Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (5): 759-765.doi: 10.12307/2024.256
Previous Articles Next Articles
Research advance in preparation and functional modification of porous polytetrafluoroethylene artificial blood vessels
Xu Rong1, Wang Haojie1, Geng Mengxiang1, Meng Kai1, 2, Wang Hui1, 2, Zhang Keqin1, 2, Zhao Huijing1, 2
- 1College of Textile and Clothing Engineering, Soochow University, Suzhou 215021, Jiangsu Province, China; 2National Engineering Laboratory for Modern Silk (Suzhou), Suzhou 215123, Jiangsu Province, China
-
Received:2023-02-22Accepted:2023-04-07Online:2024-02-18Published:2023-08-17 -
Contact:Zhao Huijing, PhD, Professor, College of Textile and Clothing Engineering, Soochow University, Suzhou 215021, Jiangsu Province, China; National Engineering Laboratory for Modern Silk (Suzhou), Suzhou 215123, Jiangsu Province, China -
About author:Xu Rong, Master candidate, College of Textile and Clothing Engineering, Soochow University, Suzhou 215021, Jiangsu Province, China -
Supported by:Science and Technology Guidance Project of China National Textile and Apparel Council, No. 2021035 (to ZKQ, ZHJ)
CLC Number:
Cite this article
Xu Rong, Wang Haojie, Geng Mengxiang, Meng Kai, Wang Hui, Zhang Keqin, Zhao Huijing. Research advance in preparation and functional modification of porous polytetrafluoroethylene artificial blood vessels[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 759-765.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
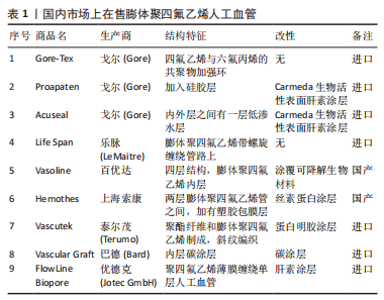
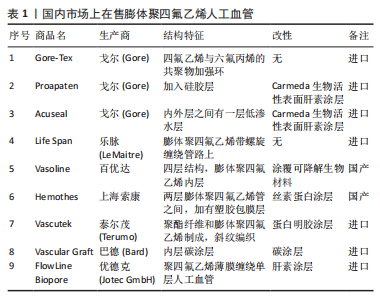
2.1 多孔聚四氟乙烯人工血管的制备 孔隙给组织细胞的侵入和迁移提供了三维空间,有利于血管组织细胞的黏附[17];此外,孔隙决定了血管的通透性,便于血液和组织细胞之间进行物质交换,将液体、氧气、营养物质、生物活性分子等物质通过孔隙扩散到组织细胞,滋养血管组织细胞使其增殖和生长,同时,组织细胞代谢产生的二氧化碳等废物通过孔隙扩散到血液,随血液输送到其他器官排泄,从而保证身体的正常代谢的进行[18]。根据成孔机制不同,目前有两种方法可以制备聚四氟乙烯人工血管,分别是快速热拉伸法和静电纺丝法,其中快速热拉伸法是制备多孔聚四氟乙烯人工血管的传统方法,表1为国内市场在售的膨体聚四氟乙烯血管汇总,其中大部分膨体聚四氟乙烯人工血管来自进口,国内只有百优达和上海索康生产膨体聚四氟乙烯人工血管。此外,对于改性处理方面,大部分都在生产时就进行了功能化改性处理,即使没有进行改性处理也需要在移植前进行抗凝处理,如乐脉的Life Span人工血管需在移植前使用肝素钠进行抗凝。在使用年限上,其中Gore的Proapaten人工血管膝下移植9年通畅率达到41%。在有效期上,乐脉的Life Span人工血管从灭菌起可以提供7年有效屏障。静电纺丝技术是制备多孔聚四氟乙烯人工血管的较新型方法,具有较广阔的前景。"
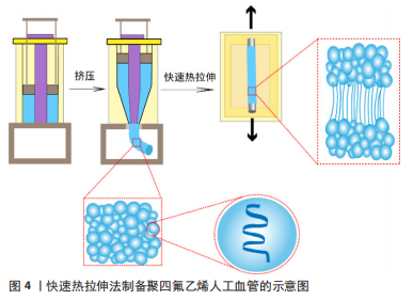
2.1.1 快速热拉伸法 聚四氟乙烯螺旋形的分子链构象使得分子链可以高效地折叠形成片晶,而后堆积成带状多晶结构,折叠的带状结构卷成球状或椭球状粒子[19]。由于聚四氟乙烯分子链间相互作用力弱,所以在一定温度和拉伸作用下折叠的带状结构很容易被打开,沿着拉伸的方向从聚四氟乙烯分散树脂颗粒中抽出细长的原纤维,没有被抽出的结晶区域则以结点形式与原纤维连接,形成独特的“原纤维-结点”多孔结构[20]。 聚四氟乙烯人工血管是一种满足人工血管厚度、孔隙率、强度等要求的多孔聚四氟乙烯管状膜,其制备的典型过程如图4所示:原料使用超高分子质量分散聚四氟乙烯树脂,过筛后与石脑油等有机易挥发的助挤剂按照一定比例配置成糊状物,压成无空隙的厚壁圆筒状预制件;将预制件送入柱塞挤出机,通过特定的锥形模具挤压出聚四氟乙烯管状膜;随后,对聚四氟乙烯管状膜进行热处理,去除助挤剂,并在低于聚四氟乙烯熔点温度(327 ℃)下进行轴向的快速热拉伸,一部分树脂被拉伸成原纤维,另一部分形成结点,形成多孔结构的聚四氟乙烯管状膜[8]。为了提高多孔聚四氟乙烯管状膜的力学强度,往往还需要在一定应力和高于聚四氟乙烯熔点的温度下进行高温烧结[21],使分散的单个树脂颗粒通过互相扩散熔融黏结成一个连续的整体,冷却后即得到柔韧的多孔聚四氟乙烯人工血管。"
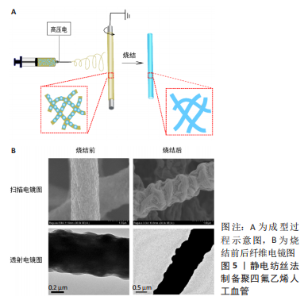
