Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (22): 3597-3602.doi: 10.12307/2024.477
Previous Articles Next Articles
Polymer-based antibiotic sustained-release carrier in treatment of chronic osteomyelitis
Ilham • Maimaitiabudula1, Huang Xiaoxia1, 2, Li Luyao1, 2, Teng Yong2
- 1Graduate School of Xinjiang Medical University, Urumqi 830017, Xinjiang Uygur Autonomous Region, China; 2General Hospital of Xinjiang Military Region of Chinese People’s Liberation Army, Urumqi 830099, Xinjiang Uygur Autonomous Region, China
-
Received:2023-08-07Accepted:2023-09-15Online:2024-08-08Published:2024-01-20 -
Contact:Teng Yong, Chief physician, Doctoral supervisor, General Hospital of Xinjiang Military Region of Chinese People’s Liberation Army, Urumqi 830099, Xinjiang Uygur Autonomous Region, China -
About author:Ilham • Maimaitiabudula, Master candidate, Graduate School of Xinjiang Medical University, Urumqi 830017, Xinjiang Uygur Autonomous Region, China Huang Xiaoxia, PhD candidate, Graduate School of Xinjiang Medical University, Urumqi 830017, Xinjiang Uygur Autonomous Region, China; General Hospital of Xinjiang Military Region of Chinese People’s Liberation Army, Urumqi 830099, Xinjiang Uygur Autonomous Region, China -
Supported by:Natural Science Foundaton of Xinjiang Uygur Autonomous Region, No. 2023D01C93 (to TY); Xinjiang Uygur Autonomous Region Regional Collaborative Innovation Project (Technology Aid to Xinjiang), No. 2019E0277 (to TY)
CLC Number:
Cite this article
Ilham • Maimaitiabudula, Huang Xiaoxia, Li Luyao, Teng Yong. Polymer-based antibiotic sustained-release carrier in treatment of chronic osteomyelitis[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3597-3602.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

聚合物基抗生素缓释载体是以天然或人工合成方法获得的可生物降解载体材料,通过合适的制备工艺设计成纳米微球、纳米纤维、骨水泥、水凝胶、海绵、涂层膜、三维多孔支架等不同形式载体,提供可持续释放的有效浓度抗生素来控制感染[22-24],并且聚合物基载体在降解过程中促进骨细胞的黏附与生长,可为骨缺损修复提供有效的结构支撑[25]。 2.1 天然有机高分子聚合物基载体 天然聚合物因其良好的生物相容性、生物降解性、生物活性、分解产物无毒性及易于获取等优点而被广泛应用于组织工程[26-27]。当前,常用于骨组织工程材料的天然聚合物包括天然多糖类(如壳聚糖、透明质酸、海藻酸盐等)和天然蛋白质或肽类(如胶原蛋白、丝素蛋白、纤维蛋白、明胶等)。上述材料具有一些特殊优点赋予他们独特的性质,同样存在的一些缺点需要改进,见表1。"


2.1.1 壳聚糖及其衍生物 壳聚糖为一种天然衍生的氨基多糖(来源于甲壳素的部分脱乙酰化),其分子结构与胶原蛋白相似,可用于模拟细胞外基质有机成分。壳聚糖基材料的主要优点是具有生物降解性、生物相容性、低免疫原性、无毒性等综合特性[28-29]。此外,壳聚糖还具有一些特殊性质赋予它多元化的性能:①壳聚糖具有丰富的生物活性羟基和氨基。壳聚糖结构中的功能氨基提供了与醛酮进行乙酰化、季铵化、缩合反应、烷基化等反应的可能性。到目前为止,壳聚糖已经通过化学改性产生了硫酸壳聚糖、三甲基壳聚糖、硫醇壳聚糖、羟烷基壳聚糖、羧烷基壳聚糖和磷酸化壳聚糖,这决定了它多变的理化性质及抗菌性能[30-31]。②主链中游离胺基的存在赋予了壳聚糖独特的多阳离子特性,确保了它可以结合带负电荷的材料及蛋白质或抗生素类药物。③具有特殊的吸水和溶胀特性,可以制备成水凝胶等载体。④具有内在的抗菌性能[32]。这些特殊特性决定了它具有多变的溶解度、黏附性、可修饰性及内在的抗菌性能等物理化学反应特点,被广泛用于组织工程和药物递送系统。 TAO等[33]设计了一种基于壳聚糖的温敏性水凝胶来制备万古霉素纳米粒子/凝胶局部给药系统,以带正电荷季铵化壳聚糖和带负电荷羧化壳聚糖/万古霉素纳米颗粒为载体,通过正负电荷吸附形成壳聚糖/万古霉素纳米颗粒,得到的纳米颗粒包封率和载药率相对较高,分别为(60.1±2.1)% 和(24.1±0.84)%;该纳米颗粒在26 d内持续释放万古霉素。将万古霉素纳米粒子包封在壳聚糖基水凝胶中可以保持纳米粒子的粒径大小和形态,随着水凝胶基质的扩散和降解,纳米粒子中的抗生素缓慢稳定释放,并且还保留了水凝胶的可注射性和热敏性,可以有效填充骨缺损。纳米颗粒与水凝胶的结合表现出抗感染和修复不规则骨缺损的双重功能。YANG等[34]建立的季铵化壳聚糖接枝的3D 打印聚乳酸-羟基乙酸/羟基磷灰石复合支架结合了不同材料的优点,表现出良好的细胞相容性、骨诱导和骨传导性及体外抗菌性能,动物实验进一步证实该支架在感染性骨缺损动物模型中表现出显著的抗感染和骨再生效果。因此,除了壳聚糖材料本身优异的特性之外,可以通过物理化学修饰、交联、复合其他材料等方法改善壳聚糖基材料机械性能差、水溶性差、抗生素缓释过快等缺点。另外,通过不同的制备工艺可以合成纳米颗粒、水凝胶、涂层膜及三维多孔支架等,有望成为理想的抗生素缓释载体。 2.1.2 胶原蛋白及其衍生物 胶原蛋白是由多肽链和明胶组成的三螺旋结构(来源于脊柱动物体内的细胞外基质),是人体主要的细胞外基质成分,其在体内不仅提供局部骨组织结构的完整性,还调节细胞的增殖和分化能力,促进骨髓间充质干细胞的黏附与生长。胶原蛋白因具有良好的亲水性、高拉伸强度和柔韧性、生物降解性能、低抗原活性、低毒性、易于改性和理想的组织再生潜力等特殊优点而成为理想的药物递送系统候选材料之一[35]。同样,胶原蛋白基材料存在的主要问题有生理条件下的稳定性较差、机械性能较低等。交联(包括酶交联、物理交联和化学交联)和复合其他材料是改性聚合物材料最常见的方法,如GRABSKA-ZIELI?SKA 等[36]通过N-(3-二甲基氨基丙基)N’-乙基碳二亚胺盐酸盐和N-羟基琥珀酰亚胺化学交联,采用冻干法制备了以胶原、壳聚糖和丝素蛋白3种混合材料为基础的多孔支架,3种材料具有较高的溶胀度,力学性能可以根据材料的组成而改变;支架中所有材料均与 MG-63成骨样细胞具有细胞相容性,说明交联特性在一定程度上解决了不同聚合物材料之间的结合问题,可以通过调节比例得到预期生物力学性能的复合载体材料。