Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (3): 420-426.doi: 10.12307/2022.835
Previous Articles Next Articles
Meniscal injury repair methods for non-blood supply area
Ning Ziwen, Wang Xu, Shi Zhengliang, Qin Yihua, Wang Guoliang, Jia Di, Wang Yang, Li Yanlin
- Department of Sports Medicine, First Affiliated Hospital of Kunming Medical University, Kunming 650032, Yunnan Province, China
-
Received:2021-10-12Accepted:2021-11-13Online:2023-01-28Published:2022-05-24 -
Contact:Li Yanlin, Chief physician, Professor, Department of Sports Medicine, First Affiliated Hospital of Kunming Medical University, Kunming 650032, Yunnan Province, China -
About author:Ning Ziwen, Master candidate, Department of Sports Medicine, First Affiliated Hospital of Kunming Medical University, Kunming 650032, Yunnan Province, China -
Supported by:National Natural Science Foundation of China, No. 81760403, 81960409 (to LYL); Yunnan Provincial Medical Leadership Project, No. L-201601 (to LYL); Yunnan Provincial Bone and Joint Disease Clinical Medicine Center Project, No. ZX2019-03-04 (to LYL)
CLC Number:
Cite this article
Ning Ziwen, Wang Xu, Shi Zhengliang, Qin Yihua, Wang Guoliang, Jia Di, Wang Yang, Li Yanlin. Meniscal injury repair methods for non-blood supply area[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 420-426.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
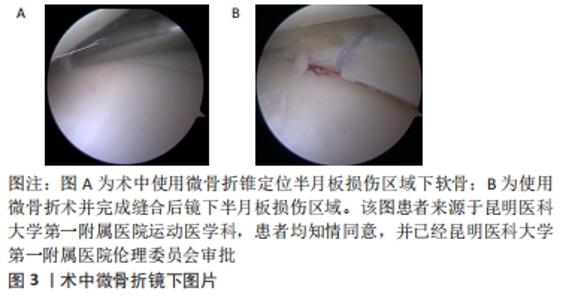
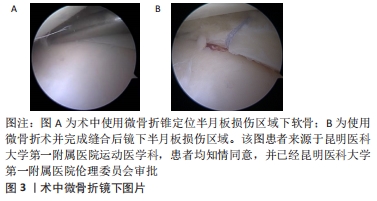
