Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (17): 2659-2666.doi: 10.12307/2024.450
Previous Articles Next Articles
Platelet-camouflaged silver nanoparticle hydrogel accelerates wound healing in type 1 diabetic rats
Wang Jinlei1, Li Ke2, Zhao Liang1
- 1School of Pharmacy, 2Life Science Institute, Jinzhou Medical University, Jinzhou 121001, Liaoning Province, China
-
Received:2023-06-27Accepted:2023-08-04Online:2024-06-18Published:2023-12-14 -
Contact:Zhao Liang, MD, Professor, School of Pharmacy, Jinzhou Medical University, Jinzhou 121001, Liaoning Province, China -
About author:Wang Jinlei, Master candidate, School of Pharmacy, Jinzhou Medical University, Jinzhou 121001, Liaoning Province, China Li Ke, Master candidate, Life Science Institute, Jinzhou Medical University, Jinzhou 121001, Liaoning Province, China
CLC Number:
Cite this article
Wang Jinlei, Li Ke, Zhao Liang. Platelet-camouflaged silver nanoparticle hydrogel accelerates wound healing in type 1 diabetic rats[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2659-2666.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
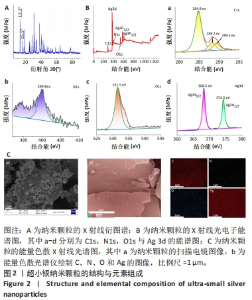
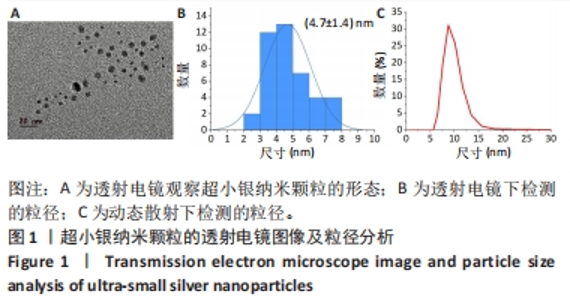
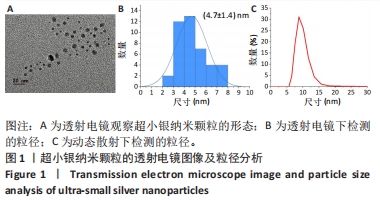
为了确定usAgNPs中Ag元素的化学状态,基于X射线光电子能谱测试研究了usAgNPs样品中的4种元素(C、N、O和Ag),C1s发射光谱可分为3个主峰,288.2 eV中的峰来自于O=C-OH中的羧酸碳,而284.8 eV和289.3 eV中的峰与来自苯环中的芳基碳有关;N1s信号图显示,399.8 eV的峰分别可归因于配体中的-NH2基团;O1s发射光谱图显示531.5 eV处的组分是典型的金属-氧(Ag-O)键;Ag 3d水平的光谱显示,Ag 3d3/2和Ag 3d5/2自旋-轨道光电子的结合能为368.5 eV和374.5 eV(图2B)。根据之前的报告[23],峰值在368.5 eV和374.5 eV处归因于金属银,这2个自旋能量分离为6.0 eV的峰,进一步表明usAgNPs样品中存在Ag[24]。 通过能量色散X射线光谱检测到usAgNPs中C、N、O和Ag的存在(图2C),结果表明Ag元素均匀分散到usAgNPs中。"
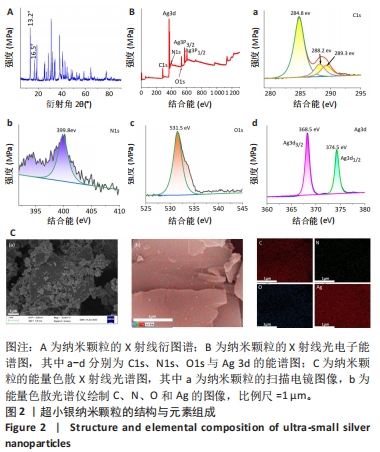
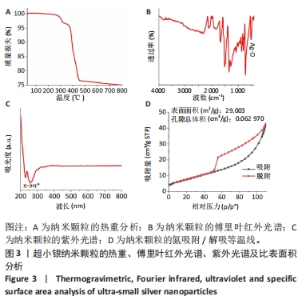
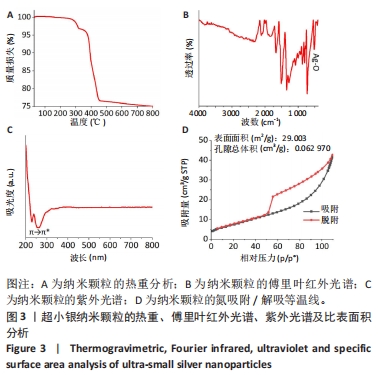
图3A热重分析曲线显示,usAgNPs降解分2个步骤:当温度在260 ℃时水分蒸发,usAgNPs从300 ℃开始分解,在400 ℃下完成羟基、羧基和吡啶环的分解,质量损失约为25%;当温度达到400 ℃后,usAgNPs的质量不再降低[25]。 图3B傅里叶红外光谱中显示,1 517 cm?1处的峰值归因于C-N拉伸,1 729 cm?1处的峰值归因于N-H拉伸;在1 288 cm?1和1 350 cm?1处发现的2个峰很可能分别对应于C=C和C≡O拉伸,1 288 cm?1处的峰值是由C-O-C对称拉伸引起的;1 082 cm?1处的峰值与C-O拉伸有关;881-762 cm?1范围内的几个峰可能归因于C-H键合,特别是摇摆振动;512 cm?1属于500-577 cm?1之间的细峰,这可能与Ag-O的振动[26],779 cm-1除的峰值也归因于Ag-O振动。 图3C紫外光谱显示,usAgNPs在252-310 nm处具有较强的吸附带,表明吡啶环的π → π*跃迁[27]。由于材料的吸附能力与其表面积、孔隙体积和多孔结构密切相关[28],为了证明usAgNPs可以吸附PL,分析了其氮吸附/解吸等温线。在图3D中,usAgNPs具有典型Ⅳ等温线,N2吸附下usAgNPs的表面积为29.003 m2/g、孔容为0.062 970 cm3/g,结果表明usAgNPs具有良好的吸附能力。"