ALEGRETE 等[37]合成了一种载有万古霉素的肝素化纳米羟基磷灰石/胶原生物复合材料载体,在体外研究表明万古霉素的释放模式具有一个强烈的初始爆发,然后是缓慢持续释放,其浓度高于最小抑菌浓度,该颗粒并未损害细胞活力;在动物模型中进行体内毒理学测试未见相关的不良反应,该支架被证明是一种有效的药物缓释载体,可以控制耐甲氧西林金黄色葡萄球菌感染,同时在动物模型中修复骨缺损。因此,通过交联或复合其他材料得到的多功能复合支架可以弥补单纯胶原蛋白基载体的缺陷。 综合近年来的研究成果,天然高分子材料作为组织工程材料具有许多优点,然而以天然聚合物为基础的骨替代物普遍存在生物力学性能差等一些特定的问题,最主要的是天然聚合物的特性局限于软组织,难以与骨组织的力学强度、生物降解速率和稳定性等相匹配[27,48-49]。 2.2 人工合成有机高分子聚合物基载体 与天然聚合物材料相比,合成聚合物材料可以在更可控的条件下合成,其理化性质及微观结构更易于控制,通过合理设计聚合物的官能团或调节制备工艺参数获得预期的性能,适合大批量生产和临床应用。这些聚合物可以在体内通过非酶解降解产生无毒降解产物,被广泛用于生物组织工程[49],多功能性是它们最大的优势。聚乳酸、聚乳酸-羟基乙酸、 聚己内酯、聚富马酸丙二醇酯是研究最多的合成聚合物,其中聚己内酯、聚乳酸-羟基乙酸已经通过美国食品药品管理局批准,是研究最充分的聚合物。这些材料具有一些特殊优点及缺点,可根据需要去选择合适的材料,并通过合适的方法改进材料缺点来获得预期的抗生素缓释载体(表2)。"

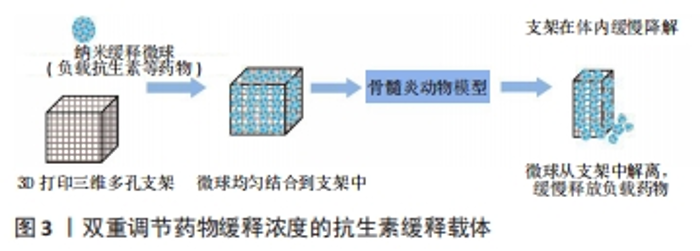
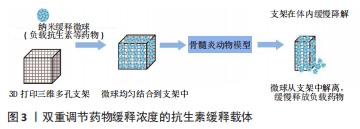
2.2.1 聚乳酸 聚乳酸是一种生物可降解的热塑性聚合物(来源于玉米淀粉和甘蔗等),具有生物相容性、生物降解性、热稳定性和降解产物无毒等骨组织工程所需要的基本特性。然而,单纯的聚乳酸材料缺乏骨组织所需要的机械强度及生物活性,并且分解产生的代谢产物——乳酸的积累很容易引起炎症反应。LV等[50]合成了负载万古霉素的纳米羟基磷灰石/聚乳酸复合材料载体,其在不同酸性溶液中保持良好的稳定性,纳米羟基磷灰石/聚乳酸将万古霉素的半衰期延长了6倍,有利于其体外长期保存;渗透实验证实该缓释载体的渗透效率较高,其能够有效地将万古霉素转运至骨髓组织,从而更好地抑制细菌活性并降低小鼠慢性骨髓炎模型病变区域 CD4、CD8、CD19和 CD20的表达;在成骨实验中,纳米羟基磷灰石的加入更有效地维持了成骨细胞的活力,促进了成骨细胞的增殖和迁移,从而更好地修复了缺损的骨组织;在小鼠慢性骨髓炎模型中,该缓释载体能较好地抑制病灶炎症反应,增加骨小梁的数量和厚度,并且骨组织最大载荷、弹性载荷、骨缺损区刚度系数均得到较大程度的恢复。因此,复合其他材料可以弥补聚乳酸的局限性。 2.2.2 聚乳酸-羟基乙酸共聚物 聚乳酸-羟基乙酸共聚物是一种开发最成功的合成聚合物之一,也是研究最充分的药物递送载体。基于聚乳酸-羟基乙酸共聚物材料的主要优势在于其良好的生物相容性、可生物降解性、热稳定性及很容易被加工成任何尺寸和形状[55-56]。聚乳酸-羟基乙酸共聚物是通过两种单体(丙交酯和乙交酯)的缩聚反应或开环聚合反应合成的,其理化性质可以通过改变组成(丙交酯与乙交酯摩尔比)、分子质量、溶解度等来调节[52,56]。一般来说,增加聚乳酸-羟基乙酸聚合物中丙交酯单体的含量可以提高聚合物的亲水性,加快聚合物的降解速度,共聚物在摩尔比50∶50(丙交酯∶乙交酯)下表现出高降解率;溶解度会影响聚乳酸-羟基乙酸的降解速率、力学性能及溶胀行为,其玻璃化转变温度在45-55 ℃,玻璃化转变温度也与降解速率呈反比关系。药物从聚合物中释放的速率随着聚合物玻璃化转变温度的升高而降低;聚合物的分子质量与降解呈负相关,随着分子质量的减少,聚合物的降解速率增加,从而药物释放增加,通常来说5-150 kD范围内的分子质量最适合用作药物递送系统[57]。因此,聚乳酸-羟基乙酸共聚物的这些理化性质会直接影响聚乳酸-羟基乙酸共聚物微球的降解速率和药物释放动力学。同样,聚乳酸-羟基乙酸材料的主要问题在于生物活性不足、机械性能较差、缺乏细胞识别信号、分解产物会产生炎症反应等。LI等[58]首先制备了万古霉素/聚乳酸-乙醇酸缓释微球,然后通过双喷嘴三维打印技术将缓释微球均匀载入纳米羟基磷灰石/聚乳酸支架的孔隙中,按一定比例打印出复合支架,既能促进骨修复又能控制局部感染。MISTRY等[59]将载有抗生素的聚乳酸-羟基乙酸/双相磷酸钙复合骨水泥用于治疗实验性骨髓炎也得到了相似的结果。LI等[61]采用双乳液溶剂挥发法制备了盐酸万古霉素聚乳酸-羟基乙酸共聚物微球,考察了不同浓度丝素蛋白包被后药物微球的体外释放行为,结果表明微球载药量为(24.11±1.72)%、包封率为(48.21±3.44)%,体外药物释放实验表明载药微球具有明显的初始爆发释放特性,丝素蛋白包被可以不同程度地延缓载药微球初始爆发释放行为、降低药物的累积释放量,其中以包覆0.1%丝素蛋白的微球效果最好;不同载药微球组的体外释药时间可达28 d,0.1%丝素蛋白和0%丝素蛋白包被微球的累积释放量分别为(82.50±3.51)%和(67.70±3.81)%。因此,万古霉素微球表面包覆丝素蛋白可以延缓初始爆发释放、减少药物累积释放、延长药物作用时间、提高抗感染效果。作者认为聚乳酸-羟基乙酸基抗生素缓释载体是临床上治疗慢性骨髓炎最有希望的候选载体之一。除了优化聚乳酸-羟基乙酸本身配方之外,可以通过表面修饰及复合其他材料来改善缓释载体的性能。 2.2.3 聚己内酯 聚己内酯是一种半结晶、无毒的可生物降解聚酯,是骨组织工程最常见的合成聚合物之一。与其他聚酯相比,基于聚己内酯材料的主要优势在于具有良好的生物相容性、较强的力学性能、缓慢的降解速度、低熔点、酸性分解产物较少及成本相对较低且具有承重潜力,是最常用的3D打印组织工程支架材料之一[64]。然而,纯聚己内酯材料缺乏骨诱导及骨再生潜力。WANG等[65]设计了聚己内酯/β-磷酸三钙交叉尺度结构的支架,该支架由熔融沉积型3D打印技术印刷的直径500 μm粗纤维构成,在粗纤维的孔隙中,采用熔融静电纺丝方式填充直径约10 μm的超高精度细纤维网格;细胞可以生长在粗纤维表面,同时细纤维网状结构为细胞生长提供了一个桥梁,使细胞能够通过粗纤维的孔隙;复合材料中的β-磷酸三钙为支架提供了Ca2+和 PO43-,有效地促进了支架上细胞的成骨分化。与传统支架相比,交叉支架的成骨性能有了很大提高,不仅在支架表面形成了骨样沉淀,而且在支架孔隙中也出现了明显的碱性磷酸酶表达及有效钙沉淀。因此,无机材料的骨诱导、骨传导性能可以弥补聚己内酯材料的局限性。另外,聚己内酯是疏水性的,缺乏细胞结合位点。LóPEZ-GONZáLEZ等[66]将载有不同万古霉素浓度的壳聚糖水凝胶结合到基于聚己内酯的3D 打印支架中,该支架结合了壳聚糖和聚己内酯的优异性能,并发现冷等离子体处理可使聚己内酯水接触角降低20°;体外实验表明,该支架具有生物相容性、生物活性及体外抗菌性能,对细胞没有毒性作用。因此,聚合物表面改性及复合其他材料也是一种改善基础材料局限性的方法。 2.2.4 聚富马酸丙二醇酯 聚富马酸丙二醇酯是一种不饱和线性聚酯,是一种用于骨缺损修复的新型可注射医用生物材料。近30年来,在聚富马酸丙二醇酯基材料的合成、交联、复合材料改性、支架制造、功能化等方面取得了良好进展[67]。低分子质量聚富马酸丙二醇酯在室温下是一种高黏性液体,可通过热交联和/或光交联制备固化的聚富马酸丙二醇酯基可吸收载体。杨峰等[68]采用两步法合成了聚富马酸丙二醇酯骨水泥,并与商品化聚甲基丙烯酸甲酯骨水泥相比较,发现该骨水泥具有更好的生物相容性及力学性能,并具有较低的固化温度(53 ℃),是一种有发展潜力的新型可注射骨水泥材料。然而,热交联合成的聚合物可能因会残留未反应的交联剂、引发剂和促进剂等降低材料的生物相容性,阻碍了利用3D打印技术合成聚富马酸丙二醇酯支架。