Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (5): 766-770.doi: 10.12307/2024.257
Previous Articles Next Articles
Application of core-shell structured nanofibers in oral tissue regeneration
Yin Tong1, Yang Jilei2, Li Yourui1, Liu Zhuoran1, Jiang Ming3
- 1Department of Stomatology, Affiliated Hospital of Binzhou Medical University, Binzhou 256600, Shandong Province, China; 2Department of Stomatology, Dezhou Hospital of Traditional Chinese Medicine, Dezhou 253000, Shandong Province, China; 3First Department of Dental and Pulp Diseases, Jinan Stomatological Hospital, Jinan 250000, Shandong Province, China
-
Received:2023-02-20Accepted:2023-04-07Online:2024-02-18Published:2023-08-17 -
Contact:Jiang Ming, MD, Associate chief physician, First Department of Dental and Pulp Diseases, Jinan Stomatological Hospital, Jinan 250000, Shandong Province, China -
About author:Yin Tong, Master candidate, Department of Stomatology, Affiliated Hospital of Binzhou Medical University, Binzhou 256600, Shandong Province, China -
Supported by:Shandong Province Medical and Health Technology Development Project, No. 2017WS753 (to LYR); Science and Technology Plan Project of Binzhou Medical University, No. 2016KJ06 (to LYR)
CLC Number:
Cite this article
Yin Tong, Yang Jilei, Li Yourui, Liu Zhuoran, Jiang Ming. Application of core-shell structured nanofibers in oral tissue regeneration[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 766-770.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
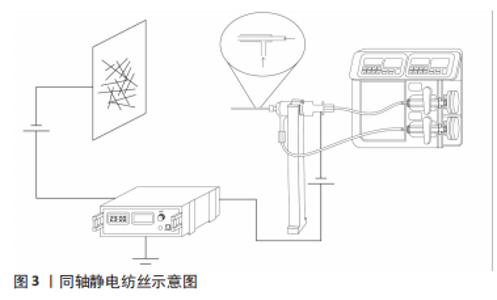
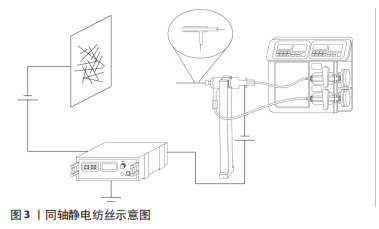
2.1.1 同轴法静电纺丝 2002年,LOSCERTALES等[17]初次报道了一种使用2个不同直径的毛细血管构成同心喷头的静电喷雾装置,生产出直径在微米至纳米范围内的纤维。同轴静电纺丝是在传统静电纺丝技术的基础上发展而来,二者纺丝原理相同,只是改变了喷嘴的结构。在此基础上,SUN等[11]制备出了核壳结构纳米纤维,并在透射电镜下观察到了核壳结构。在纺丝过程中,同轴法旨将两种独立的聚合物溶液分别放置在2个不同直径的同心毛细血管中,在喷嘴处聚集形成复合液滴,并在电场力作用下形成泰勒锥,经过拉伸、固化从而形成了具有核壳结构的纳米纤维[18],见图3。已有研究表明,核壳结构纤维的壳层能够决定药物的释放速率,而核层又能够决定药物负载的量[19]。因此,同轴法静电纺丝可以通过选择结合不同聚合物的优势和装载不同类型生物制剂以实现理想的组织再生。例如,感染是牙周炎缺损治疗失败的主要原因,HE等[15]基于牙周炎的病理学特点,利用同轴静电纺丝分别将柚苷皮和甲硝唑同时负载至具有核壳结构的纳米纤维中,结果显示出短期释放的甲硝唑和长期释放的柚苷皮在抑制炎症的同时特异性促进了细胞的黏附和增殖,以实现感染控制下的牙周组织再生。因此,同轴法静电纺丝通过负载生物活性物质更好地仿生了细胞外基质的特性,并且以可控的方式向细胞传递生化信号,以满足缺损区组织生长所需特性[20]。"
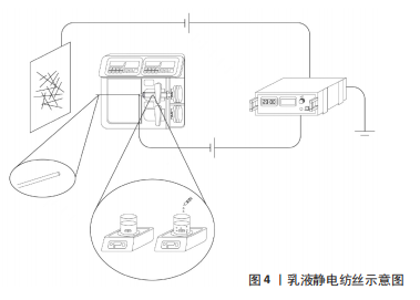
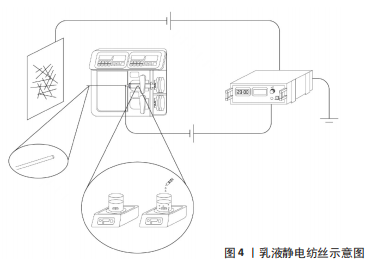
2.1.2 乳液法静电纺丝 乳液法静电纺丝的溶液可分为油包水或水包油两种类型[21]。2003年,SANDERS等[13]将牛血清白蛋白和聚乙烯-醋酸乙烯酯作为两相形成的混悬液进行了电纺,通过光学显微镜观察到了荧光标记的两相,首次提出两相溶液进行静电纺丝以及添加表面活性剂来控制液滴的方法。以油包水型乳液为例是指将药物或蛋白质以水溶液的形式(水相)置于聚合物溶液(油相)中,添加一定量乳化剂/表面活性剂,在磁力搅拌作用下形成稳定均匀的乳液,运用单喷头的静电纺丝装置即可制备出核壳结构的纳米纤维,见图4。YAN等[14]采用乳液法静电纺丝包埋牛血清白蛋白和罗丹明B两种试剂在镜下均观察到了光滑、均匀的纳米纤维,且纤维支架也表现出可控的释放性能,进一步验证了乳液静电纺丝制备核壳结构纤维的可行性。在制备乳液时,添加的乳化剂/表面活性剂不仅在乳化过程中充当油相和水相的隔离层,防止相分离[22],并且通过在静电纺过程中重新分布排列改善了纤维支架的亲水性能[23],因此乳液静电纺丝也被认为是制备生物活性组织工程支架的一种有效措施[24]。此外,有研究认为乳液静电纺核壳纳米纤维尤其适用于携带亲疏水双重药物和蛋白质[25],因而该方法也有望应用于生物医学或制药领域,如组织工程支架、药物输送载体等。"

