Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (7): 1089-1095.doi: 10.12307/2023.104
Previous Articles Next Articles
Regeneration of facial nerve injury repaired by biomaterial nerve conduits
Xu Cong1, 2, 3, Zhao He1, 2, 3, Sun Yan1, 2, 3
- 1Second Clinical Medical College, Binzhou Medical University, Yantai 264000, Shandong Province, China; 2Department of Otorhinolaryngology, Head and Neck Surgery, Yantai Yuhuangding Hospital, Qingdao University, Yantai 264000, Shandong Province, China; 3Shandong Provincial Clinical Research Center for Otorhinolaryngologic Diseases, Yantai 264000, Shandong Province, China
-
Received:2022-02-24Accepted:2022-04-23Online:2023-03-08Published:2022-07-19 -
Contact:Sun Yan, Chief physician, Second Clinical Medical College, Binzhou Medical University, Yantai 264000, Shandong Province, China; Department of Otorhinolaryngology, Head and Neck Surgery, Yantai Yuhuangding Hospital, Qingdao University, Yantai 264000, Shandong Province, China; Shandong Provincial Clinical Research Center for Otorhinolaryngologic Diseases, Yantai 264000, Shandong Province, China -
About author:Xu Cong, Master candidate, Second Clinical Medical College, Binzhou Medical University, Yantai 264000, Shandong Province, China; Department of Otorhinolaryngology, Head and Neck Surgery, Yantai Yuhuangding Hospital, Qingdao University, Yantai 264000, Shandong Province, China; Shandong Provincial Clinical Research Center for Otorhinolaryngologic Diseases, Yantai 264000, Shandong Province, China -
Supported by:Natural Science Foundation of Shandong Province, No. ZR2021MH378 (to SY); Yantai Science and Technology Program Project, No. 2021MSGY046 (to SY)
CLC Number:
Cite this article
Xu Cong, Zhao He, Sun Yan. Regeneration of facial nerve injury repaired by biomaterial nerve conduits[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1089-1095.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
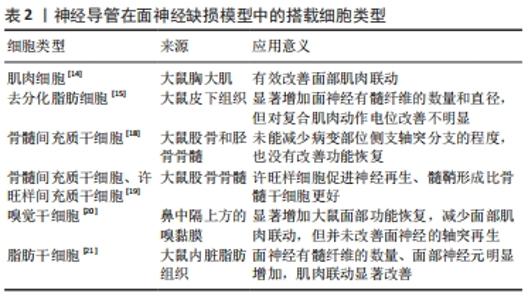
神经导管的体内实验动物模型多采用大鼠和兔,因为这两种动物的面神经直径更易与导管匹配,而小鼠的面神经过细,对导管制备及手术能力要求较高,因此小鼠使用较少。面神经颊支易暴露、长度长,是最常用的造模神经;而面神经干位于耳后,周围有颈外静脉,暴露困难,因此较少用于造模。导管连接神经导管搭载营养因子或细胞是常见且有效的应用模式,既能诱导神经定向生长,还能释放因子加快轴突再生,增加髓鞘厚度。另一种常见应用模式是中空导管中填充促生长凝胶,这种模式对凝胶要求较高,凝胶需能降解吸收,且降解时间不能过长,否则会阻碍神经生长。 有研究者对搭载细胞的神经导管在面神经缺损模型中的修复情况进行了广泛研究,可促进周围神经再生并缩短恢复时间[10,12,18-21],见表2。 "
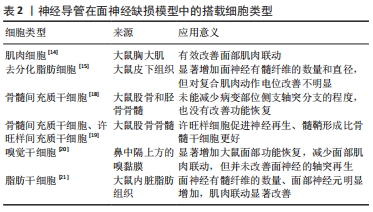
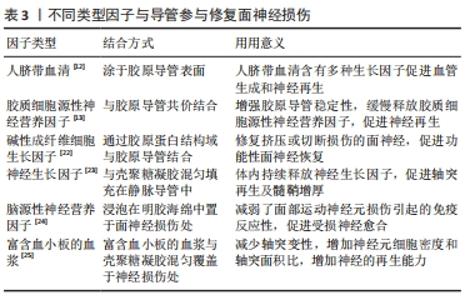
间充质细胞及脂肪干细胞取材方便,增殖迅速,可以培养分化成许旺细胞,为神经纤维提供支持和引导,还可以分泌神经营养因子,包括神经生长因子、脑源性神经营养因子和神经胶质细胞源性神经营养因子等,促进面神经生长,防止神经元死亡,因此应用较为广泛。但是去分化脂肪细胞对神经电传导无明显改善。此外,如嗅觉干细胞和肌细胞等特殊细胞同样对面神经修复有促进作用。肌细胞周围的基底板与神经管外的基底板类似,因此被用于修复受损神经,但仅在肌细胞与静脉导管结合时效果较好,应用较为局限。嗅觉干细胞取材困难,目前同样尚未广泛应用。 神经营养因子可促进轴突从近端到远端的再生以及调节轴突分支,减少面部肌肉联动。同时还可以为远端神经残端的细胞提供营养因子,并保持再生轴突营养充足,直到远端轴突恢复营养供应。胶质源性神经营养因子、脑源性神经营养因子可以促进感觉和运动神经元的存活,将其递送至面神受损处可增强损伤后的再生,但是胶质源性神经营养因子作用时间较短,只有包裹在神经导管中缓慢释放才会对面神经再生有促进意义。人脐带血清及富含血小板的血浆含有多种生长因子,促进血管生成,有利于氧气及营养物质的供应,促进神经再生。碱性成纤维细胞生长因子作为成纤维细胞和血管内皮细胞的调节剂,可以增强神经损伤区域的血管化和细胞化,对神经再生至关重要。多种类型因子可与导管参与修复面神经损伤[12-13,22-25],见表3。 "