与热交联方法相比,光交联技术对聚合的时间和空间有更大控制,在支架植入过程中有更大的灵活性[67]。 光交联聚富马酸丙二醇酯的力学性能与骨小梁相似,目前研究最多的两种基于光交联的技术是立体光刻技术和连续数字光处理技术[69]。但是,聚富马酸丙二醇酯本身作为组织工程材料时(分子质量< 3 500 Da)是一种高黏性聚合物[70],含有纯聚富马酸丙二醇酯的配方因黏度太高而不能用于基于光交联的3D打印技术,因为高黏度特性阻碍了层厚度的精确控制,并增加了固化时间。此外,用于光交联的树脂混合物是单体/低聚物、溶剂和光引发剂的复合物,其他成分(如分散剂、乳化剂或光衰减剂等)可以根据需要添加[71]。综上,基于聚富马酸丙二醇酯基材料的骨水泥有望成为临床上新型可注射骨水泥的候选材料。而聚富马酸丙二醇酯材料的高黏度特性限制了它在光交联3D打印技术的合成,因此,优化聚富马酸丙二醇酯基树脂组分及3D打印参数来打破可加工树脂的高浓度限制是后续研究的关键。 2.3 聚合物基有机-无机复合材料型载体 为了选择最合适的生物材料,了解天然骨的物理化学结构以及相关的生物力学特征是至关重要的。骨组织由不同类型的骨细胞和细胞外间质组成,胶原蛋白是细胞外基质主要的有机成分,磷灰石作为细胞外基质主要的无机部分占总骨量的65%[27]。因此,许多研究通过模拟天然骨的微环境成分来寻找最佳骨替代材料,即有机(天然或合成聚合物)-无机(羟基磷灰石、β-磷酸三钙、生物活性玻璃、金属纳米颗粒和碳基纳米材料等)复合材料用于骨组织工程得到了良好的实验证据。CHENG等[72]将万古霉素载入介孔生物活性玻璃中形成局部抗生素释放微球系统,然后采用冷冻干燥法制备介孔生物活性玻璃与聚乳酸-羟基乙酸复合骨组织工程支架,体外降解和水接触角分析表明,介孔生物活性玻璃/聚乳酸-羟基乙酸支架具有可控的降解性,稳定了周围环境的 pH值,提高了支架的亲水性;此外,介孔生物活性玻璃的存在为人骨髓来源间充质干细胞的附着、扩散和增殖提供了良好的相互联通的孔隙结构,上调了成骨标志物的表达;体外实验表明负载万古霉素的支架可以持续释放抗生素8周以上,促进抗菌活性、抑制生物膜的形成,而对细胞无不利影响。因此,作者认为无机生物陶瓷类材料成分可以提高聚合物基材料的机械强度和生物活性,而天然或合成高分子聚合物有机材料成分可以增加韧性和可加工性[73]。 此外,无机金属氧化物、金属离子及碳基纳米颗粒材料等由于其独特的物理化学性质,可以弥补聚合物材料机械强度、生物活性、抗菌性、骨诱导及骨传导性方面的不足[49,74-78]。近些年,骨组织工程领域的迅速发展有望解决骨缺损的修复问题,该领域常用的生物活性细胞及因子可以加入到抗生素缓释载体中,有效改善载体的生物活性、促进骨细胞的黏附与生长,为制备具有抗感染及骨再生双重功能药物缓释载体提供理论基础[75,79-80]。其中,新型3D生物打印技术能够将生物活性细胞及因子加入到骨组织工程支架的3D打印程序里面,有望实现双重功能支架的有效打印[81]。 2.4 聚合物基抗生素缓释载体的制备工艺 高分子材料加工技术的发展促进了组织工程高分子材料的功能化发展,除了材料本身特性之外,制备工艺对药物缓释载体的合成来说也是一个重要内容。不同的制备工艺可以合成不同结构形式的载体,供临床医生根据需要去选择。目前,溶剂铸造和微粒浸出、气体发泡、热诱导相分离、冷冻干燥法是合成高分子聚合物缓释载体最简单的方法,通过物理化学途径得到的载体具有较高孔隙率和比较均匀的内部孔隙结构。另外,静电纺丝是一种比较流行的支架制造技术,可以设计和制造类似于天然细胞外基质的纳米纤维支架[26]。 但是,这些传统的制造技术很难实现对微结构的精确控制。相比之下,个性化定制的3D打印支架在骨组织工程领域有着重要作用。3D打印支架可以精确控制载体孔隙的大小、几何形状和空间分布等,以及操纵各种材料、生物活性细胞及因子来复制天然组织的形式和功能,并能负载有效浓度的抗生素做到控制释放[82]。而且,高分子聚合物基材料因具有较高的热稳定性、可塑性、易加工性及易于获取等优点最适合用于3D打印[63]。目前存在的主要问题在于如何根据临床需要找到最佳工艺参数来制备预期的抗生素缓释载体。 2.5 聚合物基抗生素缓释载体的多重功能 聚合物基抗生素缓释载体具有生物相容性、生物降解性、生物活性、促进细胞黏附与生长、药物缓释性及抗菌性能等多重功能。一般认为,天然聚合物载体比合成聚合物载体具有更好的生物相容性和生物活性,但是合成聚合物比天然聚合物具有更好的可控机械性和生物降解性。在骨髓炎治疗中,药物缓释性及抗菌性能是人们重点关注的内容,这与材料类型及载体的结构设计有着直接的关系。超小尺寸的纳米粒子(大小范围在1-100 nm之间)表现出更大的比表面积,将抗生素类药物包封在纳米粒子中可以防止化学降解并改善其药代动力学性质,从而实现长期控制释放[83]。此外,抗生素耐药和细菌生物膜形成是抗生素缓释载体治疗慢性骨髓炎最重要的屏障,BIRK等[84]认为抗生素缓释载体处理生物膜的重要参数包括载药量、药物释放动力学、生物膜中载体颗粒大小及细胞迁移率等,这些参数主要受缓释载体的孔隙率、孔径大小、连通性、亲水性、比表面积等结构因素的影响。 针对抗菌效果而言,除了抗生素缓释载体材料和结构之外,抗生素的选择也是需要去关注的内容。首先,所选的抗生素必须覆盖包括耐甲氧西林金黄色葡萄球菌在内的常见致病菌;其次,抗生素浓度要在较长时间内达到或超过致病病原体的最小抑菌浓度,且不易产生耐药和生物膜形成等;另外,抗生素在生理条件下以及载体制备过程中能够保持稳定、结构不被破坏等[19,85-86]。 最后,抗生素与缓释载体的结合方式也是一个重要的工程。传统的结合方式包括物理吸附、化学吸附、直接包载等[75],这样会导致抗生素在载体中的分布不均匀及严重的初始爆发释放等问题。作者认为将负载抗生素的纳米缓释微球均匀填充到3D打印三维多孔支架或水凝胶等载体中,结合一些表面改性或涂层等方法调节抗生素的释放速率,得到提供预期抗生素控释浓度的载体是一种较为理想的方法[33,58,87]。其中,支架或水凝胶可以通过占据骨缺损处的死腔提供机械支撑,支架的多孔隙结构可以促进细胞的黏附与生长,并增强成骨过程而修复骨缺损;同时,随着支架或水凝胶在体内生理条件下缓慢降解,纳米微球逐渐从支架中解离并递送具有可调节释放动力学的抗生素,这种具有抗感染和骨再生双重功能的抗生素缓释载体可能最适合用于感染性骨缺损的治疗(图3)。"
| [1] METSEMAKERS WJ, MORGENSTERN M, SENNEVILLE E, et al. General treatment principles for fracture-related infection: recommendations from an international expert group. Arch Orthop Trauma Surg. 2020;140(8):1013-1027. [2] 化昊天,王新卫,张磊,等.慢性骨髓炎的诊疗研究进展[J].中国骨与关节杂志,2022,11(2): 132-136. [3] 郝建强,韩涛,李闯兵,等.四肢长骨慢性骨髓炎清创研究进展[J].中国医学创新,2023, 20(13):179-182. [4] LIU Y, YUSHAN M, LIU Z, et al. Complications of bone transport technique using the Ilizarov method in the lower extremity: a retrospective analysis of 282 consecutive cases over 10 years. BMC Musculoskelet Disord. 2020;21(1):354. [5] 金磊,艾合买提江•玉素甫.长骨骨缺损手术治疗进展[J].中国骨与关节杂志,2021,10(9): 673-677. [6] 邢浩,张永红,王栋.长骨大段骨缺损修复方法的优势与不足[J].中国组织工程研究,2021, 25(3):426-430. [7] 李德满.负压封闭引流术治疗慢性化脓性骨髓炎患者的临床疗效观察[J].