Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (7): 1096-1102.doi: 10.12307/2023.105
Previous Articles Next Articles
Current status and prospects of bioprosthetic heart valves
Chen Shisong, Liu Xiaohong, Xu Zhiyun
- Department of Cardiovascular Surgery, Changhai Hospital Affiliated to Naval Medical University, Shanghai 200433, China
-
Received:2022-03-24Accepted:2022-05-06Online:2023-03-08Published:2022-07-19 -
Contact:Liu Xiaohong, MD, Associate professor, Department of Cardiovascular Surgery, Changhai Hospital Affiliated to Naval Medical University, Shanghai 200433, China Xu Zhiyun, Professor, Department of Cardiovascular Surgery, Changhai Hospital Affiliated to Naval Medical University, Shanghai 200433, China -
About author:Chen Shisong, Master candidate, Department of Cardiovascular Surgery, Changhai Hospital Affiliated to Naval Medical University, Shanghai 200433, China -
Supported by:National Research and Development Program of China, No. 2016YFC1100900 (to XZY); Key Research and Development Program of Ningbo, No. 2018B10092 (to LXH)
CLC Number:
Cite this article
Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
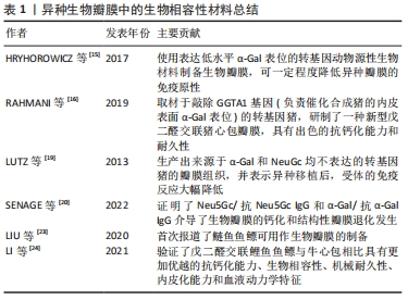
2.1 生物瓣膜的类型 2.1.1 自体瓣膜 自体瓣膜是来源于患者自身其他部位的瓣膜。1962年,Donald Ross首次提出Ross手术,并于1967年成功实施[5]。该手术最初是将自体肺动脉瓣植入主动脉瓣环内,但由于操作复杂以及自体移植物或周边血管的进行性扩张导致其再手术率较高,其术式经历了多次修改。目前,Ross手术大多选择使用肺动脉根置换技术来进行,即将肺动脉根部和肺动脉瓣作为整体移植到主动脉根部,将左、右冠状动脉分别吻合于移植物的相应部位,再利用肺同种移植物重建右室-肺动脉通道[6]。 自体瓣膜具有极佳的免疫相容性和血流动力学性能,并且血栓形成的风险低,术后无需抗凝[7]。此外,自体瓣膜会随患者的生长发育而变化,具有优异的长期生存率[6],对于严重瓣膜病的儿童及青年患者意义重大[7]。虽然有研究表示,Ross手术在成年患者中也取得了良好的长期效果[8-9],但目前Ross手术仍面临着难度高、患者创伤大和继发性瓣膜功能不全等问题[10],故目前自体瓣膜置换的受众群体仍多限于儿童及较年轻的患者[11]。 2.1.2 同种异体瓣膜 同种异体瓣膜来源于人体,指从尸体上切除或在心脏移植过程中获得的主动脉根部和肺动脉根部。与自体瓣膜类似,同种异体瓣膜也具有优越的血流动力学性能,无需抗凝[12]。但同种异体瓣膜植入人体前需要进行脱细胞处理,并需进行抗生素处理、冷冻储存或在4 ℃下短期储存[12]。此外,受材料来源限制,同种异体瓣膜无法大规模生产制造,并且随着时间的推移易发生SVD,其临床应用受到严重限制[12]。 2.1.3 异种瓣膜 由于动物来源的生物材料来源广泛,取材丰富,异种瓣膜是目前研究的重点。市面上常见的生物瓣膜多为异种瓣膜,主要是脱细胞以及交联处理后的牛/猪心包或猪主动脉瓣。经过脱细胞处理后的生物材料,其免疫原性显著降低,避免了移植物抗宿主反应的发生。此外,目前的市售生物瓣膜大都经过戊二醛交联固定,其机械性能、耐久性都显著提高。 异种瓣膜又可以分为2种亚型,即经手术植入瓣膜和经导管植入瓣膜。手术植入瓣膜有多种结构类型,包括有支架、无支架和无缝合瓣膜[13],其中无缝合主动脉瓣可在主动脉瓣环处快速展开,用不超过3个缝合线固定在主动脉环内,可以显著减少手术时间,同时促进微创手术和复杂的心脏干预[13]。经导管植入瓣膜由一个球囊扩张式或自扩张式的金属支架和内部的生物材料叶片组成,通过插入股动脉或桡动脉的导管或通过肋骨之间的小切口(经心尖)输送到心脏,然后该瓣膜在受影响的天然瓣膜或退化的生物瓣膜处展开。 2.2 生物相容性材料 生物瓣膜的耐久性受到异种生物瓣膜中不同抗原表位(如α-Gal)的表达情况影响[14]。通过交联和脱细胞处理可以一定程度上灭活或掩蔽生物材料的抗原表位,并且其效率是影响生物瓣膜临床转化的关键因素。HRYHOROWICZ等[15]提出使用表达低水平α-Gal表位的转基因动物源性生物材料制备生物瓣膜,可一定程度降低异种瓣膜的免疫原性;RAHMANI等[16]取材于敲除GGTA1基因(负责催化合成猪的内皮表面α-Gal表位)的转基因猪,研制了一种新型戊二醛交联猪心包瓣膜,具有出色的抗钙化能力和耐久性。然而,α-Gal基因敲除并不能完全消除异种移植物的免疫原性[17]。 Neu5Gc被认为是一种强免疫原性的非α-Gal聚糖类异种抗原,在用于生产人工瓣膜的部分生物材料组织(牛和马的心包膜或猪的主动脉瓣)中广泛存在,其中牛心包组织的表达水平最高[18],人体中存在的抗Neu5Gc抗体可与之产生免疫炎性反应。在过去,LUTZ等[19]就生产出来源于α-Gal和NeuGc均不表达的转基因猪的瓣膜组织,并表示异种移植后受体的免疫反应大幅降低。目前,SENAGE等[20]通过一个15年的纵向前瞻性研究,证明了Neu5Gc/抗Neu5Gc IgG和α-Gal/抗α-Gal IgG介导了生物瓣膜的钙化和SVD发生,并表示对缺乏主要聚糖异种抗原的生物相容性材料的临床研究可以降低SVD的发生率。 弹性蛋白是瓣膜组织的一种重要细胞外基质蛋白,参与构成弹性纤维并维持瓣膜弹性机械性能。除了机械效应,弹性蛋白及其降解产物还发挥信号作用[21],弹性蛋白不足或弹性纤维组装不当可能会影响瓣膜血流动力学[22]。鱼鳔主要由弹性蛋白、糖胺多糖和Ⅰ型胶原蛋白组成,其弹性蛋白含量丰富,弹性模量高于牛心包组织。此外,鱼鳔具有良好的生物相容性,并且其来源广泛取材丰富,还规避了宗教信仰因素的限制,因此鱼鳔衍生材料可能是理想的心血管生物材料。在这方面,LIU等[23]首次报道了鲢鱼鱼鳔可用作生物瓣膜的制备,并且各项性能显著提高。LI等[24]验证了戊二醛交联鲤鱼鱼鳔与牛心包相比具有更加优越的抗钙化能力、生物相容性、机械耐久性、内皮化能力和血液动力学特征。此外,鱼鳔制备的生物瓣膜除了可用于普通生物瓣膜制备之外,得益于其较薄的厚度还可以用作经导管主动脉瓣置换装置中需要折叠的生物瓣膜材料[23]。但目前关于鱼鳔植入后的物理特性、长期性能和安全性的相关研究数量较少,仍有待进一步开发。异种生物瓣膜中的生物相容性材料总结,见表1。 "