2.1 半月板的组织结构和血液供应 半月板是由液体和固体成分组成的双相软组织[11]。液相主要是水(60%-70%,湿质量),固相主要由有机物组成,包括蛋白多糖(1%-2%,湿质量)和Ⅰ型胶原(15%-25%,湿质量)。半月板横截面可分为3层:第1层是由覆盖半月板的薄纤维组成的网络结构;第2层,即第1层下面的所谓片层,是由以不同角度交叉的胶原纤维片组成。 同时,在第2层中央主层中存在沿圆周方向排列的胶原纤维束;而在第3层中,放射状胶原纤维束与沿圆周方向排列的胶原纤维束相互交叉。这种结构能有效地将半月板的垂直应力转化为圆周应力,使半月板在膝关节屈曲-伸展活动中受到应力时能够更均匀地分散在半月板上,从而防止局部应力引起的软骨损伤[12]。 半月板的血供主要来源于膝内、外侧及膝中动脉等血管[13],这些小血管在滑膜及关节囊组织中形成半月板周围血管丛及环状血管网进入半月板,为半月板的外侧缘提供血供,同时,膝内、外侧动脉还会发出细小的分支进入半月板前后角形成毛细血管网。半月板周围血管丛发出入板小动脉分上、中、下3层进入半月板的体部,这些小动脉在半月板体部内反复分支,终末形成毛细血管,支配半月板外侧缘25%-30%区域的血液供应。 半月板前后角的血供同样由半月板周围血管丛支配,半月板前后角的毛细血管网分布于整个角部区域。而体部内侧的65%-70%的区域无血管分布,其营养主要依靠表面分布的具有血管内皮细胞性质的滑膜层分泌滑液来提供[14]。 这与以往学术界对半月板血供区域分类通用的Arnoczky分区法结论基本一致,对于半月板内侧2/3的区域来说,损伤后愈合概率较低,同时由于血供不足,仍常出现难以吸收半月板缝线的情况,致使患者需行二次手术取出异物。 2.2 微骨折辅助半月板非血供区损伤修复 一些临床研究发现,前交叉韧带损伤合并半月板损伤的患者修复术后半月板愈合率和临床疗效均优于单纯半月板损伤修复患者[15-19],其中,与前交叉韧带重建相关的半月板修复的愈合率为92%,而与前交叉韧带重建无关的半月板撕裂修复的愈合率仅为63%。这一原因可能是由于前交叉韧带重建需在胫骨端和股骨端建立骨隧道,随后从骨隧道中溢出的骨髓间充质干细胞以及各种生长因子(如血小板衍生生长因子、纤维连接蛋白和血管生成素等)有助于促进半月板损伤的愈合,同时,这些溢出的物质还会形成纤维蛋白凝块,也起到了部分辅助半月板损伤愈合的效果。 因此,在对半月板红-白区、白-白区的损伤进行缝合时,为提高半月板愈合率,临床上通常采用微骨折技术辅助半月板缝合的外科手术,即在半月板缝合手术完成后,使用微骨折锥反复穿刺后交叉韧带股骨髁间窝止点附近,每个穿刺点间隔4.0 mm,深3.0-4.0 mm,直至镜下可见脂滴溢出。通过这样的方式模拟合并前交叉韧带断裂时建立的骨隧道后,使其释放各种生子因子辅助半月板的愈合。近年来,一些国内学者使用Fast-Fix技术结合微骨折技术对半月板损伤患者进行手术并进行病例对照研究发现[20-21],试验组术后MRI评估效果和临床功能评分显著高于对照组。DEAN等[22]则通过对采用微骨折技术辅助半月板损伤缝合愈合的患者与半月板损伤缝合合并前十字韧带重建的患者进行了长达2年的随访研究,发现使用微骨折技术和同时合并有前十字韧带重建的患者在行半月板缝合术后半月板修复效果均有所改善,结果验证了微骨折技术在辅助半月板愈合方面的技术可行性。 但在微骨折辅助手术中,使用微骨折锥穿刺会损伤股骨髁非负重区的软骨,并且有可能导致患者术后疼痛和骨关节炎[23],这可能是由于需要多次穿刺和进行微骨折时穿刺深度较深。BOSTAN等[24]将新西兰兔行微骨折辅助半月板损伤修复,分别使用0.8 mm,1.5 mm和4.0 mm作为微骨折穿刺直径,发现4.0 mm穿刺直径能够释放更多的骨髓间充质干细胞和各种生长因子,但与0.8 mm和1.5 mm的穿刺直径相比没有达到更好的治疗效果。该研究说明目前临床实践中微骨折穿刺辅助半月板损伤愈合的技术仍不成熟,钻孔次数过多,钻孔直径过大可能是患者术后疼痛和骨关节炎发生的原因。模拟前交叉韧带重建时制备骨道的方法,设计新的定位器,直接将钻孔部位定于半月板损伤部位下的胫骨平台上以此来缩小钻孔直径,减少钻孔数量的微骨折技术可能会减少微骨折技术的术后不良反应,并利于患者的术后快速康复。 图3为术中微骨折镜下图片,图中显示采用微骨折锥于半月板损伤部位定位,并进行钻孔,使脂肪滴及血液流出。 "