WANG等[22]将商业细聚四氟乙烯粉末(由约80 nm纳米颗粒组成的微尺度聚集体)与助挤剂(体积分数70%乙醇)均匀混合,通过使用圆柱形模具形成预制件;然后通过施加所需压力将预制件插入糊状挤出机中,获得口径为6 mm的聚四氟乙烯管状膜;在200 ℃下对挤压出来的聚四氟乙烯管状膜进行拉伸膨化,在拉伸比300%的情况下获得了原纤维长度为20-35 μm的多孔聚四氟乙烯人工血管。 快速热拉伸法制备的多孔聚四氟乙烯人工血管又称为膨体聚四氟乙烯人工血管,其原纤维长度平均30 μm,孔隙率可达76%,已被报道用于腹主动脉和下腔静脉重建[23]、血液透析动静脉造瘘等[24]。SINGHAL等[25]在右叶活体供肝移植中使用膨体聚四氟乙烯重建静脉流出道,1个月和6个月血管通畅率分别为97.6%和84.4%,而使用自体静脉1个月和6个月血管通畅率分别为96.4%和78.1%,说明在同种或自体血管移植物使用受限的情况下,使用膨体聚四氟乙烯是一种可行的选择,具有良好的短期通畅性和患者预后。膨体聚四氟乙烯已经实现商业化生产,在临床上使用广泛,但限于原材料和工艺技术壁垒,中国发展较慢,如上海索康医用材料有限公司这一家企业于2005年上市了膨体聚四氟乙烯人工血管产品,目前临床上仍依赖于进口,价格较为昂贵。 2.1.2 静电纺丝法 静电纺丝技术可制备与天然细胞外基质相似的三维纤维结构,在聚四氟乙烯人工血管的制备方面具有很大的发展潜力。然而,聚四氟乙烯几乎不溶于有机溶剂[26],难以直接单独静电纺丝。在聚四氟乙烯水分散液中混入少量成纤聚合物作为助纺剂,进行乳液静电纺丝,在电场力的作用下,成纤聚合物载着聚四氟乙烯分散颗粒被拉伸形成微纳米纤维,纤维在三维空间上杂乱堆砌形成孔隙。由于助纺剂的分解温度低于聚四氟乙烯的熔点,所以高温煅烧过程中助纺剂被去除,同时聚四氟乙烯颗粒熔融流动,填补助纺剂空隙,颗粒间相互融合形成纯聚四氟乙烯纤维,最终得到纯的多孔聚四氟乙烯管状膜,同时提高了强力。静电纺丝法制备聚四氟乙烯人工血管的过程见图5。"
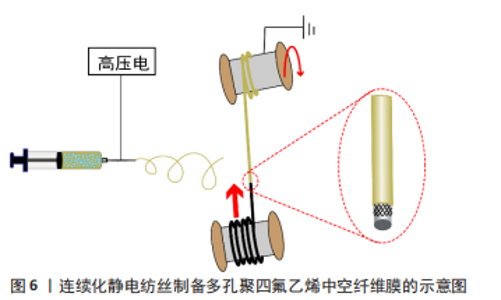
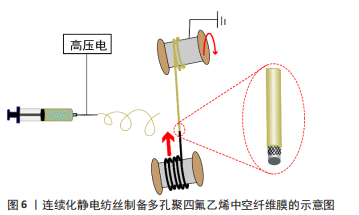
静电纺丝制备聚四氟乙烯管状膜所用到的原料是高浓度的聚四氟乙烯分散液以及高分子质量的助纺剂,助纺剂主要有聚乙烯醇、聚氧化乙烯等。HUANG等[27-28]以聚乙烯醇作为助纺剂,将玻璃纤维编织管套在旋转芯轴上作为收集装置,为解决烧结后管产生裂缝的问题,共纺聚丙烯腈作为前体管的支撑框架,以1 ℃/min的加热速率升温至380 ℃烧结10 min,制得的聚四氟乙烯管状膜孔径分布均匀,孔隙率较高(> 70%)。ZHOU等[29]以聚乙烯醇为助纺剂,用一束平行的铁丝构成直径8 mm的可移动集电体作为静电纺丝的收集装置,通过控制收集时间获得了不同厚度的聚乙烯醇/聚四氟乙烯管状膜,将聚乙烯醇/聚四氟乙烯管状膜与收集器一起干燥后于管式炉中烧结,在氮气气氛中加热至380 ℃保温30 min,冷却后去除铁丝,得到了厚度87-346 μm的多孔聚四氟乙烯管状膜。 LUO等[30]用500万分子质量的聚氧化乙烯作为助纺剂、不锈钢编织管套在直径4 mm的金属芯轴上作为旋转接收装置,使用聚氧化乙烯与聚四氟乙烯质量比为3∶97的均匀混合液进行乳液静电纺丝,380 ℃烧结10 min后得到了厚度80-140 μm、孔隙率65%-75%的聚四氟乙烯管状膜,厚度越小孔隙率越大;生物相容性测试结果显示,该方法制备的多孔聚四氟乙烯管状膜具有良好的血液相容性和细胞相容性。 SU等[31-32]以聚氧化乙烯为助纺剂,将多孔支撑管固定在直径2 mm的软电极上作为接收聚氧化乙烯/聚四氟乙烯管的装置,在旋转阀芯的牵引下软电极和支撑管连续向前移动,而不是旋转,最终得到了高孔隙率(> 82%)的聚四氟乙烯中空纤维膜,制备过程如图6所示,与传统所报道的旋转式电纺收集装置相比,这种收集方式有大规模连续化生产的潜力。"

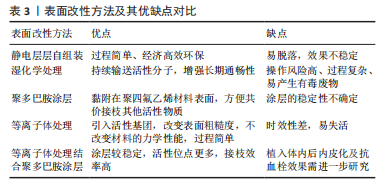
近年来,越来越多学者开始研究利用静电纺丝技术制备聚四氟乙烯管状膜,大部分是研究外径很小(低于3 mm)的管状膜,即聚四氟乙烯中空纤维膜,应用在膜蒸馏、过滤等领域,除了骆宇[33]将静电纺丝技术制备的多孔聚四氟乙烯管状膜用于血管支架覆膜外,少有研究报道将静电纺丝制备的多孔聚四氟乙烯管状膜用于人工血管领域。用静电纺丝技术制备多孔聚四氟乙烯人工血管将是很有潜力的一种方法,不仅可以通过调整静电纺丝参数来控制纤维膜的孔隙结构,而且通过设计接收装置还可以实现连续化收集。与传统热拉伸制备的多孔聚四氟乙烯人工血管相比,采用静电纺丝技术制备的多孔聚四氟乙烯人工血管过程更加简单、高效,且所需成本更低。 2.2 多孔聚四氟乙烯人工血管的功能化改性 大口径聚四氟乙烯人工血管已经在临床得到广泛应用,但小口径聚四氟乙烯人工血管的远期通畅率极低,将内径6 mm的聚四氟乙烯人工血管用于膝下旁路时,3年时通畅率仅为20%[34],这主要是因为血管内表面缺失内皮细胞层。小口径血管内血液流动速度缓慢,致使血液直接与聚四氟乙烯材料表面紧密接触,容易发生血小板黏附和血浆蛋白吸附,引发凝血级联反应,从而导致血栓形成[35]。在生理条件下,内皮细胞层可以防止血小板活化和凝血级联反应[36],血管内皮细胞表面的血栓调节蛋白通过与凝血酶形成复合体将凝血酶的促凝活性转化为抗凝血剂,从而将蛋白C激活为活化蛋白C,活化蛋白C使凝血辅因子失活,进一步抑制凝血酶的生成[37];内皮细胞分泌的一氧化氮和前列环素是天然的抗凝血剂,能够阻止血小板的黏附和聚集。因此,在聚四氟乙烯人工血管内表面快速内皮化对于保证其通畅性至关重要。常用的膨体聚四氟乙烯人工血管改性药品见表2。"

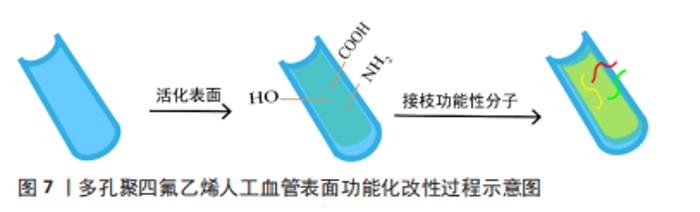
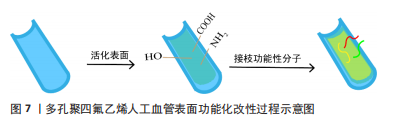
聚四氟乙烯材料表面缺乏活性位点,内皮细胞在纯聚四氟乙烯人工血管上的生长潜能较低,难以快速在纯聚四氟乙烯表面原位形成完整的内皮细胞层。为了解决这一问题,目前已经提出了一种组织工程方法,即用自体内皮细胞预植入人工血管[65],然而其临床的广泛应用受到高生产成本、复杂程序的限制。故制备利于快速原位内皮化的小口径聚四氟乙烯人工血管是目前的一个重点研究方向,通过对纯多孔聚四氟乙烯人工血管材料进行改性,赋予血管内表面更好的生物活性,提高其血液相容性和细胞相容性[66],从而提高聚四氟乙烯小口径人工血管的远期通畅率。 2.2.1 表面改性 多孔聚四氟乙烯人工血管表面功能化改性的过程如图7所示。首先,通过共价或非共价作用在化学稳定性好的膨体聚四氟乙烯表面引入新的官能团,如氨基、羧基、羟基,以此形成活化的表面,这不仅能提高膨体聚四氟乙烯材料表面的亲水性和黏附性,也提供了反应位点;在交联剂的作用下,活化的膨体聚四氟乙烯表面可以与生物活性分子以酰胺键等形式共价结合,赋予膨体聚四氟乙烯人工血管抗血栓、促内皮化等生物学功能。"