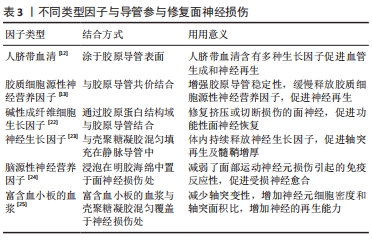

2.2 脱细胞材料 脱细胞材料是指通过物理、化学和生物等方法将人体或动物的组织、器官去除细胞而保留其活性成分的基质材料,能够诱导并促进细胞的黏附、增殖、分化及组织形成[26]。许多人体或动物组织、器官已被脱细胞用于修复面神经,例如肌肉和动静脉血管等。 2.2.1 动脉脱细胞材料 在1891年,有学者进行了动脉移植物首次用于修复周围神经的动物实验,但单纯动脉导管修复面神经效果不理想[27]。SUN等[21]将脂肪干细胞填充动脉导管,桥接神经远端及近端,大鼠再生轴突纤维数量及髓鞘厚度接近自体移植神经,胡须运动功能基本恢复。然而,脂肪干细胞可导致有髓纤维过度增长,这与其丰富的营养功能有关[18]。许多采用舌下-面神经吻合术修复面神经的患者,会出现面部肌肉的异常同步运动,有研究取大鼠腹主动脉及其分叉做成“Y”型导管,舌下神经近端穿入“Y”型导管的长支,将颧神经和颊神经分支的远端残端插入“Y”型导管的短支中,减少面神经轴突侧支生长,避免了面部肌肉同步运动[11,28]。 2.2.2 静脉脱细胞材料 静脉移植物在1909年首次尝试用于桥接人类周围神经缺损,成功地修复了1例男性患者的正中神经,取得了满意的效果,随后开始用于修复面神经损伤。静脉导管的优点十分明显:①静脉的组织成分与神经组织相似,引发的炎症反应较少[29];②静脉壁可以阻挡瘢痕生长,并且具有良好的孔隙率,允许营养物质和氧气扩散到损伤部位支持细胞修复[30];③静脉内皮的基膜富含层粘连蛋白和中层,外膜主要由胶原组成,为轴突生长提供所需的微环境[31] ;④静脉移植物供应充足,成本低,损伤程度相对较小。有研究分析发现静脉移植在神经缺损小于3 cm时是有效的,神经缺损小于1 cm时,静脉移植物不易塌陷,更利于神经生长[32]。然而,有研究通过向大鼠面神经分支断端注入逆行神经追踪剂,证明了相比单纯缝合,自体静脉套扎不能改善再生轴突的质量[33]。还有研究结果显示,神经营养因子在控制神经细胞的存活、迁移、增殖和分化方面发挥着至关重要的作用,是治疗周围神经损伤的潜在药物[34]。静脉导管植入神经营养因子、间充质干细胞和嗅觉干细胞等,加速了面神经愈合再生[20,34-37]。 2.2.3 肌肉脱细胞材料 移植自体横纹肌恢复神经损伤的方法早在20世纪40年代就有记载,其优点在于肌肉细胞周围的基板和神经管外的基板具有相似性[38],新鲜的肌肉组织还可以为移植神经提供充足营养[39]。有研究采用肌肉-静脉结合的方式,将大鼠胸大肌置入静脉管道内,与面神经两侧断端缝合,以此促进单神经支配程度,改善面部肌肉的协调活动[16]。但肌肉神经导管仅适用于修复短的神经间隙,且容易发生神经瘤,因此单纯的肌肉神经导管并不多见[40]。 2.3 可降解材料 可降解材料主要是指一定时间内可于体内降解的生物材料,主要包括胶原蛋白、聚乙醇酸、聚己内酯、丝蛋白、壳聚糖和石墨烯等,这些材料具有优越的机械性能及生物相容性,且由于技术工艺的进步,在一定条件下均可达到体内降解。 2.3.1 胶原蛋白 是制作导管最常用的一种天然生物材料,体内普遍存在,具有对神经再生至关重要的细胞黏附性[41]、生物相容性强,一定条件下可体内降解无需二次取出[42]。目前胶原蛋白神经导管技术非常成熟,在修复短间隙神经缺损中取得了较好的结果,已批准应用于临床。普通胶原导管机械性能较差,体内长时间存在会出现溶胀现象,中空管道会变得狭窄甚至闭死,阻碍轴突再生。纵行多通道结构可以极大程度避免这种溶胀现象,同时为再生轴突提供固定通道,避免轴突分散异常支配周围神经和肌肉[41]。将各类细胞因子加载到神经导管上可以促进损伤部位细胞的存活和迁移,受损神经恢复效果明显。胶质细胞源性神经营养因子、碱性成纤维细胞生长因子和睫状神经营养因子等营养因子偶联胶原导管[13,22,43],以缓释的方式持续促进近端神经纤维生长,维持远端神经营养供应,再生轴突数量明显增加。加入肝素的胶原导管固定神经营养因子更加牢固,作用时间更长[44-45]。人脐血清含有多种生长因子,表层包裹人脐血清的导管为受损神经提供了丰富的营养、适宜微环境,加快神经纤维恢复速度,有利于面部肌肉震颤恢复[12]。GROSHEVA等[18]发现胶原导管搭载骨髓间充质干细胞没有改善面神经有髓纤维的数量、直径以及功能修复。WATANABE等[46]向胶原凝胶中加入脂肪干细胞与许旺细胞,其面瘫修复效果无明显差别。而基质血管成分比脂肪干细胞更加实用,修复面神经能力更强[15]。 2.3.2 聚乙醇酸 又称为聚羟基乙酸,易水解,酸碱作用下可降解为无毒性的CO2和H2O,其渗透性允许营养物质和氧气的交换,为神经再生创造了理想的微环境。Neurotube导管是第一种应用于修复面神经的聚乙醇酸神经导管,6个月内可完全水解[47]。有研究对4例周围神经瘤切除后聚乙醇酸导管重建的患者进行评估,静息性疼痛、神经根性疼痛和感觉障碍在术后均得到不同程度的改善,但此4例病例中并无面神经瘤患者[48]。有研究对2例聚乙醇酸导管修复面神经额支损伤患者进行追踪随访发现,尽管患者出现了一些肌肉萎缩,但额部肌肉运动的改善十分明显[49]。还有研究比较了聚乙醇酸导管和胶原导管的修复能力,修复坐骨神经10 mm缺损时,聚乙醇酸导管组再生轴突的密度和组织化程度更低[50],尽管并无实验对比2种导管修复面神经缺损的效果,该作者认为该实验具有一定的参考价值。此外,管壁内外涂有胶原蛋白的聚乙醇酸导管搭载去分化脂肪细胞能明显促进面神经再生,胡须摆动恢复良好[15]。 2.3.3 聚己内酯 是一种由ε-己内酯聚合而成的高分子有机聚合物,具有良好降解率、优异的机械性能优异、高细胞黏附性,生物相容性好,可以减少瘢痕形成[12,51-52]。复合聚己内酯导管表面覆盖聚吡咯后具有优异的导电性,增强神经细胞间电信号传导,加速神经再生[53]。ASSAF等[54]发现聚己内酯导管内加入碳纳米颗粒和石墨烯纳米颗粒会具有更好的细胞黏附性,再生神经有髓轴突的数量增加了2倍,对靶肌肉有一定保护作用。SARHANE等[55]采用静电纺丝技术研制的聚己内酯导管能促使巨噬细胞向M2型极化,M2巨噬细胞通过产生促进基质重塑和血管生成的抗炎细胞因子,使微环境支持组织修复,有利于轴突增殖、迁移及髓鞘形成。加载三维许旺细胞基质或纤维蛋白基质的聚己内酯导管能有效促进轴突再生,许旺细胞基质或纤维蛋白基质中加入白血病抑制因子能显著预防面神经神经过度分化[56]。 2.3.4 丝蛋白 又称为丝素,是一种天然高分子材料,主要来源于蚕丝和蜘蛛丝,具有独特的生物医学特性,一直被用于伤口愈合和外科缝合。