Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (14): 2254-2260.doi: 10.12307/2024.288
Previous Articles Next Articles
The role of reactive oxygen species in periodontitis and periodontal tissue regeneration
Zhai Haoyan, Zhao Yuan, Fan Dengying, Liu Chunyan
- Department of Orthodontics, School and Hospital of Stomatology, Hebei Medical University, Hebei Key Laboratory of Stomatology, Hebei Clinical Research Center for Oral Diseases, Shijiazhuang 050017, Hebei Province, China
-
Received:2023-03-22Accepted:2023-04-10Online:2024-05-18Published:2023-07-28 -
Contact:Liu Chunyan, MD, Chief physician, Associate professor, Department of Orthodontics, School and Hospital of Stomatology, Hebei Medical University, Hebei Key Laboratory of Stomatology, Hebei Clinical Research Center for Oral Diseases, Shijiazhuang 050017, Hebei Province, China -
About author:Zhai Haoyan, Master candidate, Department of Orthodontics, School and Hospital of Stomatology, Hebei Medical University, Hebei Key Laboratory of Stomatology, Hebei Clinical Research Center for Oral Diseases, Shijiazhuang 050017, Hebei Province, China -
Supported by:2021 Provincial Government-funded Medical Excellence Project, No. 361029 (to LCY); 2022 Grant Program for the Introduction of Overseas Students, No. C20220353 (to LCY)
CLC Number:
Cite this article
Zhai Haoyan, Zhao Yuan, Fan Dengying, Liu Chunyan. The role of reactive oxygen species in periodontitis and periodontal tissue regeneration[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(14): 2254-2260.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
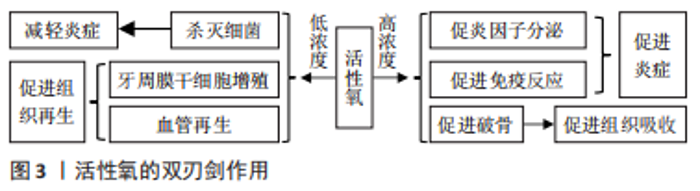
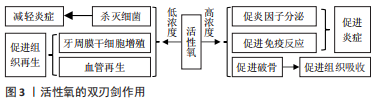
2.1 活性氧的来源及作用 活性氧包括羟基自由基、超氧阴离子、单线态氧和过氧化氢等,在生物体内,活性氧产生于定位于质膜、胞质溶胶、过氧化物酶体、线粒体和内质网膜上的多种细胞系统[12-18]。线粒体在呼吸底物存在下产生过氧化氢,过氧化氢是一种温和的活性氧分子,可以导致氧化还原蛋白的可逆性改变。在细胞质中,硫氧还蛋白通过还原过氧化氢来抵消细胞氧化应激,再由还原酶维持活性,实现胞内的氧化还原缓冲[19]。生物体内的活性氧主要来源于中性粒细胞的呼吸爆发过程中NADPH氧化酶产生的超氧阴离子[20],反应活性高的超氧阴离子可能对低特异性的大分子造成不可逆的损伤[19]。体内还有一部分活性氧来自外源的辐射及氧化还原循环等过程[21],每个细胞中每天产生的细胞内活性氧量达到 10 亿个分子[22]。 活性氧在生物体发挥重要作用,在细胞水平,活性氧调节生长、凋亡和信号传导;在系统水平,活性氧有助于复杂的功能调节,包括免疫调节等。活性氧具有双重作用,见图3。低浓度的活性氧在信号传导过程中作为调节介质发挥重要作用,然而在中高浓度时,它们对生物体有害,使重要的细胞分子失活[19]。"

2.2 活性氧在牙周炎发生发展机制中的作用 牙周炎是牙周组织的炎症破坏性疾病,菌斑是牙周炎发生的始动因子。细菌入侵后,中性粒细胞发生呼吸爆发,产生大量活性氧,杀灭细菌,但同时过量的活性氧则会导致组织的损伤[23]。活性氧在牙周炎中起着双重作用,它们不仅可以引起氧化损伤和组织功能障碍,还可以作为分子信号激活对生物体有益的应激反应[19]。以上证据表明,活性氧水平的升高与抗氧化能力降低可能是导致牙周炎患者组织破坏的重要因素[24],活性氧是牙周炎发生发展过程中的重要因素。 2.2.1 低浓度活性氧杀灭病原菌 口腔微生物由数百种不同的细菌组成,它们能够与胞外聚合物基质形成三维结构,称为生物膜。与浮游状态相比,生物被膜中的细菌对抗菌剂以及宿主的保护性免疫反应更有抵抗力。口腔链球菌在生物膜早期形成阶段占主导地位。生物膜中牙龈卟啉单胞菌、伴放线放线杆菌等致病菌成分较高则可导致牙周炎。口腔固有免疫由中性粒细胞主导,当牙周组织中存在病原菌时,中性粒细胞通过吞噬作用产生活性氧,活性氧可以对微生物造成直接的氧化损伤,例如蛋氨酸残基的氧化、脂质过氧化、碱氧化和脱氨以及DNA链断裂等[25]。研究发现具核梭杆菌通过激活人牙龈成纤维细胞中的蛋白激酶B/丝裂原活化激酶信号通路促进活性氧的产生和炎症细胞因子的产生[26]。口腔链球菌、放线菌和牙龈卟啉单胞菌可以诱导中性粒细胞产生活性氧,而在同一免疫细胞中,口腔链球菌诱导活性氧产生,但当活性氧水平过高时,可能通过产生抗氧化剂来限制氧化损伤[27]。研究发现,牙周炎患者局部和全身的活性氧水平升高,与牙周炎严重程度呈正相关[22]。以上证据表明,活性氧是有效对抗微生物所需的关键成分。 2.2.2 高浓度活性氧促进牙周炎症 促进炎症因子分泌:NOD样受体热蛋白结构域相关蛋白3(nucleotide‐ binding leucine‐rich repeat protein 3,NLRP3)炎性小体是由感应蛋白NLRP3、衔接蛋白和半胱氨酸蛋白酶原1组成的复合物。