静电层层自组装活化表面:聚四氟乙烯表面带负电,通过静电引力的作用吸附相反电荷的聚电解质进行层层组装,可以在聚四氟乙烯表面引入活性基团,便于固定生物活性分子。如SHAN等[39]将膨体聚四氟乙烯膜浸入聚乙烯亚胺溶液中,使膜表面带正电;清洗干燥后将膜浸泡在带负电的肝素溶液中,再次清洗干燥后浸入带正电的胶原蛋白溶液,重复交替此浸泡过程5次,利用静电层层自组装制备了肝素/胶原多层膜;在戊二醛的共价交联作用下,将精氨酸-谷氨酸-天冬氨酸固定在活化的膜表面,功能化的膨体聚四氟乙烯膜对早期内皮细胞黏附、后期细胞增殖和细胞活性均有积极影响,改善了膨体聚四氟乙烯薄膜的体外血液相容性、抗凝血性和细胞相容性。MANABE等[40]通过研究浸渍溶液的pH值和交联剂的综合影响,在聚四氟乙烯基底上开发了由白蛋白和肝素组成的稳定多层膜,并表现出抗血栓形成能力;添加了交联剂戊二醛的多层膜可增强对PBS的抵抗力,而没有交联剂的多层膜在浸入PBS中48 h后完全消失;此外,用白蛋白与弹性蛋白的复合溶液可使多层膜的弹性得到显著改善,该研究有可能构建复杂的血管移植物并防止由于针刺而导致血管移植物的整体塌陷。Gore公司采用Carmeda生物活性表面改性(CBAS?)技术,将膨体聚四氟乙烯人工血管逐层沉积带正电的聚乙烯亚胺和带负电的硫酸葡聚糖,然后把肝素共价连接到还原端的聚乙烯亚胺上,制备出市售的肝素键合人工血管产品[41]。 静电层层自组装在环境温度和压力下即可进行,过程简单、用途广泛、经济高效且环保,还能够在微观和纳米尺度上控制涂层的表面纹理和厚度。但单纯的静电层层自组装活化表面依靠的是静电作用力,而非共价键合,在复杂的生物环境内较易脱落,效果不稳定。 湿化学处理活化表面:湿化学改性是基于溶液与聚四氟乙烯之间的反应,高活性溶剂使碳氟键(C-F)断裂,从而引入活性基团[67]。用于聚四氟乙烯改性的化学溶液包括呋喃碱溶液、萘钠溶液、强酸和强氧化盐等。AL MESLMANI等[68]使用过氧化氢/硫酸溶液将膨体聚四氟乙烯表面氧化生成羟基,再通过使用3-氨基丙基三乙氧基硅烷对活化羟基进行氨解反应,将游离氨基引入膨体聚四氟乙烯表面;产生的氨基被进一步用作共价固定聚乳酸-乙醇酸纳米粒子的锚定位点,纳米粒子中载有药物异硫氰酸荧光素葡聚糖以便持续输送活性分子,有利于增强膨体聚四氟乙烯的长期通畅性。湿化学处理依靠的是共价键合,但操作风险较高、过程较复杂且可能产生有毒废物。 聚多巴胺涂层活化表面:多巴胺在弱碱环境下会发生自聚合形成聚多巴胺,黏附在聚四氟乙烯材料表面,聚多巴胺涂层后的聚四氟乙烯人工血管表面产生氨基等活性基团,可以与其他生物活性物质进行共价接枝。KOPE?等[54]对内径6 mm小口径膨体聚四氟乙烯人工血管表面进行聚多巴胺涂层,结果显示聚多巴胺涂层后提高了内皮细胞的定植率,有利于小口径聚四氟乙烯人工血管的内皮化。XING等[55]将膨体聚四氟乙烯人工血管浸入多巴胺的缓冲溶液中之后,用1-乙基-(3-二甲基氨基丙基)碳酰二亚胺/N-羟基硫代琥珀酰亚胺(EDC/NHS)为交联剂,共价接枝了抗凝剂比伐卢定和内皮细胞选择性黏附肽精氨酸-谷氨酸-天冬氨酸-缬氨酸(REDV),制备的比伐卢定/REDV共修饰的膨体聚四氟乙烯血管在植入后12周具有良好的通畅率(66.7%)和较高的内皮细胞覆盖率(70%)。贾山山[42]对膨体聚四氟乙烯人工血管进行多巴胺共混聚乙烯亚胺处理,利用聚乙烯亚胺中的大量氨基方便二次接枝肝素,进行血小板黏附实验,结果显示改性后的人工血管血小板黏附明显较少。黄彩标[44]对含有多巴胺自聚涂层的膨体聚四氟乙烯小口径人工血管表面接枝比伐卢定,利用比伐卢定抑制凝血酶的活性进行改性,结果显示改性处理的人工血管表面血小板黏附少,能显著延长凝血时间,具有良好的血液相容性。 聚多巴胺涂层聚四氟乙烯的最新进展包括其他分子与多巴胺的共沉积[56],多巴胺涂层后通过聚多巴胺中的活性基团接枝第二涂层,或这两种的结合。FOWLER等[57]将聚乙烯亚胺和聚多巴胺的共沉积涂层,以在膨体聚四氟乙烯表面产生胺基,这些来自聚乙烯亚胺的氨基通过第二涂层中的开环反应与两性离子共聚物的环氧基团偶联,该涂层能够抵抗81.57%来自人全血的血细胞附着。YU等[58]将聚乙烯亚胺和聚多巴胺共沉积涂层在聚四氟乙烯表面,然后接种人脐静脉内皮细胞和血管内皮细胞,共培养后进行脱细胞操作,获得细胞外基质涂层的膨体聚四氟乙烯,具有抑制血栓形成、防止内膜增生、促进血管内皮化的功能,可用于开发小口径人工血管。 等离子体处理活化表面:等离子体处理可以裂解聚四氟乙烯材料表面的C-F键,增大聚四氟乙烯材料的表面能,并引入多种活性基团如羟基、氨基活化原本惰性的表面,处理过程简单、成本低。PU等[69]用氨气对聚四氟乙烯进行等离子体处理后发现可改善内皮细胞的黏附和生长,并且没有表现出炎症作用。GAO等[70]用氮气等离子体处理聚四氟乙烯表面,表面被纳米结构粗糙化,产生的大量自由基不断迁移到表面并与环境分子发生反应;而后通过简单的溶液浸渍将具有不同功能的各种生物分子稳定地固定在聚四氟乙烯表面,促进了内皮祖细胞的募集,有效减少血栓的形成。 等离子体处理结合聚多巴胺涂层:聚多巴胺在聚四氟乙烯上的涂层稳定性尚未得到充分研究。CHENG等[48,59]在聚多巴胺涂层之前使用氩等离子体处理作为预处理,通过调整等离子体能量、气体压力和处理时间等处理参数,在聚四氟乙烯表面构建了稳定的聚多巴胺涂层;该涂层具有大量反应性官能团,作为进一步功能化改性聚四氟乙烯的反应平台,接枝2-甲基丙烯酰氧基乙基磷酰胆碱提高了聚四氟乙烯的生物相容性。MI等[60]使用氧等离子体处理聚四氟乙烯表面,之后以聚多巴胺涂层、聚乙烯亚胺接枝、精氨酸-甘氨酸-天冬氨酸(RGD)固定或RGD/肝素固定,多巴胺和RGD修饰后血小板黏附增加,但引入肝素后显著降低;由于内皮细胞和RGD之间强烈的细胞-底物相互作用,RGD/肝素移植的聚四氟乙烯表现出高内皮细胞亲和力。 2.2.2 本体改性 传统方法制备膨体四氟乙烯人工血管时使用石脑油或其他类似化学品作为润滑的助挤剂,但这些助挤剂不能溶解和分散许多潜在有用的生物大分子和药物[71]。最近有学者开始创新性地尝试乙醇/水助挤剂体系,在四氟乙烯糊料中填充溶于水的生物大分子和药物,实现了本体改性处理。YAN等[46,72]以乙醇/水混合物作为助挤剂,将蛋壳膜提取物和肝素成功地嵌入膨体四氟乙烯,包埋的蛋壳膜提取物和肝素改善了膨体四氟乙烯亲水性和细胞相容性;之后又在乙醇/水/助挤剂体系下将丝素溶液和水杨苷溶液与聚四氟乙烯颗粒混合,经膏体挤压和膨胀后产生了具有生物相容性和抗血栓的柔性膨体四氟乙烯人工血管。WANG等[73]在用糊料挤出法制备膨体四氟乙烯人工血管时,把RGD、肝素和硒代胱胺溶解在助挤剂中,通过活体和死体测定、细胞形态学、CD31染色、一氧化氮释放和抗凝试验证明了膨体四氟乙烯人工血管的生物学功能得到了极大改善。"

| [1] 贾慧莹,马建伟,陈韶娟.聚四氟乙烯纤维的制备技术及其进展[J].产业用纺织品,2018,36(7):1-6. [2] PUTS GJ, CROUSE P, AMEDURI BM. Polytetrafluoroethylene: synthesis and characterization of the original extreme polymer. Chem Rev. 2019;119(3): 1763-1805. [3] KORZINSKAS T, JUNG O, SMEETS R, et al. In Vivo Analysis of the Biocompatibility and Macrophage Response of a Non-Resorbable PTFE Membrane for Guided Bone Regeneration. Int J Mol Sci. 2018;19(10):2952. [4] ZHANG M, ZHOU Z, YUN J, et al. Effect of Different Membranes on Vertical Bone Regeneration: A Systematic Review and Network Meta-Analysis. Biomed Res Int. 2022;2022:7742687. [5] SHIRAZI MMA, KARGARI A, TABATABAEI M. Evaluation of commercial PTFE membranes in desalination by direct contact membrane distillation. Chem Eng Process. 