Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (20): 3219-3225.doi: 10.3969/j.issn.2095-4344.3210
Previous Articles Next Articles
Irisin mediates protective effects on failing heart via integrin alpha V receptors
Wang Jing, Wu Jiangbo
- Tianjin University of Sport, Tianjin 301617, China
-
Received:2020-08-04Revised:2020-08-07Accepted:2020-09-26Online:2021-07-18Published:2021-01-15 -
Contact:Wu Jiangbo, MD candidate, Assistant researcher, Tianjin University of Sport, Tianjin 301617, China -
About author:Wang Jing, Master, Experimentalist, Tianjin University of Sport, Tianjin 301617, China -
Supported by:the Youth Project of Tianjin Natural Science Foundation, No. 18JCQNJC12400 (to WJB), the Third Level of “131” Innovative Talents Training Project in Tianjin (to WJB)
CLC Number:
Cite this article
Wang Jing, Wu Jiangbo. Irisin mediates protective effects on failing heart via integrin alpha V receptors[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3219-3225.
share this article
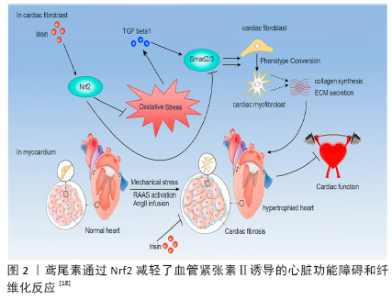
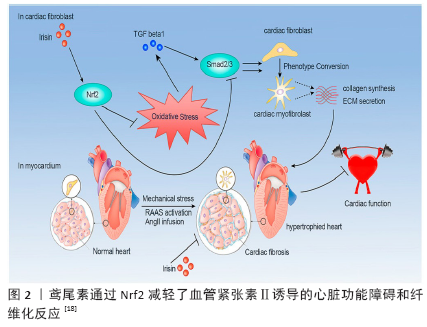
2.1 鸢尾素的产生及其心肌保护效应 鸢尾素可由骨骼肌释放,尤其在运动之后。运动会提高活性氧(reactive oxygen species,ROS)水平,继而活性氧可以激活p38丝裂原活化蛋白激酶(p38 mitogen-activated protein kinase,p38MAPK),而p38MAPK又进一步调控过氧化物酶体增殖物激活受体γ激活因子1α(peroxisome proliferator-activated receptor gamma co-activator 1-alpha,PGC-1α)。PGC-1α上调了FNDC5表达,使鸢尾素分泌增加[10]。活性氧→p38MAPK→PGC-1α→FNDC5/Irisin途径是运动产生鸢尾素的分子机制。 2.1.1 鸢尾素的特异性受体 BOSTR?M等[1]推测鸢尾素受体是一种细胞膜受体,因为鸢尾素在体内可形成二聚体,而二聚体形式有利于鸢尾素作为配体与其受体结合并激活受体。XIE等[11]也证实了H9C2心肌细胞膜上存在鸢尾素的特异性受体,然而此时其受体尚未确定。直到2018-12-13,KIM等[12]发现了整合素αV是骨细胞和脂肪组织中的鸢尾素受体,鸢尾素能作为一种信号在肌肉和骨骼组织之间传递,启动新骨细胞替换旧的或受损骨细胞的过程。 2.1.2 鸢尾素的心肌保护作用机制 鸢尾素对维持正常的心功能有重要作用,鸢尾素诱导的心脏保护与线粒体功能改善有关。XIE等[11]实验证实鸢尾素会抑制H9C2细胞增殖,但促进了其生长。此外,鸢尾素会激活PI3K-AKT信号通路。WANG等[13]发现鸢尾素可通过降低乳酸脱氢酶漏出和心肌细胞凋亡减轻缺氧再复氧的损害。 (1)鸢尾素抑制心肌活性氧生成:鸢尾素可以抑制心肌氧化应激,且这种作用可能是由UCP2、锰超氧化物歧化酶和Nrf2介导的。 UCP2是解偶联蛋白家族的重要一员,广泛存在于骨骼肌、心肌、胎盘、肝脏、胰腺、肾脏、肺、胃和小肠等组织。UCP2的主要生理功能是氧化还原调节,减少氧自由基产生和免疫反应[14]。线粒体解偶联蛋白表达增加可能是通过“解偶联生存”减少活性氧的代偿机制,即线粒体膜电位降低可降低线粒体释放的活性氧。MCLEOD等[15]发现,心肌缺血预处理诱导UCP2 mRNA表达升高,减轻了氧自由基损伤,增强了心肌线粒体对缺氧再复氧的耐受能力,从而减轻了心肌缺血再灌注损伤。MCLEOD等[16]进一步研究发现,UCP2基因敲除的心肌细胞对缺氧再复氧耐受能力降低了,这进一步证实了UCP2在心肌缺血中的保护作用。在应激状态下,鸢尾素可以直接作用于UCP2,参与调节线粒体功能,调节活性氧和能量代谢。CHEN等[3]研究发现,经过缺血再灌注处理的小鼠肺中UCP2表达下降,而外源性鸢尾素能部分恢复UCP2表达。鸢尾素可与UCP2发生物理性结合进而有效防止UCP2降解,这可能与鸢尾素减轻氧化应激和改善线粒体功能有关。 锰超氧化物歧化酶在维持活性氧水平中起重要作用,但它降低活性氧产生的能量成本较高,因为其依赖于ATP和NADPH。鸢尾素在心肌缺血再灌注中对心肌有保护作用,其主要机制可能是鸢尾素能靶向性地到达受损细胞的线粒体,并与锰超氧化物歧化酶共定位,抑制锰超氧化物歧化酶降解,减少活性氧生成[4]。BOSS等[17]发现4周跑台有氧训练后大鼠心肌组织中UCP2 mRNA下降,有氧运动下调了UCP2表达,作用机制可能是有氧运动提高了机体的抗氧化酶活性,使活性氧生成减少,UCP2表达降低。 此外,CHEN等[18]研究证实鸢尾素可以通过Nrf2抑制ROS /TGFβ1/ Smad2 /3信号轴减轻血管紧张素Ⅱ诱导的心脏纤维化(图2)。 (2)鸢尾素改善心肌能量代谢:生理条件下,UCP2正常表达是保证心肌能量供应和维持心脏正常功能的重要保障。UCP2参与游离脂肪酸转运,心力衰竭和运动可能会通过调控UCP2的表达影响心肌能量代谢[19]。李楠等[20]发现,心力衰竭大鼠血清游离脂肪酸水平升高的同时UCP2表达增加,且UCP2表达量与血清游离脂肪酸浓度存在显著正相关。 耐力运动诱导的鸢尾素可能通过提高心肌抗氧化酶活性减少活性氧生成,继而下调UCP2表达[17]。BO等[21]发现未训练大鼠进行一次性亚极量急性运动中,在45 min时心肌UCP2 mRNA达到峰值;在急性运动恢复期,UCP2 mRNA在 3 h后显著升高;耐力训练后UCP2 mRNA较训练前显著下降。有氧运动可以通过鸢尾素改善机体的氧化应激,提高机体抗氧化酶活性,减少了活性氧产生,从而下调运动相关的心肌UCP2表达。UCP2表达下降抑制了游离脂肪酸向线粒体内转运,从而降低了游离脂肪酸对心肌线粒体的毒性作用,有利于心力衰竭康复[19]。 "