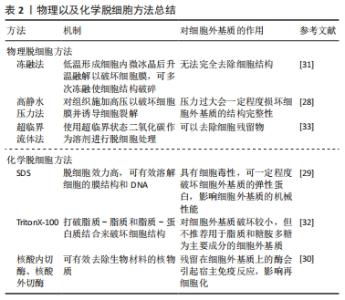
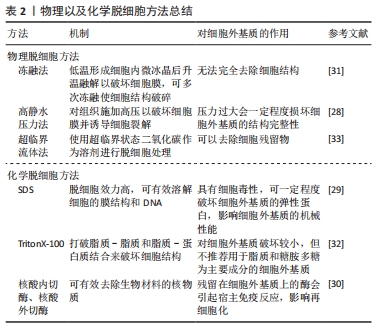
2.3 生物材料脱细胞 异种来源的生物材料所制成的生物瓣膜,具有一定免疫原性。MEEZAN等[25]首次提出对生物材料进行脱细胞处理,即去除细胞外基质中的细胞和核酸,进而降低非自体来源生物材料的免疫原性。如此,可得到保留固有几何构型的细胞外基质支架,并且其组成成分生物相容性高,可促进细胞的迁移和分化,进而推进内皮化进展;同时,生物材料的各向异性不受较大影响。 目前通常以物理、化学(或酶)或组合方法对生物材料进行脱细胞处理,需要裂解和去除细胞、去除遗传物质,并保留细胞外基质的组成和结构。在2011年,业内达成了脱细胞标准化的普遍共识[26]:①每毫克细胞外基质干质量中的DNA含量应低于50 ng;②残余DNA片段的长度应短于200个碱基对;③组织切片经DAPI或苏木精-伊红染色后缺乏可见核物质。第一和第二条标准可以分别通过PicoGreen荧光染料等dsDNA插入剂和凝胶电泳进行量化检测;第三条标准通过苏木精-伊红组织染色或DAPI免疫荧光法进行评估,并可作为前2个标准的定性验证。 物理方法包括冻融法、高静水压力法、以及超临界流体法等。物理方法对细胞外基质结构破坏较小,但无法达到有效去除生物材料免疫原性的目的[27]。例如:冻融法通过低温(-80 ℃)形成细胞内微冰晶后再升至室温(37 ℃)融解,进而破坏细胞膜来释放细胞内容物,并可多次反复冻融使细胞结构破碎,但无法完全去除细胞结构。高静水压力法通过对组织施加高压以破坏细胞膜并诱导细胞裂解,但同时压力过大会一定程度损坏细胞外基质的结构完整性[28]。超临界流体法使用具有类似于气体的强穿透力和类似于液体的高溶解度的超临界状态(处于液态和气态之间)二氧化碳作为溶剂进行脱细胞处理,可以去除细胞残留物。 化学脱细胞方法通常使用表面活性剂[十二烷基硫酸钠(SDS)和TritonX-100、Tween 20、TnBP、CHAPS]、酸、碱和酶(胰酶和胰蛋白酶)进行,这些化学试剂可有效降低组织的免疫原性,但具有一定细胞毒性,并对细胞外基质的结构蛋白产生破坏[27]。例如:SDS的脱细胞效力高,可有效溶解细胞的膜结构和DNA,但同时其细胞毒性与浓度密切相关,使用其低浓度试剂(0.01%)脱细胞后,即使多次洗涤细胞外基质,仍可检测出SDS残留,并可一定程度降解细胞外基质的弹性蛋白,对细胞外基质的机械性能产生负面影响[29]。TritonX-100对细胞外基质结构完整性的破坏较小,可以在保持蛋白质-蛋白质的相互作用不变的前提下,打破脂质-脂质和脂质-蛋白质结合来破坏细胞结构,但不推荐用于脂质和糖胺多糖作为重要组成成分的细胞外基质的脱细胞过程。核酸内切酶和核酸外切酶可有效去除生物材料的核物质,但残留在细胞外基质上的酶可能会引起宿主免疫反应,影响细胞外基质再细胞化[30]。 目前,多项研究发现使用联合脱细胞方法可以在保护细胞外基质的基础上提高脱细胞效率。例如:LIU等[31]提出真空脱细胞法作为辅助脱细胞方法可以使组织形成连续的多孔结构使得脱细胞试剂能够到达组织的深层,并且联合使用真空-冻融循环方法可以提高低浓度SDS的脱细胞效率并降低其细胞毒性。RAMM等[32]观察到在脱细胞过程中Triton X-100与胰蛋白酶联合使用可提高细胞碎片清除率并达到预期降低免疫原性效果。此外,他们还提出肽N-糖苷酶F(PNGase F)和脱氧核糖核酸酶I(DNase I)与Triton X-100和SDS的联合使用也可以提高脱细胞效率。GIL-RAMíREZ等[33]表示加压CO2-乙醇流体可为细胞外基质有效脱脂,而加压CO2-柠檬烯流体合并Benzonase核酸酶处理可有效去除DNA。 物理及化学脱细胞方法总结,见表2。 "

2.4 生物材料交联方法 生物材料脱细胞后需要进行交联固定以提高其稳定性、机械性能以及抗钙化能力,从而提高耐久性以及对原有瓣膜的替代性。交联试剂主要包括化学交联剂或天然交联剂等。 2.4.1 化学交联剂 半世纪以来,戊二醛固定剂是最广泛使用的化学交联剂。戊二醛的醛基与胶原蛋白中赖氨酸(或羟赖氨酸)的ε-氨基反应形成席夫碱键,两分子以五碳桥的形式连接,实现分子间和分子内形成共价键,因而交联固定;而对于弹性蛋白,由于其赖氨酸衍生氨基酸的含量较低,戊二醛难以对其进行交联。 戊二醛作为固定剂具有不可否认的优势,如水溶性强、化学反应速率高、交联性能优越、成本效益好等。然而,戊二醛交联会导致瓣膜进行性钙化,进而发生SVD,其确切机制尚不清楚。目前,存在的解释有:戊二醛溶于水发生醛醇缩合形成聚合物,存在游离的醛基,会在交联组织间隙中发挥持续的细胞毒性作用;戊二醛交联的生物材料,其表面带有负电荷,会吸引宿主血浆中带正电的钙离子与之结合进而产生钙化。 通过对传统戊二醛交联方法进行改进,可有效缓解生物瓣膜的钙化程度。TRANTINA-YATES等[34]提出,乌拉唑对戊二醛具有一定解毒作用,同时使用可减轻瓣叶钙化。FLAMENG等[35] 通过使用聚山梨酯-80、乙醇和甲醛的混合溶液预处理标准Perimount瓣膜以封闭游离醛基,结果显示原位瓣膜植入的幼羊模型的血流动力学和抗钙化特性明显提高。SHANG等[36]将戊二醛交联的牛心包进行氨基油酸预处理以阻断残留的醛基,再包埋在兔子肌肉内,发现瓣膜钙化减少了95%。 环氧化合物被提议作为戊二醛的替代品,一定程度上能够起到戊二醛相似的效果。与戊二醛对照组相比,环氧化合物处理的牛心包植入兔子皮下后,钙化程度明显降低[37]。在结构上,环氧化合物的环氧基团能够与ε-氨基发生亲核反应形成稳定共价键,进行胶原分子内的交联。作为交联固定剂,环氧化合物的优点包括低毒性、高溶解度,以及额外结合胶原蛋白的羟基和羧基的能力,但其抗酶解能力和稳定性较差。 一些技术可通过利用羧基的激活进行交联。碳二亚胺或1-乙基-3-(3-二甲基氨基丙基)碳二亚胺盐酸盐(EDC)可与羧基反应形成酰胺键,该处理方式下的生物瓣膜具有良好机械性能[38]。其他相关交联方案包括在酸性条件下(pH=4.5)利用1,4-丁二醇二缩水甘油醚与羧基反应进行交联[39],或利用醛类防止羧基活化后与羟基形成酯键[40],以及通过外源氨基供体(精氨酸或赖氨酸)提供更多交联位点使碳二亚胺可与弹性蛋白的羧基部分偶联等[41]。 2.4.2 天然交联剂 化学交联剂多具有一定细胞毒性,能引起瓣膜的钙化。而天然交联剂细胞毒性低,具有更高的人体相容性,受到了研究者的更多关注。 (1)京尼平:京尼平属环烯醚萜类化合物,可由栀子果实提取的京尼平苷经过β-葡萄糖苷酶水解得到。在碱性环境中,京尼平可形成单个醛基中间体,可发生自聚合形成长链聚合体,其末端的醛基可与生物材料的ε-氨基生成席夫碱键,形成一种可靠的环状交联结构。兔肌肉包埋模型表明,京尼平交联固定的牛心包钙化程度较戊二醛对照组降低了30%[42]。此外,京尼平交联剂还表现出降低移植后瓣膜炎症反应的作用[43]。伴随低温3D生物打印技术的兴起,京尼平在胶原/壳聚糖支架交联领域也拥有出色表现[44-45]。 (2)多酚类化合物:多酚类化合物中的酚羟基可与羟基、羧基、ε-氨基等基团形成氢键,使生物瓣膜材料中的胶原纤维及细胞外基质的弹性纤维和黏多糖等以氢键连接,从而实现交联固定。 LIU等[46]将姜黄素交联处理后的生物瓣膜同传统生物瓣膜进行对比,发现姜黄素交联处理后的牛心包的细胞毒性和钙化水平明显降低,机械性能和生物相容性明显提升;同时,在植入体内后其表面有大量细胞生长覆盖,说明姜黄素可能还具有促进组织修复再生的功能。此外,姜黄素具有心肌保护作用[47-48],并且使用姜黄素交联的胶原凝胶具有一定的促血管作用[49]。 有研究表示,单宁酸可以抑制与生物瓣膜钙化相关的基质金属蛋白酶9的表达[50-51]。ISENBURG等[52]发现单宁酸和戊二醛具有化学相容性,在戊二醛交联处理牛心包的基础上使用单宁酸处理,生物材料的抗酶解以及抗钙化能力大幅提高;但刘其静[53]发现戊二醛-单宁酸交联处理后瓣膜的抗钙化能力不够理想,并且细胞毒性需要进一步控制。易亮等[54]提出了一种复合交联方法,尝试改善了单宁酸交联的缺点,该方法使用核黄素光氧化和单宁酸及新霉素协同,对脱细胞牛心包进行交联,其成品生物瓣膜的细胞外基质结构良好,力学性能优秀,抗钙化能力强且厚度较薄。然而,单宁酸在人体内会发生小分子化水解,可能会导致恶心、肝损伤、消化酶活性降低等反应[55]。 王云兵等[56]发现利用茶多酚提供酚羟基,在辣根过氧化物酶/过氧化氢条件下可对猪或牛的心包膜进行酶交联,能提高生物材料弹性蛋白的结构稳定性和抗钙化性能,进而有效提升生物瓣膜的整体结构稳定性以及抗钙化性能,延长其使用寿命。 2.5 促内皮化涂层加工 生物瓣膜移植后可能发生血栓形成、凝血反应以及免疫反应,导致生物瓣膜功能障碍[57-58]。由于天然内皮层具有固有的抗凝血和抗炎作用,研究者开始关注生物瓣膜表面的快速内皮化[59]。 GULDNER等[60]提出柠檬酸、醛脱氢酶和钛涂层协同使用,对交联牛心包有良好解毒效果,并促进内皮化过程。岩藻糖胶/血管内皮生长因子聚电解质多层膜、肝素壳聚糖多层膜和纤维连接蛋白表面涂层,也被证实可增强脱细胞细胞外基质的血液相容性、抗血栓能力和内皮化能力[61-63]。 CHENG等[64]开发了一种聚丙烯酰胺/透明质酸双层网状水凝胶涂层,与脱细胞猪心包结合后,其表面内皮化加速,钙化程度减轻,并且表现出了良好的生物相容性和血液相容性。值得关注的是,其长期抗疲劳性和细胞外基质稳定性优异,经过2×108次疲劳循环后,瓣膜的表面完整性、抗钙化和抗血栓能力保持良好,符合作为生物瓣膜的长期耐久性要求。 WANG等[65]开发了一种具有长期抗血栓性能并能增强生物瓣膜内皮化的多合一涂层。该涂层接触血液内的葡萄糖后,触发底层纳米凝胶固定的葡萄糖氧化酶,产生过氧化氢并造成局部酸性环境[66]。底层的水凝胶对活性氧敏感,受过氧化氢刺激后释放负载的利伐沙班,和表层聚乙二醇协同作用抵消置换初期的剧烈凝血反应;其后,局部酸性环境刺激连接聚乙二醇的酸碱性敏感键断裂,聚乙二醇涂层逐渐脱落,进而增强细胞亲和力并允许随后的内皮化过程。 2.6 生物介入瓣膜的发展 生物材料加工结束后会修剪成合适的瓣叶形状,或与钴铬合金等金属支架进行缝合和固定,制备成完整、可供移植的生物心脏瓣膜。心脏瓣膜病的传统治疗方法是在体外循环辅助下的瓣膜置换术或瓣膜成形术,但在2002年全球首例经导管主动脉瓣置换术(transcatheter aortic valve replacement,TAVR)成功操作之后,心脏瓣膜病治疗产生了介入治疗的新方向。目前,所有TAVR均使用生物瓣,该技术采用腔内导管运载人工瓣膜,途经外周血管或心尖,于主动脉瓣处进行置换。TAVR无需进行体外循环和开胸手术,使手术风险得到大幅降低,并且,由于创伤小、恢复快等优势,在无并发症的前提下,住院时间也得以明显缩短。 目前,越来越多的新型瓣膜正在研发或投入临床应用。例如,输送系统轻巧的球囊扩张式Sapien 3瓣膜[67],不受原瓣膜钙化影响的Jena Valve瓣膜[68],具有可回收功能且无需球囊辅助的一代CoreValve瓣膜以及瓣周漏和冠脉闭塞发生率显著降低的二代CoreValve Evolut R系统[69]。 国产二代介入瓣膜可回收功能成为常态化,干式预装瓣膜、球囊扩张瓣膜、定位键辅助系统等新型产品也在不断研发中。在介入二尖瓣和三尖瓣治疗领域:中国医学科学院阜外医院团队结合了人工腱索置入和缘对缘技术,研发出经心尖二尖瓣修复装置MitralStitch,能有效治疗原发和继发性二尖瓣反流[70];复旦大学附属中山医院团队通过简化二尖瓣缘对缘技术,研发出经心尖Valve Clamp系统;浙江大学医学院附属第二医院团队研发出经导管缘对缘修复二尖瓣的Dragonfly?系统,初步临床应用良好,并尝试应用于三尖瓣反流病例[71]。上海长海医院心外科团队首次提出了非径向支撑力依赖的新理念和室间隔锚定新技术,研发出首款拥有自主知识产权的经导管三尖瓣置换瓣膜LuX-Valve,并可能成为全球首款上市的经导管三尖瓣置换瓣膜[72]。陈思等[73]对LuX-Valve治疗的重度三尖瓣关闭不全患者的短期临床效果的回顾性分析结果显示,该瓣膜围术期病死率以及术后残余三尖瓣关闭不全发生率均为3.8%,证实该介入生物瓣膜可降低手术风险和术后残余三尖瓣关闭不全风险。 由于介入瓣膜技术相对较新,并且目前使用介入干预的多为高龄或合并一定基础疾病的患者,因此对介入瓣膜的耐久性研究较少。尽管临床研究报告表示,主动脉位置的介入瓣膜和传统的生物瓣膜在移植后5-10年的耐久性相似[74-75],但目前缺乏关于介入瓣膜在更长时间内的耐久性数据[76]。 "