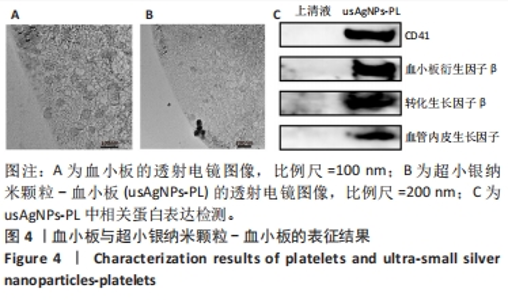
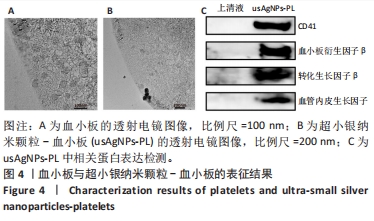
2.2 usAgNPs-PL的表征结果 透射电镜证明了PL表面成功吸附了usAgNPs,图4A显示了单纯PL的结构特征,外形为无色,圆盘状小体,部分表现为不规则的形状,无细胞核,展现出良好的结构[29]。当PL表面吸附usAgNPs后,通过透射电镜观察到usAgNPs在PL中的分布情况(图4B)。透射电镜表明了在(未染色的)PL表面能观察到黑色球形颗粒,说明usAgNPs都被空心状囊泡吸附[30-31]。当usAgNPs与PL混合后,收集小球获得usAgNPs-PL,并选择残留的上清液作为对照,Western blot检测结果显示(图4C),与上清液相比,usAgNPs-PL被证实含有血小板衍生因子β、转化生长因子β、血管内皮生长因子和CD41。"
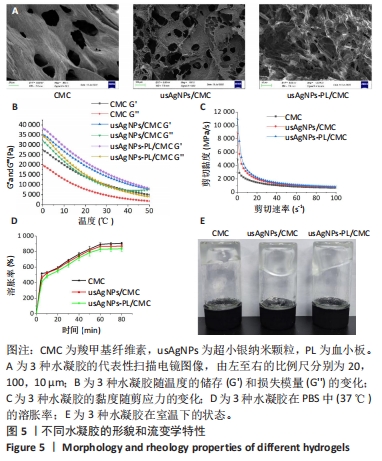
2.3 usAgNPs-PL/CMC水凝胶的表征结果 扫描电镜下可见3种水凝胶都具有均匀多孔的三维网状结构,usAgNPs-PL的加入不影响凝胶体系中的微观结构,见图5A。为了研究水凝胶的流变行为,进行了动态温度扫描流变测试,计算水凝胶储能模量(G’)和损耗模量(G’’),图5B显示3种水凝胶的G’和G’’在20-40 ℃之间相对稳定,各组水凝胶的G’值始终大于G’’,所以水凝胶仍然是一个弹性网络。当剪切速率轻微增加时,3种水凝胶的黏度迅速下降;当剪切速率逐渐增加时,3种水凝胶的黏度缓慢下降,表现出剪切稀化行为(图5C)。图5D显示CMC水凝胶、usAgNPs/CMC和usAgNPs-PL/CMC水凝胶在80 min后的溶胀率分别为907.3%,870.7%和836%,结果表明3种水凝胶都具有较强吸水能力,有助于吸收创面表面渗出液、保持创面干燥、加速伤口愈合过程、控制药物的释放。图5E显示3种水凝胶在室温下的状态,表明所有水凝胶都处于凝胶状态。"
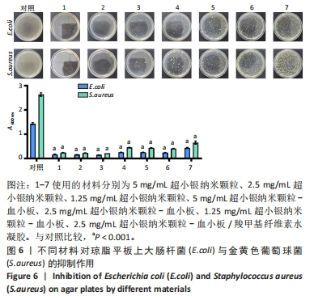
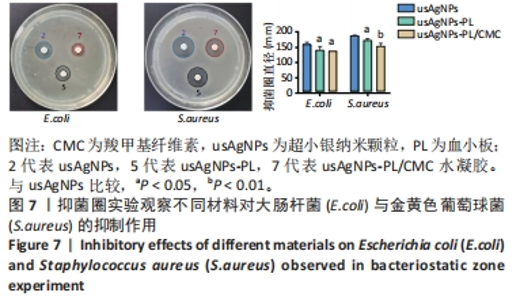
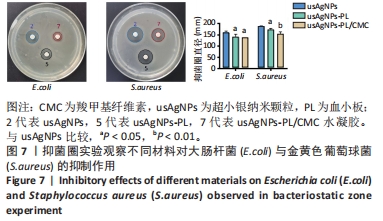
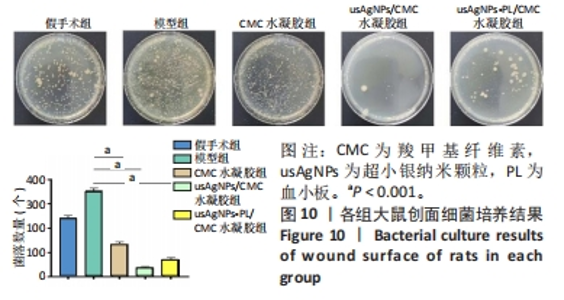
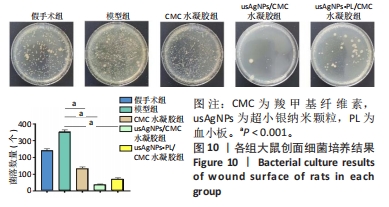
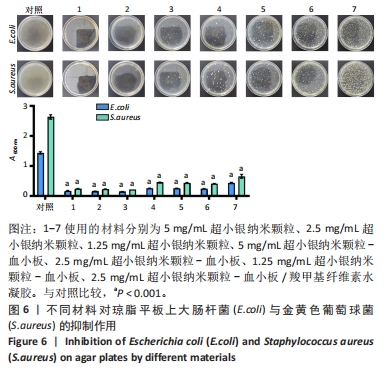
2.4 usAgNPs-PL/CMC水凝胶的体外抗菌性能 UsAgNPs和usAgNPs-PL通常作为创面愈合的液体形式给药,然而这些传统剂型很难停留在创面表面,并且存在涂抹后药物容易丢失和排出、可用性差且时间短等问题,对创面愈合产生不利影响[32]。相比之下,水凝胶表现出了兼容性,可以延缓药物释放,从而避免多次、重复给药。因此,实验选择CMC水凝胶作为载体来加载usAgNPs-PL复合物。 为了解usAgNPs-PL/CMC水凝胶的最佳抗菌效果,筛选不同质量浓度的usAgNPs和usAgNPs-PL,以此确定最终装载的usAgNPs-PL/CMC水凝胶的浓度,研究其对革兰阴性和革兰阳性菌的抗菌活性。如图6所示,usAgNPs-PL/CMC水凝胶明显抑制大肠杆菌和金黄色葡萄球菌的生长。由于高浓度的Ag具有显著的毒性,因此装载水凝胶的最佳杀菌浓度是含有2.5 mg/mL的usAgNPs。可以看到usAgNPs、usAgNPs-PL和usAgNPs-PL/CMC水凝胶都明显抑制细菌的生长,经过usAgNPs-PL(其中usAgNPs的质量浓度为2.5 mg/mL)和usAgNPs-PL/CMC水凝胶处理后存活的细菌比usAgNPs多,虽然与usAgNPs相比,usAgNPs-PL和usAgNPs-PL/CMC水凝胶具有较低的抗菌效果,但usAgNPs-PL和usAgNPs-PL/CMC水凝胶均对细菌具有有效的抑制率。抑菌圈实验发现,usAgNPs/CMC水凝胶显著增加大肠杆菌和金黄色葡萄球菌的抑菌范围(图7)。"
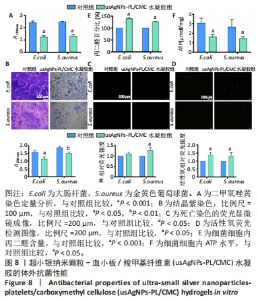
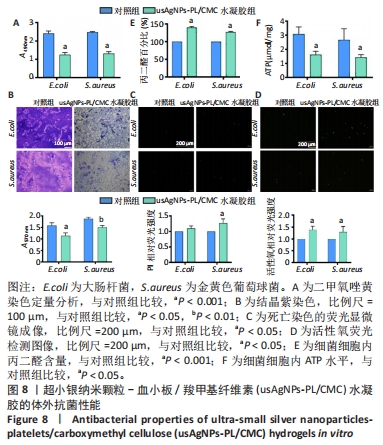