2.3 间充质干细胞辅助半月板非血供区损伤修复 国外学者在研究膝关节内软组织之间的关系时,分析了从同一患者的滑膜、半月板、前十字韧带、肌肉、脂肪组织和骨髓中提取的间充质干细胞,发现滑膜间充质干细胞和半月板间充质干细胞的基因表达谱基本相似[25]。因此,使用滑膜间充质干细胞来替代半月板非血供区的损伤组织或增强其愈合潜力是可能的。 NAKAGAWA等[26]使用同种异体间充质干细胞辅助修复猪半月板非血供区损伤,发现滑膜间充质干细胞促使半月板修复效果明显提高,结果证明了滑膜间充质干细胞对于改善半月板的非血供区损伤后的愈合效果是有效的。SEKIYA等[27]在理论研究的基础上首次于临床上使用滑膜间充质干细胞与Fast-fix技术相结合的方式来辅助修复半月板损伤,在缝合半月板损伤后再将准备好的滑膜间充质干细胞悬液通过注射器注射于患者半月板撕裂部位,术后对患者进行随访发现患者临床评分较术前明显改善,使用3D MRI技术对患者术后2年进行评估,发现半月板撕裂部位基本愈合,验证了在临床手术中使用滑膜间充质干细胞辅助修复的可行性。SUZUKI等[28]则通过悬滴技术募集滑膜间充质干细胞,发现募集后的间充质干细胞由于表面张力的增加,有助于间充质干细胞依附于损伤部位,代替了纤维蛋白骨架的功能,可减少直接注射时干细胞的流失,从流程上减少间充质干细胞辅助手术的复杂程度;同时,相较于单层培养的间充质干细胞,募集后的滑膜间充质干细胞具有更强的成软骨潜力。KONDO等[29]在对食蟹猴进行实验时使用间充质干细胞募集物代替滑膜间充质干细胞悬浮液进行移植,发现滑膜间充质干细胞可直接分化为半月板细胞以促进半月板再生和修复,同时还诱导滑膜组织聚集到半月板损伤部位。然而,目前在临床上仍没有使用间充质干细胞聚集体来辅助半月板非血供区损伤修复的相关研究,未来进行对比募集后的间充质干细胞与间充质干细胞之间疗效差距的研究将是必要的。 YUN等[30]研究了半月板细胞外基质,通过生化和蛋白质分析将半月板细胞外基质分为3个来源,其中内层的糖胺聚糖含量最高,而中层的胶原蛋白含量最高,随后该研究通过将间充质干细胞与内源性半月板细胞外基质进行共培养,发现半月板细胞外基质与人半月板纤维软骨细胞具有良好的生物相容性。此外,发现不同层面的半月板细胞外基质与间充质干细胞共培养会出现不同的组织,加入内层半月板细胞外基质可形成更多的软骨样组织,而加入外层半月板细胞外基质则会形成更多的纤维组织。有研究采用纤维蛋白支架辅助半月板修复技术以模拟半月板特殊的组织结构和生理学特性并设计了不同的化学浓度梯度[31],但这样的方法不能完全模拟半月板的组织结构,其修复效果并不理想。因此,若使用不同来源的半月板细胞外基质搭建生物支架,再将准备好的间充质干细胞加入进行共培养以更好地模仿半月板的组织结构或许可以取得更好的半月板修复效果。 而通过超滤法从人间充质干细胞中分离出细胞外泌体的方法经研究表明可以辅助关节软骨损伤的修复[32-33]。KAWATA等[34]尝试将间充质干细胞来源的细胞外泌体应用于小鼠的半月板损伤修复,在切除小鼠半月板后立即注射间充质干细胞来源的细胞外泌体,并每间隔1周之后再次注射,发现间充质干细胞来源的细胞外泌体的注射可帮助修复半月板,增加软骨细胞和滑膜间充质干细胞的生长和迁移,此研究首次证实了间充质干细胞来源的细胞外泌体不仅可以起到修复软骨损伤的作用,同样可以起到辅助半月板损伤修复及再生的作用。因此,在今后的研究中继续探讨间充质干细胞来源的细胞外泌体的作用机制,或可为半月板损伤的修复提供新的方向。 文章列举了一些典型的间充质干细胞辅助半月板损伤修复相关的实验性研究成骨[26-30,34],见表1。 "
