Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (22): 3603-3608.doi: 10.12307/2024.532
Research and development of nanoparticles with active targeting ability in nonneoplastic kidney disease
Liu Chang, Li Linhua, Fu Ping
- Department of Nephrology, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, China
-
Received:2023-09-16Accepted:2023-11-10Online:2024-08-08Published:2024-01-20 -
Contact:Fu Ping, PhD, Professor, Doctoral supervisor, Department of Nephrology, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, China -
About author:Liu Chang, Master candidate, Department of Nephrology, West China Hospital, Sichuan University, Chengdu 610041, Sichuan Province, China -
Supported by:Science and Technology Plan Program of Sichuan Province, No. 2023NSFSC1586 (to LLH)
CLC Number:
Cite this article
Liu Chang, Li Linhua, Fu Ping . Research and development of nanoparticles with active targeting ability in nonneoplastic kidney disease[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3603-3608.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 急性肾损伤 急性肾损伤是指由各种病因引起的短时间内出现肾脏损伤及肾功能急剧减退的临床综合征[19]。急性肾损伤的病因众多,包括肾脏缺血、肾毒性药物、脓毒血症及泌尿系统梗阻等[20-21]。不同病因引起的急性肾损伤所涉及的病理生理学机制有所不同,其共同机制包括血流动力学异常、氧化应激损伤、肾组织炎症及免疫反应等,但主要病理表现为肾小管细胞亚致死性和致死性损伤引起的肾结构迅速破坏或肾功能损害[22]。急性肾损伤发病急,临床上要求药物起效快且较为准确地发挥作用,因此靶向肾小管上皮细胞的纳米药物递送系统在急性肾损伤治疗中有较多研究。 2.1.1 肾小管损伤分子1与丝氨酸 肾小管损伤分子1是一种跨膜蛋白,在各种原因导致损伤的肾脏尤其是肾近端小管表达增加。肾小管损伤分子1在临床中通常被认为是一种可靠的近端肾小管损伤早期生物标志物,因为它可以在肾损伤早期阶段被特异性地检测和识别[23]。L-丝氨酸是一种有效的肾小管靶向修饰剂,据研究证实其可能是肾小管损伤分子1的靶向药物[24]。肾小管损伤分子1与丝氨酸相互作用是急性肾损伤中药物主动靶向的研究热点,与肾小管损伤分子1和丝氨酸相关的肾主动靶向策略研究历程如图3所示。LIU等[25]合成了以壳聚糖为基础的载药体系,通过修饰丝氨酸可以有效靶向急性肾损伤肾脏,尤其是肾小管;通过活性氧自由基敏感的连接子将线粒体靶向抗氧化剂SS31与丝氨酸连接,合成了具有精确逐步靶向作用的前药,该药物在急性肾损伤肾脏中积累并靶向运输到肾小管中,被受损的肾小管上皮细胞内化,响应性地释放SS31,进一步靶向线粒体发挥抗氧化作用,减轻线粒体损伤,进而缓解急性肾损伤,这种双重靶向体系在精准靶向和灵敏释放的基础上显著增强了SS31的治疗效果。"

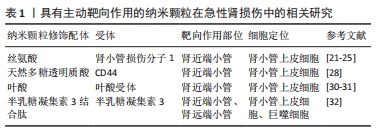
MATSUURA等[24]合成了L-丝氨酸修饰的聚酰胺树状大分子作为有效的肾脏靶向药物载体,静脉注射后3 h后约82%的药物剂量在肾脏中累积,证明L-丝氨酸修饰的聚酰胺树状大分子是一种有前途的肾脏靶向药物载体用于治疗肾脏疾病。 LI等[26]也设计了一种有效递送迷迭香酸的纳米颗粒并在纳米颗粒上进行L-丝氨酸修饰,通过肾小管损伤分子1和丝氨酸之间的特异性相互作用最终产生了一种急性肾损伤肾脏靶向纳米颗粒。在缺血-再灌注诱导的急性肾损伤肾脏中,这种纳米颗粒可迅速积累并长期保留,特别是在受损的肾小管上皮细胞中,对缺血再灌注引起的急性肾损伤发挥保护作用。 在以上研究的基础上,KONG等[27]首次报道手性材料用于急性肾损伤的诊断和治疗。合成了L-丝氨酸和D-丝氨酸修饰的聚酰胺树状大分子并分别结合过氧化氢荧光探针,发现D-丝氨酸修饰荧光探针在急性肾损伤小鼠肾脏中具有较强的积累能力,通过荧光探针可检测到过量产生的过氧化氢,从而来诊断急性肾损伤。随后,研究者将抗炎药锰五碳酸溴装载入D-丝氨酸修饰的聚酰胺树状大分子中,获得具有治疗功能的纳米颗粒。可见分子的手性也是可能影响主动靶向作用的因素。 以上研究表明在急性肾损伤发生时,肾小管上皮细胞上表达上调的肾小管损伤分子1可以作为药物主动靶向的有效靶点,目前将L-丝氨酸作为肾小管损伤分子1的靶向剂进行载体修饰的研究较多,但最近也有研究证明了D-丝氨酸同样具有肾小管损伤分子1的靶向能力,为未来研究中靶向修饰剂的选择提供了更多可能性。 2.1.2 CD44与天然多糖透明质酸 CD44是一种在细胞膜上广泛表达的跨膜蛋白,在多种炎症过程中承担重要的功能,在急性肾损伤肾脏中过表达[28-29]。 