2014;76:16-25. [6] YU S, KANG G, ZHU Z, et al. Nafion-PTFE hollow fiber composite membranes for improvement of anti-fouling and anti-wetting properties in vacuum membrane distillation. J Membrane Sci. 2021;620:118915. [7] WANG A, LI X, HOU T, et al. High efficiency, low resistance and high temperature resistance PTFE porous fibrous membrane for air filtration. Mater Lett. 2021;295:129831.1-129831.4. [8] WANG D, XU Y, LI Q, et al. Artificial small-diameter blood vessels: materials, fabrication, surface modification, mechanical properties, and bioactive functionalities. J Mater Chem B. 2020;8(9):1801-1822. [9] GORE RW. Process for producing porous products: U.S. Patent 3,953,566. 1976-4-27. [10] CAMPBELL CD, BROOKS DH, WEBSTER MW, et al. Expanded microporous polytetrafluoroethylene as a vascular substitute: a two year follow-up. Surgery. 1979;85(2):177-183. [11] LAVERY KS, RHODES C, MCGRAW A, et al. Anti-thrombotic technologies for medical devices. Adv Drug Deliv Rev. 2017;112:2-11. [12] LIU Y, MUNISSO MC, MAHARA A, et al. A surface graft polymerization process on chemically stable medical ePTFE for suppressing platelet adhesion and activation. Biomater Sci. 2018;6(7):1908-1915. [13] ZOU XP, KANG ET, NEOH KG. Plasma-induced graft polymerization of poly (ethylene glycol) methyl ether methacrylate on poly (tetrafluoroethylene) films for reduction in protein adsorption. Surf Coat Technol. 2002;149(2-3): 119-128. [14] GABRIEL M, NIEDERER K, BECKER M, et al. Tailoring novel PTFE surface properties: promoting cell adhesion and antifouling properties via a wet chemical approach. Bioconjug Chem. 2016;27(5):1216-1221. [15] BAX DV, WANG Y, LI Z, et al. Binding of the cell adhesive protein tropoelastin to PTFE through plasma immersion ion implantation treatment. Biomaterials. 2011;32(22):5100-5111. [16] PARK C, PARK S, KIM J, et al. Enhanced endothelial cell activity induced by incorporation of nano-thick tantalum layer in artificial vascular grafts[J]. Appl Surf Sci. 2020;508:144801. [17] 陈晓洁,籍晓萍,王璐,等.ePTFE人工血管管壁结构及其体外性能的试验研究[J].东华大学学报(自然科学版),2014,40(4):424-429. [18] ZHOU X, NOWICKI M, SUN H, et al. 3D Bioprinting-Tunable Small-Diameter Blood Vessels with Biomimetic Biphasic Cell Layers. ACS Appl Mater Interfaces. 2020;12(41):45904-45915. [19] RANJBARZADEH-DIBAZAR A, BARZIN J, SHOKROLLAHI P. Microstructure crystalline domains disorder critically controls formation of nano-porous/long fibrillar morphology of ePTFE membranes. Polymer. 2017;121:75-87. [20] JIA J, KANG G, ZOU T, et al. Sintering process investigation during polytetrafluoroethylene hollow fibre membrane fabrication by extrusion method. High Perform Polym. 2017;29(9):1069-1082. [21] WANG H, DING S, ZHU H, et al. Effect of stretching ratio and heating temperature on structure and performance of PTFE hollow fiber membrane in VMD for RO brine[. Sep Purif Technol. 