加入丝素的神经导管具有更好的物理性能、生物相容性和生物降解性,可避免神经瘢痕形成[19,57]。研究表明,种植在掺杂有丝素的纳米纤维上,成纤维细胞分泌的血管内皮生长因子会增加 7倍以上,进而促进神经血管生成,为神经再生提供充足的营养物质[58]。有研究在丝素纳米纤维表面雕刻纵向排列微通道,使轴突沿着排列的纤维方向穿过导管管腔,表明丝素纳米纤维具有促进神经突起生长和控制定向突起延伸的能力[59]。有研究证明了具有抗氧化活性的黑色素与丝素组合促进神经元生长和再生,但体内修复效果仍需进一步验证[60]。 2.3.5 壳聚糖 是一种自然生成的甲壳素衍生物,具有更高的生物降解性和生物相容性,在体内可自动降解为可吸收的氨基葡萄糖[14]。壳聚糖可通过降低caspase-3、caspase-9和Bax 活性和增加 Bcl-2 活性,减少许旺细胞凋亡[61];下调许旺细胞的miR-327来刺激CCL2表达,增加受损区域的巨噬细胞聚集,积极促进神经愈合[62]。面神经或其他周围组织造成损伤,可能导致面神经周围瘢痕形成,瘢痕持续压迫可能导致轴突和髓鞘变性,影响神经血供,从而影响神经功能的恢复[63]。壳聚糖的高度脱乙酰化可以抑制产生胶原蛋白的成纤维细胞的收缩,结合神经溶解剂-透明质酸,可有效减少瘢痕形成[14]。神经生长因子缓释微球植入壳聚糖导管修复面神经颊支10 mm缺损,术后90 d发现神经传导速度和波幅以及髓鞘厚度明显高于神经生长因子组[10]。钾离子阻滞剂4-氨基吡啶应用于神经挤压伤可增加髓鞘再生和神经传导速度,有研究将4-氨基吡啶加入埃洛石/壳聚糖溶液中,制备成复合神经导管,促进神经髓鞘增厚,且肝脏未发现药物累积和脂肪变性等不良影响[64]。血小板含有多种生长因子,如血小板衍生生长因子、转化生长因子B和血管内皮生长因子,壳聚糖凝胶浸附富含血小板的血浆极大促进神经愈合再生[25]。 2.3.6 石墨烯 是一种新型的碳纳米材料,具有比表面积大、电子迁移率高、延展性好等特点,对生物传感、诊断、药物输送、抗菌活性和癌症治疗。石墨烯还具有很强的细胞黏附力以及促进细胞增殖的特点,种植在石墨烯表面的神经干细胞5 d后便长出轴突,且排列整齐[65]。氧化石墨烯是石墨烯经过氧化后的产物,不仅保留了石墨烯的优越特性,还具备了丰富的表面官能团,可以使性质更加活泼,毒性更低。有学者观察了加入氧化石墨烯的明胶/聚己内酯纳米纤维支架培养的许旺细胞,发现其Sox2基因表达水平明显升高,这表明氧化石墨烯可提高许旺细胞中Sox2基因表达水平,将许旺细胞重新编程为侵袭性间充质样细胞,进而促进大鼠面神经再生和功能恢复[17,66]。氧化石墨烯是一种天然抗氧化剂,可以通过减少巨噬细胞内的活性氧来减轻巨噬细胞(M1)的炎症极化,同时其特殊的物理和化学性质使其可以调控巨噬细胞表型,将M1巨噬细胞极化为促愈合的M2巨噬细胞,此类细胞已证明可释放营养因子促进受损神经再生[45,67-68]。"

| [1] GAUDIN R, KNIPFER C, HENNINGSEN A, et al. Approaches to peripheral nerve repair: generations of biomaterial conduits yielding to replacing autologous nerve grafts in craniomaxillofacial surgery. Biomed Res Int. 2016;2016:3856262. [2] SUN Y, LIU L, HAN Y, et al. The role of great auricular-facial nerve neurorrhaphy in facial nerve damage. Int J Clin Exp Med. 2015;8(8): 12970-12976. [3] CHRZASZCZ P, DERBISZ K, SUSZYNSKI K, et al. Application of peripheral nerve conduits in clinical practice: a literature review. Neurol Neurochir Pol. 2018;52(4):427-435. [4] JANDALI D, REVENAUGH PC. Facial reanimation:an update on nerve transfers in facial paralysis. Curr Opin Otolaryngol Head Neck Surg. 2019;27(4):231-236. [5] 李煜,於子卫.神经干细胞与许旺细胞联合应用于神经损伤修复的研究进展[J].临床耳鼻咽喉头颈外科杂志,2014,28(13):1006-1009. [6] LI X, YANG W, XIE H, et al. CNT/sericin conductive nerve guidance conduit promotes functional recovery of transected peripheral nerve injury in a rat model. ACS Appl Mater Interfaces. 2020;12(33):36860-36872. [7] WANG J, CHENG Y, WANG H, et al. Biomimetic and hierarchical nerve conduits from multifunctional nanofibers for guided peripheral nerve regeneration. Acta Biomater. 2020;117:180-191. [8] BENGUR FB, STOY C, BINKO MA, et al. Facial nerve repair: bioengineering approaches in preclinical models. Tissue Eng Part B Rev. 2022;28(2):364-378. [9] HU A, ZUO B, ZHANG F, et al. Evaluation of electronspun silk fibroin-based transplants used for facial nerve repair. Otol Neurotol. 