2.2.1 药物 创口感染是手术患者最常见的并发症之一,术后使用抗生素用于杀灭和抑制细菌来预防感染已成为临床最常用的治疗策略。CAI等[26]首次提出利用Pickering乳液静电纺实现盐酸万古霉素包封,与共混静电纺相比,通过乳液法静电纺制备的纤维不仅具有更优异的机械性能,而且纤维包封的药物表现出更长的缓释时间和优异的抗菌性能。然而随着细菌对抗生素的耐药性问题愈来愈严峻,研究人员开始寻求新的抗菌剂。近期研究文献表明,精油具备广泛的生物特性,如抗菌、镇痛、抗炎、抗糖尿病、抗肿瘤等[27],但其化学性质不稳定。通过乳液法和同轴法静电纺两种方式可成功地将精油封装到核壳纳米纤维的核层,不但提高了精油的物理化学特性,也防止精油与有机溶剂或表面活性剂接触,有望用于天然抗菌剂的系统或局部递送[28]。此外,通过乳液静电纺丝法制备了包载抗肿瘤药物的核壳结构纤维,位于纤维核内的抗肿瘤药物比游离的药物表现出更好的抗肿瘤活性以及更少的不良反应[29],因此核壳纤维也被认为是一种优异的递送抗癌药物的方式。除了抗生素、抗癌药物的包裹负载之外,中药成分类药物也被广泛用于生物医学领域,如姜黄素不仅无毒,还具有抗炎、抗氧化和抗菌活性[30],KIM等[31]使用了三同心喷头制造了负载姜黄素的多层核壳结构纤维支架,与传统同轴法相比可获得更慢的释放率。因此,核壳结构纤维作为药物储备的同时,通过选择合适的聚合物材料以提供与组织生长相匹配的生物学性能和力学性能,满足组织生长所需的特性,实现缺损组织的修复再生[32]。 2.2.2 生长因子 在组织的修复再生过程中,生长因子在细胞增殖、迁移和分化中发挥着重要作用[33-34],然而,生长因子在体内的半衰期短以及快速释放限制了其在促进愈合和组织再生中的临床应用。因此,基于静电纺丝制备的生物支架材料作为生长因子的缓释系统,可以在生物体内获得活性分子持续可控的释放。有学者将血小板衍生生长因子直接装载至壳聚糖-纤维蛋白支架上,生长因子的释放只维持了48 h[35]。为实现长期有效的释放,YANG等[36]利用乳液静电纺丝的优点成功将碱性成纤维细胞生长因子包埋至具有核壳结构的纳米纤维中,表现出低突释率且在接下来的4周内仍持续释放,体内实验结果表明,持续释放的生长因子可促进组织的理想再生。此外,还可通过将细胞-生物材料相结合以适应细胞微环境,进一步促进组织再生[37]。ZHAO等[38]通过乳液静电纺丝和同轴细胞电喷雾二者同时进行制备出了含活细胞的3D纳米纤维,且表现出了良好的细胞存活率以及持续稳定释放的生长因子协同促进了血管的生成,实现了支架、细胞和生物活性物质的三者结合,为制备复杂的仿生结构开辟了新途径。 2.2.3 蛋白质 蛋白质作为细胞的基本组成成分,参与机体酶催化、代谢过程、信号转导和免疫反应等作用[22]。与小分子药物相较,基于蛋白质的治疗药物具有高的特异性,而且能够减少小分子药物不可避免的不良反应。但由于蛋白质制剂半衰期短,一旦进入血液容易被体内免疫细胞或肝/肾脏所清除[39],阻碍了临床的进一步应用,需要可靠的递送系统在保持蛋白质生物活性的同时控制其持续释放,以降低应用剂量和减少给药频率,克服蛋白质的低生物利用率。因此,乳液法或同轴法静电纺丝制备的核壳结构纳米纤维不失为是一种最优选,将蛋白质包裹至纤维的核层可很大程度上避免它们发生化学和物理降解[40]。例如,人血清白蛋白是人血浆中含量最丰富的蛋白之一,由于它自身特性常被用作药物输送剂。牛血清白蛋白的结构与人血清白蛋白相似,在多数研究中以牛血清白蛋白作为模型蛋白进行电纺,比较研究了混合型纤维和核壳型纤维的形态和结构、蛋白质分布和释放曲线,结果表明两种纤维类型的释放曲线虽都观察到了爆发性的释放效果,但后者表现出更持久的蛋白质释放。此外,体外细胞毒性实验也表明,构建的支架具有良好的生物相容性且无细胞毒性[20]。因此,核壳结构纳米纤维可作为一种有前景的蛋白质掺入方法,来保持支架中负载蛋白质的生物活性。 2.2.4 基因 在组织工程的应用研究中,采用核壳结构纤维将质粒DNA (pDNA)传递给细胞以促进原位生产编码的生长因子、信号分子和不溶性生物活性分子的分化已逐渐变得可行[41]。LUU等[42]第一次成功的将质粒DNA利用静电纺丝整合到纳米纤维中,结果也证实了从纤维支架中释放的DNA是完整的。为实现基因的成功传递,需保证在组织再生的时间范围内释放基因,SARAF等[43]由聚乙内酯和聚乙烯亚胺-透明质酸(PEI-HA)作为壳层包裹质粒DNA而形成核壳纤维,与LUU等观察到的前2 h爆发释放之后急剧下降的释放曲线不同的是,通过壳层包裹的基因持续释放的时间延长,为组织再生过程提供了更有利的条件。因此,核壳结构的基因递送具有显著的优势。此外,影响基因传递的另一个重要因素是调节转染效率。XIE等[44]采用同轴静电纺丝将骨形成蛋白2质粒负载至核壳纤维的芯层,实验结果表明带有骨形成蛋白2质粒的支架并没有对细胞增殖产生抑制作用,而且该支架在组织再生及结构降解过程中提供了足够的机械稳定性。与单喷头的纤维支架相比,同轴电纺用于基因传递明显提高了其转染率,有效地促进了组织的修复再生。因此,基因传递与核壳结构纤维支架相结合的基因激活基质被认为将基因传递有效应用于组织工程的一种独特方式。 2.3 核壳结构纤维负载药物的释放原理 运用静电纺丝装置制备的核壳结构纳米纤维,药物的扩散一定程度上取决于材料的性能。根据受损部位选择合适的天然或合成聚合物材料,制造出具有特异性机械强度的支架以进一步满足机体再生需求。当输送材料为不可降解聚合物时,药物的释放以扩散为主导;而对于可降解聚合物的核壳纳米纤维,药物的释放机制受扩散、降解、壳层厚度3个因素的影响,其中增加壳层聚合物的浓度会降低核内药物的释放速率[20]。通常核壳结构纳米纤维的药物释放包括最初的部分突释和长时间的持续释放2个部分。这种初始释放的原因是纤维表面吸附少量的药物,而相对高浓度的药物则位于核层,需要通过在聚合物机体的扩散和聚合物的降解控制实现的[45]。这种药物输送系统应用于体内后,初始突释的药物可以缓解症状,在之后的几小时甚至几天内持续释放药物以达到最终疗效[46]。有研究者采用乳液静电纺丝所得到的包裹血管内皮生长因子的聚L-丙交酯-乙交酯的核壳结构纳米纤维,其体外释放研究显示释放曲线分为前24 h的突释和后648 h的持续释放,并在镜下观察到28 d后的纤维平均直径较释放前减小,还观察到了核壳纳米纤维出现断裂[25],这验证了上述结论并为进一步应用于体内研究提供了理论基础。 2.4 载药核壳结构纤维在口腔组织再生中的应用 口腔组织再生包括颅颌面骨骼再生、牙周软硬组织再生以及软骨再生等,然而其组织的再生修复能力有限且涉及复杂的生理过程,仅依靠单纯的生物支架和单一的药物或生物制剂难以达到预期的治疗效果。此外,口腔是个绝对有菌的环境,这就决定了口腔组织再生修复不应仅局限于如何增强组织的再生能力,还应注重局部免疫微环境对其的影响。运用静电纺丝技术可以制得具有高孔隙率、可调节机械强度及良好的生物相容性及降解性能的纳米纤维支架[47],利于养分和氧气的扩散、促进细胞的黏附、增殖和迁移以及维持组织再生空间。