中国药物经济学,2018,13(8):106-108. [8] WANG X, WEI F, LUO F, et al. Induction of granulation tissue for the secretion of growth factors and the promotion of bone defect repair. J Orthop Surg Res. 2015;10:147. [9] 陈英华,黄巍峰,韦武,等.膜诱导技术在慢性创伤性骨髓炎中的应用进展[J].中外医疗, 2023,42(14):194-198. [10] ALFORD AI, NICOLAOU D, HAKE M, et al. Masquelet’s induced membrane technique: Review of current concepts and future directions. J Orthop Res. 2021;39(4):707-718. [11] LIODAKIS E, PACHA TO, AKTAS G, et al. [Biological reconstruction of large bone defects : Masquelet technique and new procedures]. Unfallchirurgie (Heidelb). 2023;126(3):184-189. [12] HSU CA, CHEN SH, CHAN SY, et al. The Induced Membrane Technique for the Management of Segmental Tibial Defect or Nonunion: A Systematic Review and Meta-Analysis. Biomed Res Int. 2020;2020:5893642. [13] 中华医学会骨科学分会,沈杰,陈林,等.膜诱导技术治疗感染性骨缺损临床循证指南(2023版)[J]. 中华创伤杂志,2023,39(2):107-120. [14] GEURTS JAP, VAN VUGT TAG, ARTS JJC. Use of contemporary biomaterials in chronic osteomyelitis treatment: Clinical lessons learned and literature review. J Orthop Res. 2021; 39(2):258-264. [15] WASSIF RK, ELKAYAL M, SHAMMA RN, et al. Recent advances in the local antibiotics delivery systems for management of osteomyelitis. Drug Deliv. 2021;28(1):2392-2414. [16] SHI X, WU Y, NI H, et al. Antibiotic-loaded calcium sulfate in clinical treatment of chronic osteomyelitis: a systematic review and meta-analysis. J Orthop Surg Res. 2022;17(1): 104. [17] MEREDDY P, NALLAMILLI SR, GOWDA VP, et al. The use of Stimulan in bone and joint infections. Bone Jt Open. 2023;4(7):516-522. [18] SAMELIS PV, PAPAGRIGORAKIS E, SAMELI E, et al. Current Concepts on the Application, Pharmacokinetics and Complications of Antibiotic-Loaded Cement Spacers in the Treatment of Prosthetic Joint Infections. Cureus. 2022;14(1):e20968. [19] VAN VUGT TAG, ARTS JJ, GEURTS JAP. Antibiotic-Loaded Polymethylmethacrylate Beads and Spacers in Treatment of Orthopedic Infections and the Role of Biofilm Formation. Front Microbiol. 2019;10:1626. [20] XU T, WU KL, JIE K. Comprehensive meta-analysis of antibiotic-impregnated bone cement versus plain bone cement in primary total knee arthroplasty for preventing periprosthetic joint infection. Chin J Traumatol. 2022;25(6):325-330. [21] LIU Y, LI X, LIANG A. Current research progress of local drug delivery systems based on biodegradable polymers in treating chronic osteomyelitis. Front Bioeng Biotechnol. 2022;10:1042128. [22] COBB LH, MCCABE EM, PRIDDY LB. Therapeutics and delivery vehicles for local treatment of osteomyelitis. J Orthop Res. 2020;38(10): 2091-2103. [23] GUILLÉN-CARVAJAL K, VALDEZ-SALAS B, BELTRÁN-PARTIDA E, et al. Chitosan, Gelatin, and Collagen Hydrogels for Bone Regeneration. Polymers (Basel). 2023;15(13):2762. [24] SMITH M, ROBERTS M, AL-KASSAS R. Implantable drug delivery systems for the treatment of osteomyelitis. Drug Dev Ind Pharm. 2022;48(10):511-527. [25] KRISHNAN AG, BISWAS R, MENON D, et al. Biodegradable nanocomposite fibrous scaffold mediated local delivery of vancomycin for the treatment of MRSA infected experimental osteomyelitis. Biomater Sci. 2020;8(9):2653-2665. [26] YE B, WU B, SU Y, et al. Recent Advances in the Application of Natural and Synthetic Polymer-Based Scaffolds in Musculoskeletal Regeneration. Polymers (Basel). 2022;14(21):4566. [27] FAN J, ABEDI-DORCHEH K, SADAT VAZIRI A, et al. A Review of Recent Advances in Natural Polymer-Based Scaffolds for Musculoskeletal Tissue Engineering. Polymers (Basel). 2022; 14(10):2097. [28] DESAI N, RANA D, SALAVE S, et al. Chitosan: A Potential Biopolymer in Drug Delivery and Biomedical Applications. Pharmaceutics. 