Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (5): 783-788.doi: 10.12307/2024.260
Previous Articles Next Articles
Application of metal-organic frameworks in implant surface modification
Yin Kaiwen, Li Yunfeng
- Department of Orthognathic and Temporomandibular Joint Surgery, West China Hospital of Stomatology, Sichuan University & State Key Laboratory of Oral Diseases & National Clinical Research Center for Oral Diseases, Chengdu 610041, Sichuan Province, China
-
Received:2023-03-02Accepted:2023-04-18Online:2024-02-18Published:2023-08-17 -
Contact:Li Yunfeng, Master’s supervisor, Associate chief physician, Department of Orthognathic and Temporomandibular Joint Surgery, West China Hospital of Stomatology, Sichuan University & State Key Laboratory of Oral Diseases & National Clinical Research Center for Oral Diseases, Chengdu 610041, Sichuan Province, China -
About author:Yin Kaiwen, Master candidate, Department of Orthognathic and Temporomandibular Joint Surgery, West China Hospital of Stomatology, Sichuan University & State Key Laboratory of Oral Diseases & National Clinical Research Center for Oral Diseases, Chengdu 610041, Sichuan Province, China -
Supported by:National Key Research & Development Program of China, No. 2022YFB3804500 (to LYF); Natural Science Foundation of Sichuan Province (General Program), No. 2023NSFSC0570 (to LYF)
CLC Number:
Cite this article
Yin Kaiwen, Li Yunfeng. Application of metal-organic frameworks in implant surface modification[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 783-788.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
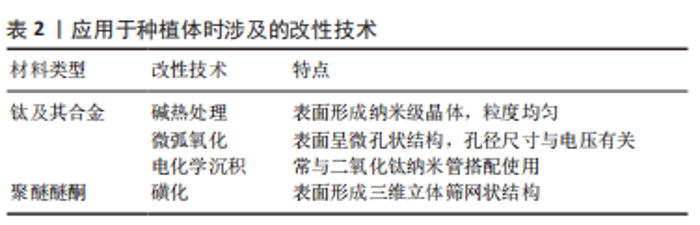
2.1.1 类沸石骨架材料 ZIFs是一种四面体框架结构,由2价金属离子与咪唑类配体配位产生,金属离子包括钴(Co2+)、锌(Zn2+)、铜(Cu2+)、铁(Fe2+)等,而其中的咪唑基团充当连接金属中心的桥梁[10]。ZIFs主要通过溶剂热法制备而成,可利用合成后修饰以及包封目标分子来调控其功能[10],并且能够人为改变其孔隙开口度和体积尺寸[11]。 ZIF系列在药物输送方面的应用较为多见,孔径大小可控允许其负载各种不同的药物分子,易于在酸性环境下降解的特点有助于实现药物在特定微环境下的可控性释放,同时对小分子药物具有优越的缓释能力。由Zn2+和2-甲基咪唑构成的ZIF-8因合成简单、水热稳定性好及生物安全性高等优势[12],在该方面受到了广泛关注。SHU等[13]用一锅合成法将阿奇霉素封装到ZIF-8中,同时完成MOFs合成和药物封装两道程序,实验显示材料在酸性环境下的释药效率明显高于中性条件,实现药物的pH响应性释放。GALLIS等[14]将头孢他啶包封在ZIF-8中,通过实验观察到ZIF-8在长达1周的时间内保持结构基本稳定,可在细胞内中直接发生内化和药物释放,表明ZIF-8可作为细胞内感染的有效治疗缓释载体。除了作为药物载体,有研究证明ZIFs还可负载多种生物分子如蛋白质及多糖等,保护其正常发挥功能,增强稳定性[15]。 2.1.2 MIL系列 MIL系列通常由羧酸盐和三价金属阳离子组成,如铁(Fe3+)、铝(Al3+)、镓(Ga3+)和铬(Cr3+)等,具有稳定性高、孔隙率持久及比表面积大等特性,突出特点是其结构能够在外界刺激的影响下发生灵活变化[16];制备MIL家族最常使用溶剂热和水热合成的方法,温度是决定合成时间长短的重要影响因素之一[17]。 MIL系列的大比表面积和高孔隙率,使其成为优秀的药物载体材料。HORCAJADA等[18]用MIL-53(Cr)和MIL-53(Fe)吸附药物,在体外模拟实验中所装载的药物持续释放长达3周。构成MIL系列的金属元素大多存在多种价态形式,得益于此,MIL系列可具有一定的酶活性,用作纳米酶催化化学反应。Hu等[19]分别把葡萄糖氧化酶和乳酸氧化酶固定到具有过氧化物酶活性的AuNPs@MIL-101上,这两种酶分别与AuNPs@MIL-101形成级联反应,用于检测葡萄糖和乳酸的变化。 2.1.3 Bio-MOFs Bio-MOFs是利用生物内源性有机配体制备的,包括碱基、氨基酸、多肽、蛋白质和糖类等。作为MOFs的一个分支,Bio-MOFs既具有MOFs的结构特性,又具有生物分子配体的生理功能[20]。 Bio-MOFs的低毒性和优异的生物相容性是其最为显著的优势,使其在生物医学领域的应用逐渐增多,包括生物传感、生物成像、疾病治疗等。值得注意的是,Bio-MOFs的结构通常不太稳定,因此,ZHU等[21]在Fe3+和阿奇霉素自组装形成的稳定性较差的生物MOF外围构建了另一种生物活性Gd-MOF,前者可在Gd-MOF的保护下在肿瘤部位有效积聚和释放药物。Bio-MOFs中有机配体的选择多样性赋予其各种独特性能,卟啉的存在使MOFs具有光响应效应,ZHANG等[22]利用卟啉基Zr-MOF在近红外光的照射下高效产生单线态氧,杀死肿瘤细胞。 2.2 种植体表面MOFs的制备工艺 利用MOFs对骨内种植体表面进行改性,材料的制备成为首先需要考虑的问题。当前应用MOFs较多的植入生物材料主要有钛及其合金和聚醚醚酮,由于它们的表面特性,想要将MOFs应用在种植体上,通常涉及一些表面改性技术。按钛及其合金和聚醚醚酮两种植入材料类型分别进行归纳,涉及的改性技术总结见表2。"