核壳结构纳米纤维是基于对传统静电纺丝方法的基础上进行改良,能够包封不同类型的生物活性制剂并可通过调节纤维结构来控制药物释放,以适应组织修复愈合的过程[48]。因此,在口腔组织再生中采用核壳结构纳米纤维既能够提高组织的再生修复能力,又可以显著改善创口感染问题、降低复发率和失败率,促进组织的理想再生。 2.4.1 颌骨的修复再生 创伤、炎症、肿瘤以及手术等都易引起牙槽骨及颌骨的吸收甚至缺损,严重影响患者的健康和生活质量。然而,牙槽骨内源性的修复再生能力有限,加之牙周组织率先长入缺损区以及炎症因子的影响,牙槽骨缺损的完全再生是非常困难的。目前组织工程可以将细胞和生长因子输送至缺陷部位并保持其活性等优势而被广泛应用于骨的修复再生[49]。虽然牙槽骨与机体的其他骨骼相比具有不同的发育来源和骨化过程,但是在骨缺损的修复再生过程中血管生成都是骨形成的先决条件[50]。CHENG等[51]通过核壳结构实现骨形成蛋白2和结缔组织生长因子的双生长因子负载及缓释,细胞培养及临界骨缺损实验显示,结缔组织生长因子的突释在成骨分化早期抑制了骨形成蛋白2的诱导成骨,但瞬时释放的结缔组织生长因子并不会阻碍成骨分化的全程,反而在成骨分化后期结缔组织生长因子的促成血管的作用与骨形成蛋白2的持续释放协同增强了骨形成,促进了早期骨的再生。此外,天然骨细胞外基质呈现出有机和无机成分构成的纳米纤维网络[52]。ZHANG等[53]通过同轴电纺将无机材料钙钛矿和生物活性药物淫羊藿分别引入纤维的壳层和核层,提高了骨纤维支架的生物活性,还解决了淫羊藿不能够单独电纺的问题;经过体外细胞培养验证了钙钛矿和缓慢释放的淫羊藿有利益于矿物质的沉积,并且具有协同促进成骨的作用。因此在骨组织工程中,用于多重药物递送的核壳纤维支架有效支撑了细胞的生长和充当了药物、生长因子等诱导骨形成因素的储存库,促进颌骨的修复再生[54]。 2.4.2 软骨的修复再生 颞下颌关节是口腔颌面部软骨组织所在的主要部位,针对颞下颌关节器质性病变的临床治疗措施多采取保守治疗和必要时的介入治疗[55]。由于软骨的细胞外基质主要是由Ⅱ型胶原蛋白和蛋白多糖组成,且软骨的自我修复能力很差,因此,可以通过组织工程的方法为治疗颞下颌关节病变开辟一种新的治疗措施[56]。MENEZES等[57]通过传统静电纺丝制备了含有部分硫酸化纤维素(巯基糖胺聚糖的模拟物)的明胶纤维支架,虽有利于人间充质干细胞的软骨分化,但在第28天后细胞数量未见明显增殖。因此,为了延长药物和生长因子的作用,有研究者通过使用同轴静电纺丝将硫酸氨基葡萄糖封装在聚乙内酯纳米纤维的核心中,细胞实验表明位于核层硫酸氨基葡萄糖的缓慢释放刺激了软骨细胞外基质中蛋白多糖和胶原蛋白的合成,展示了同轴静电纺丝技术在软骨组织工程支架方面的应用前景[58]。目前,组织工程应用于软骨的再生修复主要集中于膝关节[59],虽然对于如何更好地应用于颞下颌关节仍需要进一步探索研究,但也为颞下颌关节病变的修复治疗提供了新思路。 2.4.3 牙周软硬组织的修复再生 牙周炎是一种慢性炎性疾病,细菌是诱发牙周炎的始动因子,随着时间的推移缓慢不可逆地造成软组织损伤和牙槽骨吸收,最终影响牙齿的功能及美观。为治疗牙周疾病,在20世纪末曾有学者提出利用物理屏障即引导组织再生术来防止牙周组织修复再生过程中上皮组织和结缔组织率先长入缺损区域,阻碍牙周膜的重新附着和骨再生[60-61],或者在缺损区域放置骨填充物防止骨丧失等方法[62]。但是在大范围的牙周组织缺损中,引导组织再生术使用的膜材料作为一个隔离的物理屏障缺乏抑制细菌和主动诱导组织再生的能力;单纯的骨移植材料可以促进骨再生,但并不能促进软组织再生,所以单一的生物材料无法充分指导不同类型组织的协调生长和发育[63]。因此,利用核壳结构纳米纤维的优势,结合生物活性因子、药物、基因治疗或细胞递送,以重建牙周组织结构和功能[64]。但是由于生物支架材料、生物支架的制备方式和负载的生物活性因子的不同,会产生不同的效果。过去许多研究主要集中于单组分抑菌剂的包载或促进软硬组织再生的生物因子的传递[65-66]。最近,ZHANG等[63]通过乳液静电纺丝制备了包裹奥硝唑和碱性成纤维细胞生长因子的核壳纳米纤维,其中外壳的奥硝唑抑制了细菌生长,核心层的碱性成纤维生长因子有助于组织再生,充分发挥药物的作用,更有效地诱导组织再生。此外,LIU等[67]通过同轴静电纺丝也构建了多种药物有序释放的核壳纳米纤维,在纤维膜长达4周的体外释放过程中,最开始只检测到了壳层药物的释放,直到第12天才检测到骨形成蛋白2的释放,这种有序释放不仅发挥了抑制炎症反应的作用,还通过促成骨药物来诱导骨再生。近期,国内外学者的研究也验证了这种具有成骨和抗菌特性的核壳纳米纤维膜既可以缓解和控制牙周炎症[15,68-70],也在一定程度上修复骨和软组织缺损,作用于牙周组织再生是非常理想的。"

| [1] DATTANI S, LI X, LAMPA C, et al. A Comparative Study on Micelles, Liposomes and Solid Lipid Nanoparticles for Paclitaxel Delivery. Int J Phar. 2023;631:122464. [2] ZHONG J, WEN W, WANG J, et al. Bone-Targeted Dual Functional Lipid-coated Drug Delivery System for Osteosarcoma Therapy. Pharm Res. 2023;40(1):231-243. [3] TEFAS LR, TOMA I, SESARMAN A, et al. Co-delivery of gemcitabine and salinomycin in PEGylated liposomes for enhanced anticancer efficacy against colorectal cancer. J Liposome Res. 2022;15:1-7. [4] LI S, DONG S, XU W, et al. Antibacterial Hydrogels. Adv Sci. 2018;5(5):1700527. [5] NING C, GUO Y, YAN L, et al. On-Demand Prolongation of Peripheral Nerve Blockade through Bupivacaine-Loaded Hydrogels with Suitable Residence Periods.ACS Biomater Sci Eng. 2019;5(2):696-709. [6] BAYER IS. A Review of Sustained Drug Release Studies from Nanofiber Hydrogels. Biomedicines. 