WANG等[30]根据天然多糖透明质酸与损伤肾近端小管中过表达的CD44受体具有强结合能力的特性,开发了一种具有肾靶向作用的活性氧响应性药物递送系统,将细胞内钙螯合剂2-氨基苯氧乙烷-N,N,N,N-四乙酸乙酰氧基甲酯(BAPTA-AM,BA)递送进入细胞,目的是通过靶向清除肾小管细胞内超载的钙离子来挽救肾细胞损伤,实验结果证明该药物对急性肾损伤有较好的保护作用。 2.1.3 叶酸受体与叶酸 由于肾近端小管细胞表达高水平的叶酸受体,利用叶酸与其受体可以特异性结合的原理,叶酸偶联也可被用于肾靶向药物传递[31-32]。 KNIGHT等[32]开发了一种靶向治疗方法,靶向高表达叶酸受体的肾近端小管,构建了叶酸-抗氧化剂缀合物——氮氧自由基哌啶醇-叶酸缀合物用以靶向叶酸受体,在小鼠肾脏缺血再灌注损伤模型中证实氮氧自由基哌啶醇-叶酸缀合物比单独替酚更有效降低肾脏超氧化物水平。 XIE等[33]设计了具有肾靶向作用的纳米颗粒用于递送含有脯氨酸羟化酶结构域蛋白2的小干扰RNA(siRNA),经研究证明该纳米系统可减轻肾缺血/再灌注损伤;研究中使用叶酸修饰的小干扰RNA载体聚酰胺树突状分子5递送低氧感受器蛋白,即脯氨酸羟化酶2-小干扰RNA,研究证实注射该系统可显著减轻肾缺血再灌注损伤,与对照组相比可抑制血清肌酐和血尿素氮升高,改善组织学损伤。 2.1.4 半乳糖凝集素3与半乳糖凝集素3结合肽 肾脏中的半乳糖凝集素3在发育过程中促进肾的形成。半乳糖凝集素3表达的增加与肾纤维化和急性肾小管损伤相关。研究者们利用组合噬菌体展示技术鉴定了半乳糖凝集素3识别结构域的特定肽[30]。半乳糖凝集素3与半乳糖凝集素3结合肽在小鼠肾脏特别是肾小管中显示大量积累。与单独使用药物血管紧张素转换酶抑制剂卡托普利相比,用半乳糖凝集素3与半乳糖凝集素3结合肽修饰药物后药物肾脏释放的曲线下面积增加了2.7倍,说明半乳糖凝集素3与半乳糖凝集素3结合肽在急性肾损伤中具有较强的肾靶向作用。尽管该肽被鉴定为半乳糖凝集素3结合肽,但由于其具有正电荷,借助巨噬细胞表达受体进行的肾脏重吸收也可能在其肾小管靶向过程中发挥作用[34]。目前半乳糖凝集素3与半乳糖凝集素3结合肽的肾脏靶向的机制尚不完全清楚。 2.1.5 总结 目前研究者们针对急性肾损伤中纳米颗粒主动靶向策略的实现进行了以上4种探索,4种配体-受体结合体系特性总结如表1。纳米颗粒经过连接靶向剂改造后比单一药物对急性肾损伤的保护效果更好,但上述研究并没有对连接靶向剂后纳米颗粒肾脏外沉积水平进行定量检测,即暂未进行纳米颗粒靶向效率检测及脱靶毒性相关检测,因此不同靶向剂的靶向效率对比有待进一步研究验证。"
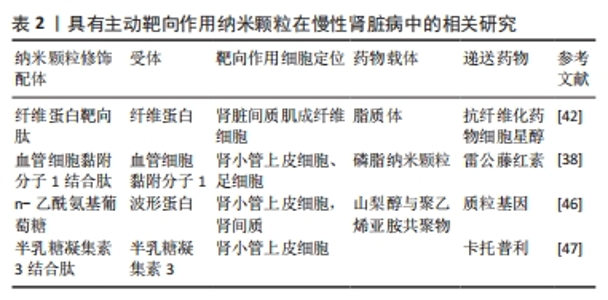
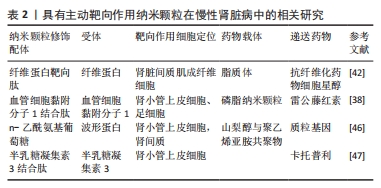
2.2 慢性肾脏病 慢性肾脏病定义为一种持续3个月以上的肾脏结构或功能异常[35]。在全球范围内,慢性肾脏病是一种不可逆的进展性疾病,患病率为13.4%(11.7%-15.1%),大多数慢性肾脏病患者可能进展到需要移植或透析的终末期肾病[36-37],目前已被确定是一个与高心血管风险相关的重要公共卫生问题[38-39]。 慢性肾脏病的病因是复杂多样的,肾小球或肾小管的损伤都有可能引起慢性肾脏病的发生或进展[40]。慢性肾脏病的主要病理结局表现为肾小球硬化、内皮功能障碍和损伤以及慢性的炎症[40],因此,靶向递送药物到肾小球以改善肾小球内皮损伤可能有帮助缓解慢性肾脏病。 纤维化是一种细胞外基质在实体器官中过度积累的病理过程,可作为对各种类型损伤的一种适应性反应[41-42]。肾纤维化是慢性肾脏病进展到后期的重要病理特征,改善纤维化也可以有助于慢性肾脏病的治疗[43]。 2.2.1 纤维蛋白靶向肽与肌成纤维细胞 纤维蛋白靶向肽是一个5肽(Cys-Arg-Glu-Lys-Ala)的序列,已被证明可以靶向各种纤维蛋白疾病,包括转移性癌症、肾纤维化和血栓症。 LI等[44]合成了靶向纤维化肾脏的纤维蛋白靶向肽-聚乙二醇化脂质体作为载体,将具有较严重全身毒性的抗纤维化药物细胞星醇特异性传递到肾脏间质肌成纤维细胞,可以显著减轻小鼠单侧输尿管梗阻引起的肾纤维化、损伤和炎症;但在研究中也观察到大量未修饰纤维蛋白靶向肽的脂质体摄取,可见对这种细胞类型的更精确靶向策略仍有待改进。 2.2.2 血管细胞黏附分子1结合肽与血管细胞黏附分子1 据研究证实,当肾小球内皮细胞和足细胞受到炎症等刺激时血管细胞黏附分子1的表达会上调[45]。血管细胞黏附分子1结合肽(VHPKQHRGGSKGC)是一种线性多肽,可以结合血管细胞黏附分子1用于靶向肾小管上皮细胞和足细胞。 WU等[40]设计了一种名为肽偶联的雷公藤红素-磷脂纳米颗粒靶向递送系统,通过修饰VHPKQHRGGSKGC肽段有效地将雷公藤红素递送至肾小球中受损的内皮细胞和足细胞,起到“一箭双雕”的作用,用于慢性肾脏病的治疗。研究证实,肽偶联的雷公藤红素-磷脂纳米颗粒可以有效抑制炎症、减少内皮损伤,减轻慢性肾脏病严重程度。除此之外,肽偶联的雷公藤红素-磷脂纳米颗粒靶向递送系统还可以通过靶向递送降低药物剂量,减轻雷公藤系统使用带来的不良反应。 2.2.3 波形蛋白与N-乙酰氨基葡萄糖 波形蛋白是一种中间丝蛋白,通常是慢性肾脏病中肾小管上皮细胞向间充质转化的标志物,可作为是肾小管损伤或肾功能障碍的标志物[46]。有研究表明,波形蛋白具有类似凝集素的结合特性,并与N-乙酰氨基葡萄糖(GlcNAc)特异性结合[47]。MATHEW等[48]开发了一种基于山梨醇和聚乙烯亚胺共聚物的近端肾小管靶向基因递送系统,以波形蛋白特异性靶向剂修饰,用于靶向肾脏中表达波形蛋白的细胞,在体外研究证实人近端肾小管上皮细胞系(HK-2)对药物的特异性摄取增加,也在体内奥尔波特综合征小鼠模型中验证了制备的基因载体具有较高靶向效率。 2.2.4 半乳糖凝集素3与半乳糖凝集素3结合肽 半乳糖凝集素3与半乳糖凝集素3结合肽在慢性肾脏病中也有作为肾靶向给药合适候选载体的潜力。GENG等[49]开发了一种针对肾脏靶向递送血管紧张素Ⅰ转化酶抑制剂卡托普利的肽-药物偶联物,并使用半乳糖凝集素3与半乳糖凝集素3结合肽进行修饰,静脉注射后半乳糖凝集素3与半乳糖凝集素3结合肽可以特异性地在肾脏中积累;半乳糖凝集素3与半乳糖凝集素3结合肽-FITC的荧光成像显示,在静脉注射后不久这种积累主要是由近端肾小管细胞对肽的重吸收实现。 