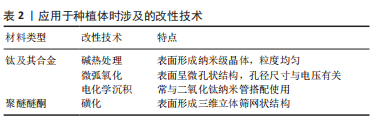
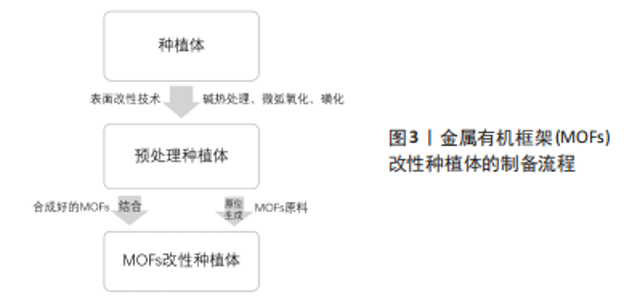
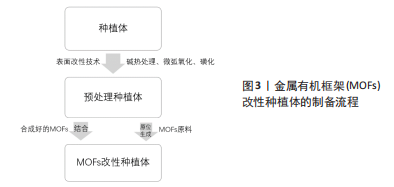
钛及其合金因其优异的力学特性、较强的耐腐蚀性和良好的生物相容性,成为硬组织修复和骨骼替代物的首选材料[23]。但其本质上是生物惰性材料,表面不存在有机官能团,因此MOFs不能直接加载在其上[24],需要对其进行预处理。目前最常见的处理方法是碱热处理,将钛片在氢氧化钠溶液中浸泡后在高温环境下保温一段时间。研究表明,碱热处理后的钛合金表面粗糙,产生 Ti-OH 活性基团,呈纳米针状结构[23]。这一系列改变为后续原位合成MOFs提供了更多的位点,也有助于已合成MOFs与钛表面的紧密结合。除此之外,也可用微弧氧化技术对钛合金进行预处理。简单概括,就是将钛合金作为阳极放置在电解液中,在其和惰性阴极之间施加高电压,使材料表面发生微弧放电,从而原位生成氧化陶瓷膜[25]。使用微弧氧化法处理后的钛合金表面呈粗糙的多孔状结构,其中大小不一的微孔无规律分布[26],较高的孔隙率和较大的孔隙直径为MOFs的制备和结合提供了有利条件。 聚醚醚酮具有弹性模量与骨骼相当、理化性质稳定及天然辐射透明等优势[27],有望成为可替代钛及其合金的新型种植体材料。然而,聚醚醚酮作为种植体的缺点也很明显,它存在表面惰性,生物活性差[28]。磺化是聚醚醚酮表面制备或结合MOFs时最常应用的改性技术。聚醚醚酮被浸入浓硫酸中一定时间,利用浓硫酸的强腐蚀作用发生亲电取代反应,在聚醚醚酮表面生成磺酸基团,形成三维立体筛网状结构[29],该操作提高了材料表面的微观粗糙度,使得MOFs与聚醚醚酮之间拥有更多的有效结合位点,有利于两者形成高结合力的机械嵌合。 对植入材料进行预处理后,后续制备过程大致分为两类:一种是将合成好的MOFs结合到预处理种植体上,另一种是在处理后的种植体上原位合成MOFs,见图3。"
2.2.1 MOFs与种植体结合 MOFs的获得可借助水热/溶剂热法、微波辅助法、机械化学法和电化学法等。 水热法:是指在密闭容器内,以水为溶剂,在一定温度和压力下原始混合物发生反应的一种合成方法,而溶剂热法与其的区别在于将水换为有机溶剂。这两种方法的优点在于过程相对简单且易于控制,产物的产率和结晶度较为优秀,是MOFs合成中最常见的方法。PAN等[30]报道了首次在水溶液中合成ZIFs,室温下即可进行,反应快、产率高,产物具有优异的稳定性。 微波辅助法:此方法能够为反应体系快速施加能量,显著缩短反应时间,提高反应速率至几倍、几十倍甚至更多。在合成过程中可以防止晶型转变和晶粒间聚集,因而易于得到体积小、形状规则的产物,还可通过调节参数实现产物尺寸的控制[31]。JACOBSEN等[32]在微波加热的辅助下通过溶剂热法合成了具有MIL-140结构的Ce(IV)基MOFs,开辟了合成目标MOFs的新途径。 机械化学法:就是把两种或两种以上的反应物混合后在一起,然后研磨或者球磨,利用机械能促进化学反应的产生。一般不添加溶剂,反应简单、环保、快速,是大量生产MOFs的优质选择。RAUTENBERG等[33]用机械化学法合成MOFs,通过优化磨削参数,可以获得多晶型。 电化学法:电化学法合成MOFs,金属离子由工作电极而不是金属化合物提供,反应条件温和,化学试剂消耗少。KANG等[34]发现可在室温下通过电化学法快速合成介孔MOFs,获得的产物稳定性好、催化效率高。 声化学法:利用超声波来促进反应产物发生化学反应。超声空化现象能够在很大程度上加快反应速度,与传统的合成方法相比,该方法更加绿色环保。RAZAVI等[35]提出了超声辅助交换连接剂的方法合成MOFs,不仅缩短了反应时间,还提高了产物的比表面积和孔体积。 合成好的MOFs结合到预处理种植体上,多采用电化学沉积和聚多巴胺黏结。电化学沉积是指在外加电场的作用下,正负电极之间发生氧化还原反应,使电解液中的离子发生迁移形成镀层[36]。在常规电解液中加入制备的MOFs纳米颗粒,植入材料充当负极进行电化学沉积,获得MOFs涂层。单体自聚合形成的贻贝激发聚多巴胺由于富含3,4-二羟基苯丙氨酸而具有优异的黏附性和生物相容性,可以均匀地黏附在具有不同特征的表面上,包括有机、陶瓷和金属[37]。有实验证明随着3,4-二羟基苯丙氨酸浓度的增加,贻贝激发肽膜对二氧化钛表面的黏附能增加,在接触1 h后薄膜对二氧化钛的黏附能可高达0.86 mJ m?2[38],因此,聚多巴胺可以充当钛表面与MOFs之间的粘结剂,将两者紧密结合在一起。此外,与钛表面操作相似,研究者们也会将具有强黏附性和螯合能力的聚多巴胺作为中间层,借助聚多巴胺的黏附力,在聚醚醚酮表面构建牢固的MOFs涂层[39]。 2.2.2 种植体表面原位合成MOFs 预处理后的种植体表面粗糙度增加,将其置入合成MOFs的反应体系中,MOFs直接在材料表面原位合成。原位合成法简捷方便,操作难度小,主要适用于一些构成简单的MOFs涂层。除了常规方法外,种子生长法作为一种环保温和的方法开始引起人们关注。采用上文提到的典型方法生成MOFs,制得种子混悬液,将材料置于混悬液中使其表面覆盖MOFs,随后再将其置入合成MOFs的反应溶液中原位合成[40]。首次贴附在材料表面的MOFs作为种子,为后续直接在种植体上生成MOFs提供锚定成核点,同时能够增强植入材料与MOFs的结合。 2.3 MOFs改性种植体的作用机制 鉴于骨整合不良和细菌感染是种植体失败的主要原因,学者对于骨科种植体表面MOFs改性的研究主要围绕设计特定MOFs以实现抑制感染和促进骨整合的目标这一主题进行。MOFs的独有特性使之可以通过多种机制发挥功能:构建高度多孔的结构,使其包封输送释放药物;引入治疗分子作为组成部分,使其溶解释放组分发挥作用;选择价态可变的金属中心,使其发挥酶活性催化底物反应;此外,MOFs可以与其他材料结合,协同发挥作用,取得理想的效果,见图4。"
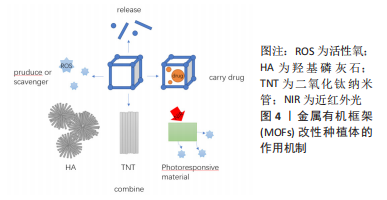