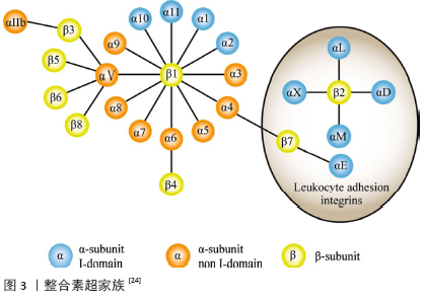
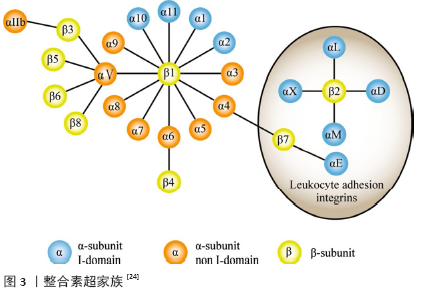
2.2 整合素的结构与功能 2.2.1 整合素结构与类别 整合素是一类跨膜蛋白,它们由以不同结构域为特征的长胞外段以及短的跨膜和胞质段构成。迄今为止,整合素是由α亚基(120-180 kD)和β亚基(90-110 kD)以非共价键连接形成的一个异二聚体跨膜受体家族,每一个都包括了一个大的胞外区、一个单一的跨膜区和通常有一个短的胞质尾。 α和β两个亚单位均有多种,αV只是α亚单位中一种,它可以和不同β亚单位构成不同的整合素。脊椎动物表达24种不同的整合素,包括18种不同的α亚基和8种不同的β亚基[22]。β亚基将整合素靶向细胞外基质细胞黏附位点,而α亚基决定了配体与各种细胞外基质蛋白(例如纤连蛋白,层黏连蛋白和胶原蛋白)结合的特异性[23]。特定的α和β胞质域选择性加工进一步增加了整合素功能多样性。如图3所示,多个α亚基可以与单个β亚基结合从而产生组合的配体专一性,反之亦然。 "
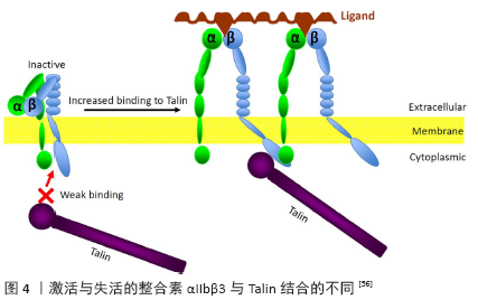
2.2.2 整合素的功能 整合素是一组细胞表面蛋白,在不同的细胞中有多种作用。整合素在与其他特定蛋白质接触时被激活,它们和其他与蛋白质结合的分子一样被统称为“配体”。整合素介导的黏附和信号传导活动实际上调节着所有细胞的生长和分化,而整合素的失调则与癌症的发病机制、自身免疫状况和血管血栓生成有关。整合素异二聚体识别其他细胞、细胞外基质或病原体上重叠的不同配体[24]。大多数整合素以不结合配体或信号的非激活状态存在于细胞表面。整合素被通过内-外信号过程的胞内刺激和由通过外-内信号把细胞外信号转导到细胞质的配体占有率激活[25]。 2.3 整合素在心肌的表达与功能 心肌细胞之间以及其与细胞外基质之间的主要锚定蛋白是心肌细胞膜中的整合素,他们对维持心肌组织的结构和心脏内的机械传导非常重要。心肌细胞中主要表达α1、α3、α5、α6、α7、α9、α10、αV和β1、β3、β5等整合素[26]。整合素在心肌的所有细胞区室中都有表达,它们对其形态和功能至关重要,且它们在调节细胞过程中是必不可少的[27]。完整的整合素功能与抗凋亡信号和细胞存活[28]、梗死后细胞迁移和心肌修复的诱导[29]、上皮-间质转化的激活和再生以及整个细胞周期中心肌细胞的正常过程有关[30-31]。整个心肌细胞周期的进展可能取决于通过整合素β1的细胞附着,并且与β1剪接变异体及其各自α亚型的变化相关[31]。不同整合素亚型表达和激活在心肌肥大和重构的不同阶段发挥作用[32]。 心衰患者安装左心室辅助装置可引起心肌重构逆转,某些整合素的mRNA表达发生了显著变化[33]。血小板整合素αIIbβ3在血栓形成和止血过程中起重要作用,整合素αIIbβ3脱落可用于改善连续流左心室辅助装置患者的出血风险[34]。整合素αVβ3在静止的内皮细胞表达水平低,而在梗死后心肌血管生成状态下显著上调[35-36],而血管生成是缺血性损伤后修复过程中必不可少的部分[37]。对雄性大鼠进行短暂的左冠状动脉阻断20 min后再灌注处理,结果发现心肌梗死后整合素αVβ3表达上调,在1-3周达到峰值,直至再灌注后6个月仍可检测到[36]。临床前和临床研究表明,活化的心肌成纤维细胞和巨噬细胞在边缘化和趋化性过程中均表达整合素αVβ3。整合素αVβ3受体是心脏修复的关键介质和决定因素,心肌整合素αVβ3是一个持续心脏修复的标志物。18F-Fluciclatide是一种新型的αVβ3选择性放射性示踪剂,它可以作为心脏修复和预测恢复区域的生物标记物,且18F-Fluciclatide在新的心肌梗死的部位摄取增加[38]。 此外,整合素β3也在心肌内表达且发挥重要作用。盐皮质激素受体拮抗剂螺内酯通过盐皮质激素受体增加了大鼠心肌细胞整合素β3的表达,从而降低了心力衰竭的死亡率[39]。 2.4 心肌中整合素相关信号通路 整合素是一类异二聚体跨膜受体,它们在包括心肌细胞的所有细胞中均有表达。它们参与多种关键的细胞过程,包括黏附、细胞外基质组织、信号传导、存活和增殖。整合素和收缩的肌细胞密切相关,它们是机械转化器将机械信息转化为生化信息。整合素是与细胞外基质结合的重要黏附性跨膜受体,它们的激活影响了细胞骨架重构和其他细胞内信号途径[40]。整合素信号和整合素与细胞骨架之间的联系是由许多蛋白介导的,包括整合素连接激酶(integrin-linked kinase,ILK)、踝蛋白(talin,TLN)和黏着斑激酶(focal adhesion kinase,FAK)[41]。 2.4.1 整合素连接激酶 整合素连接激酶是一种连接整合素与细胞内细胞骨架和激酶信号级联的接头蛋白和丝氨酸-苏氨酸激酶。整合素连接激酶在哺乳动物组织中广泛表达,它最初被鉴定为整合素β1胞尾的结合伴侣[42-43]。整合素连接激酶可被整合素或生长因子以依赖于PI3K的方式激活,继而作用于下游PKB(protein kinase B)/AKT和GSK-3(glycogen synthase-3),调控细胞的生长、分化、迁移等[44]。整合素连接激酶还与连接蛋白Parvin、Pinch和Paxillin(Pax)结合,从而提供一个整合素信号平台[45]。 