| [1] KOSTYUNIN A, MUKHAMADIYAROV R, GLUSHKOVA T, et al. Ultrastructural Pathology of Atherosclerosis, Calcific Aortic Valve Disease, and Bioprosthetic Heart Valve Degeneration: Commonalities and Differences. Int J Mol Sci. 2020;21(20):7434. [2] YANG Y, WANG Z, CHEN Z, et al. Current status and etiology of valvular heart disease in China: a population-based survey. BMC Cardiovasc Disord. 2021;21(1):339. [3] HARKEN DE, SOROFF HS, TAYLOR WJ, et al. Partial and complete prostheses in aortic insufficiency. J Thorac Cardiovasc Surg, 1960;40(6): 744-762. [4] FATIMA B, MOHANANEY D, KHAN F W, et al. Durability Data for Bioprosthetic Surgical Aortic Valve: A Systematic Review. JAMA Cardiol, 2019;4(1):71-80. [5] SOMERVILLE J. The origins of the Ross operation. Images Paediatr Cardiol. 2012;14(2):3-5. [6] VAN HOOF L, VERBRUGGHE P, JONES E AV, et al. Understanding Pulmonary Autograft Remodeling After the Ross Procedure: Stick to the Facts. Front Cardiovasc Med. 2022;9:829120. [7] MAZINE A, EL-HAMAMSY I, VERMA S, et al. Ross Procedure in Adults for Cardiologists and Cardiac Surgeons: JACC State-of-the-Art Review.J Am Coll Cardiol. 2018;72(22):2761-2777. [8] RYAN WH, SQUIERS JJ, HARRINGTON KB, et al. Long-term outcomes of the Ross procedure in adults. Ann Cardiothorac Surg. 2021;10(4):499-508. [9] STARNES VA, BOWDISH ME, COHEN RG, et al. The Ross procedure utilizing the pulmonary autograft inclusion technique in adults. JTCVS Tech. 2021;10:372-376. [10] LUCIANI GB, FAVARO A, CASALI G, et al. Ross operation in the young: a ten-year experience. Ann Thorac Surg. 2005;80(6):2271-2277. [11] HAGE A, HAGE F, VALDIS M, et al. The Ross procedure is the optimal solution for young adults with unrepairable aortic valve disease. Ann Cardiothorac Surg. 2021;10(4):454-462. [12] LISY M, KALENDER G, SCHENKE-LAYLAND K, et al. Allograft Heart Valves: Current Aspects and Future Applications. Biopreserv Biobank. 2017; 15(2):148-157. [13] RODRIGUEZ-GABELLA T, VOISINE P, PURI R, et al. Aortic Bioprosthetic Valve Durability: Incidence, Mechanisms, Predictors, and Management of Surgical and Transcatheter Valve Degeneration. J Am Coll Cardiol. 2017;70(8):1013-1028. [14] DALGLIESH AJ, PARVIZI M, LOPERA-HIGUITA M, et al. Graft-specific immune tolerance is determined by residual antigenicity of xenogeneic extracellular matrix scaffolds. Acta Biomater. 2018;79:253-264. [15] HRYHOROWICZ M, ZEYLAND J, SLOMSKI R, et al. Genetically Modified Pigs as Organ Donors for Xenotransplantation. Mol Biotechnol. 2017; 59(9-10):435-444. [16] RAHMANI B, MCGREGOR C, BYRNE G, et al. A Durable Porcine Pericardial Surgical Bioprosthetic Heart Valve: a Proof of Concept. J Cardiovasc Transl Res. 2019;12(4):331-337. [17] MANJI RA, EKSER B, MENKIS AH, et al. Bioprosthetic heart valves of the future. Xenotransplantation. 2014;21(1):1-10. [18] JIN C, CHERIAN RM, LIU J, et al. Identification by mass spectrometry and immunoblotting of xenogeneic antigens in the N- and O-glycomes of porcine, bovine and equine heart tissues. Glycoconj J. 2020;37(4):485-498. [19] LUTZ AJ, LI P, ESTRADA JL, et al. Double knockout pigs deficient in N-glycolylneuraminic acid and galactose alpha-1,3-galactose reduce the humoral barrier to xenotransplantation. Xenotransplantation. 2013;20(1):27-35. [20] SENAGE T, PAUL A, LE TOURNEAU T, et al. The role of antibody responses against glycans in bioprosthetic heart valve calcification and deterioration. Nat Med. 2022;28(2):283-294. [21] COCCIOLONE AJ, HAWES JZ, STAICULESCU MC, et al. Elastin, arterial mechanics, and cardiovascular disease. Am J Physiol Heart Circ Physiol. 