2.2.5 其他机制 肌成纤维细胞是纤维化过程的直接贡献者。CHENG等[50]采用体外-体内组合噬菌体展示技术来识别肽配体,以增强单侧输尿管梗阻诱导的纤维化肾脏中肌成纤维细胞对纳米载体的摄取,合成了具有肌成纤维细胞靶向作用的纳米颗粒,并且将多激酶抑制剂索拉非尼装载在纳米颗粒中,发现该纳米颗粒可以抑制人肌成纤维细胞的胶原合成;当纳米颗粒静脉注射给单侧输尿管梗阻诱导的肾纤维化小鼠时,装载在肌成纤维细胞靶向纳米颗粒中的索拉非尼显著改善了肾纤维化,为肾纤维化提供了一种有效治疗选择。 2.2.6 总结 慢性肾脏病的病程长、病理机制复杂,包括了炎症、凋亡、氧化应激、纤维化等多种机制的相互影响,肾脏的多个部位如肾小球、肾小管、肾间质均会有一定程度地受累,因此针对慢性肾脏病主动靶向策略设计的目标靶向细胞可以尝试在肾脏不同部位发挥不同作用的细胞,针对不同病理机制递送的药物也会有更多选择,有广阔的研究前景。具有主动靶向作用纳米颗粒在慢性肾脏病中的相关研究,见表2。"

2.3 多囊肾 常染色体显性遗传性多囊肾病是最常见的遗传性肾脏疾病,其特征是肾囊肿形成和最终发展为终末期肾病[51]。治疗常染色体显性遗传性多囊肾病的一种方法是抑制哺乳动物雷帕霉素靶蛋白通路,该通路与细胞过度增殖有关,有肾囊肿扩张的作用[52]。然而,哺乳动物雷帕霉素靶蛋白抑制剂包括雷帕霉素[53-54]、依维莫司等[55],系统用药具有多种不良反应,包括脱靶毒性、免疫抑制等。研究者们针对具有主动靶向作用纳米颗粒的研究为常染色体显性遗传性多囊肾病治疗提供了新的可能。 2.3.1 巨细胞蛋白与巨蛋白结合肽 肾小管上皮细胞在常染色体显性遗传性多囊肾病早期参与囊肿形成,巨细胞蛋白是肾小管上皮细胞上高表达的配体/受体,其与巨蛋白结合肽有特异性结合作用[56]。 COX等[57]据上述原理合成了皮质收集管靶向肽修饰的两亲性胶束纳米颗粒,并得出较高的药物包封效率(> 92.6%),体外分析表明使用两亲性胶束包封药物可以增强雷帕霉素、西罗莫司等药物对人肾小管上皮细胞的抗增殖作用。JIANG等[56]通过将巨蛋白结合肽结合到胶束表面开发了一种基于肽两亲性胶束的肾脏靶向药物传递系统,将该系统静脉注射到野生型小鼠体内,发现该胶束表现出显著的肾脏积累和较高的生物相容性;由于胶束具有两亲性结构,研究者使用6种疏水药物(包括沙拉酸盐、雷帕霉素、托伐坦、奥曲肽、甲基巴多索酮和吡格列酮)通过疏水相互作用有效地装载到胶束的脂质核中,实现了药物较为有效的肾脏靶向运输。 2.3.2 总结 常染色体显性遗传性多囊肾是遗传性肾病,在临床上多见于成人,早期病变隐匿,多因体检发现或已进展到肾功能不全阶段,能否在疾病早期阶段对遗传学上的高危患者做出干预、全程防控延缓疾病进展是目前医疗领域要攻克的难题。目前关于巨细胞蛋白与巨蛋白结合肽靶向策略在常染色体显性遗传性多囊肾病中的相关研究较少,对该种策略的安全性及脱靶毒性还未进行具体评估,以及该靶向策略对幼龄动物是否有影响需更多研究证实。 2.4 肾病综合征 肾病综合征主要表现为蛋白尿(24 h尿蛋白> 3.5 g)、低白蛋白血症(< 30 g/L)和外周性水肿,主要病因包括微小的肾小球损伤、局灶性阶段性肾小球硬化症和非膜性肾小球肾炎,次要病因是糖尿病、高血压和淀粉样变性等原因继发[58]。肾病综合征的主要病理损伤包括肾小球损伤及小管间质纤维化。 2.4.1 半乳糖凝集素3与半乳糖凝集素3结合肽 已有研究证明,半乳糖凝集素3与半乳糖凝集素3结合肽修饰的卡托普利在肾脏中的积累是游离卡托普利积累的2.7倍,这表明半乳糖凝集素3与半乳糖凝集素3结合肽有可能作为一种新的肾脏靶向载体[49]。LI等[26]据此合成了一种新型的二维材料,利用半乳糖凝集素3与半乳糖凝集素3结合肽主动靶向肾近端小管细胞的性能,通过半乳糖凝集素3与半乳糖凝集素3结合肽的修饰促进了细胞对黑磷纳米片的摄取;纳米片中的小分子化合物鲁索利替尼磷酸盐通过抑制细胞炎症和减少活性氧产生在大鼠肾病综合征模型中发挥治疗作用。因此,半乳糖凝集素3与半乳糖凝集素3结合肽在靶向肾脏的性能在肾病综合征中也得到了验证。 2.4.2 总结 肾病综合征是以大量蛋白尿为特征的临床肾脏疾病,主要由肾小球病变引起,表现为肾小球基底膜通透性增加,目前对于肾病综合征中具有主动靶向能力的纳米颗粒相关研究较少,关于肾小球基底膜上相关细胞如血管内皮细胞、足细胞的靶向策略尚未有研究报道。 2.5 系膜细胞介导的肾小球肾炎 系膜细胞介导的肾小球肾炎是指一类免疫介导的肾小球损伤,其特征是肾小球炎症细胞浸润、系膜细胞增殖,后期常见的病理表现为肾纤维化,并伴有细胞外基质的过度积累[59]。目前对于系膜细胞介导的肾小球肾炎的治疗方案包括糖皮质激素和免疫抑制剂治疗,但药物的脱靶分布会导致严重的全身毒性[60],因此“抗炎/抗纤维化”药物特异性共递送到肾小球系膜细胞区域有望产生更好的治疗效果。 2.5.1 α8整合素 SCINDIA等[61]的体外研究表明,以系膜细胞区抗体分子α8整合素作为靶向分子制备的免疫复合物具有良好的系膜细胞靶向特性。ZHOU等[62]据此构建了靶向肾小球系膜细胞的新型肾靶向纳米载体给药系统,将α8整合素抗体修饰在脂质体-聚乳酸羟基乙酸聚合物纳米颗粒上,使地塞米松和卡托普利靶向递送到肾小球系膜区发挥抗炎/ 抗纤维化作用,治疗肾小球肾炎。 2.5.2 总结 系膜细胞介导的肾小球肾炎是临床免疫相关性肾小球肾炎中的一类,主要病变部位在肾小球系膜区。除系膜细胞介导的肾小球肾炎外,临床上有很多其他肾炎类型如以弥漫性足突融合为特征的微小病变型肾炎等目前研究中未涉及,不同肾炎病理病变部位不同,有广阔的研究前景。 "