2.3.1 MOFs释放组分发挥功能 一些类型的亚稳态MOFs容易在液体中解构,然后释放出金属离子和有机配体。大量文献表明,许多金属离子如Zn2+、Mg2+等在抗菌、抗炎和促进骨整合等方面发挥有利作用[41-42]。WU等[43]制备了由Zn2+和腺嘌呤构成的Bio-MOF涂层,通过释放Zn2+促进骨整合。值得注意的是,MOFs发生快速降解时会在短时间内释放出大量的金属离子和有机配体,从而产生明显的生理毒性;另一方面,金属离子需要持续释放约4周才能产生实质性效果[23]。因此,种植体植入后其表面MOFs在体内环境中的稳定性问题引起人们的重视。SHEN等[44]在碱热处理钛表面构建了掺杂 Zn2+的Mg-MOF74复合涂层,Zn2+的加入能够明显改善Mg-MOF74涂层的稳定性。MOFs不仅可以自身直接发挥功效,还可以充当通过高温处理获得的纳米结构的优秀模板。很多MOFs衍生物具有优于前体MOFs的机械和化学稳定性,Si等[24]将MOFs衍生的CuO@ZnO复合材料结合在钛表面,实现稳定可控地释放金属离子Cu2+和Zn2+,从而取得抗菌、促进骨整合以及血管生成的效果。 除了利用金属离子发挥作用外,有学者在MOFs有机配体的选择上加以设计,将药物和抗氧化剂等引入MOFs,通过释放特定有机组分实现植入材料功能化。YAN等[45]选用抗氧化剂富马酸作为配体,氢氟酸作为调节剂,在钛表面构建Zr-MOF膜。氟离子和富马酸的联合释放,使材料展现出良好的抗菌、抗炎和促进成骨的性能。在研究过程中,关注MOFs组分功用的同时需要将MOFs涂层的物理性质考虑在内。MOFs晶体的大小以及MOFs涂层的密度和厚度可能会干扰基材的生物性能。CHEN等[40]进行了将MOFs薄膜应用于钛表面改性的首次尝试,分别制备了纳米级和微米级ZIF-8薄膜,发现微米级ZIF-8薄膜产生明显的细胞毒性,而纳米级ZIF-8薄膜达到抗菌和促进骨整合的效果。 2.3.2 MOFs作为载体发挥功能 大比表面积和高孔隙率有利于MOFs具有良好的药物加载能力,而如何在恰当的时间释放所需量的药物成为另一重要的问题。细菌的代谢作用导致周围环境呈微酸性,骨质疏松等骨病也会使微环境略偏酸性,这正与MOFs的pH敏感性相呼应,利于改性后的种植体实现药物的响应性释放。YU等[46]在钛合金表面加载了包封中药柚皮苷的MOFs颗粒,实现体液环境下可控性释放药物,达到抑菌、促进骨整合的效果。SANDOMIERSKI等[47]借助MOFs实现金属钛对利塞膦酸盐的控释,有效避免其他给药方式产生的不良反应。聚电解质多层膜是通过逐层沉积带有正负电荷的聚电解质形成的,将其涂覆在MOFs上能够进一步帮助MOFs有效避免药物的爆发性释放,并且对MOFs原有结构无影响。TAO等[48]将MOF@左氧氟沙星纳米颗粒沉积在钛底物上,并进行多层壳聚糖/凝胶涂覆处理,发现涂覆多层膜的材料释放左氧氟沙星的速率明显低于未处理样品。 由于人体内环境复杂多变,实现环境响应性释放目前仍是药物载体系统面对的一大挑战[41]。现阶段基于MOFs的载药系统的环境响应条件主要聚焦在pH值、温度及氧化还原等方面[42]。如何根据植入术后周围的微环境特点,实现种植体表面MOFs吸附的药物的可控性释放成为未来研究的重点。 2.3.3 MOFs作为纳米酶发挥功能 MOFs可以通过金属组分或在有机配体上锚定官能团来引入活性位点,因其结构特点而具有均相催化的优势[50],在纳米催化领域有着可观的发展前景。常见的纳米酶具有氧化酶、过氧化物酶、超氧化物歧化酶和(或)过氧化氢酶活性,前两者可催化产生活性氧,杀灭细菌[51];后两者可有效清除活性氧,利于骨整合[52]。CHEN等[53]在碱热钛表面构建了骨微环境响应的金属有机框架(Ce/SrMOF)涂层,金属Ce和Sr离子的存在使种植体显示出超氧化物歧化酶和过氧化氢酶样催化活性,可快速分解间充质干细胞中的活性氧并恢复其线粒体功能。对于骨质疏松症患者而言,间充质干细胞功能的恢复有利于促进新骨形成,改善骨整合。ZHANG等[54]在医用钛表面制备了具有氧化酶样活性的铈基金属有机框架(Ce-BTC)涂层,并进行了氢等离子体浸没离子注入处理。处理后,Ce-BTC得以暴露出更多的金属活性位点,明显提高了其ATP剥夺能力和氧化酶样活性,从而抑制生物膜的形成,响应细菌介导的酸性微环境杀灭细菌。 纳米酶的活性不同,催化的反应底物也不尽相同。如何丰富种植体表面构建的MOFs纳米酶的活性类型以及提高其活性将成为未来研究的重点问题之一。 2.3.4 MOFs与其他结构联合 羟基磷灰石:羟基磷灰石与人类骨骼系统中的矿物成分具有相似的化学成分和晶体结构,在骨和牙齿替代方面享有优势[55]。羟基磷灰石涂层利用其生物相容性好以及与骨骼的化学成分相似的特点,可促进骨组织与种植体的融合,同时有助于保持金属基材的机械性能。 ZHANG等[56]在种植体表面构建了镁介导的金属有机框架(Mg-MOF74)和锶取代的羟基磷灰石复合涂层,Mg-MOF74在感染情况下能快速解构并高效清除周围的细菌,而涂覆在MOFs表面的羟基磷灰石涂层具有促进成骨细胞增殖和分化的潜能。ZHANG等[57]对钛进行改性时将MIL@Ce和羟基磷灰石涂层共沉积在TiO2-Ti表面,合理推测在电化学共沉积过程中MIL@Ce纳米颗粒充当了成核中心,而羟基磷灰石作为外壳起保护和固定作用。因此,羟基磷灰石的存在有助于MOFs缓慢释放金属离子,起到成骨抑菌作用。羟基磷灰石与MOFs联合应用时,两者发挥协同作用,互相弥补不足,赋予种植体功能多样化和效果显著化的特点,使其成为理想的多功能植入材料。 二氧化钛纳米管:二氧化钛纳米管是种植体表面改性的常见选择,主要通过阳极氧化工艺制备而成。二氧化钛纳米管主要呈锐钛矿相,提供了微观的纳米纹理和加大的比表面积,有利于周围细胞的黏附和与之相互作用,自身还可作为载药平台传递释放药物,因此其在促进骨整合、细胞分化和抗菌等方面都有一定的积极作用[58]。 TAO等[59]利用钛箔表面制备了二氧化钛纳米管,然后在其上沉积形成负载成骨生长肽的ZIF-67涂层,实验结果证实,该材料能够在早期有效地发挥抗菌和抗炎作用,并在随后的骨整合中产生积极影响。在二氧化钛纳米管上进行MOFs电化学沉积的效果要优于钛表面,因此将二氧化钛纳米管与MOFs相结合,既有利于MOFs涂层的制备,又能显著增强种植体的功能。 光响应材料:光疗杀菌是指使用适当波长的光照射,激活光响应材料,使其吸收光能,通过产生活性氧或/和热量在短时间内有效杀死病原体[60]。MOFs自身可作为光响应材料,接受光照产生热和或自由基[61]。但在目前关于种植体表面MOFs改性与光疗的研究中,学者更倾向于将MOFs与其他的光敏材料联合应用。 聚多巴胺不仅具有前文提到的黏附性能,还拥有优异的光响应性。它吸收光谱宽,光热转换快,同时具有半导体性质和电荷转移能力[62-63]。CHEN等[64]将聚多巴胺和ZIF-8结合到磺化聚醚醚酮底物上,有近红外光照射,聚多巴胺产生热和自由基,与释放的大量Zn2+协同杀菌;无近红外光照射,ZIF-8持续缓慢释放Zn2+,促进成骨。DENG等[39]构建了由负载辛伐他汀的MOFs和聚多巴胺纳米层组成的异质结构涂层,辛伐他汀的加入提高了材料整体的成骨能力。在种植体MOFs改性操作中,光敏剂与MOFs结合能够显著增强种植体抑制细菌感染的能力,同时,光敏剂的加入提供了近红外光这一“开关”,使人们可以根据需要人为控制种植体在不同环境下发挥的具体功能,实现多模式治疗。 近年来,MOFs在种植体表面改性领域的应用逐渐增多,关于种植体表面MOFs改性研究的时间脉络总结见表3。"