整合素连接激酶在转导细胞-基质相互作用诱导的生物力学信号中发挥着重要作用,这些信号调节了细胞骨架重构、血管生成、细胞生长、增殖、存活和分化。果蝇中的整合素连接激酶和整合素β1共定位于心肌细胞连接处和Z带附近,它们二者功能下降抑制了野生果蝇心脏老化的典型性变化,例如心律失常。老年野生果蝇的整合素β1蛋白水平高于年轻果蝇,且年轻果蝇整合素β1的心脏特异性过表达导致了老年化心脏功能障碍。此外,整合素/整合素连接激酶途径相关基因的心脏特异性中度敲低抑制了随着年龄下降的心功能[46]。心肌细胞特异性敲除整合素连接激酶导致了以心肌细胞死亡、纤维化和炎症为特征的致死性心肌病。心肌和骨骼肌整合素连接激酶敲除小鼠发展为伴随着细胞结构和纤维化被破坏的心力衰竭,同时黏着斑激酶和AKT磷酸化降低[47]。靶向敲除整合素连接激酶可引起6周龄小鼠原发性心肌病和心力衰竭。整合素连接激酶敲除导致心肌细胞分解,这与整合素β1/黏着斑激酶复合物黏附信号的破坏有关[48]。整合素连接激酶可以减轻大鼠和猪心肌梗死后的心室重构,改善心功能[49]。 整合素连接激酶是心肌牵张传感器的重要组成部分,它调节心肌收缩、代偿性肥大、存活和修复。整合素连接激酶在保护哺乳动物心脏免受心肌病和衰竭方面起着重要作用,整合素连接激酶治疗可能是治疗心力衰竭的一种新方法。 2.4.2 踝蛋白 踝蛋白(270 kD)是一种二聚的黏着斑/肋状蛋白,它结合整合素β亚基的胞质域并激活整合素以调节细胞外基质结合。踝蛋白通过直接连接肌动蛋白和通过其与纽蛋白的结合间接地将整合素连接到细胞骨架[50]。踝蛋白对于整合素与细胞外基质的正确黏附是必需的,其缺失或下调会损害黏着斑的形成[51]。对非肌肉细胞的研究表明,踝蛋白是力传递和传导的关键调节因子,是心肌的一个特别重要的特性,心肌是一个在基础状态下持续承受机械力的器官,这必须适应生理应激或病理条件下的进一步机械变化[52-53]。踝蛋白是使心肌细胞膜上整合素β1D稳定表达和维持适当心肌细胞完整性所必需的。踝蛋白的正常表达对于在心肌细胞中维持整合素黏附复合物及其伴随蛋白,以及最终对于正常心肌细胞和整个心脏的功能是必要的。踝蛋白1和踝蛋白 2的同时敲除使心肌不稳定,导致心力衰竭[54]。 整合素是由α和β亚单位组成的Ⅰ型跨膜结构域蛋白,每个亚单位都有一个大的胞外结构域、一个单通道跨膜结构域和一个短的胞质尾[55]。细胞质蛋白踝蛋白参与了由内向外和由外向内的信号传递[56]。踝蛋白与整合素胞质尾结合并引起整合素β Ⅰ型跨膜结构域的倾斜角改变,这使α和βⅠ型跨膜结构域相互作用不稳定并激活整合素[57]。踝蛋白结合也与α细胞质尾部的膜近端区域发生空间冲突,并进一步干扰α和β Ⅰ型跨膜结构域堆积[58]。踝蛋白与整合素的结合是整合素双向信号转导中的重要事件,其结合至少受两种机制的调控:通过踝蛋白对整合素尾部的募集和踝蛋白与整合素之间亲和力的调节。踝蛋白与整合素的结合通过踝蛋白的募集、靶向和构象变化在时间和空间上受到其他蛋白质的调节。 整合素在失活时与踝蛋白的结合较弱,但在激活时与踝蛋白的结合明显增强。无论αIIbβ3如何被激活,失活的αIIbβ3一旦被激活都会显著增加其与踝蛋白的结合。此外,与踝蛋白的结合增加是β3尾依赖性的:在失活的整合素中,αIIb和β3胞质尾相互作用,从而降低β3尾与踝蛋白之间的亲和力,在激活的整合素中,αIIb和β3尾是分开的,使得β3尾可以自由接近踝蛋白,并在αIIbβ3和踝蛋白之间提供更强的相互作用(图4)[56]。 Kindlin和踝蛋白是整合素激活过程中的整合素结合蛋白。尽管踝蛋白可以诱导整联蛋白的构象变化,但它本身不足以激活整合素,还需要Kindlin的作用。Kindlin协同踝蛋白通过增强踝蛋白与整合素β3膜近端区域的相互作用促进整合素激活,而且Kindlin修饰了在整合素aIIbβ3尾部由内而外信号传导的分子机制[59]。 2.4.3 黏着斑激酶 黏着斑激酶是一种胞质非受体酪氨酸激酶,为整合素信号通路中的关键分子,又与细胞黏附密切相关,在细胞迁移、黏附和浸润等方面起着至关重要的作用。黏着斑激酶可以将细胞外的信号经整合素受体传导至细胞内。αVβ3和αVβ5通过激活Akt激活了生存信号通路[60-62],而AKT在保护小鼠心肌细胞免受缺血再灌注损伤中起着重要作用[63]。当整合素被激活后,黏着斑激酶作为接头蛋白,将PI3K通过其p85停泊。p85亚基的SH2和SH3结构域均在一个磷酸化位点与接头蛋白结合。PI3K募集到活化的受体后,起始多种PI中间体的磷酸化,PI3K转化PIP2为PIP3。PI3K激活的结果是在质膜上产生第二信使PIP3,它与细胞内的AKT和3-磷酸肌醇依赖性蛋白激酶1结合,促使磷酸肌醇依赖性蛋白激酶1磷酸化AKT蛋白的Ser308导致AKT活化。AKT是PI3K下游主要的效应物,它通过下游多种途径对靶蛋白进行磷酸化而发挥作用。在脂肪和骨细胞中发现了一组称为整合素αV的鸢尾素受体,继而用10 nmol/L的鸢尾素对MLO-Y4细胞进行处理,引起了黏着斑激酶(Tyr397)和AKT (Ser308)的磷酸化[12]。此外,H9C2细胞中整合素αVβ3和/或αVβ5激活后可通过下游FAK/AKT通路抑制缺氧诱导的心肌细胞凋亡[6]。 胡明珠等[64]研究发现PI3K/AKT/SIRT1信号通路参与了硫化氢(H2S)后处理对大鼠缺血心肌的保护作用,且H2S后处理的心肌保护作用与Sirt1/PGC-1α的激活有关[65]。用siRNA干扰人脐静脉内皮细胞内SIRT1蛋白的表达,干扰48 h后发现锰超氧化物歧化酶蛋白水平降低。染色质免疫共沉淀发现SIRT1结合在锰超氧化物歧化酶启动子上,过表达SIRT1可以上调锰超氧化物歧化酶启动子活性,且呈现剂量-效应关系[66]。SIRT1的小分子激活剂Comp5通过SIRT1-PINK1-Parkin通路诱导了胶质母细胞瘤细胞的线粒体自噬[67]。此外,耐力运动训练还可以激活大鼠心脏SIRT1信号通路,上调SIRT1的表达,调节FoxO1的去乙酰化水平,抑制心肌细胞凋亡,从而发挥心肌保护作用[68]。 综上所述,整合素αV是鸢尾素的特异性受体,鸢尾素可使其表达增加。整合素αV可通过激活整合素相关蛋白激酶(整合素连接激酶和/或黏着斑激酶)继而激活下游的AKT/SIRT1通路,从而引起了包括线粒体氧化应激、生物合成和自噬等线粒体稳态的改变,从而发挥心肌保护作用。 "