2023;15(4):1313. [29] TAO F, MA S, TAO H, et al. Chitosan-based drug delivery systems: From synthesis strategy to osteomyelitis treatment - A review. Carbohydr Polym. 2021;251:117063. [30] QIN Y, LI P. Antimicrobial Chitosan Conjugates: Current Synthetic Strategies and Potential Applications. Int J Mol Sci. 2020;21(2):499. [31] CONFEDERAT LG, TUCHILUS CG, DRAGAN M, et al. Preparation and Antimicrobial Activity of Chitosan and Its Derivatives: A Concise Review. Molecules. 2021;26(12):3694. [32] KONG M, CHEN X G, XING K, et al. Antimicrobial properties of chitosan and mode of action: a state of the art review. Int J Food Microbiol. 2010;144(1):51-63. [33] TAO J, ZHANG Y, SHEN A, et al. Injectable Chitosan-Based Thermosensitive Hydrogel/Nanoparticle-Loaded System for Local Delivery of Vancomycin in the Treatment of Osteomyelitis. Int J Nanomedicine. 2020;15:5855-5871. [34] YANG Y, CHU L, YANG S, et al. Dual-functional 3D-printed composite scaffold for inhibiting bacterial infection and promoting bone regeneration in infected bone defect models. Acta Biomater. 2018;79:265-275. [35] ZHENG M, WANG X, CHEN Y, et al. A Review of Recent Progress on Collagen-Based Biomaterials. Adv Healthc Mater. 2023;12(16):e2202042. [36] GRABSKA-ZIELIŃSKA S, SIONKOWSKA A, CARVALHO Â, et al. Biomaterials with Potential Use in Bone Tissue Regeneration-Collagen/Ch:itosan/Silk Fibroin Scaffolds Cross-Linked by EDC/NHS. Materials (Basel). 2021;14(5):1105. [37] ALEGRETE N, SOUSA SR, PADRÃO T, et al. Vancomycin-Loaded, Nanohydroxyapatite-Based Scaffold for Osteomyelitis Treatment: In Vivo Rabbit Toxicological Tests and In Vivo Efficacy Tests in a Sheep Model. Bioengineering (Basel). 2023;10(2):206. [38] WU H, LIN K, ZHAO C, et al. Silk fibroin scaffolds: A promising candidate for bone regeneration. Front Bioeng Biotechnol. 2022;10:1054379. [39] ZHANG L, ZHANG W, HU Y, et al. Systematic Review of Silk Scaffolds in Musculoskeletal Tissue Engineering Applications in the Recent Decade. ACS Biomater Sci Eng. 2021;7(3):817-840. [40] SHABBIRAHMED AM, SEKAR R, GOMEZ LA, et al. Recent Developments of Silk-Based Scaffolds for Tissue Engineering and Regenerative Medicine Applications: A Special Focus on the Advancement of 3D Printing. Biomimetics (Basel). 2023;8(1):16. [41] LI M, YOU J, QIN Q, et al. A Comprehensive Review on Silk Fibroin as a Persuasive Biomaterial for Bone Tissue Engineering. Int J Mol Sci. 2023;24(3):2660. [42] ZHAI X, WU Y, TAN H. Gelatin-based Targeted Delivery Systems for Tissue Engineering. Curr Drug Targets. 2023;24(8):673-687. [43] BELLO AB, KIM D, KIM D, et al. Engineering and Functionalization of Gelatin Biomaterials: From Cell Culture to Medical Applications. Tissue Eng Part B Rev. 2020;26(2):164-180. [44] BAYER IS. Hyaluronic Acid and Controlled Release: A Review. Molecules. 2020;25(11):2649. [45] HWANG HS, LEE CS. Recent Progress in Hyaluronic-Acid-Based Hydrogels for Bone Tissue Engineering. Gels. 2023;9(7):588. [46] ZHANG H, CHENG J, AO Q. Preparation of Alginate-Based Biomaterials and Their Applications in Biomedicine. Mar Drugs. 2021;19(5):264. [47] FARSHIDFAR N, IRAVANI S, VARMA RS. Alginate-Based Biomaterials in Tissue Engineering and Regenerative Medicine. Mar Drugs. 2023;21(3):189. [48] SHARMA S, SUDHAKARA P, SINGH J, et al. Critical Review of Biodegradable and Bioactive Polymer Composites for Bone Tissue Engineering and Drug Delivery Applications. Polymers (Basel). 