Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (17): 2759-2765.doi: 10.12307/2024.444
Previous Articles Next Articles
Research and application of electrospinning drug delivery systems containing traditional Chinese medicine
Zhang Shuzhi, Qu Pengfei, Han Junquan, Wang Hong
- Department of General Surgery, Second Teaching Hospital of Tianjin University of Traditional Chinese Medicine, Tianjin 300250, China
-
Received:2023-07-08Accepted:2023-08-14Online:2024-06-18Published:2023-12-16 -
Contact:Han Junquan, Master, Associate chief physician, Department of General Surgery, Second Teaching Hospital of Tianjin University of Traditional Chinese Medicine, Tianjin 300250, China -
About author:Zhang Shuzhi, Master, Attending physician, Department of General Surgery, Second Teaching Hospital of Tianjin University of Traditional Chinese Medicine, Tianjin 300250, China -
Supported by:National Natural Science Foundation of China, No. 82274522 (to WH)
CLC Number:
Cite this article
Zhang Shuzhi, Qu Pengfei, Han Junquan, Wang Hong. Research and application of electrospinning drug delivery systems containing traditional Chinese medicine[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2759-2765.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

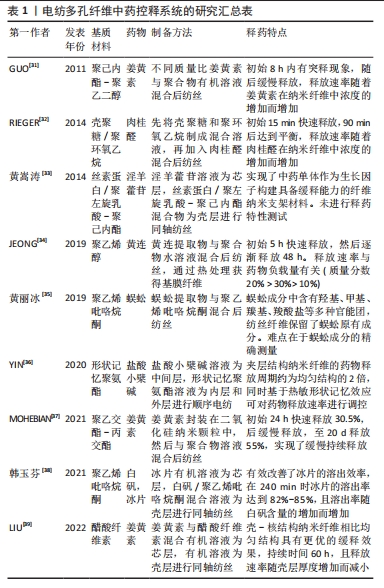
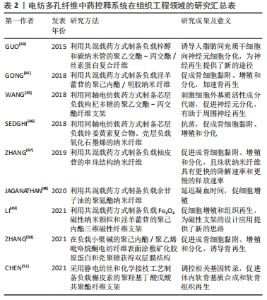
2.1 中药控释系统 中药在中国具有几千年的传承历史,并且很早就出现了中药释药系统的雏形——丸剂和膏剂。但是由于中药的组成复杂,对药剂基础组成物质的准确判断非常困难,体内突释等因素迟滞了其更广泛的应用。现代科技和医学的发展促使中药控释系统技术得以不断提升并实现不同的给药方式和药物释放特征。 根据释药特点的不同分类:①定速释药系统:药剂以零级或接近零级释放并能长效维持血药浓度的相对稳定,从而实现减少服药次数并增加服药的顺应性[12],目前已在临床应用的缓控释制剂大部分是这一类型。②定位释药系统:通过将药物在体内指定部位进行释放实现局部治疗强化,如将五味子木脂素类成分药剂制成胃漂浮片增加其在胃肠道的停留时间和生物利用度[13]。③定时释药系统:可依据人体生物节律变化按临床治疗需要定时单次或多次释放药物,如以杜仲提取物速释片芯为崩解剂制备的药片可实现瞬间起漂,并维持至六七小时后脉冲释放药物,从而为节律性疾病的治疗提供了新的思路方法[14]。 根据载体或剂型的不同分类:①纳米粒子释药系统,药物可通过包裹、吸附及溶解等多种方法负载于纳米粒子的表面和内部,其释放行为主要由粒子载体的物理化学性质和作用的位置所决定,在提高药物稳定性、控制释放及靶向给药等方面均展示出一定的优势[15]。②水凝胶释药系统,具有生物相容性好、载药效率高、稳定性好及制备简单等优势,同时也可通过调控水凝胶载体的成分及结构实现药物控释[16],如壳聚糖季铵盐/氧化魔芋葡甘露聚糖水凝胶即表现出了良好的抗溶血性能和长效抗菌性。③微球(丸)释药系统,一般为几微米到几百微米的球丸状高聚物,药物被包封于微球内部使其免于被胃酸或其他组织液破坏,适合于靶向给药[17]。④电纺纤维释药系统,可以有效提高中药的分散性和稳定性,降低毒副反应,合成靶向药剂[18-19]。"

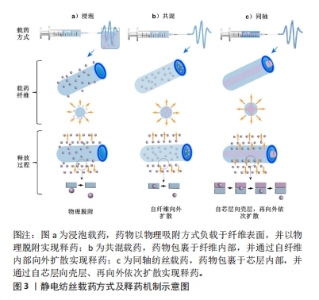