2013; 34(2):311-318. [10] LIU H, WEN W, HU M, et al. Chitosan conduits combined with nerve growth factor microspheres repair facial nerve defects. Neural Regen Res. 2013;8(33):3139-3147. [11] KAYA Y, OZSOY U, TURHAN M, et al. Hypoglossal-facial nerve reconstruction using a Y-tube-conduit reduces aberrant synkinetic movements of the orbicularis oculi and vibrissal muscles in rats. Biomed Res Int. 2014. doi:10.1155/2014/543020. [12] JANG CH, LEE H, KIM M, et al. Effect of polycaprolactone/collagen/hUCS microfiber nerve conduit on facial nerve regeneration. Int J Biol Macromol. 2016;93(Pt B):1575-1582. [13] MA F, XU F, LI R, et al. Sustained delivery of glial cell-derived neurotrophic factors in collagen conduits for facial nerve regeneration. Acta Biomater. 2018;69:146-155. [14] LIU H, HUANG H, BI W, et al. Effect of chitosan combined with hyaluronate on promoting the recovery of postoperative facial nerve regeneration and function in rabbits. Exp Ther Med. 2018;16(2):739-745. [15] FUJIMAKI H, MATSUMINE H, OSAKI H, et al. Corrigendum to “Dedifferentiated fat cells in polyglycolic acid-collagen nerve conduits promote rat facial nerve regeneration” [Regen Ther 11 (2019) 240-248]. Regen Ther. 2020;15:35-43. [16] MANTHOU ME, GENCHEVA D, SINIS N, et al. Facial nerve repair by muscle-vein conduit in rats:functional recovery and muscle reinnervation. Tissue Eng Part A. 2021;27(5-6):351-361. [17] AGHAJANIAN S, TAGHI DOULABI A, AKHBARI M, et al. Facial nerve regeneration using silicone conduits filled with ammonia-functionalized graphene oxide and frankincense-embedded hydrogel. Inflamm Regen. 2021;41(1):13. [18] GROSHEVA M, GUNTINAS-LICHIUS O, ARNHOLD S, et al. Bone marrow-derived mesenchymal stem cell transplantation does not improve quality of muscle reinnervation or recovery of motor function after facial nerve transection in rats. Biol Chem. 2008;389(7):873-888. [19] COSTA HJ, BENTO RF, SALOMONE R, et al. Mesenchymal bone marrow stem cells within polyglycolic acid tube observed in vivo after six weeks enhance facial nerve regeneration. Brain Res. 2013;1510:10-21. [20] BENSE F, MONTAVA M, DUCLOS C, et al. Syngeneic transplantation of rat olfactory stem cells in a vein conduit improves facial movements and reduces synkinesis after facial nerve injury. Plast Reconstr Surg. 2020;146(6):1295-1305. [21] SUN F, ZHOU K, MI WJ, et al. Repair of facial nerve defects with decellularized artery allografts containing autologous adipose-derived stem cells in a rat model. Neurosci Lett. 2011;499(2):104-108. [22] WANG P, ZHAO H, YAO Y, et al. Repair of facial nerve crush injury in rabbits using collagen plus basic fibroblast growth factor. J Biomed Mater Res A. 2020;108(6):1329-1337. [23] CHAO X, XU L, LI J, et al. Facilitation of facial nerve regeneration using chitosan-beta-glycerophosphate-nerve growth factor hydrogel. Acta Otolaryngol. 2016;136(6):585-591. [24] SERPE CJ, BYRAM SC, SANDERS VM, et al. Brain-derived neurotrophic factor supports facial motoneuron survival after facial nerve transection in immunodeficient mice. Brain Behav Immun. 