| [1] BONNER R, ALBAJRAMI O, HUDSPETH J, et al. Diabetic kidney disease. Prim Care. 2020;47(4):645-659. [2] KOVESDY CP. Epidemiology of chronic kidney disease: an update 2022. Kidney Int Suppl. 2022;12(1):7-11. [3] WANNER N, BECHTEL-WALZ W. Epigenetics of kidney disease. Cell Tissue Res. 2017;369(1):75-92. [4] KIM BY, RUTKA JT, CHAN WC. Nanomedicine. N Engl J Med. 2010;363(25): 2434-2443. [5] PATRA JK, DAS G, FRACETO LF, et al. Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnology. 2018; 16(1):1-33. [6] WANG J, MASEHI-LANO JJ, CHUNG EJ. Peptide and antibody ligands for renal targeting: nanomedicine strategies for kidney disease. Biomater Sci. 2017;5(8):1450-1459. [7] 肖贵英,李昕,邓阳,等.抗菌药物的肾毒性机制及危险因素研究进展[J].中国抗生素杂志,2022,47(8):779-785. [8] 伍敏,刘必成,程守勤.合理使用解热镇痛药警惕不良反应发生[N].健康报,2023-02-06(008). doi:10.28415/n.cnki.njika.2023.000320. [9] ZHAO J, WANG X, YANG Y, et al. Upregulated miR-101 inhibits acute kidney injury-chronic kidney disease transition by regulating epithelial-mesenchymal transition. Hum Exp Toxicol. 2020;39(12):1628-1638. [10] BRUNO BJ, MILLER GD, LIM CS. Basics and recent advances in peptide and protein drug delivery. Ther Deliv. 2013;4(11):1443-1467. [11] ARSLAN FB, OZTURK K, CALIS S. Antibody-mediated drug delivery. Int J Pharmaceut. 2021;596:120268. [12] GISBERT-GARZARÁN M, VALLET-REGÍ M. Nanoparticles for Bio-Medical Applications. Nanomaterials (Basel). 2022;12(7):1189. [13] MARY SM, RAMAKRISHNAN M, SUDALAIMANI PAULPANDIAN SD, et al. Application of nanoparticles in Dentistry. Bioinformation. 2023;19(1):14-18. [14] PEARCE AK, O’REILLY RK. Insights into active targeting of nanoparticles in drug delivery: advances in clinical studies and design considerations for cancer nanomedicine. Bioconjug Chem. 2019;30(9):2300-2311. [15] 李珍,刘宏宝.纳米粒子肾靶向的影响因素及机制[J].中国组织工程研究,2023,27(3):453-460. [16] HUANG Y, WANG J, JIANG K, et al. Improving kidney targeting: The influence of nanoparticle physicochemical properties on kidney interactions. J Control Release. 2021;334:127-137. [17] HE L, WEI Q, LIU J, et al. AKI on CKD: heightened injury, suppressed repair, and the underlying mechanisms. Kidney Int. 2017;92(5):1071-1083. [18] YAN MT, CHAO CT, LIN SH. Chronic kidney disease: Strategies to retard progression. Int J Mol Sci. 2021;22(18):10084. [19] CHAWLA LS, EGGERS PW, STAR RA, et al. Acute kidney injury and chronic kidney disease as interconnected syndromes. N Engl J Med. 2014;371(1): 58-66. [20] ABUELO JG. Normotensive ischemic acute renal failure. New Engl J Med. 2007;357(8):797-805. [21] POSTON JT, KOYNER JL. Sepsis associated acute kidney injury. BMJ. 2019; 364:k4891. [22] BUGGER H, PFEIL K. Mitochondrial ROS in myocardial ischemia reperfusion and remodeling. Biochim Biophys Acta Mol Basis Dis. 2020;1866(7):165768. [23] ICHIMURA T, ASSELDONK EJ, HUMPHREYS BD, et al. Kidney injury molecule-1 is a phosphatidylserine receptor that confers a phagocytic phenotype on epithelial cells. J Clin Invest. 