生物膜的产生是导致抗生素耐药性和治疗效果差的原因之一,作者认为usAgNPs-PL/CMC水凝胶对生物膜具有高渗透性,对大肠杆菌和金黄色葡萄球菌的生物膜形成具有良好的抑制和清除作用,因此实验采用结晶紫染色和XTT测定两种方法来评价细菌生物膜的形成。XTT定量分析结果显示,usAgNPs-PL/CMC水凝胶对大肠杆菌和金黄色葡萄球菌生物膜的破坏作用较强,能够抑制细菌繁殖(图8A)。然后通过结晶紫染色确定生物膜的生物量,结晶紫染料可以与细菌细胞膜表面的分子和多糖结合。从图8B可以看出,对照组呈深紫色,表明细菌形成了相对完整的生物膜;然而,usAgNPs-PL/CMC水凝胶组形成了几乎透明的薰衣草色,这证明了usAgNPs-PL/ CMC水凝胶可以抑制细菌形成生物膜和生长繁殖。核酸染料PI能与死细菌结合后发出红色荧光,图8C显示对照组表现出微弱的红色荧光,相比之下,usAgNPs-PL/CMC水凝胶组显示较强的红色荧光,说明usAgNPs-PL/CMC水凝胶具有更重要的抗菌能力。基于上述抗菌实验结果,作者尝试探索usAgNPs-PL/CMC水凝胶的抗菌机制,进行了活性氧、丙二醛和ATP水平检测,其中,细菌产生大量活性氧是导致其死亡的主要原因。图8D显示usAgNPs-PL/CMC水凝胶诱导大量活性氧,细胞内活性氧积累的主要后果之一是逐渐建立的氧化应激对膜完整性的损害。因此继续使用丙二醛方法检测细胞脂质过氧化,以确定膜损伤的程度,如图8E显示,usAgNPs-PL/CMC水凝胶组活性氧诱导的丙二醛在大肠杆菌中达到149.94%、在金黄色葡萄球菌中达到127.08%,均高于对照组。为了评估usAgNPs-PL/CMC水凝胶对细菌代谢的影响,测定了细胞内ATP水平,如图8F所示,usAgNPs-PL/CMC水凝胶组中的ATP水平明显低于对照组,表明usAgNPs-PL/CMC水凝胶能够抑制细菌(大肠杆菌和金黄色葡萄球菌)ATP水平。"
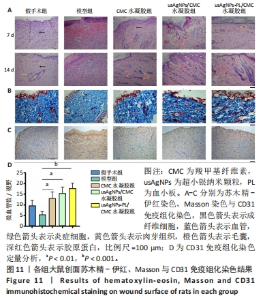
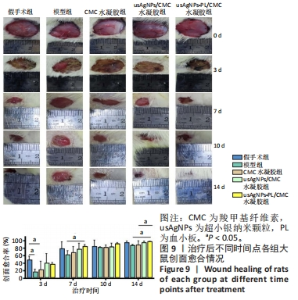
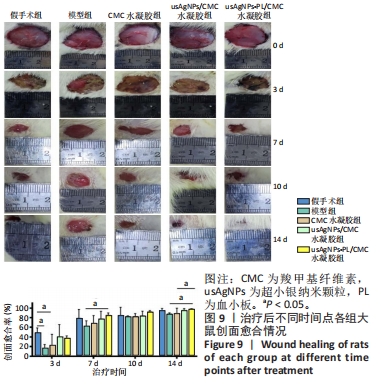
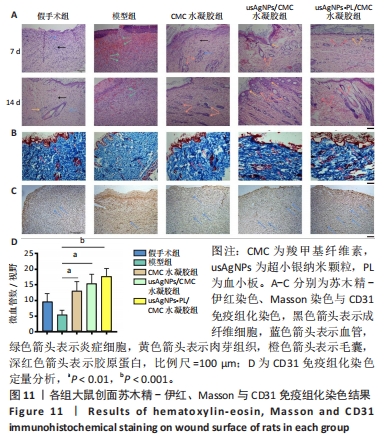
2.5.3 各组大鼠创面愈合质量 采用苏木精-伊红染色研究不同给药大鼠糖尿病创面的病理结构变化[33-34],结果见图11A所示。苏木精-伊红染色显示,治疗7 d时,模型组创面边缘有较多中性粒细胞浸润,肉芽组织疏松,成纤维细胞较少,没有表皮形状;usAgNPs-PL/CMC水凝胶处理后,创面部位炎症细胞较少,肉芽组织和血管形成较多,出现更多的再上皮化和完整的表皮。治疗14 d时,各治疗组出现了再生真皮的组织结构,新生肉芽组织表皮均完整且厚,而模型组未观察到上述现象,在第14天仍有明显的炎症细胞浸润。 由于适当的胶原蛋白沉积和重塑可以改变组织的拉伸强度[35],能产生更好的治疗效果,因此进一步通过Masson染色评估新生的胶原蛋白,评估真皮形成的效率,结果见图11B。Masson染色结果显示,与模型组和CMC水凝胶组相比,usAgNPs-PL/CMC水凝胶组和usAgNPs/CMC水凝胶组治疗第14天创面有更多形成和排列整齐的蓝色胶原纤维,其中usAgNPs-PL/CMC水凝胶组可见堆积的平行的成熟胶原纤维,而在其他治疗组中仅观察到部分增殖的胶原纤维。Masson染色结果表明,usAgNPs-PL/CMC水凝胶持续释放因子可以改善创面的胶原蛋白沉积,从而促进皮肤再生和糖尿病全层创面的修复。 血管生成在整个创面修复过程中至关重要,血管提供祖细胞、氧气和营养物质,以维持创面的增殖和重塑[36],许多研究表明血管生成受损会导致糖尿病患者的慢性创面不愈合[37]。 CD31是一种血管生成标志物,在内皮细胞表面表达[38]。治疗第7天的CD31免疫组化染色显示,CMC水凝胶组和usAgNPs/CMC水凝胶组可见中等数量的阳性细胞,usAgNPs-PL/CMC水凝胶组阳性细胞最多(图11C、D)。"
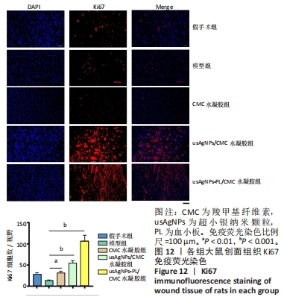
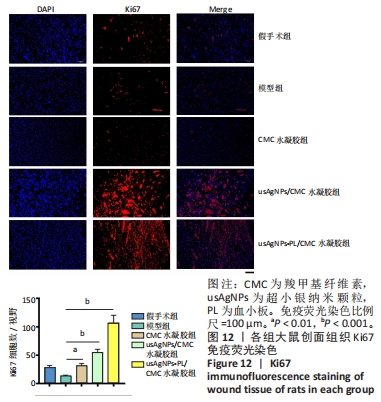
为了研究局部应用usAgNPs-PL/CMC水凝胶影响肉芽组织细胞的潜在机制,实验对增殖细胞标志物Ki67进行了免疫荧光染色[39],结果见图12。Ki67免疫荧光染色显示,治疗第7天时, usAgNPs-PL/CMC水凝胶组创面组织中Ki67的表达丰富,而CMC水凝胶组创面组织中Ki67的表达增加不明显;此外,usAgNPs/CMC水凝胶组创面组织中Ki67表达水平(阳性细胞)显著高于CMC水凝胶组,CMC水凝胶组Ki67表达和模型组相比略有升高,表明usAgNPs-PL/CMC水凝胶可以通过控制成纤维细胞在伤口组织中的增殖而加速创面愈合。这些结果与创面愈合率、创面组织学和血管生成结果一致,表明usAgNPs-PL/CMC水凝胶可以作为支持usAgNPs-PL的支架,延长其体外存活时间,并将其原位送至糖尿病创面。"