2.2 电纺多孔纤维中药控释系统的构建 2.2.1 基质材料选择 静电纺丝的基质材料作为理想的药物载体和控制药物释放的关键,应具有以下特点:生物相容性好,无毒无刺激,性质稳定,具有一定的强度、弹性和可塑性,能与药物配伍,不影响药物的药理作用和含量测定,具有抗菌作用和符合要求的黏度、渗透性、亲水性和溶解性等,可生物降解,降解时间可调,降解后形成无毒的产物。整体而言,静电纺丝纤维基材可分为两大类:天然高分子和合成高分子。 天然高分子聚合物主要包括壳聚糖、明胶、丝素蛋白、白蛋白、海藻酸盐和淀粉等[20-22]。天然聚合物生物相容性优良,展现出了高细胞亲和力,有利于生物细胞在其表面攀附和增殖,同时其也容易进行各种改性处理进一步提高性能。不足之处在于天然聚合物的力学强度较差,在生物体内稳定性较差导致其降解较快,同时在纺丝过程中部分天然聚合物会因为蛋白变性等现象影响其纤维性能。 人工合成高分子聚合物具有较高的机械强度和稳定性,且在合成和改性方面具有很大的灵活性,可以通过工艺优化调整其形态、结构、降解性能和力学性能等从而扩大应用范围,主要包括聚乳酸、聚乙二醇、聚己内酯、聚乙交酯-丙交酯和聚乙烯醇等[23-26]。但是合成聚合物与细胞亲和力较低,表面缺乏细胞识别位点导致细胞黏附和增殖能力降低。近年来,为了克服单一材料的缺点,研究者们选择将天然与合成聚合物进行共混制备复合基质材料,从而发挥二者的优势[27-28]。 2.2.2 负载中药的选择 载药纳米多孔纤维医用材料的治疗效果主要取决于其所负载的物质,然而传统中药种类繁多,导致其分类理论和方法众多。一般按照药物的来源和性质大致可分为3类:①植物中药,如板蓝根、黄连和金银花等;②动物中药,如水蛭、蜈蚣和羚羊角等;③矿物中药,如白矾、芒硝和朱砂等。总体而言,植物中药在中国传统中药系统中占80%以上,目前在电纺纤维中药控释系统中所研究应用的药剂也是以各类植物中药提取物为主,这主要得益于植物提取物成分单纯,易溶于有机溶剂,容易制备纺丝溶液。其他两类用在电纺纤维载药系统的报道较少,特别是动物中药。已有研究人员初步探索了负载蜈蚣提取物、白矾等药物纳米纤维的制备方法,但由于动物和矿物药成分复杂、药用成分提取和质量控制困难、缺乏精确有效的测试手段等因素,仍需进一步对其药物控制释放及疗效等进行研究并开发动物药新剂型。 2.2.3 载药方式的选择 基于负载物质多样性的特点及其在纳米纤维上的分布要求,静电纺丝纳米多孔纤维也具有多种载药方式,从而导致药物的释放结果也存在差异,目前常用的载药方式包括浸泡载药、共混载药和同轴纺丝载药等方式,见图3。"
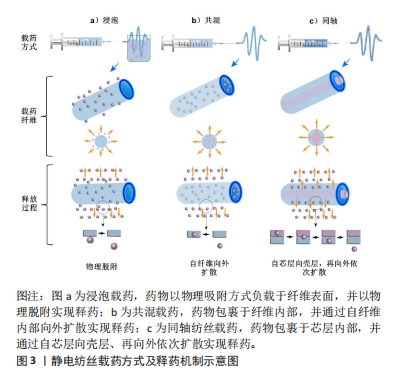

浸泡载药:首先采用静电纺丝制备纳米多孔纤维毡,再将其放在含有药物或载药纳米粒子的溶液中浸渍一定时间获得载药纤维。基于纳米纤维高比表面积特性可实现较高的载药量,然而负载中药主要通过物理吸附方式与电纺纤维结合,初期药物突释现象明显且药物持续释放时间较短。 共混载药:相对于浸泡式载药,它最大的特点是先将药物或载药纳米颗粒混合在聚合物溶液中,再用静电纺丝制备成载药纳米纤维。由于该方法的载药效率、药物分布及释放特征等与基质材料的理化性质及其与药物的相互作用息息相关,因此在共混载药时要求药物与基质材料能共溶解于同种有机溶剂或水溶液,减少由于药物不均匀分布导致的初始突释现象。共混载药方法可以实现将药物更均匀地分布于纺丝纤维中,对中药活性成分突释现象的控制较浸泡式更好,但可能影响包裹生物活性分子的活性,且药物包封率较低[29]。同时,共混方式只能制备单层纳米纤维,这种单一成分结构导致其混合药物一般会在48 h内释放。近年来有研究人员先将药物封装到介孔纳米微球中,再与聚合物混合进行纺丝,从而在满足不同特性药物与聚合物共混需求的同时,进一步延长了药物释放时间。 同轴纺丝载药:控制高聚物在静电纺丝过程中与药物或载药粒子溶液分别进入2个同心的喷丝头管,在内外层界面拉伸力和剪切应力作用下制备出具有壳-核结构的双层纳米纤维[30]。一般以可降解高分子材料为壳层,中药及其他活性成分作为芯层,随着聚合物的降解,芯层的药物实现平稳释放。相对于前述载药方式,同轴纺丝纤维的外层聚合物纤维可起到对内层中药活性成分的保护作用,从而进一步增强其缓释效果,且载药量更大,同时也可通过改变壳层厚度对药物的释放行为进行调控。但该方法需要壳层和芯层原料溶液必须在纺丝过程中同步固化,技术敏感性较高。 其他载药方式:顺序电纺可制备具有三明治结构的多层纳米纤维,通过在不同层上负载不同药物或活性成分实现多功能化,并对药物释放率进行调控。在同轴电纺的基础上改进的多轴电纺可实现药物同时负载在多轴载药纤维的不同层中并呈现梯度分布,同时多层结构也具有调控各层药物浓度和药物释放速率的作用,有助于拖尾释放现象。文章总结了近年来电纺多孔纤维中药控释系统的相关研究进展[31-39],见表1。前期主要采用于浸泡和共混方式,这得益于其工艺较简单,对设备要求较低,但是药物与基质材料需共溶解于同种有机溶剂,因负载药物均为植物提取物,在研究中集中于通过对溶液理化性质的优化不断扩展其释药特性和适应场景,如采用水做溶剂并结合热处理方式制备载药纤维,避免了有机溶剂的引入,为水溶性药物的负载提供了新的思路。近年来同轴、顺序电纺等更先进工艺被不断采用制备多层结构载药纤维,外层能够对内层中药活性成分起到保护作用,且通过改变壳层厚度对药物的释放行为进行调控,从而使其持续释药时间显著增加;同时也对动物和矿物中药进行了研究,但主要集中于载药工艺方面的探索。"
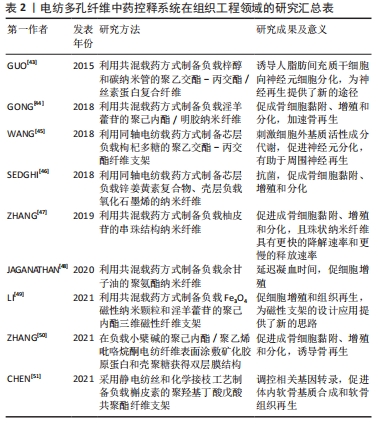
2.3 电纺多孔纤维中药控释系统在医学领域的应用 静电纺丝多孔纳米纤维接近于天然的细胞外基质,有利于蛋白的吸附和细胞的黏附,同时通过负载中药活性成分并以可控的方式释放,成功制备出多种电纺多孔纤维中药控释系统,并应用在骨、软骨、神经和皮肤修复等领域[40]。 2.3.1 组织工程领域 组织工程是利用特定组织细胞、活性成分和生物材料的结合,在体外或体内维持、修复、再生或改善损伤组织和器官功能。对于骨组织修复而言,静电纺丝中药控释系统负载药剂的选择原则是能够促进成骨细胞的黏附、增殖和分化,具有良好的抗菌性能,不影响其他活性成分或生长因子作用等,也有研究表明部分中药活性成分能够作为骨向分化的替代生长因子[41]。从构建策略角度来看,一种方法是将中药单体或提取物直接负载在电纺纤维上,其优势是工艺较简单,单体提取物的作用机制较为清晰,如传统中药提取物淫羊藿苷、黄芩苷和柚皮苷等均展示出了促进成骨诱导的骨髓间充质干细胞增殖和成骨分化,加速骨再生的效果,与电纺纳米纤维联合构建控释系统在组织工程方面有着较大的潜力。