2014;126:82-94. [22] WANG D, XU Y, WANG L, et al. Long-term nitric oxide release for rapid endothelialization in expanded polytetrafluoroethylene small-diameter artificial blood vessel grafts. Appl Surf Sci. 2020;507:145028. [23] WANG J, YUAN D, LU Y, et al. A large pheochromocytoma requiring aortic and inferior vena caval reconstruction: A case report. Medicine (Baltimore). 2019;98(29):e16494. [24] DRIESSEN W, VAN DER MEIJDEN W, WANTEN G, et al. Long-term patency rate of the translocated autologous saphenous vein versus prosthetic material in vascular access surgery for haemodialysis and parenteral nutrition. J Vasc Access. 2021;11297298211013133. doi: 10.1177/11297298211013133. [25] SINGHAL A, MAKKI K, CHORASIYA V, et al. Venous outflow reconstruction using a polytetrafluoroethylene (PTFE) graft in right lobe living donor liver transplantation: A single center study. Surgery. 2021;169(6):1500-1509. [26] RENFREW MM, LEWIS EE. Polytetrafluoroethylene. Heat resistant, chemically inert plastic. Ind Eng Chem Res. 1946;38(9):870-877. [27] HUANG Q, HUANG Y, GAO S, et al. Novel Ultrafine Fibrous Poly(tetrafluoroethylene) Hollow Fiber Membrane Fabricated by Electrospinning. Polymers. 2018;10(5):464. [28] HUANG Y, XIAO C, HUANG Q, et al. Robust preparation of tubular PTFE/FEP ultrafine fibers-covered porous membrane by electrospinning for continuous highly effective oil/water separation. J Membrane Sci. 2018;568: 87-96. [29] ZHOU T, LI J, GUO X, et al. Freestanding PTFE electrospun tubular membrane for reverse osmosis brine concentration by vacuum membrane distillation. Desalin Water Treat. 2019;165:63-72. [30] LUO Y, GONG X S, XU Z, et al. PTFE Electrospun Stent Graft—Preparation, Properties and Its Industrialization Prospect. Chem Res Chin Univ. 2021;37(3):589-597. [31] SU C, LI Y, CAO H, et al. Novel PTFE hollow fiber membrane fabricated by emulsion electrospinning and sintering for membrane distillation. J Membrane Sci. 2019;583:200-208. [32] SU C, LU C, CAO H, et al. Fabrication of a novel nanofibers-covered hollow fiber membrane via continuous electrospinning with non-rotational collectors. Mater Lett. 2017;204: 8-11. [33] 骆宇.聚四氟乙烯电纺膜及覆膜支架的制备与性能研究[D].苏州:苏州大学,2021. [34] KASHYAP MS, AHN SS, QUINONES-BALDRICH WJ, et al. Infrapopliteal-Lower Extremity Revascularization with Prosthetic Conduit: A 20-Year Experience. Vasc Endovasc Surg. 2002;36(4):255-262. [35] ZHANG J, WANG Y, LIU C, et al. Polyurethane/polyurethane nanopartic‐modified expanded poly (tetrafluoroethylene) vascular patches promote endothelialization. J Biomed Mater Res A. 2018;106(8):2131-2140. [36] NEUBAUER K, ZIEGER B. Endothelial cells and coagulation. Cell Tissue Res. 2022;387(3):391-398. [37] WANG Y, WU H, ZHOU Z, et al. A thrombin-triggered self-regulating anticoagulant strategy combined with anti-inflammatory capacity for blood-contacting implants. Sci Adv. 2022;8(9):3378. [38] TABORSKA J, RIEDELOVA Z, BRYNDA E, et al. Endothelialization of an ePTFE vessel prosthesis modified with an antithrombogenic fibrin/heparin coating enriched with bound growth factors. RSC Adv. 2021;11(11):5903-5913. [39] SHAN Y, JIA B, YE M, et al. Application of Heparin/Collagen-REDV Selective Active Interface on ePTFE Films to Enhance Endothelialization and Anticoagulation. Artif Organs. 