2018;315(2):H189-H205. [22] WAHART A, HOCINE T, ALBRECHT C, et al. Role of elastin peptides and elastin receptor complex in metabolic and cardiovascular diseases. FEBS J. 2019;286(15):2980-2993. [23] LIU J, LI B, JING H, et al. Swim Bladder as a Novel Biomaterial for Cardiovascular Materials with Anti-Calcification Properties. Adv Healthc Mater. 2020;9(2):e1901154. [24] LI N, LI X, MA Y, et al. Swim bladder as an alternative biomaterial for bioprosthetic valves. Biomater Sci. 2021;9(24):8356-8365. [25] MEEZAN E, HJELLE JT, BRENDEL K, et al. A simple, versatile, nondisruptive method for the isolation of morphologically and chemically pure basement membranes from several tissues. Life Sci. 1975;17(11):1721-1732. [26] CRAPO PM, GILBERT TW, BADYLAK SF. An overview of tissue and whole organ decellularization processes. Biomaterials. 2011;32(12):3233-3243. [27] TAGHIZADEH B, GHAVAMI L, DERAKHSHANKHAH H, et al. Biomaterials in Valvular Heart Diseases. Front Bioeng Biotechnol. 2020;8:529244. [28] WALETZKO J, DAU M, SEYFARTH A, et al. Devitalizing Effect of High Hydrostatic Pressure on Human Cells-Influence on Cell Death in Osteoblasts and Chondrocytes. Int J Mol Sci. 2020;21(11):3836. [29] NASO F, GANDAGLIA A. Different approaches to heart valve decellularization: A comprehensive overview of the past 30 years. Xenotransplantation. 2018;25(1). doi: 10.1111/xen.12354. [30] ROTHRAUFF BB, TUAN RS. Decellularized bone extracellular matrix in skeletal tissue engineering. Biochem Soc Trans. 2020;48(3):755-764. [31] LIU X, CAI Y, XIA C, et al. An innovative method to obtain porous porcine aorta scaffolds for tissue engineering. Artif Organs. 2019;43(12):1162-1169. [32] RAMM R, GOECKE T, THEODORIDIS K, et al. Decellularization combined with enzymatic removal of N‐linked glycans and residual DNA reduces inflammatory response and improves performance of porcine xenogeneic pulmonary heart valves in an ovine in vivo model. Xenotransplantation. 2020;27(2):e12571. [33] GIL-RAMíREZ A, ROSMARK O, SPéGEL P, et al. Pressurized carbon dioxide as a potential tool for decellularization of pulmonary arteries for transplant purposes. Sci Rep. 2020;10(1):4031. [34] TRANTINA-YATES AE, HUMAN P, ZILLA P. Detoxification on top of enhanced, diamine-extended glutaraldehyde fixation significantly reduces bioprosthetic root calcification in the sheep model. J Heart Valve Dis. 2003;12(1):93-100; discussion -1. [35] FLAMENG W, HERMANS H, VERBEKEN E, et al. A randomized assessment of an advanced tissue preservation technology in the juvenile sheep model. J Thorac Cardiovasc Surg. 2015;149(1):340-345. [36] SHANG H, CLAESSENS SM, TIAN B, et al. Aldehyde reduction in a novel pericardial tissue reduces calcification using rabbit intramuscular model. J Mater Sci Mater Med. 2017;28(1):16. [37] XI T, MA J, TIAN W, et al. Prevention of tissue calcification on bioprosthetic heart valve by using epoxy compounds: a study of calcification tests in vitro and in vivo. J Biomed Mater Res. 1992;26(9): 1241-1251. [38] LI J, REN N, QIU J, et al. Carbodiimide crosslinked collagen from porcine dermal matrix for high-strength tissue engineering scaffold. Int J Biol Macromol. 