2005;19(2):173-180. [25] SAHIN MM, CAYONU M, DINC SK, et al. Effects of chitosan and platelet-rich plasma on facial nerve regeneration in an animal model. Eur Arch Otorhinolaryngol. 2022;279(2):987-994. [26] 杨美蓉,潘博,赵博.整形外科的脱细胞基质材料:研究进展和临床应用的有机结合[J].中国组织工程研究,2018,22(26):4259-4264. [27] BENDELLA H, RINK S, MANTHOU M, et al. Effect of surgically guided axonal regrowth into a 3-way-conduit (isogeneic trifurcated aorta) on functional recovery after facial-nerve reconstruction: experimental study in rats. Restor Neurol Neurosci. 2019;37(2):181-196. [28] OZSOY U, DEMIREL BM, HIZAY A, et al. Hypoglossal-facial anastomosis (HFA) over a 10 mm gap bridged by a Y-tube-conduit enhances neurite regrowth and reduces collateral axonal branching at the lesion site. Restor Neurol Neurosci. 2011;29(4):227-242. [29] MARCOCCIO I, VIGASIO A. Muscle-in-vein nerve guide for secondary reconstruction in digital nerve lesions. J Hand Surg Am. 2010;35(9): 1418-1426. [30] TSENG CY, HU G, AMBRON RE, et al. Histologic analysis of Schwann cell migration and peripheral nerve regeneration in the autogenous venous nerve conduit (AVNC). J Reconstr Microsurg. 2003;19(5):331-340. [31] WANG KK, COSTAS PD, BRYAN DJ, et al. Inside-out vein graft repair compared with nerve grafting for nerve regeneration in rats. Microsurgery. 1995;16(2):65-70. [32] PAPROTTKA FJ, WOLF P, HARDER Y, et al. Sensory recovery outcome after digital nerve repair in relation to different reconstructive techniques:meta-analysis and systematic review. Plast Surg Int. 2013.doi:10.1155/2013/704589. [33] CHEN P, KNOX CJ, YAO L, et al. The effects of venous ensheathment on facial nerve repair in the rat. Laryngoscope. 2017;127(7):1558-1564. [34] SABONGI RG, FERNANDES M, DOS SANTOS JB. Peripheral nerve regeneration with conduits:use of vein tubes. Neural Regen Res. 2015; 10(4):529-533. [35] BATIOGLU-KARAALTIN A, KARAALTIN MV, OZTEL ON, et al. Human olfactory stem cells for injured facial nerve reconstruction in a rat model. Head Neck. 2016;38 Suppl 1:E2011-E2020. [36] Han GH, Peng J, Liu P, et al. Therapeutic strategies for peripheral nerve injury: decellularized nerve conduits and Schwann cell transplantation. Neural Regen Res. 2019;14(8):1343-1351. [37] WANG X, LUO E, LI Y. Schwann-like mesenchymal stem cells within vein graft facilitate facial nerve regeneration and remyelination. Brain Res. 2011;1383:71-80. [38] FAWCELTT JW, KEYNES RJ. Muscle basal lamina: a new graft material for peripheral nerve repair. J Neurosurg. 1986;65(3):354-363. [39] 周振宇,蔡锦方.构建肌肉组织床修复股神经长段缺损两例[J].中国修复重建外科杂志,2020,34(10):1352-1353. [40] 陈勇,范林,付贞,等.神经导管支架修复外周神经损伤的研究与现状[J].中国组织工程研究,2017,21(30):4901-4907. [41] YAO L, BILLIAR KL, WINDEBANK AJ, et al. Multichanneled collagen conduits for peripheral nerve regeneration: design, fabrication, and characterization. Tissue Eng Part C Methods. 