2008;118(5):1657-1668. [24] MATSUURA S, KATSUMI H, SUZUKI H, et al. l-Serine-modified polyamidoamine dendrimer as a highly potent renal targeting drug carrier. Proc Natl Acad Sci U S A. 2018;115(41):10511-10516. [25] LIU D, SHU G, JIN F, et al. ROS-responsive chitosan-SS31 prodrug for AKI therapy via rapid distribution in the kidney and long-term retention in the renal tubule. Sci Adv. 2020;6(41):eabb7422. [26] LI H, TAN X, LIU Y, et al. Ruxolitinib-loaded black phosphorus nanosheets actively target proximal tubule cells to ameliorate nephrotic syndrome by reducing inflammation and oxidative stress. Nano Today. 2022;47:101631. [27] KONG L, FAN D, ZHOU L, et al. The influence of modified molecular (D/L-serine) chirality on the theragnostics of PAMAM-based nanomedicine for acute kidney injury. J Mater Chem B. 2021;9(43):9023-9030. [28] HERRERA M, BUSSOLATI B, BRUNO S, et al. Exogenous mesenchymal stem cells localize to the kidney by means of CD44 following acute tubular injury. Kidney Int. 2007;72(4):430-441. [29] RAMPANELLI E, DESSING MC, CLAESSEN N, et al. CD44-deficiency attenuates the immunologic responses to LPS and delays the onset of endotoxic shock-induced renal inflammation and dysfunction. PLoS One. 2013;8(12): e84479. [30] WANG Y, PU M, YAN J, et al. 1,2-Bis(2-aminophenoxy)ethane-N,N,N’,N’-tetraacetic Acid Acetoxymethyl Ester Loaded Reactive Oxygen Species Responsive Hyaluronic Acid-Bilirubin Nanoparticles for Acute Kidney Injury Therapy via Alleviating Calcium Overload Mediated Endoplasmic Reticulum Stress. ACS Nano. 2023;17(1):472-491. [31] DOLMAN M, HARMSEN S, STORM G, et al. Drug targeting to the kidney: Advances in the active targeting of therapeutics to proximal tubular cells. Adv Drug Deliv Rev. 2010;62(14):1344-1357. [32] KNIGHT SF, KUNDU K, JOSEPH G, et al. Folate receptor-targeted antioxidant therapy ameliorates renal ischemia-reperfusion injury. J Am Soc Nephrol. 2012;23(5):793-800. [33] XIE D, WANG J, HU G, et al. Kidney-Targeted Delivery of Prolyl Hydroxylase Domain Protein 2 Small Interfering RNA with Nanoparticles Alleviated Renal Ischemia/Reperfusion Injury. J Pharmacol Exp Ther. 2021;378(3):235-243. [34] LI HY, YANG S, LI JC, et al. Galectin 3 inhibition attenuates renal injury progression in cisplatin-induced nephrotoxicity. Biosci Rep. 2018;38(6): BSR20181803. [35] STEVENS PE, LEVIN A, KIDNEY DISEASE: IMPROVING GLOBAL OUTCOMES CHRONIC KIDNEY DISEASE GUIDELINE DEVELOPMENT WORK GROUP MEMBERS. Evaluation and management of chronic kidney disease: synopsis of the kidney disease: improving global outcomes 2012 clinical practice guideline. Ann Intern Med. 2013;158(11):825-830. [36] AMMIRATI AL. Chronic kidney disease. Rev Assoc Med Bras. 2020;66(Suppl 1): s03-s09. [37] CHARLES C, FERRIS AH. Chronic kidney disease. Prim Care. 2020;47(4): 585-595. [38] GLASSOCK RJ, WARNOCK DG, DELANAYE P. The global burden of chronic kidney disease: estimates, variability and pitfalls. Nat Rev Nephrol. 