| [1] 韩倞,杨轶,张弛,等.骨科植入物表面抗感染修饰及其骨整合性的研究进展[J].中国临床医学,2017,24(1):134-140. [2] GOODMAN SB, GIBON E, PAJARINEN J, et al. Novel biological strategies for treatment of wear particle-induced periprosthetic osteolysis of orthopaedic implants for joint replacement. J R Soc Interface. 2014;11(93):20130962. [3] 张超群,吴一芃,徐永清.骨科内植物表面抗菌改性的研究进展[J].中国修复重建外科杂志,2022,36(4):511-516. [4] FURUKAWA H, KO N, GO YB, et al. Ultrahigh porosity in metal-organic frameworks. Science. 2010;329(5990):424-428. [5] FARHA OK, YAZAYDIN AO, ERYAZICI I, et al. De novo synthesis of a metal-organic framework material featuring ultrahigh surface area and gas storage capacities. Nat Chem. 2010;2(11): 944-948. [6] HORCAJADA P, CHALATI T, SERRE C, et al. Porous metal-organic-framework nanoscale carriers as a potential platform for drug delivery and imaging. Nat Mater. 2010;9(2):172-178. [7] LI B, ZHANG Y, MA D, et al. A strategy toward constructing a bifunctionalized MOF catalyst: post-synthetic modification of MOFs on organic ligands and coordinatively unsaturated metal sites. Chem Commun (Camb). 2012;48(49):6151-6153. [8] YANG J, YANG YW. Metal-organic frameworks for biomedical applications. Small. 2020; 16(10):e1906846. [9] HERMES S, SCHRÖDER F, CHELMOWSKI R, et al. Selective nucleation and growth of metal-organic open framework thin films on patterned COOH/CF3-terminated self-assembled monolayers on Au(111). J Am Chem Soc. 2005;127(40):13744-13745. [10] MALEKI A, SHAHBAZI MA, ALINEZHAD V, et al. The progress and prospect of zeolitic imidazolate frameworks in cancer therapy, antibacterial activity, and biomineralization. adv healthc mater. 2020;9(12):e2000248. [11] YANG J, ZHANG YB, LIU Q, et al. Principles of designing extra-large pore openings and cages in zeolitic imidazolate frameworks. J Am Chem Soc. 2017;139(18):6448-6455. [12] GUO H, LIU L, HU Q, et al. Monodisperse ZIF-8@dextran nanoparticles co-loaded with hydrophilic and hydrophobic functional cargos for combined near-infrared fluorescence imaging and photothermal therapy. Acta Biomater. 2022;137:290-304. [13] SHU F, LV D, SONG XL, et al. Fabrication of a hyaluronic acid conjugated metal organic framework for targeted drug delivery and magnetic resonance imaging. RSC Adv. 2018; 8(12):6581-6589. [14] GALLIS DFS, BUTLER KS, AGOLA JO, et al. Antibacterial countermeasures via metal-organic framework-supported sustained therapeutic release. ACS Appl Mater Interfaces. 2019;11(8):7782-7791. [15] JIANG C, ZHANG L, XU X, et al. Engineering a smart agent for enhanced immunotherapy effect by simultaneously blocking PD-L1 and CTLA-4. Adv Sci (Weinh). 2021;8(20):e2102500. [16] NEIMARK AV, COUDERT FX, TRIGUERO C, et al. Structural transitions in MIL-53 (Cr): view from outside and inside. Langmuir. 2011;27(8):4734-4741. [17] ZHANG HS, HU X, LI TX, et al. MIL series of metal organic frameworks (MOFs) as novel adsorbents for heavy metals in water: a review. J Hazard Mater. 2022;429:128271. [18] HORCAJADA P, SERRE C, MAURIN G, et al. Flexible porous metal-organic frameworks for a controlled drug delivery. J Am Chem Soc. 2008;130(21):6774-6780. [19] HU Y, CHENG H, ZHAO X, et al. Surface-enhanced raman scattering active gold nanoparticles with enzyme-mimicking activities for measuring glucose and lactate in living tissues. ACS Nano. 2017;11(6):5558-5566. [20] VAIDYA LB, NADAR SS, RATHOD VK. Biological metal organic framework (bio-MOF) of glucoamylase with enhanced stability. Colloids Surf B Biointerfaces. 