| [1] EVERETT E, MATHIOUDAKIS N. Update on management of diabetic foot ulcers. Ann N Y Acad Sci. 2018;1411(1):153-165. [2] FANG PH, LAI YY, CHEN CL, et al. Cobalt protoporphyrin promotes human keratinocyte migration under hyperglycemic conditions. Mol Med. 2022;28(1):71. [3] BUCH PJ, CHAI Y, GOLUCH ED. Treating Polymicrobial Infections in Chronic Diabetic Wounds. Clin Microbiol Rev. 2019;32(2):e00091-18. [4] GONZALEZ LS 3RD, SPENCER JP. Aminoglycosides: a practical review. Am Fam Physician. 1998; 58(8):1811-1820. [5] AMINOV RI. The role of antibiotics and antibiotic resistance in nature. Environ Microbiol. 2009;11(12):2970-2988. [6] SIRITONGSUK P, HONGSING N, THAMMAWITHAN S, et al. Two-Phase Bactericidal Mechanism of Silver Nanoparticles against Burkholderia pseudomallei. PLoS One. 2016;11(12):e0168098. [7] YUAN G, TIAN Y, WANG B, et al. Mitigation of membrane biofouling via immobilizing Ag-MOFs on composite membrane surface for extractive membrane bioreactor. Water Res. 2021;209:117940. [8] DEHNAVI SM, BARJASTEH M, AHMADI SEYEDKHANI S, et al. A Novel Silver-Based Metal-Organic Framework Incorporated into Nanofibrous Chitosan Coatings for Bone Tissue Implants. Int J Pharm. 2023;640:123047. [9] WANG D, LI S, LI F, et al. Thin film nanocomposite membrane with triple-layer structure for enhanced water flux and antibacterial capacity. Sci Total Environ. 2021;770:145370. [10] XIONG Y, CHEN L, LIU P, et al. All-in-One: Multifunctional Hydrogel Accelerates Oxidative Diabetic Wound Healing through Timed-Release of Exosome and Fibroblast Growth Factor. Small. 2022;18(1):e2104229. [11] HUSSAIN Z, THU HE, SHUID AN, et al. Recent Advances in Polymer-based Wound Dressings for the Treatment of Diabetic Foot Ulcer: An Overview of State-of-the-art. Curr Drug Targets. 2018;19(5):527-550. [12] REHMAN SRU, AUGUSTINE R, ZAHID AA, et al. Reduced Graphene Oxide Incorporated GelMA Hydrogel Promotes Angiogenesis For Wound Healing Applications. Int J Nanomedicine. 2019;14:9603-9617. [13] WASIAK J, CLELAND H, CAMPBELL F, et al. Dressings for superficial and partial thickness burns. Cochrane Database Syst Rev. 2013;2013(3):Cd002106. [14] ETULAIN J. Platelets in wound healing and regenerative medicine. Platelets. 