2021;9(11):1612. [7] MILES CE, GWIN C, ZUBRIS KAV, et al. Tyrosol Derived Poly(ester-arylate)s for Sustained Drug Delivery from Microparticles. ACS Biomater Sci Eng. 2021;7(6):2580-2591. [8] YOO J, WON YY. Phenomenology of the Initial Burst Release of Drugs from PLGA Microparticles. ACS Biomater Sci Eng. 2020;6(11):6053-6062. [9] BRANNON ER, GUEVARA MV, PACIFICI NJ, et al. Polymeric particle-based therapies for acute inflammatory diseases. Nat Rev Mater. 2022;7(10):796-813. [10] 刘威,钟伟,都有为.核/壳结构复合纳米材料研究进展[J].材料导报,2007(3):59-62. [11] SUN Z, ZUSSMAN E, YARIN AL, et al. Compound Core–Shell Polymer Nanofibers by Co-Electrospinning. Adv Mater. 2003;15(22):1929-1932. [12] WANG CY, LIU JJ, FAN CY, et al. The Effect of Aligned Core–Shell Nanofibres Delivering NGF on the Promotion of Sciatic Nerve Regeneration. J Biomater Sci Polym Ed. 2012; 23(1-4):167-184. [13] SANDERS EH, KLOEFKORN R, BOWLIN GL, et al. Two-Phase Electrospinning from a Single Electrified Jet: Microencapsulation of Aqueous Reservoirs in Poly(ethylene- co -vinyl acetate) Fibers. Macromolecules. 2003;36(11):3803-3805. [14] YAN S, XIAOQIANG L, SHUIPING L, et al. Controlled release of dual drugs from emulsion electrospun nanofibrous mats. Colloids Surf B Biointerfaces. 2009;73(2):376-381. [15] HE P, ZHONG Q, GE Y, et al. Dual drug loaded coaxial electrospun PLGA/PVP fiber for guided tissue regeneration under control of infection. Mater Sci Eng C Mater Biol Appl. 2018;90:549-556. [16] MONFARED M, TAGHIZADEH S, ZARE-HOSEINABADI A, et al. Emerging frontiers in drug release control by core-shell nanofibers: a review. Drug Metab Rev. 2019;51(4):589-611. [17] LOSCERTALES IG, BARRERO A, GUERRERO I, et al. Micro/Nano Encapsulation via Electrified Coaxial Liquid Jets. Science. 2002;295(5560):1695-1698. [18] LU Y, HUANG J, YU G, et al. Coaxial electrospun fibers: applications in drug delivery and tissue engineering. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2016;8(5):654-677. [19] ZHANG Y, HUANG ZM, XU XJ, et al. Preparation of Core−Shell Structured PCL-r-Gelatin Bi-Component Nanofibers by Coaxial Electrospinning. Chem Mater. 2004;16(18):3406-3409. [20] RAFIEI M, JOOYBAR E, ABDEKHODAIE MJ, et al. Construction of 3D fibrous PCL scaffolds by coaxial electrospinning for protein delivery. Mater Sci Eng C Mater Biol Appl. 2020;113:110913. [21] TIAN L, PRABHAKARAN MP, DING X, et al. Biocompatibility evaluation of emulsion electrospun nanofibers using osteoblasts for bone tissue engineering. J Biomater Sci Polym Ed. 2013;24(17):1952-1968. [22] KURPANIK R, LECHOWSKA-LISZKA A, MASTALSKA-POPŁAWSKA J, et al. Effect of Ionic and Non-Ionic Surfactant on Bovine Serum Albumin Encapsulation and Biological Properties of Emulsion-Electrospun Fibers. Molecules. 