另一方面,研究者们也尝试将多种中药成分混合,或将中药与其他材料混合后负载至电纺纤维。如将植物中药冰片与矿物中药白矾联合载药,将氧化石墨烯、磁性纳米颗粒、碳纳米管等纳米材料与中药联合载药等,结合同轴、多轴和顺序纺丝等工艺,进一步调控药物的释药特性,发挥药剂药效,使复合纤维同时具有促细胞黏附增殖、抗菌活性、热敏形状记忆效应等复合功能。 软骨组织由于没有血供致使其自身修复能力较弱,构建可以提高软管细胞活性、促进软骨组织形成的功能化软骨组织工程支架在治疗软骨损伤中显示出巨大潜力。其中利用中药有效成分作为外源性活性因子,联合可模拟细胞外基质结构的电纺纤维为软骨组织的高效再生提供了新的思路[42]。近年来,淫羊藿苷、槲皮素、人参皂苷、牛蒡子苷元、柚皮苷、姜黄素及丹参酮ⅡA等中药成分都已被研究证明具有促进软骨细胞增殖、分化和功能性表达,减轻细胞在炎症因子刺激下的凋亡的作用,被用作电纺纤维中药控释系统的负载药剂。同时结合表面处理工艺,也可有效改善支架亲疏水性和降解性能。可见,与蛋白多肽类活性因子相比,中药活性成分展示出了更稳定的结构、药效持续时间更长、成本更低的优势,同时结合抗炎、抗氧化、抗脂质和抗凋亡等特殊作用,可实现多重功效复合调控组织细胞的生理活动。 在神经组织工程方面,控释系统构建策略主要集中于调控神经元细胞的增殖、分化、中性生长和神经间隙弥合等行为,如负载中药提取物梓醇、枸杞多糖等电纺纤维支架具有诱导人脂肪间充质干细胞向神经元细胞分化的功能。近年来研究人员也在中医理论指导下,从中医药活血化瘀、益气、补肾填精等治法与方药方面对中医药干预干细胞诱导分化过程进行了相关探讨。梓醇和枸杞多糖均是具有神经保护功能的药物,以上研究成果也表明其用作中药控释系统可作为神经再生的一种新途径。文章总结了近年来电纺多孔纤维中药控释系统在组织工程领域的应用研究进展[43-51],见表2。"
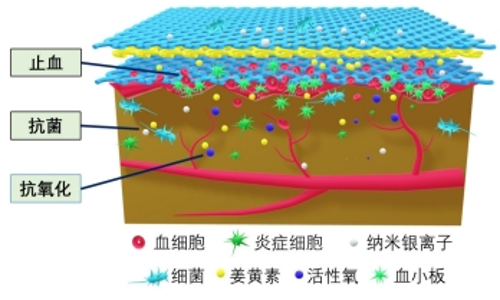
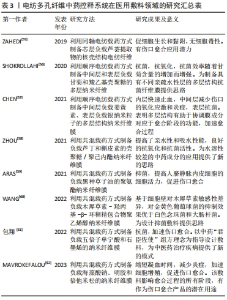
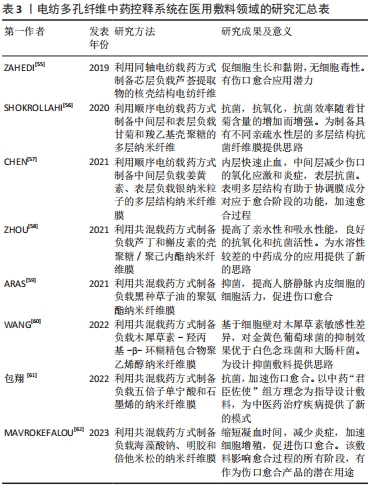
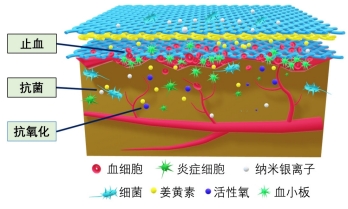
2.3.2 医用敷料 静电纺丝纳米纤维膜具有良好的机械性能、透气性和生物相容性,是制备医用敷料的理想材料,同时通过负载中药活性成分并控制释药特征,是提高药物生物利用度、对抗细菌感染、促进伤口愈合的有效手段[52]。中药活性成分在医用敷料中所起的作用大致有3类:①止血:如海藻酸钠作为医用敷料中最常用中药,与钙离子结合时可以起到很好的止血作用,研究结果也表明负载海藻酸钠的纳米纤维相对于明胶纤维膜具有更强的促进皮肤伤口愈合的能力。②抗菌:静电纺丝敷料负载姜黄素、五倍子单宁酸、槲皮素、木犀草素及甘菊等中药活性成分均展示出了良好的抑菌抗菌作用,研究表明其主要是通过损伤细胞膜、限制能量代谢、抑制细胞膜转运蛋白基因表达等作用机制实现对不同细菌的杀伤。同时,具有抗氧化性的中药成分也能够有效消除创伤部位残留积累的多余活性氧,避免形成二次损伤。③促进细胞增殖黏附:如负载芦荟提取物的电纺纤维在促进细胞生长和黏附的同时不产生任何细胞毒性作用。由于中药成分的复杂性,一种中药可能同时具有以上多种功效。同时,多种中药活性成分的混合使用也能够发挥更好的效果,如含有金盏花提取物、玉米蛋白及阿拉伯树胶的纳米纤维复合支架具有良好的抗菌性能和生物相容性,同时促进了成纤维细胞的增殖和黏附[53]。聚乙烯醇/黄芪多糖/黄芪甲苷纳米复合纤维可抑制伤口炎症的发生,同时增强胶原纤维的沉积和再生上皮的修复,有效加强糖尿病大鼠的伤口愈合[54]。同时研究者也将中药与其他材料同时负载,如中间层负载姜黄素、表层负载银纳米粒子的多层结构纳米纤维膜也展示出了更优的止血和抗菌性能,见图4。文章总结了近年来电纺多孔纤维中药控释系统在医用敷料领域的应用研究进展[55-62],见表3。电纺多孔纤维中药控释系统的早期研究集中于医用敷料方面,研究报道较多,所负载的中药成分大部分均同时具有抑菌抗菌和止血功能,近期研究发现部分药物也有促细胞黏附增殖的作用。近年来研究不断细化,如通过对中药成分的调控可实现对金黄色葡萄球菌、大肠杆菌等不同细菌的有效杀灭;调控载药纤维的亲水性和吸水性能,为水溶性较差的中药成分的应用提供了新的思路;将石墨烯、纳米颗粒、西药成分等与中药联合载药,并结合同轴、多轴和顺序纺丝等工艺,进一步调控药物的释药特性,发挥药剂药效。"
| [1] WANG WY, ZHOU H, WANG YF, et al. Current policies and measures on the development of traditional Chinese medicine in China. Pharmacol Res. 2021;163:105187. [2] WANG RN, ZHAO HC, HUANG JY, et al. Challenges and strategies in progress of drug delivery system for traditional Chinese medicine Salviae Miltiorrhizae Radix et Rhizoma (Danshen). Chin Herb Med. 2021;13(1): 78-89. [3] GAO F, HU Y, FANG G, et al. Recent developments in the field of the determination of constituents of TCMs in body fluids of animals and human. J Pharmaceut Biomed. 