2021;13(16):2623. [49] BHARADWAZ A, JAYASURIYA AC. Recent trends in the application of widely used natural and synthetic polymer nanocomposites in bone tissue regeneration. Mater Sci Eng C Mater Biol Appl. 2020;110:110698. [50] LV XF, ZHOU DM, SUN XH, et al. Nano Sized Hydroxyapatite-Polylactic Acid-Vancomycin in Alleviation of Chronic Osteomyelitis. Drug Des Devel Ther. 2022;16:1983-1993. [51] LI G, ZHAO M, XU F, et al. Synthesis and Biological Application of Polylactic Acid. Molecules. 2020;25(21):5023. [52] SU Y, ZHANG B, SUN R, et al. PLGA-based biodegradable microspheres in drug delivery: recent advances in research and application. Drug Deliv. 2021;28(1):1397-1418. [53] ZHOU S, LIU S, WANG Y, et al. Advances in the Study of Bionic Mineralized Collagen, PLGA, Magnesium Ionomer Materials, and Their Composite Scaffolds for Bone Defect Treatment. J Funct Biomater. 2023;14(8):406. [54] JIN S, XIA X, HUANG J, et al. Recent advances in PLGA-based biomaterials for bone tissue regeneration. Acta Biomater. 2021;127:56-79. [55] ESSA D, KONDIAH PPD, CHOONARA YE, et al. The Design of Poly(lactide-co-glycolide) Nanocarriers for Medical Applications. Front Bioeng Biotechnol. 2020;8:48. [56] LU Y, CHENG D, NIU B, et al. Properties of Poly (Lactic-co-Glycolic Acid) and Progress of Poly (Lactic-co-Glycolic Acid)-Based Biodegradable Materials in Biomedical Research. Pharmaceuticals (Basel). 2023;16(3):454. [57] CHAVAN YR, TAMBE SM, JAIN DD, et al. Redefining the importance of polylactide-co-glycolide acid (PLGA) in drug delivery. Ann Pharm Fr. 2022;80(5): 603-616. [58] LI J, LI K, DU Y, et al. Dual-Nozzle 3D Printed Nano-Hydroxyapatite Scaffold Loaded with Vancomycin Sustained-Release Microspheres for Enhancing Bone Regeneration. Int J Nanomedicine. 2023;18:307-322. [59] MISTRY S, ROY R, JHA AK, et al. Treatment of long bone infection by a biodegradable bone cement releasing antibiotics in human. J Control Release. 2022;346:180-192. [60] SUN F, SUN X, WANG H, et al. Application of 3D-Printed, PLGA-Based Scaffolds in Bone Tissue Engineering. Int J Mol Sci. 2022;23(10):5831. [61] LI S, SHI X, XU B, et al. In vitro drug release and antibacterial activity evaluation of silk fibroin coated vancomycin hydrochloride loaded poly (lactic-co-glycolic acid) (PLGA) sustained release microspheres. J Biomater Appl. 2022;36(9):1676-1688. [62] DWIVEDI R, KUMAR S, PANDEY R, et al. Polycaprolactone as biomaterial for bone scaffolds: Review of literature. J Oral Biol Craniofac Res. 2020;10(1):381-388. [63] GHARIBSHAHIAN M, SALEHI M, BEHESHTIZADEH N, et al. Recent advances on 3D-printed PCL-based composite scaffolds for bone tissue engineering. Front Bioeng Biotechnol. 2023;11:1168504. [64] YANG X, WANG Y, ZHOU Y, et al. The Application of Polycaprolactone in Three-Dimensional Printing Scaffolds for Bone Tissue Engineering. Polymers (Basel). 2021;13(16):2754. [65] WANG Q, YE W, MA Z, et al. 3D printed PCL/β-TCP cross-scale scaffold with high-precision fiber for providing cell growth and forming bones in the pores. Mater Sci Eng C Mater Biol Appl. 2021;127:112197. [66] LÓPEZ-GONZÁLEZ I, HERNÁNDEZ-HEREDIA AB, RODRÍGUEZ-LÓPEZ MI, et al. Evaluation of the In Vitro Antimicrobial Efficacy against Staphylococcus aureus and epidermidis of a Novel 3D-Printed Degradable Drug Delivery System Based on Polycaprolactone/Chitosan/Vancomycin-Preclinical Study. Pharmaceutics. 2023;15(6):1763. [67] CAI Z, WAN Y, BECKER ML, et al. Poly(propylene fumarate)-based materials: Synthesis, functionalization, properties, device fabrication and biomedical applications. Biomaterials. 