2018;42(8):824-834. [40] MANABE K, NARA H. Construction of stable biological albumin/heparin multilayers for elastic coatings on hydrophobic antithrombogenic artificial blood vessels. Tribol Int. 2021;156:106843. [41] BIRAN R, POND D. Heparin coatings for improving blood compatibility of medical devices. Adv Drug Deliv Rev. 2017;112:12-23. [42] 贾山山.多巴胺共混聚乙烯亚胺并接枝肝素对膨体聚四氟乙烯人工小血管表面改性的研究[D].广州:南方医科大学,2018. [43] SHAN Y, CHEN G, SHI Q, et al. Heparin/Collagen-REDV Modification of Expanded Polytetrafluoroethylene Improves Regional Anti-thrombosis and Reduces Foreign Body Reactions in Local Tissues. Front Bioeng Biotech. 2022;10: 916931. [44] 黄彩标.ePTFE人工小血管基于多巴胺自聚并接枝比伐芦定的表面改性研究[D].广州:南方医科大学,2019. [45] 马雷.PTFE膜的表面化学改性及其生物相容性的研究[D].郑州:郑州大学,2020. [46] YAN S, LI Y, JIANG YC, et al. Expanded polytetrafluoroethylene/silk fibroin/salicin vascular graft fabrication for improved endothelialization and anticoagulation. Appl Surf Sci. 2021;542:148620. [47] ONDER S, KAZMANLI K, KOK FN. Alteration of PTFE surface to increase its blood compatibility. J Biomater Sci Polym Ed. 2011;22(11):1443-1457. [48] CHENG B, INOUE Y, ISHIHARA K. Surface functionalization of polytetrafluoroethylene substrate with hybrid processes comprising plasma treatment and chemical reactions. Colloids Surf B. 2019;173:77-84. [49] LAZIC I, OBERMEIER A, DIETMAIR B, et al. Treatment of vascular graft infections: gentamicin-coated ePTFE grafts reveals strong antibacterial properties in vitro. J Mater Sci Mater Med. 2022;33(3):30. [50] KAKINOKI S, NISHIOKA S, ARICHI Y, et al. Stable and direct coating of fibronectin-derived Leu-Asp-Val peptide on ePTFE using one-pot tyrosine oxidation for endothelial cell adhesion. Colloids Surf B. 2022;216:112576. [51] WALLUSCHECK KP, STEINHOFF G, KELM S, et al. Improved endothelial cell attachment on ePTFE vascular grafts pretreated with synthetic RGD-containing peptides. Eur J Vasc Endovasc.1996;12(3):321-330. [52] 张家庆.等离子体磺酸化丝素蛋白膜膨体聚四氟乙烯复合小口径人工血管的体内实验研究[D].广州:南方医科大学,2016. [53] KIRITANI S, KANEKO J, ITO D, et al. Silk fibroin vascular graft: a promising tissue-engineered scaffold material for abdominal venous system replacement. Sci Rep. 2020;10(1):21041. [54] KOPEĆ K, WOJASIŃSKI M, EICHLER M, et al. Polydopamine and gelatin coating for rapid endothelialization of vascular scaffolds. Biomater Adv. 2022;134:112544. [55] XING Z, WU S, ZHAO C, et al. Vascular transplantation with dual-biofunctional ePTFE vascular grafts in a porcine model. J Mater Chem B. 2021;9(36):749-7422. [56] LEE S, KIM S, PARK J, et al. Universal surface modification using dopamine-hyaluronic acid conjugates for anti-biofouling. Int J Biol Macromol. 2020; 151:1314-1321. [57] FOWLER PMPT, DIZON GV, TAYO LL, et al. Surface Zwitterionization of Expanded Poly(tetrafluoroethylene) via Dopamine-Assisted Consecutive Immersion Coating. ACS Appl Mater Interfaces. 