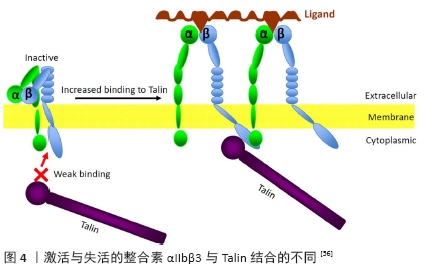
| [1] BOSTRÖM P, WU J, JEDRYCHOWSKI MP, et al. A PGC1α-dependent myokine that drives browning of white fat and thermogenesis. Nature. 2012;481(7382):463-468. [2] MARIA AS, NAGABABU C, TOMOO O, et al. The Structure of Irisin Reveals a Novel Intersubunit-Sheet Fibronectin Type III (FNIII) Dimer. J Biol Chem. 2013;288(47):33738-33744. [3] CHEN K, XU Z, LIU Y, et al. Irisin protects mitochondria function during pulmonary ischemia/reperfusion injury. Sci Transl Med. 2017; 9(418):eaao6298. [4] 王震. 不同药物干预心、肾缺血再灌注损伤的作用与机制研究[D]. 重庆: 第三军医大学,2015. [5] 吕勃. 整合素αV对内皮祖细胞治疗心肌缺血的影响[D]. 哈尔滨: 哈尔滨医科大学,2010. [6] KANAZAWA H, IMOTO K, OKADA M, et al. Canstatin inhibits hypoxia-induced apoptosis through activation of integrin/focal adhesion kinase/Akt signaling pathway in H9C2 cardiomyoblasts. Plos One. 2017;12(2):e0173051. [7] MEOLI DF, SADEGHI MM, KRASSILNIKOVA S, et al. Noninvasive imaging of myocardial angiogenesis following experimental myocardial infarction. J Clin Invest. 2004;113(12):1684-1691. [8] SARRAZY V, KOEHLER A, CHOW ML, et al. Integrins αvβ5 and αvβ3 promote latent TGF-β1 activation by human cardiac fibroblast contraction. Cardiovasc Res. 2014;102(3):407-417. [9] GRÖNMAN M, TARKIA M, KIVINIEMI T, et al. Imaging of αvβ3 integrin expression in experimental myocardial ischemia with [68Ga]NODAGA-RGD positron emission tomography. J Transl Med. 2017;15(1):144. [10] FABIAN SG, CARME PQ. The p38–PGC-1α–irisin–betatrophin axis: Exploring new pathways in insulin resistance. Adipocyte. 2013;3(1):67-68. [11] XIE C, ZHANG Y, TRAN TDN, et al. Irisin Controls Growth, Intracellular Ca2+ Signals, and Mitochondrial Thermogenesis in Cardiomyoblasts. Plos One. 2015;10(8):e0136816. [12] KIM H, WRANN CD, JEDRYCHOWSKI M, et al. Irisin Mediates Effects on Bone and Fat via αV Integrin Receptors. Cell. 2018;175(7):1756-1768. [13] Wang H, Zhao YT, Zhang S, et al. Irisin plays a pivotal role to protect the heart against ischemia and reperfusion injury. J Cell Physiol. 2017; 232(12):3775-3785. [14] Arsenijevic D, Onuma H, Pecqueur C, et al. Disruption of the uncoupling protein-2 gene in mice reveals a role in immunity and reactive oxygen species production. Nat Genet. 2000;26(4):435-439. [15] MCLEOD CJ, HOYT RF, SACK MN. UCP2: A functional target in delayed preconditioning induced cardioprotection. Cardiovasc J Afr. 2004;15(4supp1):S4-S6. [16] MCLEOD CJ, AZIZ A, HOYT RF JR, et al. Uncoupling proteins 2 and 3 function in concert to augment tolerance to cardiac ischemia. J Biol Chem. 2005;280(39):33470-33476. [17] BOSS O, SAMEC S, DESPLANCHES D, et al. Effect of endurance training on mRNA expression of uncoupling protein 1, 2, and 3 in rat. Faseb J. 1998;12(3):335-339. [18] CHEN RR, FAN XH, CHEN G, et al. Irisin attenuates angiotensin II-induced cardiac fibrosis via Nrf2 mediated inhibition of ROS/TGFβ1/Smad2/3 signaling axis. Chem Biol Interact. 