2017; 13(2):104-114. [39] LV JC, ZHANG LX. Prevalence and disease burden of chronic kidney disease. Adv Exp Med Biol. 2019;1165:3-15. [40] WU Q, WANG J, WANG Y, et al. Targeted delivery of celastrol to glomerular endothelium and podocytes for chronic kidney disease treatment. Nano Res. 2022;15(4):3556-3568. [41] LIU F, GOU R, HUANG J, et al. Effect of anaphylatoxin C3a, C5a on the tubular epithelial-myofibroblast transdifferentiation in vitro. Chin Med J. 2011;124(23):4039-4045. [42] WEISKIRCHEN R, WEISKIRCHEN S, TACKE F. Organ and tissue fibrosis: Molecular signals, cellular mechanisms and translational implications. Mol Aspects Med. 2019;65:2-15. [43] NASTASE MV, ZENG-BROUWERS J, WYGRECKA M, et al. Targeting renal fibrosis: mechanisms and drug delivery systems. Adv Drug Deliv Rev. 2018; 129:295-307. [44] LI R, LI Y, ZHANG J, et al. Targeted delivery of celastrol to renal interstitial myofibroblasts using fibronectin-binding liposomes attenuates renal fibrosis and reduces systemic toxicity. J Control Release. 2020;320:32-44. [45] SHARP CN, DOLL MA, MEGYESI J, et al. Subclinical kidney injury induced by repeated cisplatin administration results in progressive chronic kidney disease. Am J Physiol Renal. 2018;315(1):F161-F172. [46] VANSTHERTEM D, GOSSIAUX A, DECLEVES AE, et al. Expression of nestin, vimentin, and NCAM by renal interstitial cells after ischemic tubular injury. Biomed Res Int. 2010;2010:193259. [47] ASO SI, ISE H, TAKAHASHI M, et al. Effective uptake of N-acetylglucosamine-conjugated liposomes by cardiomyocytes in vitro.J Control Release. 2007; 122(2):189-198. [48] MATHEW AP, UTHAMAN S, BAE EH, et al. Vimentin Targeted Nano-gene Carrier for Treatment of Renal Diseases. J Korean Med Sci. 2021;36(49): e333. [49] GENG Q, SUN X, GONG T, et al. Peptide-drug conjugate linked via a disulfide bond for kidney targeted drug delivery. Bioconjug Chem. 2012;23(6): 1200-1210. [50] CHENG HT, HUANG HC, LEE TY, et al. Delivery of sorafenib by myofibroblast-targeted nanoparticles for the treatment of renal fibrosis. J Control Release. 2022;346:169-179. [51] CORNEC-LE GALL E, ALAM A, PERRONE RD. Autosomal dominant polycystic kidney disease. Lancet. 2019;393(10174):919-935. [52] ZOU Z, TAO T, LI H, et al. mTOR signaling pathway and mTOR inhibitors in cancer: progress and challenges. Cell Biosci. 