2014;87:241-260. [4] PARK H, OTTE A, PARK K. Evolution of drug delivery systems: from 1950 to 2020 and beyond. J Control Release. 2022;342:53-65. [5] ADEPU S, RAMAKRISHNA S. Controlled drug delivery systems: current status and future directions. Molecules. 2021;26(19):5905. [6] GORAIN B, PANDEY M, LENG NH, et al. Advanced drug delivery systems containing herbal components for wound healing. Int J Pharm. 2022;617: 121617. [7] KAPAHI H, KHAN NM, BHARDWAJ A, et al. Implication of nanofibers in oral drug delivery. Curr Pharm Des. 2015;21(15):2021-2036. [8] 高仓健,杨振,刘舒云,等.静电纺丝技术在肩袖损伤修复中的应用[J].中国组织工程研究,2022,26(4):637-642. [9] LI WJ, LAURENCIN CT, CATERSON EJ, et al. Electrospun nanofibrous structure: a novel scaffold for tissue engineering. J Biomed Mater Res. 2002;60(4):613-621. [10] LIU X, YANG Y, YU D, et al. Tunable zero-order drug delivery systems created by modified triaxial electrospinning. Chem Eng J. 2019;356:886-894. [11] HAN D, YU X, CHAI Q, et al. Stimuli-responsive self-immolative polymer nanofiber membranes formed by coaxial electrospinning. ACS Appl Mater Interfaces. 2017;9(13):11858-11865. [12] 李伟铭,于强,徐保利,等.中药缓控释制剂研究进展[J].中国中医药现代远程教育,2023,21(1):200-203. [13] 邹翔,宫甜,朱室儒,等.五味子总木脂素胃漂浮片的制备及质量评价[J].中国药房,2020,31(11):1336-1341. [14] 耿燕娜,娄婷婷,武毅君,等.杜仲漂浮型脉冲释药片的研制[J].中国药业,2020,29(7):82-86. [15] WONG PT, CHOI SK. Mechanisms of drug release in nanotherapeutic delivery systems. Chem Rev. 2015;115(9):3388-3432. [16] 毕玉杰,马笃军,彭力平,等.中医药联合医用水凝胶治疗疾病的策略及意义[J].中国组织工程研究,2024,28(3):419-425. [17] HE Q, ZHANG J, LIAO Y, et al. Current advances in microsphere based cell culture and tissue engineering. Biotechnol Adv. 2020;39:107459. [18] GANESH KR, RAJAN TP. A review on electrospinning of natural bio herbs blended with polyvinyl alcohol nanofibres for biomedical applications. J Nat Fibers. 2022;19(15):11984-12003. [19] 谢晓峰,周寒璞,赵聪,等.中药白芨/聚乙烯醇纳米静电纺丝膜的制备和生物相容性评价[J].肝胆胰外科杂志,2020,32(10):619-623. [20] TAN G, WANG L, PAN W, et al. Polysaccharide electrospun nanofibers for wound healing applications. Int J Nanomedicine. 2022;17:3913-43931. [21] CHEN K, LI Y, LI Y, et al. Silk fibroin combined with electrospinning as a promising strategy for tissue regeneration. Macromol Biosci. 2023;23(2): 2200380. [22] CAO P, WU G, YAO Z, et al. Effects of amylose and amylopectin molecular structures on starch electrospinning. Carbohydr Polym. 