2019;302:11-21. [19] 毋江波, 谢安青, 张勇. 有氧运动通过鸢尾素改善慢性心力衰竭的机制[J]. 中国组织工程研究,2019,23(11):1781-1787. [20] 李楠, 王江, 高峰, 等. 心力衰竭大鼠解偶联蛋白2表达及对线粒体功能的影响[J]. 岭南心血管病杂志,2009,15(6):474-478. [21] BO H, JIANG N, MA GD, et al. Regulation of mitochondrial uncoupling respiration during exercise in rat heart: Role of reactive oxygen species(ROS)and uncoupling protein 2. Free Radic Biol Med. 2008; 44(7):1373-1381. [22] HYNES RO. Integrins: Bidirectional, Allosteric Signaling Machines. Cell. 2002;110(6):673-687. [23] HAYASHI Y, HAIMOVICH B, RESZKA A, et al. Expression and function of chicken integrin beta 1 subunit and its cytoplasmic domain mutants in mouse NIH 3T3 cells. J Cell Biol. 1990;110(1):175-182. [24] TIAN J, ZHANG FJ, LEI GH. Role of integrins and their ligands in osteoarthritic cartilage. Rheumatol Int. 2015;35(5):787-798. [25] LUO BH, CARMAN CV, SPRINGER TA. Structural Basis of Integrin Regulation and Signaling. Annu Rev Immunol. 2007;25(1):619-647. [26] MARA B, EMILIO H, ANTONELLA N, et al. Integrin signalling: the tug-of-war in heart hypertrophy. Cardiovasc Res. 2006;70(3):422-433. [27] ROSS RS, BORG TK. Integrins and the myocardium. Circ Res. 2001; 88(11):1112-1119. [28] PFISTER R, ACKSTEINER C, BAUMGARTH J, et al. Loss of beta 1D-integrin function in human ischemic cardiomyopathy. Basic Res Cardiol. 2007; 102(3):257-264. [29] HANNIGAN GE, COLES JG, DEDHAR S. Intergin-linked kinase at the heart of cardiac contractility, repair and disease. Circ Res. 2007;100(10): 1408-1414. [30] CASTALDO C, DI MEGLIO F, NURZYNSKA D, et al. CD117-Positive Cells in Adult Human Heart Are Localized in the Subepicardium, and Their Activation Is Associated with Laminin-1 and α6 Integrin Expression. Stem Cells. 2008;26(7):1723-1731. [31] MAITRA N, FLINK IL,BAHL JJ, et al. Expression of alpha and beta integrins during terminal differentiation of cardiomyocytes. Cardiovasc Res. 2000;47(4):715-725. [32] KUZMAN J, KACIMI R, GERDES AM. 14 Integrin alterations during LV myocyte remodeling in spontaneously hypertensive heart failure (SHHF) rats. J Mol Cell Cardiol. 2002;34(7):A14. [33] DULLENS HF, SCHIPPER ME, VAN KUIK J, et al. Integrin expression during reverse remodeling in the myocardium of heart failure patients. Cardiovasc Pathol. 