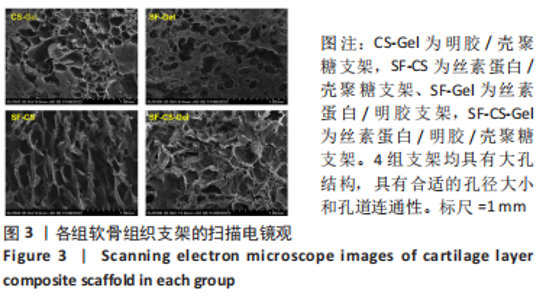
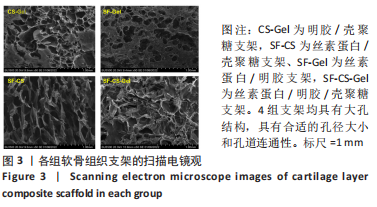
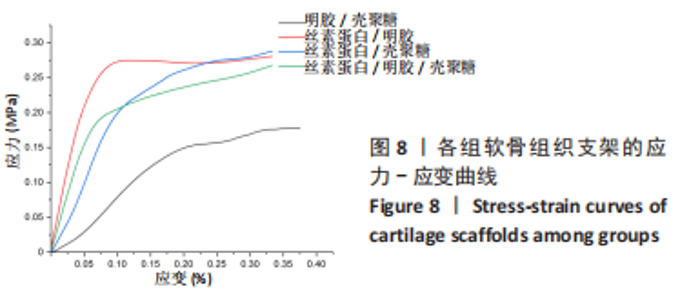
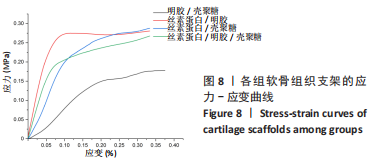
Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (3): 366-372.doi: 10.12307/2023.881
Previous Articles Next Articles
Preparation and in vitro evaluation of a three-dimensional porous cartilage scaffold made of silk fibroin/gelatin/chitosan
Gu Mingxi1, Wang Changcheng2, Tian Fengde2, An Ning2, Hao Ruihu2, Guo Lin2
- 1Shenzhen Hospital of Peking University, Shenzhen 518000, Guangdong Province, China; 2Second Department of Orthopedics, Zhongshan Hospital of Dalian University, Dalian 116001, Liaoning Province, China
-
Received:2022-04-19Accepted:2022-11-30Online:2024-01-28Published:2023-07-08 -
Contact:Guo Lin, Chief physician, Professor, MD, Master’s supervisor, Second Department of Orthopedics, Zhongshan Hospital of Dalian University, Dalian 116001, Liaoning Province, China -
About author:Gu Mingxi, Master physician, Shenzhen Hospital of Peking University, Shenzhen 518000, Guangdong Province, China
CLC Number:
Cite this article
Gu Mingxi, Wang Changcheng, Tian Fengde, An Ning, Hao Ruihu, Guo Lin. Preparation and in vitro evaluation of a three-dimensional porous cartilage scaffold made of silk fibroin/gelatin/chitosan[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 366-372.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
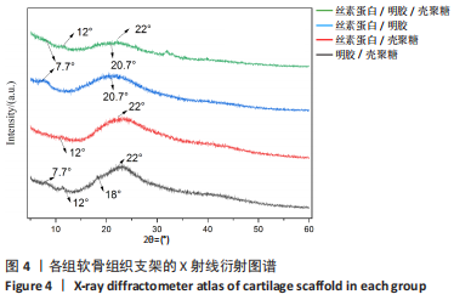
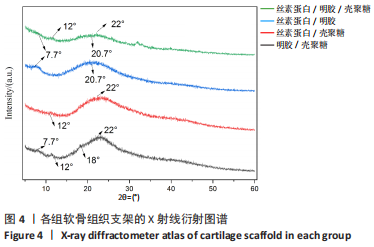
2.1.3 各组支架X射线衍射晶型分析 通过X射线衍射对各组支架进行了结构分析,4组支架的X射线衍射图谱如图4所示。明胶/壳聚糖支架的X射线衍射图谱的特征峰主要集中在2θ=7.7°,12°,18°和22°处,丝素蛋白/壳聚糖支架的X射线衍射图谱显示其特征峰集中在2θ=12°和22°。丝素蛋白/明胶支架的X射线衍射图谱显示其特征峰主要集中在2θ=7.7°和20.7°,丝素蛋白/明胶/壳聚糖支架在2θ=21°附近显示出一个宽大主峰,对应于壳聚糖、明胶和丝素蛋白的主反射的重叠。由于明胶与丝素蛋白及壳聚糖链的相互作用影响了其重组,明胶在2θ=7.7°时的低角反射特性已经消失,壳聚糖在2θ=18°时的低角反射峰消失,但是在2θ=12°时特征峰仍然存在。"
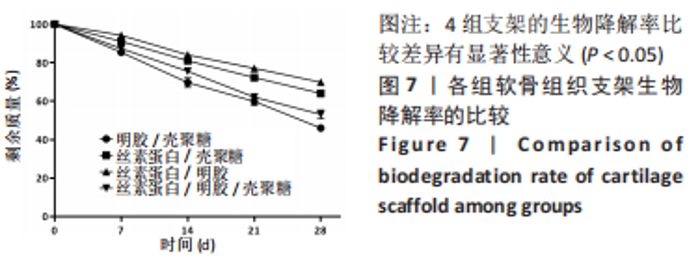
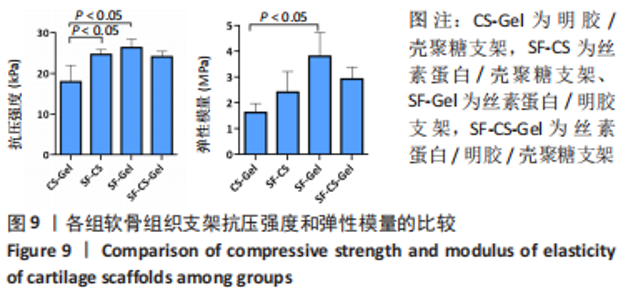
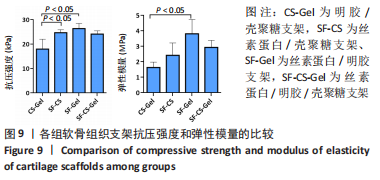
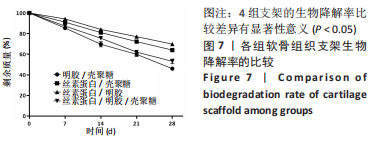
2.1.6 各组支架生物降解率 使用溶菌酶对4组支架进行了体外降解实验,如图7所示,4组支架的生物降解率比较差异有显著性意义(P < 0.05)。明胶/壳聚糖支架的降解率最高,为(53.96±2.62)%;丝素蛋白/壳聚糖支架和丝素蛋白/明胶/壳聚糖支架的降解率较低,分别为(30.15±2.71)%和(46.87±3.25)%;丝素蛋白/明胶支架的降解最为缓慢,降解率为(30.15±5.49)%。明胶/壳聚糖支架降解最快,在37 ℃环境下经过溶菌酶28 d培养后降解度可达50%以上,组织工程支架要求生物降解与软骨组织形成互相匹配才能用于修复软骨损伤,此次研究表明明胶/壳聚糖支架降解太快,明显不适合作为组织工程支架。"