2022;296(15): 119959. [23] YANG Q, GUO J, ZHANG S, et al. PVA/PEO/PVA-g-APEG nanofiber membranes with cytocompatibility and anti-cell adhesion for biomedical applications. Colloids Surf A. 2023;657:130638. [24] HUANG J, ZHOU X, SHEN Y, et al. Asiaticoside loading into polylactic-co-glycolic acid electrospun nanofibers attenuates host inflammatory response and promotes M2 macrophage polarization. J Biomed Mater Res A. 2020; 108(1):69-80. [25] ZHAO XH, NIU YN, MI CH, et al. Electrospinning nanofibers of microbial polyhydroxyalkanoates for applications in medical tissue engineering. J Polym Sci. 2021;59(18):1994-2013. [26] JI X, GUO J, GUAN F, et al. Preparation of electrospun polyvinyl alcohol/nanocellulose composite film and evaluation of its biomedical performance. Gels. 2021;7(4):223. [27] ALIMOHAMMADI M, FAKHRAEI O, MORADI A, et al. Controlled release of azithromycin from polycaprolactone/chitosan nanofibrous membranes. J Drug Deliv Sci Tec. 2023;71:103246. [28] 崔志香,司军辉,宋来瑞,等.聚乙烯醇/壳聚糖复合纳米纤维载药体系制备及释药性能[J].高分子材料科学与工程,2015,31(8):185-190. [29] CHENG H, YANG X, CHE X, et al. Biomedical application and controlled drug release of electrospun fibrous materials. Mater Sci Eng C. 2018;90:750-763. [30] 马玥珑,李佳,王虹.静电纺丝制备载药纳米纤维的研究进展[J].哈尔滨理工大学学报, 2021,26(5):130-140. [31] GUO G, FU S, ZHOU L, et al. Preparation of curcumin loaded poly (ε-caprolactone)-poly (ethylene glycol)-poly (ε-caprolactone) nanofibers and their in vitro antitumor activity against glioma 9L cells. Nanoscale. 2011;3(9):3825-3832. [32] RIEGER K, SCHIFFMAN J. Electrospinning an essential oil: cinnamaldehyde enhances the antimicrobial efficacy of chitosan/poly (ethylene oxide) nanofibers. Carbohydr Polym. 2014;113:561-568. [33] 黄嵩涛,王红,张忠芮,等.静电纺丝素/胶原/聚合物复合纤维的制备及其力学性能的研究[J].高分子通报,2014;27(6):101-106. [34] JEONG J, LEE S. Electrospun poly (vinyl alcohol) nanofibrous membranes containing coptidis rhizoma extracts for potential biomedical applications. Text Res J. 2019;89(17):3506-3518. [35] 黄丽冰,徐玉龙,曹宽,等.含中药蜈蚣成分超细纤维的制备[J].纺织导报,2019(1):63-66. [36] YIN X, TAN P, LUO H, et al. Study on the release behaviors of berberine hydrochloride based on sandwich nanostructure and shape memory effect. Mater Sci Eng C. 2020;109:110541. [37] MOHEBIAN Z, BABAZADEH M, ZARGHAMI N, et al. Anticancer efficiency of curcumin-loaded mesoporous silica nanoparticles/nanofiber composites for potential postsurgical breast cancer treatment. J Drug Deliv Sci Tec. 2021;61:102170. [38] 韩玉芬.负载中药“核壳”结构纳米纤维的制备及性能研究[D].北京:北京化工大学,2021. [39] LIU Y, CHEN X, GAO Y, et al. Electrospun core-sheath nanofibers with variable shell thickness for modifying curcumin release to achieve a better antibacterial performance. Biomolecules. 