2020;10(1):1-11. [53] LI A, FAN S, XU Y, et al. Rapamycin treatment dose-dependently improves the cystic kidney in a new ADPKD mouse model via the mTORC 1 and cell-cycle-associated CDK 1/cyclin axis. J Cell Mol Med. 2017;21(8):1619-1635. [54] ZHENG Y, JIANG Y. mTOR inhibitors at a glance. Mol Cell Pharmacolo. 2015; 7(2):15-20. [55] MACKEIGAN JP, KRUEGER DA. Differentiating the mTOR inhibitors everolimus and sirolimus in the treatment of tuberous sclerosis complex. Neuro Oncol. 2015;17(12):1550-1559. [56] JIANG K, HUANG Y, CHUNG EJ. Combining metformin and drug-loaded kidney-targeting micelles for polycystic kidney disease. Cell Mol Bioeng. 2023;16(1):55-67. [57] COX A, TUNG M, LI H, et al. In vitro delivery of mTOR inhibitors by kidney-targeted micelles for autosomal dominant polycystic kidney disease. SLAS Technol. 2023;28(4):223-229. [58] CHAUDESAYGUES E, GRASSE M, MARCHAND L, et al. Nephrotic syndrome revealed by pulmonary embolism: About four cases. Ann Cardiol Angeiol (Paris). 2014;63(5):385-388. [59] COUSER WG. Pathogenesis of glomerular damage in glomerulonephritis. Nephrol Dial Transplant. 1998;13(suppl_1):10-15. [60] FLOEGE J, RAUEN T, TANG SC. Current treatment of IgA nephropathy. Semin Immunopathol. 2021;43:717-728. [61] SCINDIA Y, DESHMUKH U, THIMMALAPURA PR, et al. Anti-alpha8 integrin immunoliposomes in glomeruli of lupus-susceptible mice: a novel system for delivery of therapeutic agents to the renal glomerulus in systemic lupus erythematosus. Arthritis Rheum. 2008;58(12):3884-3891. [62] ZHOU L, YE Z, ZHANG E, et al. Co-delivery of dexamethasone and captopril by α8 integrin antibodies modified liposome-plga nanoparticle hybrids for targeted anti-inflammatory/anti-fibrosis therapy of glomerulonephritis. Int J Nanomedicine. 2022;17:1531-1547. |
| [1] | Liu Hanfeng, Wang Jingjing, Yu Yunsheng. Artificial exosomes in treatment of myocardial infarction: current status and prospects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1118-1123. |
| [2] | Yin Tong, Yang Jilei, Li Yourui, Liu Zhuoran, Jiang Ming. Application of core-shell structured nanofibers in oral tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 766-770. |
| [3] | Tian Xin, Liu Tao, Yang Huilin, He Fan. In vitro evaluation of sustained release Kartogenin by gelatin methacryloyl microspheres for repairing nucleus pulposus degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 724-730. |
| [4] | Zhu Liwei, Wang Jiangyue, Bai Ding. Application value of nanocomposite gelatin methacryloyl hydrogels in different bone defect environments [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 753-758. |