2012;21(4):291-298. [34] MONDAL NK, CHEN Z, TRIVEDI JR, et al. Oxidative stress induced modulation of platelet integrin αIIbβ3 expression and shedding may predict the risk of major bleeding in heart failure patients supported by continuous flow left ventricular assist devices. Thromb Res. 2017; 158:140-148. [35] MEOLI DF, SADEGHI MM, KRASSILNIKOVA S, et al. Noninvasive imaging of myocardial angiogenesis following experimental myocardial infarction. J Clin Invest. 2004;113(12):1684-1691. [36] HIGUCHI T, BENGEL FM, SEIDL S, et al. Assessment of alphavbeta3 integrin expression after myocardial infarction by positron emission tomography. Cardiovasc Res. 2008;78(2):395- 403. [37] KHURANA R, SIMONS PM, MARTIN JF, et al. Role of Angiogenesis in Cardiovascular Disease: a critical appraisal. Circulation. 2005;112(12): 1813-1824. [38] JENKINS WSA, STIRRAT C, CONNELL M, et al. Cardiac αVβ3 integrin expression following acute myocardial infarction in humans. Heart. 2017;103(8):607-615. [39] CHUN TY, BLOEM L, PRATT JH. Spironolactone increases integrin β3 gene expression in kidney and heart muscle cells. Mol Cell Endocrinol. 2002;194(1):175-182. [40] DELON I, BROWN NH. Integrins and the actin cytoskeleton. Curr Opin Cell Biol. 2007;19(1):43-50. [41] GEIGER B, BERSHADSKY A, PANKOV R, et al. Transmembrane crosstalk between the extracellular matrix-cytoskeleton crosstalk. Nat Rev Mol Cell Biol. 2001;2(11):793-805. [42] HANNIGAN GE, FITZGIBBON L, COPPOLINO MG, et al. Regulation of cell adhesion and anchorage-dependent growth by a new beta1-integrin-linked protein kinase. Nature. 1996;379(6560):91-96. [43] HANNIGAN GE, COLES JG, DEDHAR S. Integrin-linked kinase at the heart of cardiac contractility, repair, and disease. Circ Res. 2007;100(10): 1408-1414. [44] 郑利民,方明镜.整合素连接激酶(ILK)参与肿瘤的形成和转移的研究进展[J].医学研究杂志,2005,34(12):60-61. [45] LEGATE KR, MONTAÑEZ E, KUDLACEK O, et al. ILK, PINCH and parvin: the tIPP of integrin signaling. Nat Rev Mol Cell Biol. 2006;7(1):20-31. [46] NISHIMURA M, KUMSTA C, KAUSHIK G, et al. A dual role for integrin-linked kinase and β1-integrin in modulating cardiac aging. Aging Cell. 2014;13(3):431-440. [47] DAI J, MATSUI T, ABEL ED, et al. Deep sequence analysis of gene expression identifies osteopontin as a downstream effector of integrin-linked kinase (ILK) in cardiac-specific ILK knockout mice. Circ Heart Fail. 2014;7(1):184-193. [48] WHITE DE, COUTU P, SHI YF, et al. Targeted ablation of ILK from the murine heart results in dilated cardiomyopathy and spontaneous heart failure.Gene Dev. 2006;20(17):2355-2360. [49] LU W, LU W, XU B. Increased expression of integrin-linked kinase improves cardiac function after myocardial infarction in the swine. Heart. 