2020;193:111052. [21] ZHU Y, XIN N, QIAO Z, et al. Bioactive MOFs based theranostic agent for highly effective combination of multimodal imaging and chemo-phototherapy. Adv Healthc Mater. 2020; 9(14):e2000205. [22] ZHANG Y, WANG F, LIU C, et al. Nanozyme decorated metal-organic frameworks for enhanced photodynamic therapy. ACS Nano. 2018;12(1):651-661. [23] KAMO M, KYOMOTO M, MIYAJI F. Time course of surface characteristics of alkali- and heat-treated titanium dental implants during vacuum storage. J Biomed Mater Res B Appl Biomater. 2017;105(6):1453-1460. [24] Si YH, Liu HY, Yu HY, et al. MOF-derived CuO@ZnO modified titanium implant for synergistic antibacterial ability, osteogenesis and angiogenesis. Colloids Surf B Biointerfaces. 2022; 219:112840. [25] WANG X, MEI L, JIANG X, et al. Hydroxyapatite-coated titanium by micro-arc oxidation and steam-hydrothermal treatment promotes osseointegration. Front Bioeng Biotechnol. 2021;9:625877. [26] 马少华.钛合金表面微弧氧化膜层性能研究[C]//.2020中国航空工业技术装备工程协会年会论文集,2020:560-563. [27] YANG X, WANG Q, ZHANG Y, et al. A dual-functional PEEK implant coating for anti-bacterial and accelerated osseointegration. Colloids Surf B Biointerfaces. 2023;224:113196. [28] WEI X, ZHOU W, TANG Z, et al. Magnesium surface-activated 3D printed porous PEEK scaffolds for in vivo osseointegration by promoting angiogenesis and osteogenesis. Bioact Mater. 2022;20:16-28. [29] WANG W, LUO CJ, HUANG J, et al. PEEK surface modification by fast ambient-temperature sulfonation for bone implant applications. J R Soc Interface. 2019;16(152):20180955. [30] PAN Y, LIU Y, ZENG G, et al. Rapid synthesis of zeolitic imidazolate framework-8 (ZIF-8) nanocrystals in an aqueous system. Chem Commun (Camb). 2011;47(7):2071-2073. [31] TADDEI M, DAU PV, COHEN SM, et al. Efficient microwave assisted synthesis of metal-organic framework UiO-66: optimization and scale up. Dalton Trans. 2015;44(31):14019-14026. [32] JACOBSEN J, WEGNER L, REINSCH H, et al. Ce-MIL-140: expanding the synthesis routes for cerium(iv) metal-organic frameworks. Dalton Trans. 2020;49(32):11396-11402. [33] RAUTENBERG M, GERNHARD M, RADNIK J, et al. Mechanochemical synthesis of fluorine-containing co-doped zeolitic imidazolate frameworks for producing electrocatalysts. Front Chem. 2022;10:840758. [34] KANG X, LYU K, LI L, et al. Integration of mesopores and crystal defects in metal-organic frameworks via templated electrosynthesis. Nat Commun. 2019;10(1):4466. [35] RAZAVI SAA, MORSALI A. Ultrasonic-Assisted Linker Exchange (USALE): a novel post-synthesis method for controlling the functionality, porosity, and morphology of MOFs. Chemistry. 2019;25(46):10876-10885. [36] LU M, CHEN H, YUAN B, et al. Electrochemical deposition of nanostructured hydroxyapatite coating on titanium with enhanced early stage osteogenic activity and osseointegration. Int J Nanomedicine. 