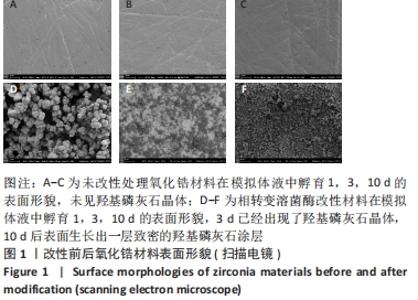
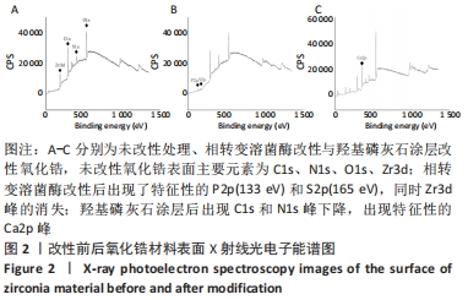
Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (7): 1043-1049.doi: 10.12307/2023.040
Previous Articles Next Articles
Bioactivity of phase-transition lysozyme for surface modification of zirconia all-ceramic implant material mediating hydroxyapatite coating
Zhang Tingting, Liu Juan, Zhang Xu
- Stomatological Hospital, Tianjin Medical University, Tianjin 300000, China
-
Received:2021-11-19Accepted:2022-02-12Online:2023-03-08Published:2022-07-18 -
Contact:Zhang Xu, Professor, Stomatological Hospital, Tianjin Medical University, Tianjin 300000, China -
About author:Zhang Tingting, Master candidate, Stomatological Hospital, Tianjin Medical University, Tianjin 300000, China -
Supported by:the National Natural Science Foundation of China, No. 2301-2KQ019 (to ZX); Scientific Research Program of Tianjin Municipal Education Commission, No. 2018KJ062 (to LJ)
CLC Number:
Cite this article
Zhang Tingting, Liu Juan, Zhang Xu. Bioactivity of phase-transition lysozyme for surface modification of zirconia all-ceramic implant material mediating hydroxyapatite coating[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1043-1049.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
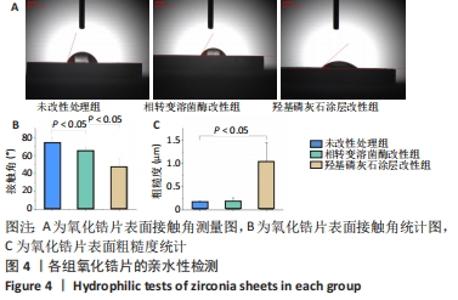
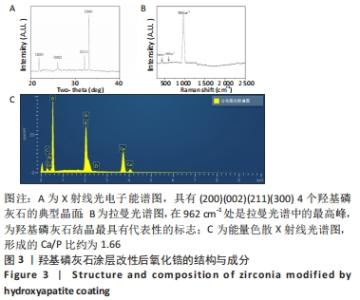
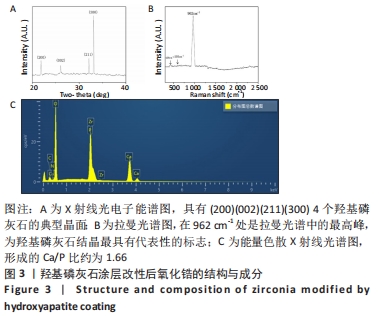
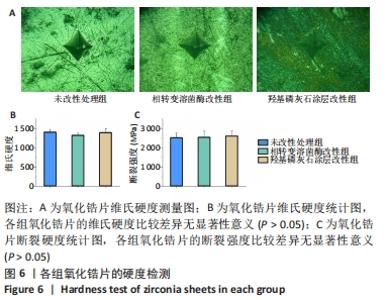
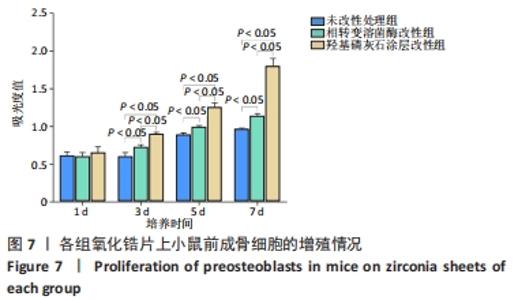
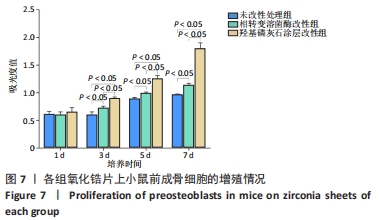
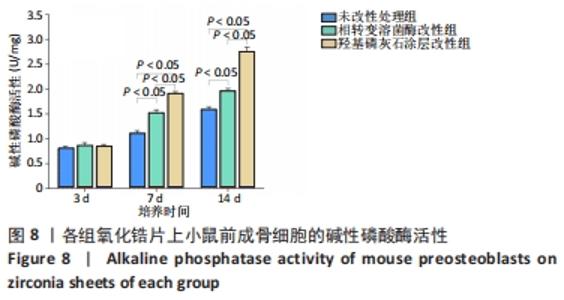
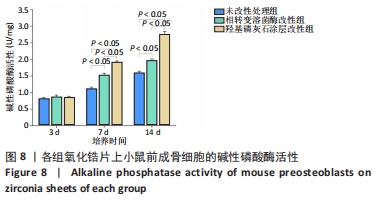
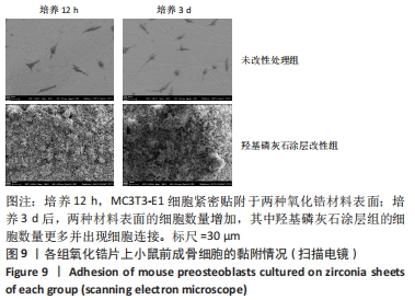
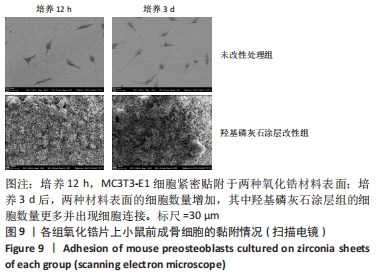
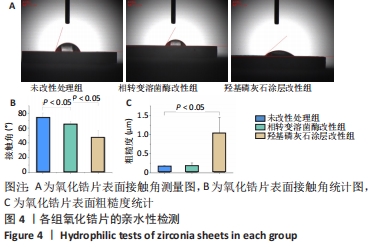
X射线多晶衍射结果显示,羟基磷灰石涂层改性后的氧化锆具有(200)(002)(211)(300)晶面,这4个晶面是羟基磷灰石中的典型晶面,说明在相转变溶菌酶表面所形成的无机物就是羟基磷灰石而不是磷酸钙等其他物质。显微共焦激光拉曼光谱可见几个峰,分别是428 cm-1处表示P-O键和593 cm-1处代表O-P-O键的弯曲振动以及962 cm-1处P-O的伸缩振动,在962 cm-1处的最高峰是羟基磷灰石晶体最具代表性标志。能量色散X射线光谱显示,羟基磷灰石涂层改性后的氧化锆表面形成的Ca/P比为1.66,该比值与天然骨组织中的羟基磷灰石中的Ca/P比接近。 2.4 改性前后氧化锆试件的表面接触角及粗糙度分析 实验结果表明,氧化锆片在经过相转变溶菌酶改性后进行羟基磷灰石矿化的过程中,相应的接触角大小也发生了显著变化。与未改性氧化锆片相比,经相转变溶菌酶改性后氧化锆片表面的接触角稍微降低,亲水性有所增加;在相转变溶菌酶改性的基础上进行羟基磷灰石矿化后,可以观察到氧化锆片表面的接触角明显缩小,增加了材料的表面亲水性。从以上结果可以看出,在相转变溶菌酶改性的基础上进行羟基磷灰石矿化,不但不会损害原有基材表面良好的亲水性,反而增加了基材表面的可湿性。表面粗糙度测试显示,改性前后材料之间的表面粗糙度比较差异有显著性意义(P < 0.05),见图4。 "
| [1] KOHAL RJ, KLAUS G, STRUB JR. Zirconia-implant-supported all-ceramic crowns withstand long-term load: a pilot investigation. Clin Oral Implants Res. 2006;17(5):565-571. [2] PICONI C, MACCAURO G. Zirconia as a ceramic biomaterial. Biomaterials. 1999;20:1-25. [3] YOSHINARI M. Future prospects of zirconia for oral implants -A review. Dent Mater J. 2020;39(1):37-45. [4] SURMENEV RA. A review of plasma-assisted methods for calcium phosphate-based coatings fabrication. Surf Coat Technol. 