2022;12(8):1057. [40] SABRA S, RAGAB DM, AGWA MM, et al. Recent advances in electrospun nanofibers for some biomedical applications. Eur J Pharm Sci. 2020;144: 105224. [41] LI D, YUAN T, ZHANG X, et al. Icariin: a potential promoting compound for cartilage tissue engineering. Osteoarthr Cartilage. 2012;20(12):1647-1656. [42] 陈微.中药有效成分功能化改性软骨组织工程支架的制备及性能研究[D].长沙:湖南大学,2021. [43] GUO JH, LIU Y, LV ZJ, et al. Potential neurogenesis of human adipose-derived stem cells on electrospun catalpol-loaded composite nanofibrous scaffolds. Ann Biomed Eng. 2015;43(10):2597-2608. [44] GONG M, CHI C, YE J, et al. Icariin-loaded electrospun PCL/gelatin nanofiber membrane as potential artificial periosteum. Colloids Surf B. 2018;170: 201-209. [45] WANG J, TIAN L, HE L, et al. Lycium barbarum polysaccharide encapsulated Poly lactic-co-glycolic acid Nanofibers: cost effective herbal medicine for potential application in peripheral nerve tissue engineering. Sci Rep. 2018;8(1):8669. [46] SEDGHI R, SAYYARI N, SHAABANI A, et al. Novel biocompatible zinc-curcumin loaded coaxial nanofibers for bone tissue engineering application. Polymer. 2018;142:244-255. [47] ZHANG Q, ZHANG Y, WATTS D, et al. Electrospun naringin-loaded beaded nanofiber with controlled release property for bone tissue engineering applications. Sci Adv Mater. 2019;11(10):1433-1442. [48] JAGANATHAN SK, MANI MP. Electrospun novel nanocomposite comprising polyurethane integrated with ayurveda amla oil for bone tissue engineering. An Acad Bras Cienc. 2020;92(1):e20180369. [49] LI K, ZHANG Y, XU J, et al. Three-dimensional magnetic fibrous scaffold with icariin expanded by supercritical CO2 for bone tissue engineering under static magnetic field. Compos B Eng. 2021;226:109304. [50] ZHANG Y, WANG T, LI J, et al. Bilayer membrane composed of mineralized collagen and chitosan cast film coated with berberine-loaded PCL/PVP electrospun nanofiber promotes bone regeneration. Front Bioeng Biotechnol. 2021;9:684335. [51] CHEN W, LI Y, HUANG Y, et al. Quercetin modified electrospun PHBV fibrous scaffold enhances cartilage regeneration. J Mater Sci Mater Med. 2021;32(8):92. [52] 陈晨,李永圆,王海霞,等.制剂技术在提高中药抗菌活性方面的应用及前景分析[J].中国实验方剂学杂志,2022,28(7):247-253. [53] PEDRAM RZ, MOKHTARI J, ABBASI M. Calendula officinalis extract/PCL/Zein/Gum arabic nanofibrous bio-composite scaffolds via suspension, two-nozzle and multilayer electrospinning for skin tissue engineering. Int J Biol Macromol. 