2020;15:6605-6618. [37] JIA L, HAN F, WANG H, et al. Polydopamine-assisted surface modification for orthopaedic implants. J Orthop Translat. 2019;17:82-95. [38] ANDERSON TH, YU J, ESTRADA A, et al. The Contribution of DOPA to substrate-peptide adhesion and internal cohesion of mussel-inspired synthetic peptide films. Adv Funct Mater. 2010;20(23):4196-4205. [39] DENG Y, SHI J, CHAN YK, et al. Heterostructured metal-organic frameworks/polydopamine coating endows polyetheretherketone implants with multimodal osteogenicity and photoswitchable disinfection. Adv Healthc Mater. 2022;11(14):e2200641. [40] CHEN J, ZHANG X, HUANG C, et al. Osteogenic activity and antibacterial effect of porous titanium modified with metal-organic framework films. J Biomed Mater Res A. 2017;105(3): 834-846. [41] MUTLU N, LIVERANI L, KURTULDU F, et al. Zinc improves antibacterial, anti-inflammatory and cell motility activity of chitosan for wound healing applications. Int J Biol Macromol. 2022;213:845-857. [42] HU T, XU H, WANG C, et al. Magnesium enhances the chondrogenic differentiation of mesenchymal stem cells by inhibiting activated macrophage-induced inflammation. Sci Rep. 2018;8(1):3406. [43] WU J, JIANG S, XIE W, et al. Surface modification of the Ti surface with nanoscale bio-MOF-1 for improving biocompatibility and osteointegration in vitro and in vivo. J Mater Chem B. 2022;10(41):8535-8548. [44] SHEN X, ZHANG Y, MA P, et al. Fabrication of magnesium/zinc-metal organic framework on titanium implants to inhibit bacterial infection and promote bone regeneration. Biomaterials. 2019;212:1-16. [45] YAN BC, TAN J, ZHANG HF, et al. Constructing fluorine-doped Zr-MOF films on titanium for antibacteria, anti-inflammation, and osteogenesis. Biomater Adv. 2022;134:112699. [46] YU M, YOU D, ZHUANG J, et al. Controlled release of naringin in metal-organic framework-loaded mineralized collagen coating to simultaneously enhance osseointegration and antibacterial activity. ACS Appl Mater Interfaces. 2017;9(23):19698-19705. [47] SANDOMIERSKI M, JAKUBOWSKI M, RATAJCZAK M, et al. Zeolitic Imidazolate Framework8 (ZIF-8) modified titanium alloy for controlled release of drugs for osteoporosis. Sci Rep. 2022;12(1):9103. [48] TAO B, ZHAO W, LIN C, et al. Surface modification of titanium implants by ZIF-8@Levo/LBL coating for inhibition of bacterial-associated infection and enhancement of in vivo osseointegration. Chemical Engineering Journal. 2020,390:124621. [49] ZHANG Y, CHAN HF, LEONG KW. Advanced materials and processing for drug delivery: the past and the future. Adv Drug Deliv Rev. 2013;65(1):104-120. [50] JIAO L, WANG Y, JIANG HL, et al. Metal-organic frameworks as platforms for catalytic applications. Adv Mater. 2018;30(37):e1703663. [51] MATTER MT, FURER LA, STARSICH FHL, et al. Engineering the bioactivity of flame-made ceria and ceria/bioglass hybrid nanoparticles. ACS Appl Mater Interfaces. 2019;11(3):2830-2839. [52] ZHAO Y, SONG S, WANG D, et al. Nanozyme-reinforced hydrogel as a H2O2-driven oxygenerator for enhancing prosthetic interface osseointegration in rheumatoid arthritis therapy. Nat Commun. 