2012;206(8-9): 2035-2056. [5] LE NIHOUANNEN D, SAFFARZADEH A, AGUADO E, et al. Osteogenic properties of calcium phosphate ceramics and fibrin glue based composites. J Mater Sci Mater Med. 2007;18(2):225-235. [6] POINERN GE, BRUNDAVANAM RK, MONDINOS N, et al. Synthesis and characterisation of nanohydroxyapatite using an ultrasound assisted method. Ultrason Sonochem. 2009;16(4):469-474. [7] ZHOU H, LEE J. Nanoscale hydroxyapatite particles for bone tissue engineering. Acta Biomater. 2011;7(7):2769-2781. [8] 凯康.牙科种植体表面改性的研究进展[J].临床医药文献电子杂志, 2019,6(102):196-198. [9] KIM HW, KOH YH, LI LH, et al. Hydroxyapatite coating on titanium substrate with titania buffer layer processed by sol-gel method. Biomaterials. 2004;25(13):2533-2538. [10] WU Z, YANG P. Simple Multipurpose Surface Functionalization by Phase Transited Protein Adhesion. Adv Mater Interfaces. 2015;2(2):1-11. [11] MALOLLARI KG, DELPARASTAN P, SOBEK C, et al. Mechanical Enhancement of Bioinspired Polydopamine Nanocoatings. ACS Appl Mater Interfaces. 2019;11(46):1-26. [12] GU J, MIAO S, YAN Z, et al. Multiplex Binding of Amyloid-like Protein Nanofilm to Different Material Surfaces. Colloid Interface Sci Commun. 2018;22:42-48. [13] HA Y, YANG J, TAO F, et al. Phase-Transited Lysozyme as a Universal Route to Bioactive Hydroxyapatite Crystalline Film. Adv Funct Mater. 2018;28(4):1-12. [14] LI C, QIN R, LIU R, et al. Functional amyloid materials at surfaces/interfaces. Biomater Sci. 2018;6(3):462-472. [15] GU J, SU Y, LIU P, et al. An Environmentally Benign Antimicrobial Coating Based on a Protein Supramolecular Assembly. ACS Appl Mater Interfaces. 2017;9(1):198-210. [16] FURLONG RJ, OSBORN JF. Fixation of hip prostheses by hydroxyapatite ceramic coating. J Bone Joint Surg. 1991;73(5):741-745. [17] CHO Y, HONG J, RYOO H, et al. Osteogenic responses to zirconia with hydroxyapatite coating by aerosol deposition. J Dent Res. 2015;94(3): 491-499. [18] PARK JW, JANG JH, LEE CS, et al. Osteoconductivity of hydrophilic microstructured titanium implants with phosphate ion chemistry. Acta Biomater. 2009;5(6):2311-2321. [19] DRELICH J, MELLER JD. The Effect of Solid Surface Heterogeneity and Roughness on the Contact AngleDrop (Bubble) Size Relationship. J Colloid Interface Sci. 1994;164(1):252-259. [20] GILJEAN S, BIGERELLE M, ANSELME K, et al. New insights on contact angle/roughness dependence on high surface energy materials. Appl Surf Sci. 2011;257(22):9631-9638. [21] GAHLERT M, GUDEHUS T, EICHHORN S, et al. Biomechanical and histomorphometric comparison between zirconia implants with varying surface textures and a titanium implant in the maxilla of miniature pigs. Clin Oral Implants Res. 2007;18(5):662-668. [22] SANTANDER S, ALCAINE C, LYAHYAI J, et al. In vitro osteoinduction of human mesenchymal stem cells in biomimetic surface modified titanium alloy implants. Dent Mater J. 2012;31(5):843-850. [23] RUPP F, SCHEIDELER L, REHBEIN D, et al. Roughness induced dynamic changes of wettability of acid etched titanium implant modifications. Biomaterials. 