研究发现活性氧是激活NLRP3炎性小体必需的上游信号[12],通过使硫氧还蛋白互作蛋白与硫氧还蛋白解离来诱导NLRP3的激活[22],NLRP3被激活后,与衔接蛋白和半胱氨酸蛋白酶原1结合,导致半胱氨酸蛋白酶的激活,进而导致白细胞介素1家族(即白细胞介素1β和白细胞介素18)促炎细胞因子的释放[28]。抑制活性氧的表达可以降低NLRP3炎症小体相关因子表达[29]。研究发现牙周炎患者的血清和唾液NLRP3浓度升高[30],牙周治疗后NLRP3水平显著降低[31]。牙龈卟啉单胞菌通过活性氧/ 硫氧还蛋白互作蛋白/ NLRP3炎症小体通路导致牙周膜干细胞的损伤,并影响其迁移,研究表明该通路诱导的炎症反应是导致慢性牙周炎所必需的关键启动机制[32]。以上证据表明,活性氧可以促进NLRP3炎症小体及白细胞介素1家族炎症因子的表达[33],从而加重牙周炎症。 促进免疫损伤:中性粒细胞是机体免疫反应的第一道防线,病原菌入侵可以诱导中性粒细胞产生活性氧分子[27],甚至在没有病原入侵的情况下,牙周炎患者体内存在“过反应”的中性粒细胞也会产生更多的活性氧分子[34],加剧的活性氧产生导致假抗原和氧化生物分子的产生[35],进而通过间接免疫损伤参与牙周炎的发生发展[14]。通过比较牙周健康者和不同程度慢性牙周炎患者的牙周指标、炎症指标、氧化应激和中性粒细胞-内皮细胞相互作用指标,发现氧化状态的不平衡和中性粒细胞功能障碍可能促进慢性牙周炎中出现过度活跃的中性粒细胞表型,但“高反应表型”的中性粒细胞占比尚未有研究报道[36]。以上证据表明,牙周炎患者牙周组织内更多的活性氧分子会加重免疫损伤。 直接损伤组织:超氧阴离子作为活性氧的一种,是一类反应活性较高的自由基,可能对低特异性的大分子造成不可逆的损伤[19],可以通过氧化损伤脂质、蛋白质、DNA链等对牙周组织造成直接损伤[37],并生成相应的氧化损伤产物,见表1,也可以导致端粒缩短及丧失[38],干扰细胞生长和细胞周期进程,诱导细胞凋亡[39],还可以通过遗传和表观遗传改变在人类多种疾病的病因和进展中发挥重要作用[40] 。研究发现牙周炎患者龈沟液的氧化损伤产物蛋白质碳基化水平显著高于牙周健康者,且其水平与临床牙周参数显著相关[8],牙周炎患者红细胞中的脂质氧化损伤产物丙二醛水平显著高于牙周健康者[41],牙周炎患者血浆内的抗氧化剂水平降低[42]。以上研究表明,牙周炎患者全身和局部的氧化损伤产物增多,说明活性氧在牙周炎患者组织内通过氧化生物大分子直接损伤牙周组织,加重牙周炎症。"


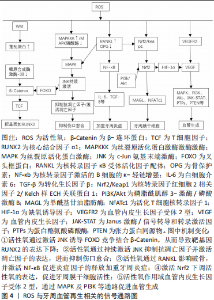
2.2.3 活性氧在牙周炎发生发展中相关信号通路 活性氧为多效性生理信号传导剂[43],参与多条细胞信号通路,介导细胞的生理病理过程。其中与牙周炎发生发展有关的信号通路包括核转录因子?B受体活化因子配体(receptor activator of nuclear factor-?B ligand,RANKL)/RANK/骨保护素信号通路,c-Jun氨基末端激酶(c-Jun N-terminal kinase,JNK)通路,Wnt/β-连环蛋白信号通路等。 JNK通路:又被称为应激活化蛋白激酶,是丝裂原活化激酶信号通路的一个重要分支,它与炎症、凋亡和生长有关。活性氧可以通过激活丝裂原活化激酶激酶激酶或使丝裂原活化激酶磷酸酶失活促使JNK的持续激活[44],进而抑制B细胞淋巴瘤2等抗凋亡因子并激活B细胞淋巴瘤2相关X蛋白等促凋亡因子促进细胞死亡并抑制伤口愈合[45]。研究发现,活性氧通过JNK通路诱导细胞凋亡是有条件的并且是细胞依赖的[22],或许与细胞分化程度有关。研究发现,牙龈卟啉单胞菌入侵后,上皮细胞中JNK参与叉头框蛋白的激活,增强了细胞的抗氧化能力[46]。另有研究发现,蓝光处理会诱发氧化应激从而激活丝裂原活化激酶信号传导,丝裂原活化激酶信号传导还具有抗炎作用并调节抗氧化防御[47]。所以活性氧和JNK信号通路在牙周炎的发生发展中发挥双重作用,活性氧可以通过JNK通路促进凋亡加重牙周组织损伤,但在某些条件下JNK可以通过激活抗氧化防御实现细胞保护作用,如何有效利用这种相互调节关系减轻牙周炎症还有待进一步研究。 RANKL/RANK/骨保护素信号通路:此信号通路维持成骨细胞成骨和破骨细胞破骨的平衡。RANKL和骨保护素是影响破骨细胞形成的关键因子[23],在牙周炎的骨吸收过程中发挥重要作用。活性氧及氧化损伤产物和肿瘤坏死因子α等细胞因子可以抑制骨保护素表达并上调RANKL表达[48],RANKL参与激活丝裂原活化激酶和核转录因子?B信号通路,促进趋化因子和炎症细胞因子的分泌,从而造成炎症反应和破骨细胞分化,而这种作用可以被抗氧化剂阻断[23]。研究发现,活性氧的负反馈调节因子也可以通过减弱RANKL诱导的核转录因子?B活化并减少活性氧的产生[49]。RANKL还可以通过抑制抗氧化防御系统进而增加活性氧的水平[50]。总之,RANKL可以促进活性氧的生成及炎症因子的表达,活性氧也可以上调RANKL的表达,两者相互调节、互相促进,可增加破骨细胞活性,加速牙周组织丧失,在牙周炎发展过程中起到至关重要的作用。 Wnt/ β-连环蛋白:该信号通路参与调控细胞的生长、迁移及分化。在Wnt通路中,胞膜外配体Wnt蛋白与受体结合,活化胞内的卷曲蛋白从而降低降解复合体中关键成分糖原合成酶激酶3β的活性,进而阻止β-连环蛋白降解,使β-连环蛋白积聚并转移入核与转录因子T细胞因子结合,激活下游靶基因[51],如核心结合因子a1,是调节成骨分化和成骨细胞活化的关键因子,Wnt/ β-连环蛋白的激活参与核心结合因子a1的基因表达,促进成骨细胞的分化并抑制其凋亡[52]。叉头框蛋白1与转录因子T细胞因子竞争结合β-连环蛋白进而抑制Wnt/ β-连环蛋白通路。研究发现,活性氧连续刺激使叉头框蛋白与β-连环蛋白结合[53],抑制细胞中的Wnt/ β-连环蛋白信号通路,损伤骨整合[54],不利于牙周缺损组织的再生。 综上所述,活性氧是一种多效性生理信号传导剂,参与的信号通路错综复杂,且这些信号通路之间相互影响,说明活性氧在牙周炎的发生发展中占据重要地位。但其具体作用尚不明确,可能是因为活性氧并不是一个具体的分子,而是包含了多种氧化物质如超氧阴离子、过氧化氢等的一类物质,而不同的活性氧对牙周炎的发生发展起到不同作用。因此,研究者们要充分认识活性氧在牙周炎中的双向作用,预防牙周炎发生及快速进展。 2.3 活性氧在牙周组织再生过程中的作用 再生被定义为身体丢失或受伤部位的重建,使丢失或受伤组织的结构和功能完全恢复。活性氧在牙周组织再生过程中发挥双重作用,在牙周炎症组织中高浓度的活性氧导致牙周膜干细胞、骨髓间充质干细胞等种子细胞的活性降低,不利于其迁移与增殖;造成组织缺损区的血管内皮损伤,不利于血管再生;并减少促进组织再生的细胞因子的生成。然而低浓度的活性氧可以诱导牙周膜干细胞增殖分化并促进血管再生,因此,可以通过降低活性氧水平减轻牙周组织及细胞中的氧化应激损伤,促进牙周组织再生。 