2022;13(1):6758. [53] CHEN MW, WANG D, LI MH, et al. Nanocatalytic biofunctional MOF coating on titanium implants promotes osteoporotic bone regeneration through cooperative pro-osteoblastogenesis MSC reprogramming. ACS Nano. 2022;16(9):15397-15412. [54] ZHANG HF, QIU JJ, XING M, et al. Hydrogen plasma treated-Ce-BTC nanorods enable enhanced antibacterial activity and soft tissue sealing ability. Chem Eng J. 2022;449:137881. [55] XU M, LIU T, QIN M, et al. Bone-like hydroxyapatite anchored on alginate microspheres for bone regeneration. Carbohydr Polym. 2022;287:119330. [56] ZHANG Y, SHEN X, MA P, et al. Composite coatings of Mg-MOF74 and Sr-substituted hydroxyapatite on titanium substrates for local antibacterial, anti-osteosarcoma and pro-osteogenesis applications. Mater Lett. 2019;241:18-22. [57] ZHANG Z, ZHANG Y, ZHANG S, et al. Synthesis of rare earth doped MOF base coating on TiO2 nanotubes arrays by electrochemical method using as antibacterial implant material. Inorganic Chem Commun. 2021;127:108484. [58] LEWANDOWSKA Z, PISZCZEK P, RADTKE A, et al. The evaluation of the impact of titania nanotube covers morphology and crystal phase on their biological properties. J Mater Sci Mater Med. 2015;26(4):163. [59] TAO B, LIN C, HE Y, et al. Osteoimmunomodulation mediating improved osteointegration by OGP-loaded cobalt-Metal organic framework on titanium implants with antibacterial property. Chem Eng J. 2021;423:130176. [60] CHEN M, LONG Z, DONG R, et al. Titanium Incorporation into Zr-Porphyrinic Metal-Organic Frameworks with Enhanced Antibacterial Activity against Multidrug-Resistant Pathogens. Small. 2020;16(7):e1906240. [61] PAN WL, TAN Y, MENG W, et al. Microenvironment-driven sequential ferroptosis, photodynamic therapy, and chemotherapy for targeted breast cancer therapy by a cancer-cell-membrane-coated nanoscale metal-organic framework. Biomaterials. 2022;283:121449. [62] D’ISCHIA M, NAPOLITANO A, BALL V, et al. Polydopamine and eumelanin: from structure-property relationships to a unified tailoring strategy. Acc Chem Res. 2014;47(12):3541-3550. [63] NAM HJ, CHA J, LEE SH, et al. A new mussel-inspired polydopamine phototransistor with high photosensitivity: signal amplification and light-controlled switching properties. Chem Commun (Camb). 2014;50(12):1458-1461. [64] CHEN Y, YANG QZ, MA DC, et al. Metal-organic frameworks/polydopamine coating endows polyetheretherketone with disinfection and osteogenicity. Inter J Polym Mater Polym Biomater. 2022;71(10):783-794. |
| [1] | Liang Cheng, Zhang Linqi, Wang Guan, Li Wen, Duan Ke, Li Zhong, Lu Xiaobo, Zhuo Naiqiang. Finite element and biomechanical analysis of different implants in repair for unilateral unstable pelvic posterior ring injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1336-1341. |
| [2] | Xiaheida·Yilaerjiang, Nijiati·Tuerxun, Reyila·Kuerban, Baibujiafu·Yelisi, Chen Xin. Three-dimensional finite element analysis of the distribution pattern of stress in bone tissues with different characteristics [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1277-1282. |