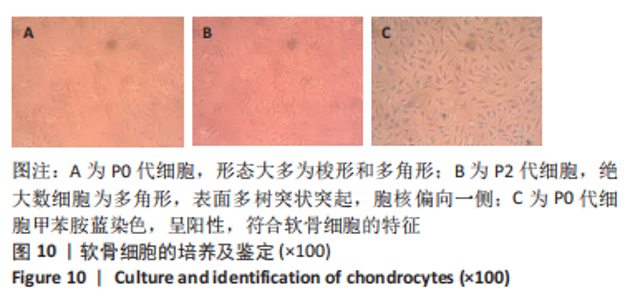
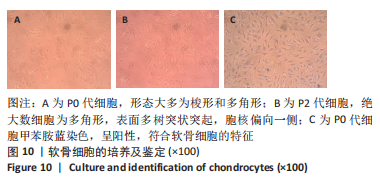
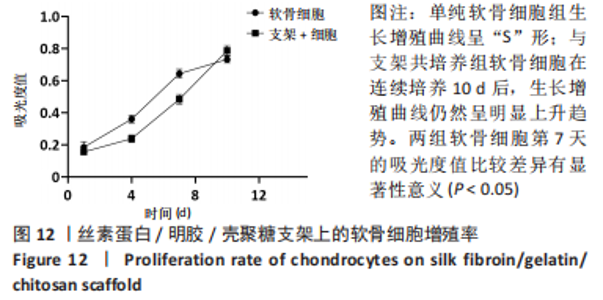
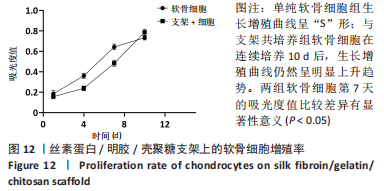
原代软骨细胞形态大多为梭形和多角形,细胞在贴壁2 d后开始很好地附着在细胞培养瓶表面并开始增殖,并在7-9 d后达到90%的汇合状态。当细胞生长达到80%-90%的汇合密度时,对细胞进行胰蛋白酶消化传代。传代细胞表现出相似的形态,绝大数细胞表现为多角形,表面多树突状突起,随着培养时间增加,树突状突起伸展为细长触丝。使用甲苯胺蓝对提取的原代细胞进行染色,细胞核被染成紫蓝色,胞核偏向一侧,未见肥大样细胞,符合软骨细胞的特征。 2.2.2 软骨组织支架上的细胞黏附率 理想的生物支架可以改善细胞黏附、细胞分化和与周围天然组织的整合[10]。此次实验观察了软骨细胞在丝素蛋白/明胶/壳聚糖支架上接种2,4,6 h后的黏附率,如图11所示,软骨细胞能与支架良好黏附,随着共培养时间的延长,细胞黏附率逐渐增大,4-6 h后达到最大,表明丝素蛋白/明胶/壳聚糖支架很好的保持材料优良的生物活性。"

| [1] THUNSIRI K, PITJAMIT S, POTHACHAROEN P, et al. The 3D-Printed Bilayer’s Bioactive-Biomaterials Scaffold for Full-Thickness Articular Cartilage Defects Treatment. Materials (Basel). 2020;13(15):3417. [2] HJELLE K, SOLHEIM E, STRAND T, et al. Articular Cartilage Defects in 1000 Knee Arthroscopies. Arthroscopy. 2002;18:730-734. [3] WEI W, DAI H. Articular cartilage and osteochondral tissue engineering techniques: Recent advances and challenges. Bioact Mater. 2021;6(12):4830-4855. [4] TSANAKTSIDOU E, KAMMONA O, KIPARISSIDES C. Recent Developments in Hyaluronic Acid-Based Hydrogels for Cartilage Tissue Engineering Applications. Polymers (Basel). 2022;14(4):839. [5] MEDVEDEVA EV, GREBENIK EA, GOMOSTAEVA SN, et al. Repair of Damaged Articular Cartilage: Current Approaches and Future Directions. Int J Mol Sci. 2018;19(8):2366. [6] DAOU F, COCHIS A, LEIGHEB M, et al. Current Advances in the Regeneration of Degenerated Articular Cartilage: A Literature Review on Tissue Engineering and Its Recent Clinical Translation. Materials (Basel). 2021;15(1):31. [7] ABPEIKAR Z, MORADI L, JAVDANI M, et al. Characterization of Macroporous Polycaprolactone/Silk Fibroin/Gelatin/Ascorbic Acid Composite Scaffolds and In Vivo Results in a Rabbit Model for Meniscus Cartilage Repair. Cartilage. 2021;13(2_suppl): 1583S-1601S. [8] MAHMOOD A, PATEL D, HICKSON B, et al. Recent Progress in Biopolymer-Based Hydrogel Materials for Biomedical Applications. Int J Mol Sci. 2022;23(3):1415. [9] ASADPOUR S, KARGOZAR S, MORADI L, et al. Natural biomacromolecule based composite scaffolds from silk fibroin, gelatin and chitosan toward tissue engineering applications. Int J Biol Macromol. 