2010;16(6):1585-1596. [42] KITAHARA AK, NISHIMURA Y, SHIMIZU Y, et al. Facial nerve repair accomplished by the interposition of a collagen nerve guide. J Neurosurg. 2000;93(1):113-120. [43] MA F, WANG H, YANG X, et al. Controlled release of ciliary neurotrophic factor from bioactive nerve grafts promotes nerve regeneration in rats with facial nerve injuries. J Biomed Mater Res A. 2022;110(4):788-796. [44] MA F, ZHU T, XU F, et al. Neural stem/progenitor cells on collagen with anchored basic fibroblast growth factor as potential natural nerve conduits for facial nerve regeneration. Acta Biomater. 2017;50:188-197. [45] LONG Q, WU B, YANG Y, et al. Nerve guidance conduit promoted peripheral nerve regeneration in rats. Artif Organs. 2021;45(6):616-624. [46] WATANABE Y, SASAKI R, MATSULMINE H, et al. Undifferentiated and differentiated adipose-derived stem cells improve nerve regeneration in a rat model of facial nerve defect. J Tissue Eng Regen Med. 2017; 11(2):362-374. [47] NAVISSANO M, MALAN F, CARNINO R, et al. Neurotube for facial nerve repair. Microsurgery. 2005;25(4):268-271. [48] ZUKKAWA M, OSADA R, KIMURA T. Clinical outcome and ultrasonographic evaluation of treatment using polyglycolic acid-collagen tube for chronic neuropathic pain after peripheral nerve injury. J Orthop Sci. 2019;24(6):1064-1067. [49] NAKAMURA Y, TAKANARI K, EBISAWA K, et al. Repair of temporal branch of the facial nerve with novel polyglycolic acid-collagen tube:a case report of two cases. Nagoya J Med Sci. 2020;82(1):123-128. [50] WAITAYAWINYU T, PARISI DM, MILLER B, et al. A comparison of polyglycolic acid versus type 1 collagen bioabsorbable nerve conduits in a rat model: an alternative to autografting. J Hand Surg Am. 2007; 32(10):1521-1529. [51] MOHAMADI F, EBRAHIMI-BAROUGH S, REZA NOURANI M, et al. Electrospun nerve guide scaffold of poly (epsilon-caprolactone)/collagen/nanobioglass:an in vitro study in peripheral nerve tissue engineering. J Biomed Mater Res A. 2017;105(7):1960-1972. [52] LOPEZ J, XIN K, QUAN A, et al. Poly (epsilon-caprolactone) nanofiber wrap improves nerve regeneration and functional outcomes after delayed nerve repair. Plast Reconstr Surg. 2019;144(1):48e-57e. [53] SUN B, ZHOU Z, LI D, et al. Polypyrrole-coated poly (l-lactic acid-co-epsilon-caprolactone)/silk fibroin nanofibrous nerve guidance conduit induced nerve regeneration in rat. Mater Sci Eng C Mater Biol Appl. 2019;94:190-199. [54] ASSAF K, LEAL CV, DERAMI MS, et al. Sciatic nerve repair using poly (epsilon-caprolactone) tubular prosthesis associated with nanoparticles of carbon and graphene. Brain Behav. 2017;7(8):e00755. [55] SARHANE KA, IBRAHIM Z, MARTIN R, et al. Macroporous nanofiber wraps promote axonal regeneration and functional recovery in nerve repair by limiting fibrosis. Acta Biomater. 