2019;135:530-543. [54] YUE Y, LIU X, PANG L, et al. Astragalus polysaccharides/PVA nanofiber membranes containing astragaloside IV-loaded liposomes and their potential use for wound healing. Evid Based Complement Altern Med. 2022;2022:9716271. [55] ZAHEDI E, ESMAEILI A, ESLAHI N, et al. Fabrication and characterization of core-Shell electrospun fibrous mats containing medicinal herbs for wound healing and skin tissue engineering. Mar Drugs. 2019;17(1):27. [56] SHOKROLLAHI M, BAHRAMI SH, NAZARPAK MH, et al. Multilayer nanofibrous patch comprising chamomile loaded carboxyethyl chitosan/poly (vinyl alcohol) and polycaprolactone as a potential wound dressing. Int J Biol Macromol. 2020;147:547-559. [57] CHEN K, PAN H, JI D, et al. Curcumin-loaded sandwich-like nanofibrous membrane prepared by electrospinning technology as wound dressing for accelerate wound healing. Mater Sci Eng C. 2021;127:112245. [58] ZHOU L, CAI L, RUAN H, et al. Electrospun chitosan oligosaccharide/polycaprolactone nanofibers loaded with wound-healing compounds of Rutin and Quercetin as antibacterial dressings. Int J Biol Macromol. 2021;183:1145-1154. [59] ARAS C, TÜMAY ÖZER E, GÖKTALAY G, et al. Evaluation of nigella sativa oil loaded electrospun polyurethane nanofibrous mat as wound dressing. J Biomater Sci Polym Ed. 2021;32(13):1718-1735. [60] WANG X, HU X, LI S, et al. Preparation of antibacterial nanofibers by electrospinning polyvinyl alcohol containing a luteolin hydroxypropyl-β-cyclodextrin inclusion complex. New J Chem. 2022;46(5):2360-2367. [61] 包翔.具有抗氧化活性的新型中药复合纳米材料促进创伤修复的应用研究[D].南京:南京中医药大学,2022. [62] MAVROKEFALOU E, MONOU PK, TZETZIS D, et al. Preparation and in vitro evaluation of electrospun sodium alginate fiber films for wound healing applications. J Drug Deliv Sci Tec. 2023;81:104298. |
| [1] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [2] | Chen Kaijia, Liu Jingyun, Cao Ning, Sun Jianbo, Zhou Yan, Mei Jianguo, Ren Qiang. Application and prospect of tissue engineering in treatment of osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1450-1456. |
| [3] | Bai Chen, Yang Wenqian, Meng Zhichao, Wang Yuze. Strategies for repairing injured anterior cruciate ligament and promoting graft healing [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1457-1463. |
| [4] | Yu Weijie, Liu Aifeng, Chen Jixin, Guo Tianci, Jia Yizhen, Feng Huichuan, Yang Jialin. Advantages and application strategies of machine learning in diagnosis and treatment of lumbar disc herniation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1426-1435. |