2.3.1 活性氧与牙周骨组织再生 低浓度活性氧促进牙周膜干细胞分化:牙周膜成纤维细胞是一类多能干细胞。2004年,施松涛团队首次从牙周组织中分离出牙周膜成纤维细胞[55],发现其具有较强的克隆形成能力及成牙骨质、成神经及成脂等多项分化能力,可成为最具牙周组织修复潜力的干细胞之一。研究发现,低浓度的活性氧可以诱导牙周膜成纤维细胞的增殖与分化[22],多项研究发现,抑制活性氧的表达[56],或激活抗氧化防御[13],可以改善氧化损伤并促进人牙周膜干细胞成骨。因此,可以通过降低活性氧水平,减轻牙周组织及细胞中的氧化应激损伤,促进成牙周膜干细胞的迁移与增殖,从而增加成骨细胞的分化和牙槽骨形成活性、促进牙周损伤组织的修复以促进牙周组织再生。 高浓度促进牙槽骨吸收:牙周炎是一种以破骨细胞持续活化和牙槽骨丢失为特征的常见疾病[57],破骨细胞的活性和数量直接关系牙周硬组织的吸收。成骨细胞生成和破骨细胞生成是控制骨重建的两个主要过程。成骨细胞和破骨细胞活性的失衡导致骨稳态失调,引起骨病理性改变[58]。过量产生的活性氧诱导氧化应激,在骨稳态和疾病中发挥作用。活性氧可以通过RANK、丝裂原活化激酶等信号通路促进破骨细胞的分化与增殖,从而加重牙周组织吸收。或诱导牙周膜成纤维细胞分泌炎症因子[12],引起破骨细胞分化[14],加重牙周骨组织吸收。细胞研究证实超氧阴离子存在于破骨细胞附近的重吸收区[59]。活性氧生成显著抑制人牙周膜干细胞成骨分化[60],活性氧也可以通过加速成骨细胞衰老,从而减少骨重建[15]。总之,活性氧分子长期作用会引起骨量减少[61]。过量的活性氧刺激会导致人牙周膜干细胞的凋亡[16]。MTT法检测培养细胞活力结果显示,浓度0.3-0.5 mmol/L的过氧化氢分子会损伤人牙周膜干细胞的活力[60],而抗氧化防御则可以降低活性氧水平,减少牙周膜干细胞的损伤。综上所述,可以通过激活抗氧化防御机制,降低体内活性氧的水平达到抑制牙槽骨吸收,促进牙周缺损修复的作用。 活性氧与牙周骨组织再生相关的通路:核转录因子红细胞2相关因子2(nuclear factor E2-related factor 2,Nrf2)对氧化应激高度敏感,通过与细胞核中的抗氧化反应元件结合来保护细胞,促进相关抗氧化基因的转录。在生理条件下,Nrf2被Kelch样ECH关联蛋白1介导的泛素化和蛋白酶体依赖性降解隔离在细胞质中,然而在氧化应激下,Nrf2与Kelch样ECH关联蛋白1的分离是由活性氧以剂量依赖性方式影响的,随后,Nrf2易位到细胞核激活了一些抗氧化靶基因的转录[62]。Nrf2与抗氧化防御系统相互作用[63],在氧化应激相关的牙周损伤中起重要的细胞保护作用[22],通过启动抗氧化酶表达,下调细胞内活性氧水平,进而抑制破骨细胞分化[23],有效抑制氧化应激带来的组织损伤和炎症,促进成骨细胞形成,抑制破骨细胞活性,这在牙周组织修复与重建中起到重要作用。活性氧与牙周膜干细胞相关研究成果[13,15,64-69],见表2。"


2.3.2 活性氧与血管再生 促进血管再生:调节诱导血管形成通常被称为治疗性血管生成,在伤口愈合、骨再生和组织工程方面具有很大的潜力。在组织工程中,及时形成发育良好的脉管系统,可以为支架中的组织提供氧气和营养,然而血管的自发发育非常缓慢,这将血液循环限制在支架上的浅表组织。因此,治疗性血管生成方法正被用于组织工程中,以诱导植入支架中的血管形成。因此,血管生成在牙周组织再生过程中十分重要[17]。血管内皮生长因子是一种关键的血管生成生长因子,主要通过内皮细胞生长因子受体2型刺激内皮细胞连接的破坏、迁移、增殖和毛细血管形成,血管内皮生长因子与质膜上的受体结合诱导受体二聚化和自磷酸化,随后受体经内吞到早期内体区室,其中持续的受体信号传导被进一步激活以驱动血管生成。在内皮细胞中,源自质膜或细胞内胞质中的NADPH氧化酶的活性氧在生长因子(如血管内皮生长因子)诱导的血管生成反应中起关键作用[70],活性氧可通过信号通路促进血管生成[71],还参与各种氧化还原信号通路以诱导血管生成的基因和转录因子[72]。多种纳米材料如氧化铈,金,氧化锌等显示出相对相似的机制,其主要基于由于活性氧或纳米颗粒产生的缺氧引起的缺氧诱导因子活化诱导血管再生[71]。一氧化氮是活性氧的一种,作为治疗性血管生成靶点显示出巨大的潜力。研究发现血管舒张、一氧化氮生成与血管生成之间存在分子/生化相关性,一氧化氮通过内皮-组成型一氧化氮合酶活化、环磷酸鸟苷升高、丝裂原活化激酶活化和成纤维细胞生长因子2表达触发和转导细胞生长和分化,从而显著促进毛细血管内皮的生存/促血管生成程序[73]。 抑制血管再生:活性氧在血管生成过程中同样发挥双刃剑作用,氧化应激依赖的血管生成作用与活性氧的浓度有关[74],研究发现低浓度过氧化氢有利于组织修复过程中的血管生成,但高浓度的过氧化氢分子则会导致内皮损伤[18,75-76],具体研究成果见表3。"

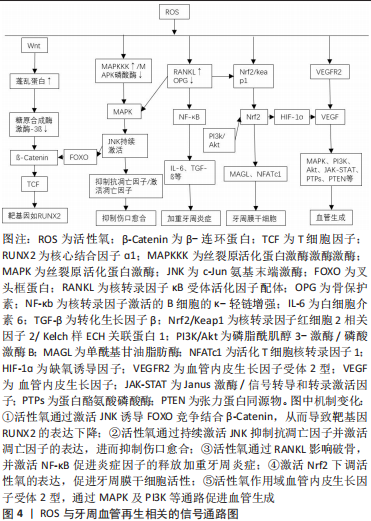
活性氧与血管再生相关的通路:许多促血管生长因子,如血管内皮生长因子和血管生成素-1组成的信号转导网络与活性氧密切相关[62]。与血管生成相关的活性氧介导的氧化还原信号涉及丝裂原活化蛋白激酶、磷脂酰肌醇-3-激酶、蛋白激酶B、Janus激酶/信号转导和转录激活因子和蛋白质酪氨酸磷酸酶,例如蛋白质酪氨酸磷酸酶1B和含SH2的蛋白质酪氨酸磷酸酶2,磷酸酶和张力蛋白同源物以及转录因子包括缺氧诱导因子1ɑ、核转录因子κB 和激活蛋白[70]。Nrf2也参与血管生成,Nrf2的靶基因包括血红素加氧酶1,激活的Nrf2通路可诱导缺氧,进而调控缺氧诱导因子1ɑ/血管内皮生长因子轴,增强血管生成[62]。研究发现高压氧促进了活性氧的产生,以及缺氧诱导因子1ɑ、核转录因子κB、基质细胞衍生因子1和血管内皮生长因子A以及人脐静脉内皮细胞中C-X-C基序趋化因子受体4、血管内皮生长因子2型受体的增殖、迁移和管形成以及表达[77]。 诱导血管生成是组织再生所必需的,但是氧化应激依赖的病理性血管生成也是慢性疾病进展的重要因素。因此,有必要深入研究活性氧诱导病理生理性血管生成的分子机制,明确病理或生理性血管生成的特异性标志物和过程,这将有助于在不久的将来利用活性氧进行治疗性血管生成,并应用于组织修复过程中。见图4。"