2019;88:332-345. [56] GALLA TJ, VEDECNIK SV, HALBGEWACHS J, et al. Fibrin/Schwann cell matrix in poly-epsilon-caprolactone conduits enhances guided nerve regeneration. Int J Artif Organs. 2004;27(2):127-136. [57] KUIHUA Z, CHUNYANG W, CUNYI F, et al. Aligned SF/P(LLA-CL)-blended nanofibers encapsulating nerve growth factor for peripheral nerve regeneration. J Biomed Mater Res A. 2014;102(8):2680-2691. [58] WANG C, JIA Y, YANG W, et al. Silk fibroin enhances peripheral nerve regeneration by improving vascularization within nerve conduits. J Biomed Mater Res A. 2018;106(7):2070-2077. [59] LI X,ZHANG Q, LUO Z, et al. Biofunctionalized silk fibroin nanofibers for directional and long neurite outgrowth. Biointerphases. 2019; 14(6):061001. [60] NUNE M, MANCHINEELLA S, GOVINDARAJU T, et al. Melanin incorporated electroactive and antioxidant silk fibroin nanofibrous scaffolds for nerve tissue engineering. Mater Sci Eng C Mater Biol Appl. 2019;94:17-25. [61] HE B, WU F, FAN L, et al. Carboxymethylated chitosan protects Schwann cells against hydrogen peroxide-induced apoptosis by inhibiting oxidative stress and mitochondria dependent pathway. Eur J Pharmacol. 2018;825:48-56. [62] ZHAO Y, WANG Y, GONG J, et al. Chitosan degradation products facilitate peripheral nerve regeneration by improving macrophage-constructed microenvironments. Biomaterials. 2017;134:64-77. [63] GAUDET AD, POPOVICH PG, RAMER MS. Wallerian degeneration:gaining perspective on inflammatory events after peripheral nerve injury. J Neuroinflammation. 2011;8:110. [64] MANOUKIAN OS, RUDRAIAH S, ARUL MR, et al. Biopolymer-nanotube nerve guidance conduit drug delivery for peripheral nerve regeneration: in vivo structural and functional assessment. Bioact Mater. 2021;6(9):2881-2893. [65] SOLANKI A, CHUENG ST, YIN PT, et al. Axonal alignment and enhanced neuronal differentiation of neural stem cells on graphene-nanoparticle hybrid structures. Adv Mater. 2013;25(38):5477-5482. [66] FANG X, GUO H, ZHANG W, et al. Reduced graphene oxide-GelMA-PCL hybrid nanofibers for peripheral nerve regeneration. J Mater Chem B. 2020;8(46):10593-10601. [67] HAN J, KIM YS, LIM MY, et al. Dual roles of graphene oxide to attenuate inflammation and elicit timely polarization of macrophage phenotypes for cardiac repair. ACS Nano. 2018;12(2):1959-1977. [68] MOKARRAM N,DYMANUS K, SRINIVASAN A, et al. Immunoengineering nerve repair. Proc Natl Acad Sci U S A. 2017;114(26):E5077-E5084. |
| [1] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [2] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [3] | Xue Ting, Zhang Xinri, Kong Xiaomei. Mesenchymal stem cell therapy for pneumoconiosis using nanomaterials combined with multi-modal molecular imaging [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1133-1140. |
| [4] | Liu Huan, Li Han, Ma Yunhao, Zhong Weijian, Ma Guowu. Osteogenic capacity of partially demineralized dentin particles in the maxillary sinus lift [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 354-359. |