| [3] | Dai Huijuan, Wang Zhaoxin, Baibujiafu·Yelisi, Sun Jiangwei, Gulizainu·Yibulayin, Nijiati·Tuerxun. Biomechanical difference between resin ceramic crown and zirconia all-ceramic crown implant restorations in three occlusal relationships [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 657-663. |
| [4] | Wang Jiani, Chen Junyu. Angiogenesis mechanism of metal ions and their application in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 804-812. |
| [5] | Zhang Qianlong, Maihemuti•Yakufu, Song Chenhui, Liu Xiuxin, Ren Zheng, Liu Yuzhe, Muyashaer•Abudushalamu, Sajidan•Aikebaier, Ran Jian. Finite element analysis of the effect of the distribution position and content of bone cement on the stress and displacement of reverse femoral intertrochanteric fracture [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 336-340. |
| [6] | Dai Jing, Liu Shasha, Shen Mingjing. Exosome-loaded injectable hydrogel for repairing bone defects around implants [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 347-354. |
| [7] | Chen Yuanyuan, Wang Wei, Zhao Lu, Annikaer·Aniwaer, Nijati·Turson. Finite element analysis of the influence of scaffold materials on the fixed restoration of edentulous maxillary implants under two designs [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 411-418. |
| [8] | Chen Jiawen, Luo Siyang, Liu Yin, Chen Guangneng, Zuo Yuwen, He Xianyu, Ma Minxian. Finite element analysis of the effect of bone on occlusal adjustment of right upper first molar implants [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(16): 2579-2586. |
| [9] | Xu Dapeng, Jing Jie, Ma Lu, Qu Aili. Finite element analysis of the optimal time for second-stage maxillary sinus floor lift [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(11): 1647-1652. |
| [10] | Wen Xinghua, Ding Huanwen, Cheng Kai, Yan Xiaonan, Peng Yuanhao, Wang Yuning, Liu Kang, Zhang Huiwu. Three-dimensional finite element model analysis of intramedullary nailing fixation design for large femoral defects in Beagle dogs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1371-1376. |
| [11] | Zhu Lin, Gu Weiping, Wang Can, Chen Gang. Biomechanical analysis of All-on-Four and pterygomaxillary implants under different maxillary bone conditions [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 985-991. |
| [12] | Sun Jiangwei, Wang Junxiang, Baibujiafu·Yellisi, Dai Huijuan, Nijati·Turson. Three-dimensional finite element analysis of stress distribution in different smooth collar implants [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1004-1011. |
| [13] | Zhang Tingting, Liu Juan, Zhang Xu. Bioactivity of phase-transition lysozyme for surface modification of zirconia all-ceramic implant material mediating hydroxyapatite coating [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1043-1049. |
| [14] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [15] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||