| [1] ALMERICH-SILLA JM, MONTIEL-COMPANY JM, PASTOR S, et al. Oxidative stress parameters in saliva and its association with periodontal disease and types of bacteria. Dis Markers. 2015;2015:653537. [2] TAMAKI N, TOMOFUJI T, EKUNI D, et al. Periodontal treatment decreases plasma oxidized LDL level and oxidative stress. Clin Oral Investig. 2011; 15(6):953-958. [3] HARMAN D. Aging: a theory based on free radical and radiation chemistry. J Gerontol. 1956;11(3):298-300. [4] MCCORD JM, FRIDOVICH I. Superoxide dismutase. J Biol Chem. 1969;244(22): 6049-6055. [5] MCCORD JM, FRIDOVICH I. Superoxide dismutases: you’ve come a long way, baby. Antioxid Redox Signal. 2014;20(10):1548-1549. [6] PANIKER NV, SRIVASTAVA SK, BEUTLER E. Glutathione metabolism of the red cells effect of glutathione reductase deficiency on the stimulation of hexose monophosphate shunt under oxidative stress. Biochim Biophys Acta. 1970;215(3):456-460. [7] D’AIUTO F, NIBALI L, PARKAR M, et al. Oxidative stress, systemic inflammation, and severe periodontitis. J Dent Res. 2010;89(11):1241-1246. [8] BALTACIOGLU E, SUKUROGLU E. Protein carbonyl levels in serum, saliva and gingival crevicular fluid in patients with chronic and aggressive periodontitis. Saudi Dent J. 2019;31(1):23-30. [9] Sczepanik FSC, Grossi ML, CASATI M, et al. Periodontitis is an inflammatory disease of oxidative stress: We should treat it that way. Periodontol 2000. 2020;84(1):45-68. [10] BABIOR BM, KIPNES RS, CURNUTTE JT. Biological defense mechanisms. the production by leukocytes of superoxide, a potential bactericidal agent. J Clin Invest. 1973;52(3):741-744. [11] MITTAL CK, MURAD F. Activation of guanylate cyclase by superoxide dismutase and hydroxyl radical: a physiological regulator of guanosine 3’,5’-monophosphate formation. Proc Natl Acad Sci U S A. 1977;74(10):4360-4364. [12] 方慧,刘琪.牙周炎与活性氧致病机制的研究进展[J].中国医药导报, 2018,15(5):22-26. [13] HUANG X, CHEN H, XIE Y, et al. FoxO1 Overexpression ameliorates TNF- α -induced oxidative damage and promotes osteogenesis of human periodontal ligament stem cells via antioxidant defense activation. Stem Cells Int. 2019;2019:2120453. [14] 夏欣怡,陈慧文,胡苡,等.硫酸吲哚酚对人牙周膜细胞增殖及炎症因子表达的影响[J].上海交通大学学报(医学版),2020,40(4):437-442. [15] GENG Q, GAO H, YANG R, et al. Pyrroloquinoline quinone prevents estrogen deficiency-induced osteoporosis by inhibiting oxidative stress and osteocyte senescence. Int J Bio Sci. 2019;15(1):58-68. [16] ZHU L, XIE H, LIU Q, et al. Klotho inhibits H2O2 ‐induced oxidative stress and apoptosis in periodontal ligament stem cells by regulating UCP2 expression. Clin Exp Pharmacol and Physiol. 2021;48(10):1412-1420. [17] 庄齐翔,董家辰,束蓉.釉基质蛋白衍生物对牙周组织再生相关细胞的生物学作用及其成血管作用的研究进展[J].上海交通大学学报(医学版),2021,41(8):1099-1102. [18] KIM YW, BYZOVA TV. Oxidative stress in angiogenesis and vascular disease. Blood. 2014;123(5):625-631. [19] DI MEO S, REED TT, Venditti P, et al. Role of ROS and RNS sources in physiological and pathological conditions. Oxid Med Cell Longev. 2016; 2016:1245049. [20] WANG Y, ANDRUKHOV O, RAUSCH-FAN X. Oxidative stress and antioxidant system in periodontitis. Front Physiol. 2017;8:910. [21] 王芃,吴立鹏.活性氧在牙周炎发生发展转归中的作用和意义综述[J].牡丹江医学院学报,2019,40(4):90-91. [22] XU X. The role of reactive oxygen species and autophagy in periodontitis and their potential linkage. Front Physiol. 