2004;25(7-8):1429-1438. [24] DELIGIANNI DD, KATSALA N, LADAS S, et al. Effect of surface roughness of the titanium alloy Ti-6Al-4V on human bone marrow cell response and on protein adsorption. Biomaterials. 2001;22:1241-1251. [25] WANG D, HA Y, GU J, et al. 2D Protein Supramolecular Nanofilm with Exceptionally Large Area and Emergent Functions. Adv Mater. 2016; 28(34):7414-7423. [26] STONE A, HERRING DH. Practical Considerations for Successful Hardness Testing. Mater Test Charact. 2006;73:83-85. [27] AMNA T. Valorization of Bone Waste of Saudi Arabia by Synthesizing Hydroxyapatite. Appl Biochem Biotechnol. 2018;186(3):779-788. [28] KIM J, KANG IG, CHEON KH, et al. Stable sol-gel hydroxyapatite coating on zirconia dental implant for improved osseointegration. J Mater Sci Mater Med. 2021;32(7):81. [29] KIM JJ, LEE JK. Bioactivity Improvement of Zirconia Substrate by Hydroxyapatite Coating Using Room Temperature Spray Processing. J Nanosci Nanotechnol. 2021;21(8):4151-4156. [30] MACAN J, SIKIRIĆ MD, DELUCA M, et al. Mechanical properties of zirconia ceramics biomimetically coated with calcium deficient hydroxyapatite. J Mech Behav Biomed Mater. 2020;111:104006. [31] AZARI A, NIKZAD S, YAZDANI A, et al. Deposition of crystalline hydroxyapatite nano-particle on zirconia ceramic: a potential solution for the poor bonding characteristic of zirconia ceramics to resin cement. J Mater Sci Mater Med. 2017;28(7):111. [32] TORSTRICK FB, LIN ASP, POTTER D, et al. Porous PEEK improves the bone-implant interface compared to plasma-sprayed titanium coating on PEEK. Biomaterials. 2018;185:106-116. [33] 王铮,丁茜,高远,等.氧化锆多孔表面显微形貌对成骨细胞增殖及分化的影响[J/OL].2021-12-16. https://www.doc88.com/p-31273042813382.html?r=1 [34] CHEVALIER J, LOH J, GREMILLARD L, et al. Low-temperature degradation in zirconia with a porous surface. Acta Biomater. 2011;7(7):2986-2993. [35] PEZZOTTI G, YAMADA K, SAKAKURA S, et al. Raman Spectroscopic Analysis of Advanced Ceramic Composite for Hip Prosthesis. J Am Chem Soc. 2008;91(4):1199-2206. [36] SANON C, CHEVALIER J, DOUILLARD T, et al. A new testing protocol for zirconia dental implants. Dent Mater. 2015;31(1):15-25. [37] ALTMANN B, RABEL K, KOHAL RJ, et al. Cellular transcriptional response to zirconia-based implant materials. Dent Mater. 2017;33(2):241-255. [38] TUNA T, WEIN M, SWAIN M, et al. Influence of ultraviolet photofunctionalization on the surface characteristics of zirconia-based dental implant materials. Dent Mater. 2015;31(2):e14-24. [39] NAKAZAWA M, YAMADA M, WAKAMURA M, et al. Activation of Osteoblastic Function on Titanium Surface with Titanium-Doped Hydroxyapatite Nanoparticle Coating: An In Vitro Study. Int J Oral Maxillofac Implants. 2017;32(4):779-791. [40] LI X, ZOU Q, LI W, et al. Intracellular Interaction of Hydroxyapatite-Based Nanocrystals with Uniform Shape and Traceable Fluorescence. Inorg Chem. 2018;57(21):13739-13748. |