2020;12(37):41000-41010. [58] YU C, YANG H, WANG L, et al. Surface modification of polytetrafluoroethylene (PTFE) with a heparin-immobilized extracellular matrix (ECM) coating for small-diameter vascular grafts applications. Mater Sci Eng C. 2021;128:112301. [59] CHENG B, INOUE Y, ISHIHARA K. Conversion of Functional Group on PTFE Surface by Argon Plasma Pre-treatment and Polydopamine Coating. Iop Conference. 2018;381(1):12039. [60] MI H, JING X, THOMSOM JA, et al. Promoting endothelial cell affinity and antithrombogenicity of polytetrafluoroethylene (PTFE) by mussel-inspired modification and RGD/heparin grafting. J Mater Chem B. 2018;6(21):3475-3485. [61] DIZON GV, TORIBIO FOWLER PM, VENAULT A, et al. Dopamine-Induced Surface Zwitterionization of Expanded Poly(tetrafluoroethylene) for Constructing Thermostable Bioinert Materials. ACS Biomater Sci Eng. 2022; 8(4):1532-1543. [62] SONG H, YU H, ZHU L, et al. Durable hydrophilic surface modification for PTFE hollow fiber membranes. React Funct Polym. 2017;114:110-117. [63] WANG P, TAN KL, KANG ET. Surface modification of poly (tetrafluoroethylene) films via grafting of poly (ethylene glycol) for reduction in protein adsorption. J Biomat Sci Polym Ed. 2000;11(2):169-186. [64] 赵康,高俊娜,束兴娟,等.电子束辐射接枝丙烯酸改性聚四氟乙烯[J].辐射研究与辐射工艺学报,2021,39(4):13-18. [65] KRAWIEC JT, VORP DA. Adult stem cell-based tissue engineered blood vessels: a review. Biomaterials. 2012;33(12):3388-3400. [66] 夏霖.聚四氟乙烯表面的仿生化修饰与生物相容性研究[D].无锡:江南大学,2013. [67] FENG S, ZHONG Z, WANG Y, et al. Progress and perspectives in PTFE membrane: Preparation, modification, and applications. J Membr Sci. 2018; 549:332-349. [68] AL MESLMANI BM, MAHMOUD GF, BAKOWSKY U. Development of expanded polytetrafluoroethylene cardiovascular graft platform based on immobilization of poly lactic- co -glycolic acid nanoparticles using a wet chemical modification technique. Int J Pharm. 2017;529(1-2):238-244. [69] PU FR, WILLIAMS RL, MARKKULA TK, et al. Expression of leukocyte–endothelial cell adhesion molecules on monocyte adhesion to human endothelial cells on plasma treated PET and PTFE in vitro. Biomaterials. 2002;23(24):4705-4718. [70] GAO A, HANG R, LI W, et al. Linker-free covalent immobilization of heparin, SDF-1α, and CD47 on PTFE surface for antithrombogenicity, endothelialization and anti-inflammation. Biomaterials. 2017;140:201-211. [71] RANJBARZADEH-DIBAZAR A, SHOKROLLAHI P, BARZIN J, et al. Lubricant facilitated thermo-mechanical stretching of PTFE and morphology of the resulting membranes. J Membr Sci. 2014;470:458-469. [72] YAN S, XU Y, LIN YJ, et al. Ethanol-lubricated expanded-polytetrafluoroethylene vascular grafts loaded with eggshell membrane extract and heparin for rapid endothelialization and anticoagulation. Appl Surf Sci. 2020;511:145565. [73] WANG D, XU Y, LIN YJ, et al. Biologically functionalized expanded polytetrafluoroethylene blood vessel grafts. Biomacromolecules. 2020; 21(9):3807-3816. |
| [1] | Yu Weijie, Liu Aifeng, Chen Jixin, Guo Tianci, Jia Yizhen, Feng Huichuan, Yang Jialin. Advantages and application strategies of machine learning in diagnosis and treatment of lumbar disc herniation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1426-1435. |