2018; 29(6):556-568. [15] CECERSKA-HERYĆ E, GOSZKA M, SERWIN N, et al. Applications of the regenerative capacity of platelets in modern medicine. Cytokine Growth Factor Rev. 2022;64: 84-94. [16] KARTIKA RW, ALWI I, SUYATNA FD, et al. The role of VEGF, PDGF and IL-6 on diabetic foot ulcer after Platelet Rich Fibrin + hyaluronic therapy. Heliyon. 2021; 7(9):e07934. [17] YU W, YIN N, YANG Y, et al. Rescuing ischemic stroke by biomimetic nanovesicles through accelerated thrombolysis and sequential ischemia-reperfusion protection. Acta Biomater. 2022;140:625-640. [18] ZHAO X, CHANG L, HU Y, et al. Preparation of Photocatalytic and Antibacterial MOF Nanozyme Used for Infected Diabetic Wound Healing. ACS Appl Mater Interfaces. 2022;14(16):18194-18208. [19] FU X, GAO Y, YAN W, et al. Preparation of eugenol nanoemulsions for antibacterial activities. RSC Adv. 2022;12(6):3180-3190. [20] GENG X, QI Y, LIU X, et al. A multifunctional antibacterial and self-healing hydrogel laden with bone marrow mesenchymal stem cell-derived exosomes for accelerating diabetic wound healing. Biomater Adv. 2022;133:112613. [21] MOGHARBEL RT, ALKHAMIS K, FELALY R, et al. Superior adsorption and removal of industrial dye from aqueous solution via magnetic silver metal-organic framework nanocomposite. Environ Technol. 2023:1-17.doi: 10.1080/ 09593330.2023.2178331. [22] GUO H, ZHANG Y, ZHENG Z, et al. Facile one-pot fabrication of Ag@MOF(Ag) nanocomposites for highly selective detection of 2,4,6-trinitrophenol in aqueous phase. Talanta. 2017;170:146-151. [23] WANG B, YU J, SUI L, et al. Rational Design of Multi-Color-Emissive Carbon Dots in a Single Reaction System by Hydrothermal. Adv Sci (Weinh). 2020;8(1):2001453. [24] LIN YH, SIVAKUMAR C, BALRAJ B, et al. Ag-Decorated Vertically Aligned ZnO Nanorods for Non-Enzymatic Glucose Sensor Applications. Nanomaterials (Basel). 2023;13(4):754. [25] ZHANG M, WANG G, ZHANG X, et al. Polyvinyl Alcohol/Chitosan and Polyvinyl Alcohol/Ag@MOF Bilayer Hydrogel for Tissue Engineering Applications. Polymers (Basel). 