2022;27(10):3232. [23] LI X, SU Y, LIU S, et al. Encapsulation of proteins in poly(l-lactide-co-caprolactone) fibers by emulsion electrospinning. Colloids Surf B Biointerfaces. 2010;75(2):418-424. [24] BRIGGS T, ARINZEH TL. Examining the formulation of emulsion electrospinning for improving the release of bioactive proteins from electrospun fibers. J Biomed Mater Res A. 2014;102(3):674-684. [25] TIAN L, PRABHAKARAN MP, DING X, et al. Emulsion electrospun vascular endothelial growth factor encapsulated poly(l-lactic acid-co-ε-caprolactone) nanofibers for sustained release in cardiac tissue engineering. J Mater Sci. 2012;47(7):3272-3281. [26] CAI N, HAN C, LUO X, et al. Fabrication of Core/Shell Nanofibers with Desirable Mechanical and Antibacterial Properties by Pickering Emulsion Electrospinning. Macromol Mater Eng. 2017;302(3):1600364. [27] PARTHENIADIS I, STATHAKIS G, TSALAVOUTI D, et al. Essential Oil—Loaded Nanofibers for Pharmaceutical and Biomedical Applications: A Systematic Mini-Review. Pharmaceutics. 2022;14(9):1799. [28] BARZEGAR S, ZARE MR, SHOJAEI F, et al. Core-shell chitosan/PVA-based nanofibrous scaffolds loaded with Satureja mutica or Oliveria decumbens essential oils as enhanced antimicrobial wound dressing. Int J Pharm. 2021;597:120288. [29] LUO X, XIE C, WANG H, et al. Antitumor activities of emulsion electrospun fibers with core loading of hydroxycamptothecin via intratumoral implantation. Int J Pharm. 2012;425(1-2):19-28. [30] ALVEN S, ADERIBIGBE BA. Efficacy of Polymer-Based Nanocarriers for Co-Delivery of Curcumin and Selected Anticancer Drugs. Nanomaterials. 2020;10(8):1556. [31] KIM H, BAE JE, SHIN MJ. Controlled release of curcumin from coaxial electrospun nanofiber mats. Bull Korean Chem Soc. 2022;43(7): 984-989. [32] HUANG ZM, HE CL, YANG A, et al. Encapsulating drugs in biodegradable ultrafine fibers through co-axial electrospinning. J Biomed Mater Res A. 2006;77(1):169-179. [33] SIMONS M, ANNEX BH, LAHAM RJ, et al. Pharmacological Treatment of Coronary Artery Disease With Recombinant Fibroblast Growth Factor-2: Double-Blind, Randomized, Controlled Clinical Trial. Circulation. 2002;105(7):788-793. [34] LEE KY, PETERS MC, MOONEY DJ. Comparison of vascular endothelial growth factor and basic fibroblast growth factor on angiogenesis in SCID mice. J Control Release. 2003;87(1-3):49-56. [35] YUAN TT, DIGEORGE FOUSHEE AM, JOHNSON MC, et al. Development of Electrospun Chitosan-Polyethylene Oxide/Fibrinogen Biocomposite for Potential Wound Healing Applications. Nanoscale Res Lett. 2018;13(1):88. [36] YANG Y, XIA T, ZHI W, et al. Promotion of skin regeneration in diabetic rats by electrospun core-sheath fibers loaded with basic fibroblast growth factor. Biomaterials. 2011;32(18):4243-4254. [37] HUANG G, LI F, ZHAO X, et al. Functional and Biomimetic Materials for Engineering of the Three-Dimensional Cell Microenvironment. Chem Rev. 2017;117(20):12764-12850. [38] ZHAO Q, ZHOU Y, WANG M. Three-dimensional endothelial cell incorporation within bioactive nanofibrous scaffolds through concurrent emulsion electrospinning and coaxial cell electrospraying. Acta Biomater. 