2017;8:439. [23] 袁一方,陈晓涛.活性氧类在慢性牙周炎所致骨吸收中的研究进展[J].医学综述,2022,28(4):666-670. [24] BASER U, GAMSIZ-ISIK H, CIFCIBASI E,et al. Plasma and salivary total antioxidant capacity in healthy controls compared with aggressive and chronic periodontitis patients. Saudi Med J. 2015;36(7):856-861. [25] JIANG Q, ZHAO Y, SHUI Y, et al. Interactions between neutrophils and periodontal pathogens in late-onset periodontitis. Front Cell Infect Microbiol. 2021;11:627328. [26] KANG W, JIA Z, TANG D, et al. Fusobacterium nucleatum facilitates apoptosis, ros generation, and inflammatory cytokine production by activating AKT/MAPK and NF-κB signaling pathways in human gingival fibroblasts. Oxid Med Cell Longev. 2019;2019:1681972. [27] MIKOLAI C, BRANITZKI-HEINEMANN K, INGENDOH-TSAKMAKIDIS A, et al. Neutrophils exhibit an individual response to different oral bacterial biofilms. J Oral Microbiol. 2020;13(1):1856565. [28] CHEAT B, TORRENS C, FODA A, et al. NLRP3 is involved in neutrophil mobilization in experimental periodontitis. Front Immunol. 2022;13:839929. [29] YIN W, LIU S, DONG M, et al. A new NLRP3 inflammasome inhibitor, dioscin, promotes osteogenesis. Small. 2020;16(1):e1905977. [30] ISOLA G, POLIZZI A, SANTONOCITO S, et al. Periodontitis activates the NLRP3 inflammasome in serum and saliva. J Periodontol. 2022;93(1):135-145. [31] ISAZA-GUZMÁN DM, MEDINA-PIEDRAHÍTA VM, GUTIÉRREZ-HENAO C, et al. Salivary levels of NLRP3 inflammasome-related proteins as potential biomarkers of periodontal clinical status. J Periodontol. 2017;88(12):1329-1338. [32] LIAN D, DAI L, XIE Z, et al. Periodontal ligament fibroblasts migration injury via ROS/TXNIP/Nlrp3 inflammasome pathway with Porphyromonas gingivalis lipopolysaccharide. Mol Immunol. 2018;103:209-219. [33] ZHU R, MI X, LI Y. Effects of hypoxic environment on periodontal tissue through the ROS/TXNIP/NLRP3 inflammasome pathway. Biomed Res Int. 2022;2022:7690960. [34] MATTHEWS JB, WRIGHT HJ, ROBERTS A, et al. Hyperactivity and reactivity of peripheral blood neutrophils in chronic periodontitis. Clin Exp Immunol. 2007;147(2):255-264. [35] BONINI MG, MALIK AB. Regulating the regulator of ROS production. Cell Res. 2014;24(8):908-909. [36] MARTINEZ‐HERRERA M, LÓPEZ‐DOMÈNECH S, SILVESTRE FJ, et al. Chronic periodontitis impairs polymorphonuclear leucocyte-endothelium cell interactions and oxidative stress in humans. J Clin Periodontol. 2018;45(12): 1429-1439. [37] CHEN M, CAI W, ZHAO S, et al. Oxidative stress‐related biomarkers in saliva and gingival crevicular fluid associated with chronic periodontitis: a systematic review and meta‐analysis. J Clin Periodontol. 2019;46(6):608-622. [38] FOUQUEREL E, BARNES RP, UTTAM S, et al. Targeted and persistent 8-oxoguanine base damage at telomeres promotes telomere loss and crisis. Mol cell. 2019;75(1):117-130.e6. [39] CHEN Y, JI Y, JIN X, et al. Mitochondrial abnormalities are involved in periodontal ligament fibroblast apoptosis induced by oxidative stress. Biochem and Biophys Res Commun. 