2021;13(18):3151. [26] SEYEDPOUR SF, DADASHI FIROUZJAEI M, RAHIMPOUR A, et al. Toward Sustainable Tackling of Biofouling Implications and Improved Performance of TFC FO Membranes Modified by Ag-MOF Nanorods. ACS Appl Mater Interfaces. 2020; 12(34):38285-38298. [27] ZHANG M, WANG D, JI N, et al. Bioinspired Design of Sericin/Chitosan/Ag@MOF/GO Hydrogels for Efficiently Combating Resistant Bacteria, Rapid Hemostasis, and Wound Healing. Polymers (Basel). 2021;13(16):2812. [28] HARANDIZADEH AH, AGHAMIRI S, HOJJAT M, et al. Adsorption of Carbon Dioxide with Ni-MOF-74 and MWCNT Incorporated Poly Acrylonitrile Nanofibers. Nanomaterials (Basel). 2022;12(3):412. [29] YAN R, GU Y, RAN J, et al. Intratendon Delivery of Leukocyte-Poor Platelet-Rich Plasma Improves Healing Compared With Leukocyte-Rich Platelet-Rich Plasma in a Rabbit Achilles Tendinopathy Model. Am J Sports Med. 2017;45(8):1909-1920. [30] BANG KH, NA YG, HUH HW, et al. The Delivery Strategy of Paclitaxel Nanostructured Lipid Carrier Coated with Platelet Membrane. Cancers (Basel). 2019;11(6):807. [31] LIU T, LI M, TANG J, et al. An acoustic strategy for gold nanoparticle loading in platelets as biomimetic multifunctional carriers. J Mater Chem B. 2019;7(13): 2138-2144. [32] NING CC, LOGSETTY S, GHUGHARE S, et al. Effect of hydrogel grafting, water and surfactant wetting on the adherence of PET wound dressings. Burns. 2014; 40(6):1164-1171. [33] WANG Z, GAO S, ZHANG W, et al. Polyvinyl alcohol/chitosan composite hydrogels with sustained release of traditional Tibetan medicine for promoting chronic diabetic wound healing. Biomater Sci. 2021;9(10):3821-3829. [34] ZHOU Y, ZHANG XL, LU ST, et al. Human adipose-derived mesenchymal stem cells-derived exosomes encapsulated in pluronic F127 hydrogel promote wound healing and regeneration. Stem Cell Res Ther. 2022;13(1):407. [35] GUIDONI M, DE SOUSA JÚNIOR AD, ARAGÃO VPM, et al. Liposomal stem cell extract formulation from Coffea canephora shows outstanding anti-inflammatory activity, increased tissue repair, neocollagenesis and neoangiogenesis. Arch Dermatol Res. 2023;315(3):491-503. [36] KULESH AA, KUKLINA EM, SHESTAKOV VV. The relationship between serum and liquor il-1β, il-6, tnfα, il-10 levels and clinical, cognitive and functional characteristics in acute ischemic stroke. Klin Med (Mosk). 