2019;208:45-71. [68] 杨峰,马春蒙,王靖,等.可注射PPF骨水泥的制备与固化性能[J].功能高分子学报, 2018,31(1):51-56. [69] GAO J, LIU X, CHENG J, et al. Application of photocrosslinkable hydrogels based on photolithography 3D bioprinting technology in bone tissue engineering. Regen Biomater. 2023; 10: rbad037.doi: 10.1093/rb/rbad037. [70] LUO Y, LE FER G, DEAN D, et al. 3D Printing of Poly(propylene fumarate) Oligomers: Evaluation of Resin Viscosity, Printing Characteristics and Mechanical Properties. Biomacromolecules. 2019;20(4):1699-1708. [71] GUERRA AJ, LARA-PADILLA H, BECKER ML, et al. Photopolymerizable Resins for 3D-Printing Solid-Cured Tissue Engineered Implants. Curr Drug Targets. 2019;20(8):823-838. [72] CHENG T, QU H, ZHANG G, et al. Osteogenic and antibacterial properties of vancomycin-laden mesoporous bioglass/PLGA composite scaffolds for bone regeneration in infected bone defects. Artif Cells Nanomed Biotechnol. 2018;46(8):1935-1947. [73] KAUR G, KUMAR V, BAINO F, et al. Mechanical properties of bioactive glasses, ceramics, glass-ceramics and composites: State-of-the-art review and future challenges. Mater Sci Eng C Mater Biol Appl. 2019;104:109895. [74] ZENG M, XU Z, SONG ZQ, et al. Diagnosis and treatment of chronic osteomyelitis based on nanomaterials. World J Orthop. 2023;14(2):42-54. [75] 马士卿,王洁,高平,等.生物降解微球在骨组织再生中的生物学优势[J].中国组织工程研究,2021,25(34):5517-5522. [76] ZHU H, ZHENG K, BOCCACCINI AR. Multi-functional silica-based mesoporous materials for simultaneous delivery of biologically active ions and therapeutic biomolecules. Acta Biomater. 2021;129:1-17. [77] BURDUȘEL AC, GHERASIM O, ANDRONESCU E, et al. Inorganic Nanoparticles in Bone Healing Applications. Pharmaceutics. 2022;14(4):770. [78] KIM SK, MURUGAN SS, DALAVI PA, et al. Biomimetic chitosan with biocomposite nanomaterials for bone tissue repair and regeneration. Beilstein J Nanotechnol. 2022;13:1051-1067. [79] V K AD, RAY S, ARORA U, et al. Dual drug delivery platforms for bone tissue engineering. Front Bioeng Biotechnol. 2022;10:969843. [80] RITSCHL L, SCHILLING P, WITTMER A, et al. Composite material consisting of microporous beta-TCP ceramic and alginate-dialdehyde-gelatin for controlled dual release of clindamycin and bone morphogenetic protein 2. J Mater Sci Mater Med. 2023;34(8):39. [81] YAZDANPANAH Z, JOHNSTON JD, COOPER DML, et al. 3D Bioprinted Scaffolds for Bone Tissue Engineering: State-Of-The-Art and Emerging Technologies. Front Bioeng Biotechnol. 2022;10: 824156. [82] ABDELAZIZ AG, NAGEH H, ABDO SM, et al. A Review of 3D Polymeric Scaffolds for Bone Tissue Engineering: Principles, Fabrication Techniques, Immunomodulatory Roles, and Challenges. Bioengineering (Basel). 2023;10(2):204. [83] TEWARI AK, UPADHYAY SC, KUMAR M, et al. Insights on Development Aspects of Polymeric Nanocarriers: The Translation from Bench to Clinic. Polymers (Basel). 2022;14(17):3545. [84] BIRK SE, BOISEN A, NIELSEN LH. Polymeric nano- and microparticulate drug delivery systems for treatment of biofilms. Adv Drug Deliv Rev. 2021;174:30-52. [85] YANG J, YAO JL, WU ZQ, et al. Current opinions on the mechanism, classification, imaging diagnosis and treatment of post-traumatic osteomyelitis. Chin J Traumatol. 2021;24(6): 320-327. [86] WANG X, ZHANG M, ZHU T, et al. Flourishing Antibacterial Strategies for Osteomyelitis Therapy. Adv Sci (Weinh). 2023;10(11): e2206154. [87] WENHAO Z, ZHANG T, YAN J, et al. In vitro and in vivo evaluation of structurally-controlled silk fibroin coatings for orthopedic infection and in-situ osteogenesis. Acta Biomater. 2020;116:223-245. |