2019;509(2):483-490. [40] PRASAD S, GUPTA SC, TYAGI AK. Reactive oxygen species (ROS) and cancer: Role of antioxidative nutraceuticals. Cancer Lett. 2017;387:95-105. [41] PATIL VIDYA S. Chronic periodontitis in type 2 diabetes mellitus: oxidative stress as a common factor in periodontal tissue injury. J Clin Diagn Res. 2016;10(4):BC12-6. [42] GHARBI A, HAMILA A, BOUGUEZZI A, et al. Biochemical parameters and oxidative stress markers in Tunisian patients with periodontal disease. BMC Oral Health. 2019;19(1):225. [43] SIES H, JONES DP. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat Rev Mol Cell Biol. 2020;21(7):363-383. [44] 李芊芊,彭双清.活性氧对NF-κB活性及JNK信号通路的调节[J].生物技术通讯,2008,19(4):611-614. [45] BURANASIN P, MIZUTANI K, IWASAKI K, et al. High glucose-induced oxidative stress impairs proliferation and migration of human gingival fibroblasts. PloS One. 2018;13(8):e0201855. [46] WANG Q, SZTUKOWSKA M, OJO A, et al. FOXO responses to Porphyromonas gingivalis in epithelial cells. Cell Microbiol. 2015;17(11):1605-1617. [47] LI H, SUN T, LIU C, et al. Photobiomodulation (450 nm) alters the infection of periodontitis bacteria via the ROS/MAPK/mTOR signaling pathway. Free Radic Biol Med. 2020;152:838-853. [48] 郝瑞,贺菲菲,梁艳,等.糖尿病合并牙周炎患者龈沟液中OPG/RANKL表达与炎症因子、自由基生成及骨代谢的相关性[J].海南医学院学报, 2018,24(5):585-588. [49] KIM JH, KIM K, KIM I, et al. NRROS negatively regulates osteoclast differentiation by inhibiting RANKL-mediated NF-κB and reactive oxygen species pathways. Mol Cells. 2015;38(10):904-910. [50] 吴赛璇,张咪,董明,等.骨稳态过程中Keap1/Nrf2/ARE信号通路的调控作用[J].中国组织工程研究,2022,26(2):271-275. [51] 沈振国,赵荣权,欧琳琳,等.微环境下Wnt/β-catenin通路参与牙周膜干细胞骨向分化机制的研究进展[J].安徽医科大学学报,2021,56(3): 497-500. [52] 程韶,舒冰,赵永见,等.氧化应激对骨重建的影响[J].中国骨质疏松杂志,2019,25(10):1478-1482. [53] GALLI C, PASSERI G, MACALUSO GM. FoxOs, Wnts and oxidative stressinduced bone loss: new players in the periodontitis arena? J Periodontal Res. 2011;46(4):397-406. [54] WANG YN, JIA TT, FENG Y, et al. Hyperlipidemia impairs osseointegration via the ROS/Wnt/β-catenin pathway. J Dent Res. 2021;100(6):658-665. [55] SEO BM, MIURA M, GRONTHOS S, et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet. 2004;364 (9429):149-155. [56] 王文娟,严妍,张凤秋.炎症微环境下牙周膜干细胞胞内ROS的表达[J].北京口腔医学,2022,30(3):171-175. [57] WENG Y, WANG H, LI L, et al. Trem2 mediated Syk-dependent ROS amplification is essential for osteoclastogenesis in periodontitis microenvironment. Redox Biol. 2021;40:101849. [58] LI X, LI B, SHI Y, et al. Targeting reactive oxygen species in stem cells for bone therapy. Drug Discov Today. 2021;26(5):1226-1244. [59] WANG ML, HAUSCHKA PV, TUAN RS, et al. Exposure to particles stimulates superoxide production by human THP-1 macrophages and avian HD-11EM osteoclasts activated by tumor necrosis factor-alpha and PMA. J Arthroplasty. 