| [5] | Lin Zeyu, Xu Lin. Research progress in gout-induced bone destruction mechanism [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1295-1300. |
| [6] | Zhang Xiaoyun, Liu Hua, Chai Yuan, Chen Feng, Zeng Hao, Gao Zhengang, Huang Yourong. Effect of Yishen Gushu Formula on bone metabolic markers and clinical efficacyn in patients with osteoporosis of kidney deficiency and blood stasis type [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1155-1160. |
| [7] | Wang Shanshan, Shu Qing, Tian Jun. Physical factors promote osteogenic differentiation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1083-1090. |
| [8] | Pan Xiaolong, Fan Feiyan, Ying Chunmiao, Liu Feixiang, Zhang Yunke. Effect and mechanism of traditional Chinese medicine on inhibiting the aging of mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1091-1098. |
| [9] | Liu Hanfeng, Wang Jingjing, Yu Yunsheng. Artificial exosomes in treatment of myocardial infarction: current status and prospects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1118-1123. |
| [10] | Ma Shuwei, He Sheng, Han Bing, Zhang Liaoyun. Exosomes derived from mesenchymal stem cells in treatment of animals with acute liver failure: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1137-1142. |
| [11] | Mei Jingyi, Liu Jiang, Xiao Cong, Liu Peng, Zhou Haohao, Lin Zhanyi. Proliferation and metabolic patterns of smooth muscle cells during construction of tissue-engineered blood vessels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1043-1049. |
| [12] | Zhang Kefan, Shi Hui. Research status and application prospect of cytokine therapy for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 961-967. |
| [13] | Shen Ziqing, Xia Tian, Shan Yibo, Zhu Ruijun, Wan Haoxin, Ding Hao, Pan Shu, Zhao Jun. Vascularized tracheal substitutes constructed by exosome-load hydrogel-modified 3D printed scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 697-705. |
| [14] | Tian Xin, Liu Tao, Yang Huilin, He Fan. In vitro evaluation of sustained release Kartogenin by gelatin methacryloyl microspheres for repairing nucleus pulposus degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 724-730. |
| [15] | Li Jiaqi, Huang Yuanli, Li Yan, Wang Chunren, Han Qianqian. Mechanism and influencing factors in molecular weight degradation of non-cross-linked hyaluronic acid [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 747-752. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||