| [1] | Cheng Weilu, Wang Zehua, Zhang Yidan, Liu Yinghui. Application and regulatory challenges of organoid technology in medical field [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(1): 202-210. |
| [2] | Wang Menghan, Qi Han, Zhang Yuan, Chen Yanzhi. Three kinds of 3D printed models assisted in treatment of Robinson type II B2 clavicle fracture [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1403-1408. |
| [3] | Liu Hanfeng, Wang Jingjing, Yu Yunsheng. Artificial exosomes in treatment of myocardial infarction: current status and prospects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1118-1123. |
| [4] | Ning Tianliang, Wang Kun, Wang Lingbiao, Han Pengfei. Finite element analysis on correction effect of varus foot orthosis based on the three-point force principle [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 891-899. |
| [5] | Yin Tong, Yang Jilei, Li Yourui, Liu Zhuoran, Jiang Ming. Application of core-shell structured nanofibers in oral tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 766-770. |
| [6] | Shen Ziqing, Xia Tian, Shan Yibo, Zhu Ruijun, Wan Haoxin, Ding Hao, Pan Shu, Zhao Jun. Vascularized tracheal substitutes constructed by exosome-load hydrogel-modified 3D printed scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 697-705. |
| [7] | Tian Xin, Liu Tao, Yang Huilin, He Fan. In vitro evaluation of sustained release Kartogenin by gelatin methacryloyl microspheres for repairing nucleus pulposus degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 724-730. |
| [8] | Wei Yuxue, Wang Di, Liu Xiaoqiu. Design, synthesis and properties of oral composite resin monomers with different photoinitiators [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 731-735. |
| [9] | Liu Penghui, Wu Fan, Wang Zejie, Wu Gaoyi, Zhou Libo. Constructing an animal model of temporomandibular joint osteoarthritis in Sprague-Dawley rats by digital technology [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(35): 5642-5648. |
| [10] | Zhang Aili, Huang Jiazheng, Fan Wen, Li Yihuan, Li Shuang, Gan Xuewen, Xiong Ying. Establishing equivalent model to verify the precision of personalized bone model rapidly [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(30): 4795-4799. |
| [11] | Xu Xiaodong, Zhou Jiping, Zhang Qi, Feng Chen, Zhu Mianshun, Shi Hongcan. 3D printing process of gelatin/oxidized nanocellulose skin scaffold with high elastic modulus and high porosity [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 398-403. |
| [12] | Yang Jie, Hu Haolei, Li Shuo, Yue Wei, Xu Tao, Li Yi. Application of bio-inks for 3D printing in tissue repair and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 445-451. |
| [13] | Kong Xiangyu, Wang Xing, Pei Zhiwei, Chang Jiale, Li Siqin, Hao Ting, He Wanxiong, Zhang Baoxin, Jia Yanfei. Biological scaffold materials and printing technology for repairing bone defects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 479-485. |
| [14] | Nie Wen, Huang Hongli, Mo Wenwen, Long Guiyue, Liao Hongbing. Applications of 3D printing in periodontal tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(29): 4671-4676. |
| [15] | Dong Bo, Li Xiaoyu, Li Birong, Li Zhen, Wang Zixuan, Yin Zhaoyi, Meng Weiyan. 3D-printed scaffolds repair infected bone defects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(29): 4685-4690. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||