2016;94(9):657-662. [37] WAN R, WEISSMAN JP, GRUNDMAN K, et al. Diabetic wound healing: The impact of diabetes on myofibroblast activity and its potential therapeutic treatments. Wound Repair Regen. 2021;29(4):573-581. [38] GOURISHETTI K, KENI R, NAYAK PG, et al. Sesamol-Loaded PLGA Nanosuspension for Accelerating Wound Healing in Diabetic Foot Ulcer in Rats. Int J Nanomedicine. 2020;15:9265-9282. [39] YANG J, CHEN Z, PAN D, et al. Umbilical Cord-Derived Mesenchymal Stem Cell-Derived Exosomes Combined Pluronic F127 Hydrogel Promote Chronic Diabetic Wound Healing and Complete Skin Regeneration. Int J Nanomedicine. 2020;15: 5911-5926. [40] HUANG L, SHI Y, LI M, et al. Plasma Exosomes Loaded pH-Responsive Carboxymethylcellulose Hydrogel Promotes Wound Repair by Activating the Vascular Endothelial Growth Factor Signaling Pathway in Type 1 Diabetic Mice. J Biomed Nanotechnol. 2021;17(10):2021-2033. [41] VILLIOU M, PAEZ JI,DEL CAMPO A. Photodegradable Hydrogels for Cell Encapsulation and Tissue Adhesion. ACS Appl Mater Interfaces. 2020;12(34): 37862-37872. |
| [1] | Feng Ruiqin, Han Na, Zhang Meng, Gu Xinyi, Zhang Fengshi. Combination of 1% platelet-rich plasma and bone marrow mesenchymal stem cells improves the recovery of peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 985-992. |
| [2] | Zhang Kefan, Shi Hui. Research status and application prospect of cytokine therapy for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 961-967. |
| [3] | Chen Xiaofang, Zheng Guoshuang, Li Maoyuan, Yu Weiting. Preparation and application of injectable sodium alginate hydrogels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 789-794. |
| [4] | Wang Wu, Fan Xiaolei, Xie Jie, Hu Yihe, Zeng Min. Hydroxyapatite-polyvinyl alcohol/collagen-chitosan-gelatin composite hydrogel for repairing rabbit osteochondral defect [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 682-689. |
| [5] | Zhang Ya, Mu Qiuju, Wang Zilin, Liu Hongjie, Zhu Lili. Hydrogel loaded with platelet-rich plasma promotes wound healing in diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 690-696. |