| [5] | Li Rui, Liu Zhen, Guo Zige, Lu Ruijie, Wang Chen. Aspirin-loaded chitosan nanoparticles and polydopamine modified titanium sheets improve osteogenic differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 374-379. |
| [6] | Jiang Haifang, Liu Rong, Hu Peng, Chen Wei, Wei Zairong, Yang Chenglan, Nie Kaiyu. Application of 3D printing technology in the precise and personalized treatment of cleft lip and palate [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 413-419. |
| [7] | Ning Ziwen, Wang Xu, Shi Zhengliang, Qin Yihua, Wang Guoliang, Jia Di, Wang Yang, Li Yanlin. Meniscal injury repair methods for non-blood supply area [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 420-426. |
| [8] | Xie Pingjin, Luo Zhen, Lu Qigui, Guo Yanxing, Chen Qunqun, Li Feilong. Effect of ligustrazine and overexpression of miR-20b-5p on synovial, cartilage and subchondral bone angiogenesis in rats with early-stage knee osteoarthritis: a histological observation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 237-245. |
| [9] | Zhang Luyao, Yang Kang. Effects of different exercises on renal interstitial fibrosis in type 2 diabetic mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 200-207. |
| [10] | Niu Zihan, Yu Yang, Ai Jiang, Bu Panpan, Li Wenbo, Suriye·Reheman, Ma Shaolin. Total flavonoids of Hippophae rhamnoides L. interfere with the regression of hypertrophic scar tissue blocks in a rabbit ear model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 258-263. |
| [11] | Yang Yan, Wang Jingxian, Zhang Ronghong, Li Chen, Fan Anran, Cui Dongbing, Wu Shumei. Effects of conditioned media of different sources on the proliferation of human dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 49-53. |
| [12] | Lan Qian, Gu Yangcong, Xiao Xin, Bi Xueting, Li Na. Human periodontal ligament stem cells-derived exosomes interfere with the proliferation and differentiation of MC3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 54-58. |
| [13] | Huang Wenjun, Zhou Yafei, Wang Jie, Li Huan, Zhang Yanmin, Zhou Rui. Establishment and identification of a protocol for highly efficient differentiation of hepatocytes from human pluripotent stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 28-33. |
| [14] | Zhao Jufen, Ma Rong, Cao Jia, Yu Chuanyang, Tao Xiang, Wang Jia, Wang Libin. Regulation of peroxisome proliferator-activated receptor gamma coactivator 1 beta on stemness expression in breast cancer stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 59-65. |
| [15] | Feng Hao, Zhang Bin, Wang Jianping. Bone marrow mesenchymal stem cell transplantation can improve bone metabolism in osteoporotic rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 72-75. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