2013;61:69-74. [39] ZEEMAN R, DIJKSTRA PJ, VAN WACHEM PB, et al. Successive epoxy and carbodiimide cross-linking of dermal sheep collagen. Biomaterials. 1999;20(10):921-931. [40] EVERAERTS F, TORRIANNI M, HENDRIKS M, et al. Biomechanical properties of carbodiimide crosslinked collagen: influence of the formation of ester crosslinks. J Biomed Mater Res A. 2008;85(2):547-555. [41] LEI Y, JIN W, LUO R, et al. Bioprosthetic heart valves’ structural integrity improvement through exogenous amino donor treatments. J Mater Res. 2018;33(17):2576-2585. [42] LIM HG, KIM SH, CHOI SY, et al. Anticalcification effects of decellularization, solvent, and detoxification treatment for genipin and glutaraldehyde fixation of bovine pericardium. Eur J Cardiothorac Surg. 2012;41(2):383-390. [43] CHANG Y, TSAI CC, LIANG HC, et al. In vivo evaluation of cellular and acellular bovine pericardia fixed with a naturally occurring crosslinking agent (genipin). Biomaterials. 2002;23(12):2447-2457. [44] 郑剑莹. EDC和京尼平交联对壳聚糖/硅酸钙/丝素蛋白支架影响的研究[D].长春:吉林大学,2021. [45] 张佳颖,索海瑞,徐铭恩,等.京尼平交联3D打印胶原/壳聚糖支架的表征[J].中国组织工程研究,2022,26(28):4477-4482. [46] LIU J, LI B, JING H, et al. Curcumin-crosslinked acellular bovine pericardium for the application of calcification inhibition heart valves. Biomed Mater. 2020;15(4):045002. [47] FANG G, CHEN S, HUANG Q, et al. Curcumin suppresses cardiac fibroblasts activities by regulating the proliferation and cell cycle via the inhibition of the p38 MAPK/ERK signaling pathway. Mol Med Rep, 2018;18(2):1433-1438. [48] CIFTCI O, TURKMEN NB, TASLIDERE A. Curcumin protects heart tissue against irinotecan-induced damage in terms of cytokine level alterations, oxidative stress, and histological damage in rats. Naunyn Schmiedebergs Arch Pharmacol. 2018;391(8):783-791. [49] BALALAIE A, REZVANI MB, MOHAMMADI BASIR M. Dual function of proanthocyanidins as both MMP inhibitor and crosslinker in dentin biomodification: A literature review. Dent Mater J. 2017;37(2):173-182. [50] SIMIONESCU A, SIMIONESCU DT, DEAC RFP. Matrix metalloproteinases in the pathology of natural and bioprosthetic cardiac valves. Cardiovasc Pathol. 1996;5(6):323-332. [51] TANIMURA S, KADOMOTO R, TANAKA T, et al. Suppression of tumor cell invasiveness by hydrolyzable tannins (plant polyphenols) via the inhibition of matrix metalloproteinase-2/-9 activity. Biochem Biophys Res Commun. 2005;330(4):1306-1313. [52] ISENBURG JC, SIMIONESCU DT, VYAVAHARE NR. Tannic acid treatment enhances biostability and reduces calcification of glutaraldehyde fixed aortic wall. Biomaterials. 2005;26(11):1237-1245. [53] 刘其静.单宁酸鞣制对牛心包材料的改性研究[D].武汉:华中农业大学,2019. [54] 易亮,宋明哲,吴忠仕,等.一种复合交联生物瓣膜及其制备方法, CN113425910A[P/OL]. [55] GHOUAS H, HADDOU B, KAMECHE M, et al. Removal of Tannic Acid From Aqueous Solution by Cloud Point Extraction and Investigation of Surfactant Regeneration by Microemulsion Extraction. J Surfact Deterg. 