2020;154:1285-1294. [10] LI J, WANG Q, GU Y, et al. Production of Composite Scaffold Containing Silk Fibroin, Chitosan, and Gelatin for 3D Cell Culture and Bone Tissue Regeneration. Med Sci Monit. 2017;23:5311-5320. [11] GRABSKA-ZIELINSKA S, SIONKOWSKA A, CARVALHO Â, et al. Biomaterials with Potential Use in Bone Tissue Regeneration-Collagen/Chitosan/Silk Fibroin Scaffolds Cross-Linked by EDC/NHS. Materials (Basel). 2021;14(5):1105. [12] DOYEL S, SNOW F, DUCHI S, et al. 3D Printed Multiphasic Scaffolds for Osteochondral Repair: Challenges and Opportunities. Int J Mol Sci. 2021;22(22):12420. [13] ZHOU Y, LIU X, SHE H, et al. A silk fibroin/chitosan/nanohydroxyapatite biomimetic bone scaffold combined with autologous concentrated growth factor promotes the proliferation and osteogenic differentiation of BMSCs and repair of critical bone defects. Regen Ther. 2022;21:307-321. [14] LI Z, JIA S, XIONG Z, et al. 3D-printed scaffolds with calcified layer for osteochondral tissue engineering. J Biosci Bioeng. 2018;126(3):389-396. [15] YE P, YU B, DENG J, et al. Application of silk fibroin/chitosan/nano-hydroxyapatite composite scaffold in the repair of rabbit radial bone defect. Exp Ther Med. 2017;14(6): 5547-5553. [16] MAGLIO M, BROGINI S, PAGANI S, et al. Current Trends in the Evaluation of Osteochondral Lesion Treatments: Histology, Histomorphometry, and Biomechanics in Preclinical Models. Biomed Res Int. 2019;2019:4040236. [17] ROGINA A, RICO P, GALLEGO FERRER G, et al. In Situ Hydroxyapatite Content Affects the Cell Differentiation on Porous Chitosan/Hydroxyapatite Scaffolds. Ann Biomed Eng. 2016;44(4):1107-1119. [18] BOZORGI A, MOZAFARI M, KHAZAEL M, et al. Fabrication, characterization, and optimization of a novel copper-incorporated chitosan/gelatin-based scaffold for bone tissue engineering applications. Bioimpacts. 2022;12(3):233-246. [19] SHANG L, MA B, WANG F, et al. Nanotextured silk fibroin/hydroxyapatite biomimetic bilayer tough structure regulated osteogenic/chondrogenic differentiation of mesenchymal stem cells for osteochondral repair. Cell Prolif. 2020;53(11):e12917. [20] ZHANG Y, TU H, WU R, et al. Programing Performance of Silk Fibroin Superstrong Scaffolds by Mesoscopic Regulation among Hierarchical Structures. Biomacromolecules. 