| [1] | Zhong Yizheng, Huang Peizhen, Cai Qunbin, Zheng Liqin, He Xingpeng, Dong Hang. Microstructural indexes that determine the trabecular bone maximum stress of micro-finite element models [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1313-1318. |
| [2] | Wu Tianliang, Tao Xiuxia, Xu Hongguang. Influence of different bone mineral densities on cage subsidence after stand-alone oblique lateral interbody fusion: three-dimensional finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1352-1358. |
| [3] | Wen Xinghua, Ding Huanwen, Cheng Kai, Yan Xiaonan, Peng Yuanhao, Wang Yuning, Liu Kang, Zhang Huiwu. Three-dimensional finite element model analysis of intramedullary nailing fixation design for large femoral defects in Beagle dogs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1371-1376. |
| [4] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [5] | Liu Jiaxin, Jia Peng, Men Yutao, Liu Lu, Wang Yeming, Ye Jinduo. Design and optimization of bone trabecular structure with triply periodic minimal surfaces [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 992-997. |
| [6] | Yuan Hucheng, Ding Yongguo, Ma Xuehua, Ma Wenxin, Sun Jianmin, Wang Zili, Jin Weidong. Sustained releasing of pyrazinamide, capreomycin, moxifloxacin and amikacin loaded bone cement in vitro [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1017-1022. |
| [7] | Zhao Wei, Feng Wei, Yang Tieyi, Ren Wei, Wang Yuxin, Lyu Huicheng, Chang Zhiqiang, Feng Xiaodong, Wang Ziheng, Guo Shibing. Antibiotic bone cement intramedullary nail prepared using 3D printed mold for the treatment of long bone infection in lower limbs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1023-1030. |
| [8] | Sun Jiangwei, Wang Junxiang, Baibujiafu·Yellisi, Dai Huijuan, Nijati·Turson. Three-dimensional finite element analysis of stress distribution in different smooth collar implants [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1004-1011. |
| [9] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [10] | Xu Cong, Zhao He, Sun Yan. Regeneration of facial nerve injury repaired by biomaterial nerve conduits [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1089-1095. |
| [11] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [12] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [13] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [14] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [15] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||