2010;96(Suppl 3):A133-A134. [50] CALDERWOOD DA, CAMPBELL ID, CRITCHLEY DR. Talins and kindlins: Partners in integrin-mediated adhesion. Nat Rev Mol Cell Biol. 2013; 14(8):503-517. [51] ZHANG X, JIANG G, CAI Y, et al. Talin depletion reveals independence of initial cell spreading from integrin activation and traction. Nat Cell Biol. 2008;10(9):1062-1068. [52] ELOSEGUI-ARTOLA A, ORIA R, CHEN Y, et al. Mechanical regulation of a molecular clutch defines force transmission and transduction in response to matrix rigidity. Nat Cell Biol. 2016;18(5):540-548. [53] AUSTEN K, RINGER P, MEHLICH A, et al. Extracellular rigidity sensing by talin isoform-specific mechanical linkages. Nat Cell Biol. 2015;17(12): 1597-1606. [54] MANSO AM, OKADA H, SAKAMOTO FM, et al. Loss of mouse cardiomyocyte talin-1 and talin-2 leads to β-1 integrin reduction, costameric instability, and dilated cardiomyopathy. Proc Natl Acad Sci USA. 2017;114(30):E6250-E6259. [55] SHIMAOKA M, TAKAGI J, SPRINGER TA. Conformational Regulation of Integrin Structure and Function. Annu Rev Biophys Biomol Struct. 2002;31(1):485-516. [56] WANG D, GUO Q, WEI A, et al. Differential Binding of Active and Inactive Integrin to Talin. Protein J. 2018;37(3):280-289. [57] KIM C, YE F, HU X, et al. Talin activates integrins by altering the topology of the beta transmembrane domain. J Cell Biol. 2012;197(5):605-611. [58] LI A, GUO Q, KIM C, et al. Integrin alphaII b tail distal of GFFKR participates in inside-out alphaII b beta3 activation. J Thromb Haemost. 2014;12(7):1145-1155. [59] HAYDARI Z, SHAMS H, JAHED Z, et al. Kindlin Cooperates with Talin for Integrin Activation, a Molecular Dynamics Approach. Biophys J. 2020; 118(3):504a. [60] HWANG S, LEE HJ, KIM G, et al. CCN1 acutely increases nitric oxide production via integrin alphavbeta3-Akt-S6K-phosphorylation of endothelial nitric oxide synthase at the serine 1177 signaling axis. Free Radic Biol Med. 2015;89:229-240. [61] RIOPEL M, STUART W, WANG R. Fibrin improves beta (INS-1) cell function, proliferation and survival through integrin alphavbeta3. Acta Biomater. 2013;9(9):8140-8148. [62] LANE D, GONCHARENKO-KHAIDER N, RANCOURT C, et al. Ovarian cancer ascites protects from TRAILinduced cell death through alphavbeta5 integrin-mediated focal adhesion kinase and Akt activation. Oncogene. 2010;29(24):3519-3531. [63] FUJIO Y, NGUYEN T, WENCKER D, et al. Akt promotes survival of cardiomyocytes in vitro and protects against ischemia-reperfusion injury in mouse heart. Circulation. 2000;101(6):660-667. [64] 胡明珠, 周波, 盛琼, 等. PI3K/Akt/Sirt1信号通路介导硫化氢后处理对大鼠缺血心肌的保护作用[J].中国药理学通报,2016,32(2): 268-273. [65] 胡明珠. PI3K/Akt/Sirt1/PGC-1α信号通路介导H2S后处理对大鼠缺血心肌的保护作用[D]. 无锡:江南大学,2016. [66] 周爽,李莉,张慧娜,等. SIRT1通过上调MnSOD的表达保护糖尿病导致的内皮细胞损伤[C]. 