| [5] | Chen Guanting, Zhang Linqi, Wang Xixi, Chen Xu. Autophagy, ferroptosis-related targets and renal function progression in patients with chronic kidney disease: bioinformatics analysis and experimental verification [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5122-5129. |
| [6] | Chen Pinrui, Pei Xibo, Xue Yiyuan. Function and advantages of magnetically responsive hydrogel in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 452-457. |
| [7] | Ilham • Maimaitiabudula, Huang Xiaoxia, Li Luyao, Teng Yong. Polymer-based antibiotic sustained-release carrier in treatment of chronic osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3597-3602. |
| [8] | Wang Zhaofei, He Guoyun, Tian Fangcan, Li Guangfeng, Cao Zhonghua, Liu Xiangfei. Antibacterial properties of silver nanoparticle-coated stainless steel prepared via active screen plasma surface modification in vitro [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3464-3471. |
| [9] | Wang Jinlei, Li Ke, Zhao Liang. Platelet-camouflaged silver nanoparticle hydrogel accelerates wound healing in type 1 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2659-2666. |
| [10] | Wang Xindong, Liang Chengzhi, Zhang Yongxian. Functional characteristics and clinical applications of MXene nanoparticles in wound healing [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2739-2746. |
| [11] | Zhang Shuzhi, Qu Pengfei, Han Junquan, Wang Hong. Research and application of electrospinning drug delivery systems containing traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2759-2765. |
| [12] | Guo Yuxin, Wang Hao, Li Mingqi, Chen Yueying, Pan Juhong, Huang Xin, Wang Zhiwen, Zhou Qing. Ultrasound-optimized hydrogel scaffold used to promote transdermal delivery of gold nanoparticles [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(15): 2330-2337. |
| [13] | Liu Chang, Zhang Wen, Zhu Can, Sun Jie, Ding Yicheng, Shi Qin. Inhibitory effect of bovine serum albumin-chitosan nanoparticles loaded with EPZ6438 on osteosarcoma [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(10): 1512-1518. |
| [14] | Bei Ying, Li Wenjing, Li Meiyun, Su Meng, Zhang Jin, Huang Yu, Zhu Yanzhao, Li Jiali, Wu Yan. Prussian blue nanoparticles promote wound healing of diabetic skin [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(10): 1526-1532. |
| [15] | Su Kunyang, Chen Bineng, Chen Yiliang, Jin Shaofeng. Strontium ranelate-loaded sodium alginate/collagen hydrogel promotes bone defect repair in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(10): 1568-1574. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||