2021;123:312-324. [39] PATEL A, CHOLKAR K, MITRA AK. Recent developments in protein and peptide parenteral delivery approaches. Ther Deliv. 2014;5(3):337-365. [40] MOREIRA A, LAWSON D, ONYEKURU L, et al. Protein encapsulation by electrospinning and electrospraying. J Control Release. 2021;329:1172-1197. [41] AHMADI S, RABIEE N, FATAHI Y, et al. Controlled Gene Delivery Systems: Nanomaterials and Chemical Approaches. J Biomed Nanotechnol. 2020;16(5):553-582. [42] LUU YK, KIM K, HSIAO BS, et al. Development of a nanostructured DNA delivery scaffold via electrospinning of PLGA and PLA–PEG block copolymers. J Control Release. 2003;89(2):341-353. [43] SARAF A, BAGGETT LS, RAPHAEL RM, et al. Regulated non-viral gene delivery from coaxial electrospun fiber mesh scaffolds. J Control Release. 2010;143(1):95-103. [44] XIE Q, JIA LN, XU HY, et al. Fabrication of Core-Shell PEI/pBMP2-PLGA Electrospun Scaffold for Gene Delivery to Periodontal Ligament Stem Cells. Stem Cells Int. 2016;2016:5385137. [45] FREDENBERG S, WAHLGREN M, RESLOW M, et al. The mechanisms of drug release in poly(lactic-co-glycolic acid)-based drug delivery systems--a review. Int J Pharm. 2011;415(1-2):34-52. [46] CRACOWSKI JL, HULOT JS, LAPORTE S, et al. Clinical pharmacology: Current innovations and future challenges. Fundam Clin Pharmacol. 2022;36(3):456-467. [47] LAN X, WANG H, BAI J, et al. Multidrug-loaded electrospun micro/nanofibrous membranes: Fabrication strategies, release behaviors and applications in regenerative medicine. J Control Release. 2021;330:1264-1287. [48] XU X, REN S, LI L, et al. Biodegradable engineered fiber scaffolds fabricated by electrospinning for periodontal tissue regeneration. J Biomater Appl. 2021;36(1):55-75. [49] KOONS GL, DIBA M, MIKOS AG. Materials design for bone-tissue engineering. Nat Rev Mater. 2020;(5):584-603. [50] RATHER HA, JHALA D, VASITA R. Dual functional approaches for osteogenesis coupled angiogenesis in bone tissue engineering. Mater Sci Eng C Mater Biol Appl. 2019;103:109761. [51] CHENG G, YIN C, TU H, et al. Controlled Co-delivery of Growth Factors through Layer-by-Layer Assembly of Core-Shell Nanofibers for Improving Bone Regeneration. ACS Nano. 2019;13(6):6372-6382. [52] SHARMA A, KOKIL GR, HE Y, et al. Inorganic/organic combination: Inorganic particles/polymer composites for tissue engineering applications. Bioact Mater. 2023;24:535-550. [53] ZHANG S, ZHANG M, BAI R, et al. Electrospun coaxial nanofibers loading with perovskite and icariin to enhance the bone scaffold-mediated osteogenesis. Mater Today Chem. 2022;26:101246. [54] MEINEL AJ, GERMERSHAUS O, LUHMANN T, et al. Electrospun matrices for localized drug delivery: Current technologies and selected biomedical applications. Eur J Pharm Biopharm. 