| [2] | Lin Zeyu, Xu Lin. Research progress in gout-induced bone destruction mechanism [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1295-1300. |
| [3] | Ma Shuwei, He Sheng, Han Bing, Zhang Liaoyun. Exosomes derived from mesenchymal stem cells in treatment of animals with acute liver failure: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1137-1142. |
| [4] | Zhang Kefan, Shi Hui. Research status and application prospect of cytokine therapy for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 961-967. |
| [5] | Cheng Jinhui, Wu Quan, Peng Min, Huang Changli, Tian Huimin, Li Yang. Preparation and properties of selective laser melting of porous titanium at a low energy density [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 664-668. |
| [6] | Li Jiaqi, Huang Yuanli, Li Yan, Wang Chunren, Han Qianqian. Mechanism and influencing factors in molecular weight degradation of non-cross-linked hyaluronic acid [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 747-752. |
| [7] | Yin Tong, Yang Jilei, Li Yourui, Liu Zhuoran, Jiang Ming. Application of core-shell structured nanofibers in oral tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 766-770. |
| [8] | Yin Kaiwen, Li Yunfeng. Application of metal-organic frameworks in implant surface modification [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 783-788. |
| [9] | Chen Xiaofang, Zheng Guoshuang, Li Maoyuan, Yu Weiting. Preparation and application of injectable sodium alginate hydrogels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 789-794. |
| [10] | Liu Chuang, Shan Shuo, Yu Tengbo, Zhou Huan, Yang Lei. Advantages, discomfort and challenges of clinical application of orthopedic hemostatic materials [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 795-803. |
| [11] | Zhang Ming, Wang Bin, Jia Fan, Chen Jie, Tang Wei. Application of brain-computer interface technology based on electroencephalogram in upper limb motor function rehabilitation of stroke patients [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 581-586. |
| [12] | He Yuanjie, Chen Yuheng, Zhao Yongchao, Wang Zhenglong. Progress in epigenetic regulation of vascular smooth muscle cell remodeling in the occurrence and development of aortic aneurysms [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 602-608. |
| [13] | Ma Sicong, Chen Jing, Li Yunqing. Functions and roles of connective tissue growth factor in nervous systems [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 615-620. |
| [14] | Yan Binghan, Li Zhichao, Su Hui, Xue Haipeng, Xu Zhanwang, Tan Guoqing. Mechanisms of traditional Chinese medicine monomers in the treatment of osteoarthritis by targeting autophagy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 627-632. |
| [15] | Shen Yurong, Nai Rentong, Zhao Ling, Liu Feigang, Yin Caoyang, Gu Yuanping, Chen Tieyi. Influence of NIC X-FILE and DENCO Pre-Shaper nickel-titanium instruments on the shaping of simulated curved canals [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 387-391. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||