2020;21(10):4169-4179. [21] MELLATI A, FAN CM, TAMAYOL A, et al. Microengineered 3D cell-laden thermoresponsive hydrogels for mimicking cell morphology and orientation in cartilage tissue engineering. Biotechnol Bioeng. 2017;114(1):217-231. [22] HAFEZI M, NOURIi KHORASANI S, ZARE M, et al. Advanced Hydrogels for Cartilage Tissue Engineering: Recent Progress and Future Directions. Polymers (Basel). 2021; 13(23):4199. [23] RUAN SQ, YAN DENG J, HUANG WL, et al. Preparation of a biphase composite scaffold and its application in tissue engineering for femoral osteochondral defects in rabbits. Int Orthop. 2017;41(9):1899-1908. [24] SARTORI M, PAGANI S, FERRARI A, et al. A new bi-layered scaffold for osteochondral tissue regeneration: in vitro and in vivo preclinical investigations. Mater Sci Eng C. 2017; 70(1):101-111. [25] SUN K, LI H, LI R, et al. Silk fibroin/collagen and silk fibroin/chitosan blended three-dimensional scaffolds for tissue engineering. Eur J Orthop Surg Traumatol. 2015;25(2):243-249. [26] ZENG S, LIU L, SHI Y, et al. Characterization of Silk Fibroin/Chitosan 3D Porous Scaffold and In Vitro Cytology. PloS One. 2015;10(6):e0128658. [27] QI X, MOU N, ZHANG J, et al. Preparation of chitosan/silk fibroin/hydroxyapatite porous scaffold and its characteristics in comparison to bi-component scaffolds. J Biomed Mater Res A. 2014;102(2):366-372. [28] DENG J, SHE R, HUANG W, et al. A silk fibroin/chitosan scaffold in combination with bone marrow-derived mesenchymal stem cells to repair cartilage defects in the rabbit knee. J Mater Sci Mater Med. 2013;24(8):2037-2046. [29] FU N, DONG T, MENG A, et al. Research Progress of the Types and Preparation Techniques of Scaffold Materials in Cartilage Tissue Engineering. Curr Stem Cell Res Ther. 2018;13(7):583-590. |
| [1] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Effect of gelatin methacryloyl hydrogel loaded with salvianolic acid B on intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 380-386. |
| [2] | Wang Xinmin, Yan Wenkai, Song Yahui, Liu Fei. Leukocyte- and platelet-rich fibrin with autologous hamstring tendon for traumatic patella dislocation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 404-410. |