2012;81(1):1-13. [55] ZHANG R, MA J, HAN J, et al. Mesenchymal stem cell related therapies for cartilage lesions and osteoarthritis. Am J Transl Res. 2019;11(10):6275-6289. [56] ÂNGELO DF, GIL FM. Tissue Engineering in Temporomandibular Joint Reconstruction. Atlas Oral Maxillofac Surg Clin North Am. 2022;30(2):235-246. [57] MENEZES R, ARINZEH TL. Comparative Study of Electrospun Scaffolds Containing Native GAGs and a GAG Mimetic for Human Mesenchymal Stem Cell Chondrogenesis. Ann Biomed Eng. 2020;48(7):2040-2052. [58] CHEN W, WANG C, GAO Y, et al. Incorporating chitin derived glucosamine sulfate into nanofibers via coaxial electrospinning for cartilage regeneration. Carbohydr Polym. 2020;229:115544. [59] BIELAJEW BJ, DONAHUE RP, ESPINOSA MG, et al. Knee orthopedics as a template for the temporomandibular joint. Cell Rep Med. 2021;2(5):100241. [60] MELCHER AH. On the repair potential of periodontal tissues. J Periodontol. 1976;47(5): 256-260. [61] GIANNOBILE WV. Commentary: Treatment of Periodontitis: Destroyed Periodontal Tissues Can Be Regenerated Under Certain Conditions. J Periodontol. 2014;85(9):1151-1154. [62] KIM CK, CHOI EJ, CHO KS, et al. Periodontal repair in intrabony defects treated with a calcium carbonate implant and guided tissue regeneration. J Periodontol. 1996; 67(12):1301-1306. [63] ZHANG W, WANG Q, WANG K, et al. Construction of an ornidazole/bFGF-loaded electrospun composite membrane with a core-shell structure for guided tissue regeneration. Mater Des. 2022;221:110960. [64] VAQUETTE C, PILIPCHUK SP, BARTOLD PM, et al. Tissue Engineered Constructs for Periodontal Regeneration: Current Status and Future Perspectives. Adv Healthc Mater. 2018;7(21):e1800457. [65] ABDELAZIZ D, HEFNAWY A, AL-WAKEEL E, et al. New biodegradable nanoparticles-in-nanofibers based membranes for guided periodontal tissue and bone regeneration with enhanced antibacterial activity. J Adv Res. 2021;28:51-62. [66] CABALLÉ-SERRANO J, ABDESLAM-MOHAMED Y, MUNAR-FRAU A, et al. Adsorption and release kinetics of growth factors on barrier membranes for guided tissue/bone regeneration: A systematic review. Arch Oral Biol. 2019;100:57-68. [67] LIU X, ZHANG W, WANG Y, et al. One-step treatment of periodontitis based on a core-shell micelle-in-nanofiber membrane with time-programmed drug release. J Control Release. 2020;320:201-213. [68] XU C, CAO Y, LEI C, et al. Polymer–Mesoporous Silica Nanoparticle Core–Shell Nanofibers as a Dual-Drug-Delivery System for Guided Tissue Regeneration. ACS Appl Nano Mater. 2020;3(2):1457-1467. [69] PENG W, REN S, ZHANG Y, et al. MgO Nanoparticles-Incorporated PCL/Gelatin-Derived Coaxial Electrospinning Nanocellulose Membranes for Periodontal Tissue Regeneration. Front Bioeng Biotechnol. 2021;9:668428. [70] HE Z, LIU S, LI Z, et al. Coaxial TP/APR electrospun nanofibers for programmed controlling inflammation and promoting bone regeneration in periodontitis-related alveolar bone defect models. Mater Today Bio. 2022;16:100438. |