2002;17(3):335-346. [60] SHI L, JI Y, ZHAO S, et al. Crosstalk between reactive oxygen species and dynamin-related protein 1 in periodontitis. Free Radic Biol and Med. 2021; 172:19-32. [61] JIAO H, XIAO E, GRAVES DT. Diabetes and Its Effect on Bone and Fracture Healing. Curr Osteoporos Rep. 2015;13(5):327-335. [62] GUO Z, MO Z. Keap1-Nrf2 signaling pathway in angiogenesis and vascular diseases. J Tissue Eng Regen Med. 2020;14(6):869-883. [63] BHATTARAI G, POUDEL SB, KOOK SH, et al. Resveratrol prevents alveolar bone loss in an experimental rat model of periodontitis. Acta Biomater. 2016;29:398-408. [64] 张纪春.牙周膜细胞来源细胞外基质在牙周组织再生的实验研究[D].上海:上海交通大学,2017. [65] ZHANG X, JIANG Y, MAO J, et al. Hydroxytyrosol prevents periodontitis-induced bone loss by regulating mitochondrial function and mitogen-activated protein kinase signaling of bone cells. Free Radic Biol Med. 2021; 176:298-311. [66] YANG N, SUN H, XUE Y, et al. Inhibition of MAGL activates the Keap1/Nrf2 pathway to attenuate glucocorticoid‐induced osteonecrosis of the femoral head. Clin Transl Med. 2021;11(6):e447. [67] JIA L, XIONG Y, ZHANG W, et al. Metformin promotes osteogenic differentiation and protects against oxidative stress-induced damage in periodontal ligament stem cells via activation of the Akt/Nrf2 signaling pathway. Exp Cell Res. 2020;386(2):111717. [68] ZHAO Y, LIU H, XI X, et al. TRIM16 protects human periodontal ligament stem cells from oxidative stress-induced damage via activation of PICOT. Exp Cell Res. 2020;397(1):112336. [69] 刘合栋,任茂贤,李杨,等.褪黑素对H₂O₂诱导的成骨细胞氧化应激损伤的保护作用[J].中国骨质疏松杂志,2022,28(8):1093-1098. [70] FUKAI T, USHIO-FUKAI M. Cross-talk between NADPH oxidase and mitochondria: role in ROS signaling and angiogenesis. Cells. 2020;9(8):1849. [71] AUGUSTINE R, PRASAD P, KHALAF IMN. Therapeutic angiogenesis: from conventional approaches to recent nanotechnology-based interventions. Materials Sci Eng C Mater Biol Appl. 2019;97:994-1008. [72] MCGARRY T, BINIECKA M, VEALE DJ, et al. Hypoxia, oxidative stress and inflammation. Free Radic Biol Med. 2018;125:15-24. [73] ZICHE M, MORBIDELLI L. Nitric oxide and angiogenesis. JNeurooncol. 2000; 50(1):139-148. [74] SCOTT E, LOYA K, MOUNTFORD J, et al. MicroRNA regulation of endothelial homeostasis and commitment-implications for vascular regeneration strategies using stem cell therapies. Free Radic Biol Med. 2013;64:52-60. [75] YASUDA M, OHZEKI Y, SHIMIZU S, et al. Stimulation of in vitro angiogenesis by hydrogen peroxide and the relation with ETS-1 in endothelial cells. Life Sci. 1999;64(4):249-258. [76] SHIMIZU S, NOMOTO M, YAMAMOTO T, et al. Reduction by NG-nitro-L-arginine of H2O2-induced endothelial cell injury. Br J Pharmacol. 1994; 113(2):564-568. [77] HUANG X, LIANG P, JIANG B, et al. Hyperbaric oxygen potentiates diabetic wound healing by promoting fibroblast cell proliferation and endothelial cell angiogenesis. Life Sci. 2020;259:118246. |
| [1] | Ouyang Beiping, Ma Xiangyang, Luo Chunshan, Zou Xiaobao, Lu Tingsheng, Chen Qiling. Three-dimensional finite element analysis of a new horizontal screw-screw crosslink in posterior atlantoaxial internal fixation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1320-1324. |