2015;19. doi: 10.1007/s11743-015-1764-9 [56] 王云兵,雷洋,訾振军.一种采用酶交联和茶多酚组合联用处理生物瓣膜的方法, CN110841110B [P/OL]. [57] BARTOLI-LEONARD F, AIKAWA E. Heart Valve Disease: Challenges and New Opportunities. Front Cardiovasc Med. 2020;7:602271. [58] KOSTYUNIN AE, YUZHALIN AE, REZVOVA MA, et al. Degeneration of Bioprosthetic Heart Valves: Update 2020. J Am Heart Assoc. 2020; 9(19):e018506. [59] LYU N, DU Z, QIU H, et al. Mimicking the Nitric Oxide-Releasing and Glycocalyx Functions of Endothelium on Vascular Stent Surfaces. Adv Sci (Weinh). 2020;7(21):2002330. [60] GULDNER NW, JASMUND I, ZIMMERMANN H, et al. Detoxification and endothelialization of glutaraldehyde-fixed bovine pericardium with titanium coating: a new technology for cardiovascular tissue engineering. Circulation. 2009;119(12):1653-1660. [61] ASSMANN A, DELFS C, MUNAKATA H, et al. Acceleration of autologous in vivo recellularization of decellularized aortic conduits by fibronectin surface coating. Biomaterials. 2013;34(25):6015-6026. [62] MARINVAL N, MORENC M, LABOUR MN, et al. Fucoidan/VEGF-based surface modification of decellularized pulmonary heart valve improves the antithrombotic and re-endothelialization potential of bioprostheses. Biomaterials. 2018;172:14-29. [63] YE X, HU X, WANG H, et al. Polyelectrolyte multilayer film on decellularized porcine aortic valve can reduce the adhesion of blood cells without affecting the growth of human circulating progenitor cells. Acta Biomater. 2012;8(3):1057-1067. [64] CHENG S, LIU X, QIAN Y, et al. Double-Network Hydrogel Armored Decellularized Porcine Pericardium as Durable Bioprosthetic Heart Valves. Adv Healthc Mater. 2022;11(8):e2102059. [65] WANG Y, MA B, LIU K, et al. A multi-in-one strategy with glucose-triggered long-term antithrombogenicity and sequentially enhanced endothelialization for biological valve leaflets. Biomaterials. 2021;275: 120981. [66] FANG C, DENG Z, CAO G, et al. Co–Ferrocene MOF/Glucose Oxidase as Cascade Nanozyme for Effective Tumor Therapy. Adv Funct Mater. 2020;30(16):1910085. [67] SOLOMONICA A, CHOUDHURY T, BAGUR R. Newer-generation of Edwards transcatheter aortic valve systems: SAPIEN 3, Centera, and SAPIEN 3 Ultra. Expert Rev Med Devices. 2019;16(2):81-87. [68] SCHäFER U, SCHIRMER J, SCHOFER N, et al. First in human implantation of a novel transfemoral self-expanding transcatheter heart valve to treat pure aortic regurgitation. EuroIntervention. 2017;13(11):1296-1299. [69] NISHIMURA RA, OTTO CM, BONOW RO, et al. 2014 AHA/ACC guideline for the management of patients with valvular heart disease: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Thorac Cardiovasc Surg. 2014;148(1): e1-e132. [70] WANG S, MENG X, LUO Z, et al. Transapical Beating-Heart Mitral Valve Repair Using a Novel Neochord Implantation System. Ann Thorac Surg. 2018;106(5):e265-e267. [71] 蒲朝霞,刘先宝,林心平,等.经食管超声心动图引导DragonFly系统缘对缘修复治疗三尖瓣反流的初步经验[J].中华超声影像学杂志,2021,30(10):843-847. [72] LU FL, YE MM, ZHAO AM, et al. First-in-Man Experience of Transcatheter Tricuspid Valve Replacement With LuX-Valve in High-Risk Tricuspid Regurgitation Patients. JACC Cardiovasc Interv. 