| [3] | Bi Yujie, Ma Dujun, Peng Liping, Zhou Ziqiong, Zhao Jing, Zhu Houjun, Zhong Qiuhui, Yang Yuxin. Strategy and significance of Chinese medicine combined with medical hydrogel for disease treatment [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 419-425. |
| [4] | Wang Xinyi, Xie Xianrui, Chen Yujie, Wang Xiaoyu, Xu Xiaoqing, Shen Yihong, Mo Xiumei. Electrospun nanofiber scaffolds for soft and hard tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 426-432. |
| [5] | Gao Xueyu, Zhang Wentao, Sun Tianze, Zhang Jing, Li Zhonghai. Application of metal ions in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 439-444. |
| [6] | Yang Jie, Hu Haolei, Li Shuo, Yue Wei, Xu Tao, Li Yi. Application of bio-inks for 3D printing in tissue repair and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 445-451. |
| [7] | Chen Pinrui, Pei Xibo, Xue Yiyuan. Function and advantages of magnetically responsive hydrogel in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 452-457. |
| [8] | Chen Junyan, Meng Qingqi, Li Siming. Cartilage targeting function in the drug delivery system by intra-articular injection for the treatment of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 458-463. |
| [9] | Long Zhirui, Huang Lei, Xiao Fang, Wang Lin, Wang Xiaobei. Characteristics of hydrogel microspheres in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 472-478. |
| [10] | Kong Xiangyu, Wang Xing, Pei Zhiwei, Chang Jiale, Li Siqin, Hao Ting, He Wanxiong, Zhang Baoxin, Jia Yanfei. Biological scaffold materials and printing technology for repairing bone defects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 479-485. |
| [11] | Xu Jing, Lyu Huixin, Bao Xin, Zhang Yi, Wang Yihan, Zhou Yanmin. Application of near infrared responsive hydrogels in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 486-492. |
| [12] | Fan Yongjing, Wang Shu, Jin Wulong. Characteristics, advantages and application of osteogenic differentiation of jaw bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 100-106. |
| [13] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [14] | Huang Linke, Wei Linhua, Jiang Jie, Liu Qian, Chen Weiwei. Effects of estrogen combined with treadmill exercise on bone mass and articular cartilage in ovariectomized mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1166-1171. |
| [15] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||