| [2] | Guo Sutong, Feng Dehong, Guo Yu, Wang Ling, Ding Yujian, Liu Yi, Qian Zhengying, Li Mingyang. Construction and finite element analysis of normal and osteoporotic hip models [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1342-1346. |
| [3] | Min Meipeng, Wu Jin, URBA RAFI, Zhang Wenjie, Gao Jia, Wang Yunhua, He Bin, Fan Lei. Role and significance of artificial intelligence preoperative planning in total hip arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1372-1377. |
| [4] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [5] | Chen Kaijia, Liu Jingyun, Cao Ning, Sun Jianbo, Zhou Yan, Mei Jianguo, Ren Qiang. Application and prospect of tissue engineering in treatment of osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1450-1456. |
| [6] | Yang Yifeng, Ye Nan, Wang Lin, Guo Shuaicheng, Huang Jian. Signaling pathway of dexmedetomidine against ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1464-1469. |
| [7] | Lou Guo, Zhang Yan, Fu Changxi. Role of endothelial nitric oxide synthase in exercise preconditioning-induced improvement of myocardial ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1283-1288. |
| [8] | Lin Zeyu, Xu Lin. Research progress in gout-induced bone destruction mechanism [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1295-1300. |
| [9] | Qi Xue, Li Jiahui, Zhu Yuanfeng, Yu Lu, Wang Peng. Abnormal modification of alpha-synuclein and its mechanism in Parkinson’s disease [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1301-1306. |
| [10] | Wang Weiqing, Zhou Yue. Chronic inflammation regulates adipose tissue fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1307-1312. |
| [11] | Huang Haoran, Fan Yinuo, Wei-Yang Wenxiang, Jiang Mengyu, Fang Hanjun, Wang Haibin, Chen Zhenqiu, Liu Yuhao, Zhou Chi. Urolithin A mediates p38/MAPK pathway to inhibit osteoclast activity [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1149-1154. |
| [12] | Zhang Xiaoyun, Liu Hua, Chai Yuan, Chen Feng, Zeng Hao, Gao Zhengang, Huang Yourong. Effect of Yishen Gushu Formula on bone metabolic markers and clinical efficacyn in patients with osteoporosis of kidney deficiency and blood stasis type [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1155-1160. |
| [13] | Shen Jiangyong, He Xi, Tang Yuting, Wang Jianjun, Liu Jinyi, Chen Yuanyuan, Wang Xinyi, Liu Tong, Sun Haoyuan. RAS-selective lethal small molecule 3 inhibits the fibrosis of pathological scar fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1168-1173. |
| [14] | Mo Jun, Luo Zongping. Effects of static traction on the nucleus pulposus and annulus fibrosus of the rat intervertebral disc [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1180-1185. |
| [15] | Dai Yuexing, Zheng Liqin, Wu Minhui, Li Zhihong, Li Shaobin, Zheng Desheng, Lin Ziling. Effect of vessel number on computational fluid dynamics in vascular networks [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1206-1210. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