2020;13(13):1614-1616. [73] 陈思,顾君君,杨帆,等.LuX-Valve系统经导管三尖瓣置换26例近期疗效分析[J].中国心血管病研究,2022,20(2):104-107. [74] BLACKMAN DJ, SARAF S, MACCARTHY PA, et al. Long-Term Durability of Transcatheter Aortic Valve Prostheses. J Am Coll Cardiol. 2019;73(5): 537-545. [75] DURAND E, SOKOLOFF A, URENA-ALCAZAR M, et al. Assessment of Long-Term Structural Deterioration of Transcatheter Aortic Bioprosthetic Valves Using the New European Definition. Circ Cardiovasc Interv. 2019;12(4):e007597. [76] SAWAYA F, JORGENSEN TH, SONDERGAARD L, et al. Transcatheter Bioprosthetic Aortic Valve Dysfunction: What We Know So Far. Front Cardiovasc Med. 2019;6:145. [77] 林振乾,黄琼,黄海伦,等.经导管主动脉瓣置换术并发症的研究进展[J].中国心血管病研究,2021,19(11):1000-1005. [78] KOLTE D, VLAHAKES GJ, PALACIOS IF, et al. Transcatheter Versus Surgical Aortic Valve Replacement in Low-Risk Patients. J Am Coll Cardiol. 2019; 74(12):1532-1540. [79] THOURANI VH, EDELMAN JJ, MEDURI CU. TAVR in TAVR. J Am Coll Cardiol. 2020;75(16):1894-1896. |
| [1] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [2] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [3] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [4] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [5] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [6] | Xu Cong, Zhao He, Sun Yan. Regeneration of facial nerve injury repaired by biomaterial nerve conduits [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1089-1095. |
| [7] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [8] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [9] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [10] | Tao Xin, Xu Yi, Song Zhiwen, Liu Jinbo. Hippo signaling pathway in the regulation of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 619-625. |
| [11] | Li Shihao, Li Qi, Li Zhen, Zhang Yuanyuan, Liu Miaomiao, Ouyang Yi, Xu Weiguo. Plantar pressure and gait analysis in patients with anterior cruciate ligament injury and reconstruction [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 626-631. |
| [12] | Liu Huan, Li Han, Ma Yunhao, Zhong Weijian, Ma Guowu. Osteogenic capacity of partially demineralized dentin particles in the maxillary sinus lift [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 354-359. |
| [13] | Jiang Haifang, Liu Rong, Hu Peng, Chen Wei, Wei Zairong, Yang Chenglan, Nie Kaiyu. Application of 3D printing technology in the precise and personalized treatment of cleft lip and palate [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 413-419. |
| [14] | Zhou Jie, Pei Xibo, Wan Qianbing. Advances and biological application of asymmetric dressings [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 434-440. |
| [15] | Chen Jingqiao, Li Ying, Meng Maohua, Xu Xingxing, Wang Qinying, Wang Huan, Lu Jing, Shu Jiayu, Dong Qiang. Research progress in platelet-rich fibrin in stomatology [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 441-446. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||