| [1] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [2] | Liu Hanfeng, Wang Jingjing, Yu Yunsheng. Artificial exosomes in treatment of myocardial infarction: current status and prospects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1118-1123. |
| [3] | Tian Xin, Liu Tao, Yang Huilin, He Fan. In vitro evaluation of sustained release Kartogenin by gelatin methacryloyl microspheres for repairing nucleus pulposus degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 724-730. |
| [4] | Zhu Liwei, Wang Jiangyue, Bai Ding. Application value of nanocomposite gelatin methacryloyl hydrogels in different bone defect environments [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 753-758. |
| [5] | Xu Rong, Wang Haojie, Geng Mengxiang, Meng Kai, Wang Hui, Zhang Keqin, Zhao Huijing. Research advance in preparation and functional modification of porous polytetrafluoroethylene artificial blood vessels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 759-765. |
| [6] | Wang Jiani, Chen Junyu. Angiogenesis mechanism of metal ions and their application in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 804-812. |
| [7] | Yang Yuqing, Chen Zhiyu. Role and application of early transient presence of M1 macrophages in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 594-601. |
| [8] | Wang Xinyi, Xie Xianrui, Chen Yujie, Wang Xiaoyu, Xu Xiaoqing, Shen Yihong, Mo Xiumei. Electrospun nanofiber scaffolds for soft and hard tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 426-432. |
| [9] | Gao Xueyu, Zhang Wentao, Sun Tianze, Zhang Jing, Li Zhonghai. Application of metal ions in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 439-444. |
| [10] | Chen Pinrui, Pei Xibo, Xue Yiyuan. Function and advantages of magnetically responsive hydrogel in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 452-457. |
| [11] | Long Zhirui, Huang Lei, Xiao Fang, Wang Lin, Wang Xiaobei. Characteristics of hydrogel microspheres in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 472-478. |
| [12] | Xu Jing, Lyu Huixin, Bao Xin, Zhang Yi, Wang Yihan, Zhou Yanmin. Application of near infrared responsive hydrogels in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 486-492. |
| [13] | Zhai Haoyan, Zhao Yuan, Fan Dengying, Liu Chunyan. The role of reactive oxygen species in periodontitis and periodontal tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(14): 2254-2260. |
| [14] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [15] | Zhang Min, Zhang Xiaoming, Liu Tongbin. Application potential of naringin in bone tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 787-792. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||