| [6] | Shen Ziqing, Xia Tian, Shan Yibo, Zhu Ruijun, Wan Haoxin, Ding Hao, Pan Shu, Zhao Jun. Vascularized tracheal substitutes constructed by exosome-load hydrogel-modified 3D printed scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 697-705. |
| [7] | Zhu Liwei, Wang Jiangyue, Bai Ding. Application value of nanocomposite gelatin methacryloyl hydrogels in different bone defect environments [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 753-758. |
| [8] | Xu Jing, Lyu Huixin, Bao Xin, Zhang Yi, Wang Yihan, Zhou Yanmin. Application of near infrared responsive hydrogels in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 486-492. |
| [9] | Dai Jing, Liu Shasha, Shen Mingjing. Exosome-loaded injectable hydrogel for repairing bone defects around implants [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 347-354. |
| [10] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Effect of gelatin methacryloyl hydrogel loaded with salvianolic acid B on intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 380-386. |
| [11] | Wang Xinmin, Yan Wenkai, Song Yahui, Liu Fei. Leukocyte- and platelet-rich fibrin with autologous hamstring tendon for traumatic patella dislocation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 404-410. |
| [12] | Bi Yujie, Ma Dujun, Peng Liping, Zhou Ziqiong, Zhao Jing, Zhu Houjun, Zhong Qiuhui, Yang Yuxin. Strategy and significance of Chinese medicine combined with medical hydrogel for disease treatment [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 419-425. |
| [13] | Chen Pinrui, Pei Xibo, Xue Yiyuan. Function and advantages of magnetically responsive hydrogel in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 452-457. |
| [14] | Long Zhirui, Huang Lei, Xiao Fang, Wang Lin, Wang Xiaobei. Characteristics of hydrogel microspheres in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 472-478. |
| [15] | Zhu Yongzhao, Fang Chao, Zhao Fang, Zhang Qing, Zhao Dan. Mechanism by which lycium barbarum polysaccharides inhibit keratinocyte apoptosis in burn wounds via autophagy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3686-3691. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||