducing intestinal permeability, and inhibiting LPS translocation (Table 4).
Adverse reaction events
No adverse reaction was found in each group during the trial.
DISCUSSION
Restriction of carbohydrate intake is considered as an effective way to reduce body weight gain. In recent years, exploring the therapeutic benefits of low-carbon diet and ketogenic diet have emerged as a hot topic[17-19]. Studies have shown that reducing carbohydrate intake increases fat utilization, reduces insulin levels, and increases insulin sensitivity[20-21]. Low-carbon diets are effective in losing weight, improving type 2 diabetes and other diseases. Carbohydrate-rich staple foods can be a good source of vitamins and minerals, and low carbohydrate intake may cause deficiency of thiamine, folate, magnesium, calcium, iron, and iodine, leading to nutrient imbalance. The Atkins diet (4%-34% carbohydrate) can decrease the levels of vitamins A and E and trace elements. A 6-month intervention using a paleolithic diet (30% carbohydrate) increases the iodine deficiency rate from 15% to 73%. Moreover, low-carbon and ketogenic diet may have adverse effects such as dizziness, fatigue, cramps, hair loss, hunger, anemia, and constipation[22].
This study analyzed the effect of an improved low-carbon diet, which provides 4184.0-4602.4 kJ of energy with 90-100 g of protein (36% of total energy, equivalent to 18%-20% of the RDA recommended for a healthy adult male), 50-60 g of carbohydrate (20%-22% of total energy, corresponding to 10%-12% of RDA), and 47-54 g of fat per day (42%-44% of total energy, equivalent to 21%-24% of RDA). The ratio of saturated, monounsaturated, and polyunsaturated fatty acids was 1.0:1.2-1.5:1.0. This dietary pattern guarantees the intake of sufficient high-quality proteins which prevents the occurrence of myodystrophy during weight-loss, and prevents the adverse effect of excessive fat intake in term of cardiovascular and long-term health. The low-carbon diet used in this study, in addition to limitation of fat intake, included a daily supplementation of multivitamin tablets 1 + calcium carbonate (300-600 mg) (male) + ferrous succinate tablets (0.2 g for male subjects and 0.4 g for female subjects). These dietary supplements can provide essential vitamins, minerals, coenzymes, and prosthetic groups, allowing the conversion of BF into glucose through gluconeogenesis. A large number of data have shown that a low-carbohydrate diet does have a unique effect on preventing obesity[23]. Our results have also demonstrated the obvious effect of low-carbohydrate diet in promoting body fat catabolism in obese patients, but its potential mechanism remains unknown. Therefore, further search for safe and effective weight-loss methods with clear mechanisms is still a hot topic worldwide.
Intestinal microecological imbalance is a hot research topic in recent years[24]. Studies have shown that a high-fat diet can alter the abundance and structure of the intestinal microbiota in mice, leading to obesity and insulin resistance, which is a key mechanism explaining the influence of the intestinal microbiome on
obesity[25-27]. The gut microbiome is altered and LPS levels and intestinal permeability are increased, allowing LPS to pass intestinal mucosal barrier into the circulation and activate an inflammatory response, which potentially leads to a substantial increase of inflammatory factors, including interleukin-6 and tumor necrosis factor-1, impairs insulin signaling, and ultimately causes insulin resistance[10, 28-29]. Other studies have shown that the ratio of Firmicutes to Bacteroides may affect energy absorption efficiency and fat conversion efficiency in humans[30]. Our previous studies showed that fecal transplantation from normal-weight mice to obese mice reduced body weight significantly in the latter group. Furthermore, the direct administration of dietary fiber or prebiotics and probiotics to obese mice significantly reduced body weight and insulin resistance. However, these studies primarily focused on animal experiments and lacked sufficient evidence in humans.
In view of this, we are the first to try to combine intestinal microecology with an improved low-carbon diet supplemented with vitamins and minerals for the clinical treatment of obesity, and to observe the effect of the combination of two treatments on body fat metabolism and possible complications of obesity, which is also the highlight of this study. We found that the combination of microecology and low-carbon diet improved BMI, body fat, lipid level, and the incidence and severity of non-alcoholic fatty liver disease, which is consistent with previous findings[10]. In addition, the occurrence of constipation, diarrhea and hair loss during weight loss in the intestinal microecology combined with improved
low-carbon diet group was also significantly lower than that in the single improved low-carbon diet group[31].
There had several limitations in present study. (1) The sample size of the study was not large enough; (2) this study only observed the conditions after 3 months of intervention to avoid loss of follow-up; (3) the potential mechanisms of an improved low-carbohydrate diet supplemented with vitamins and minerals in intestinal microecology were not well studied. Therefore, further investigations are still required.
In the present study, the improvements might relate to the effect of microecological preparation (including beneficial bacteria and prebiotics) combined with a low-carbon diet on fat metabolism and intestinal barrier function in obese patients, which can effectively prevent LPS translocation from the enteric cavity to the blood, can quickly and significantly increase the growth of native probiotics, improve intestinal immunity, and maintain normal intestinal function by optimizing intestinal microecology. Microecological preparation combined with a low-carbon diet can improve energy metabolism more effectively than a control diet in overweight/obese patients.
中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程

文题释义:#br# 肠-脑轴与肥胖症的关系:肠道微生物和大脑的相互作用可能是肥胖的后果或发病因素,肠-脑轴是它们相互作用的联络枢纽。#br# 肠-脑轴调控肥胖症发生的可能机制:肠道微生物可以通过肠道激素以及迷走神经连接(影响能量消耗和中枢神经系统中与饮食行为相关的区域)来影响肠-脑轴,从而改变宿主行为。同时,肠道微生物代谢物和其产物还可以充当信号分子并调节肠内分泌细胞的激素分泌,从而调节食欲、肠道运动、能量吸收和储存以及能量消耗等摄食相关行为,进而影响肥胖的发展。
文章探讨了以修复肠粘膜屏障损伤、改善肠道微生态失调为核心的复合肠道微生态制剂联合合理补充维生素、微量元素的改良低碳饮食通过“肠-脑轴”调控改善单纯性肥胖症患者体脂代谢及脂肪肝等代谢并发症的效果并初步分析了其可能的作用机制。对其作用机制的进一步研究将为“肠-脑轴”参与肥胖症的调控机制以及复合肠道微生态制剂防治单纯性肥胖症的临床应用提供思路和依据。
中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程#br#
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||