国际心脏研究会(ISHR)中国分会第十届暨中国病理生理学会心血管专业委员会第十三届学术会议论文摘要文集, 温州:2008. [67] YAO ZQ, ZHANG X, ZHEN Y, et al. A novel small-molecule activator of Sirtuin-1 induces autophagic cell death/mitophagy as a potential therapeutic strategy in glioblastoma. Cell Death Dis. 2018;9(7):767. [68] 李晓燕, 韩霞, 张红明, 等. 耐力运动训练通过SIRT1信号通路保护力竭运动大鼠心肌[J]. 中华心血管病杂志,2017,45(6):501-506. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhang Chao, Lü Xin. Heterotopic ossification after acetabular fracture fixation: risk factors, prevention and treatment progress [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1434-1439. |
| [3] | Zhou Jihui, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Multiple problems in the selection of implants for patellar fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1440-1445. |
| [4] | Wang Debin, Bi Zhenggang. Related problems in anatomy mechanics, injury characteristics, fixed repair and three-dimensional technology application for olecranon fracture-dislocations [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1446-1451. |
| [5] | Shen Jinbo, Zhang Lin. Micro-injury of the Achilles tendon caused by acute exhaustive exercise in rats: ultrastructural changes and mechanism [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1190-1195. |
| [6] | Wang Mengting, Gu Yanping, Ren Wenbo, Qin Qian, Bai Bingyi, Liao Yuanpeng. Research hotspots of blood flow restriction training for dyskinesia based on visualization analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1264-1269. |
| [7] | Ji Zhixiang, Lan Changgong. Polymorphism of urate transporter in gout and its correlation with gout treatment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1290-1298. |
| [8] | Yuan Mei, Zhang Xinxin, Guo Yisha, Bi Xia. Diagnostic potential of circulating microRNA in vascular cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1299-1304. |
| [9] | Wang Yongsheng, Wu Yang, Li Yanchun. Effect of acute high-intensity exercise on appetite hormones in adults: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1305-1312. |
| [10] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [11] | Wan Ran, Shi Xu, Liu Jingsong, Wang Yansong. Research progress in the treatment of spinal cord injury with mesenchymal stem cell secretome [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1088-1095. |
| [12] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [13] | Zhao Min, Feng Liuxiang, Chen Yao, Gu Xia, Wang Pingyi, Li Yimei, Li Wenhua. Exosomes as a disease marker under hypoxic conditions [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1104-1108. |
| [14] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [15] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||