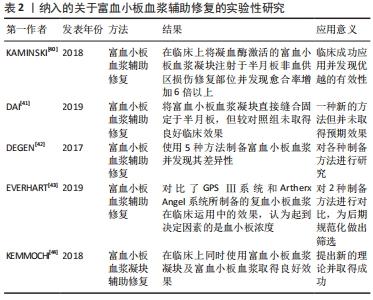
2.4 富血小板血浆辅助半月板非血供区损伤修复 富血小板血浆是一种含有高浓度血小板的自体血液产品[35]。它含有血小板释放的多种生长因子[36],血小板含有α颗粒,由包括了蛋白质、细胞因子和有助于调节愈合过程的生长因子组成[37],当血小板到达半月板损伤部位时,它们就会脱颗粒并释放有助于愈合的生物活性蛋白或生长因子[38]。BHARGAVA等[39]通过测定细胞生长因子对细胞增殖和迁移的影响发现血小板衍生生长因子可使半月板红区,红白区和白区3个区域来源的细胞迁移均增加,白细胞介素1选择性地刺激外区半月板细胞迁移,而胰岛素样生长因子1可刺激半月板细胞向中间区迁移。此结果证实了富血小板血浆可通过多种内含因子促进半月板损伤部位的修复。文章选取了5篇与富血小板血浆辅助修复半月板损伤相关的实验性研究对该部分进行综述[40-44],见表2。"
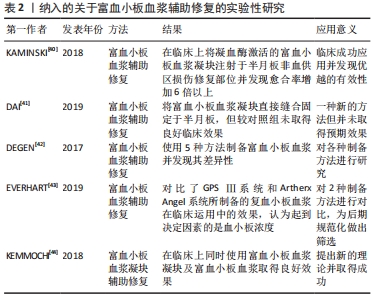

KAMINSKI等[40]在临床使用富血小板血浆辅助半月板进行修复,将凝血酶激活的富血小板血浆凝块注射于半月板修复部位,并在之后进行了病例对照研究,发现在单纯半月板撕裂的患者中,使用富血小板血浆进行半月板损伤修复的半月板愈合率显著提高,与使用安慰剂的对照组相比,实验组的半月板愈合率增加了6倍以上。DAI等[41]则在临床上对盘状半月板撕裂的患者进行半月板修整术后,对撕裂部位进行由内向外的缝合,在缝合完毕后对患者膝关节进行富血小板血浆的注射,并松开缝线使富血小板血浆充分浸润入半月板损伤部位,待富血小板血浆形成凝块后,再将缝线收紧固定,尽量避免了富血小板血浆的流失。在进行了2年的病例对照研究后发现使用富血小板血浆的实验组和未使用富血小板血浆的对照组间在临床疗效上无显著差异,这可能是由于病例数量较少,且并未进行跟踪随访,仅在12,24个月后对患者进行临床功能评分;其次该研究中所选取的损伤部位主要为红-红区和红-白区的半月板损伤,此区域的半月板自身血供相对较好,使用辅助方法的效果可能不明显。但近年来一些国外的临床研究也同样报道了使用富血小板血浆辅助半月板损伤的修复并未取得预期的效果[48-51],这表明目前的富血小板血浆辅助修复技术尚不成熟,尚待进一步探讨。 但就富血小板血浆的运用来说,不同的研究者在制备富血小板血浆时存在显著差异,DEGEN等[42]通过使用5种系统(Artherx Angel,Emcell Genesis CS,Artericell Magellan,Heavest SmartPrep和Biomet GPS Ⅲ)制备富血小板血浆并研究其差异性,发现不同系统制备出的富血小板血浆制剂的血小板浓度具有相似性,但在白细胞、中性粒细胞、红细胞浓度以及pH值来说存在显著差异。EVERHART等[43]对比了GPS Ⅲ系统和Artherx Angel系统所制备的富血小板血浆在临床运用中的效果,发现两种系统所制备的富血小板血浆均起到了辅助半月板损伤修复的作用,认为在辅助半月板修复的角度来说,白细胞、中性粒细胞、红细胞浓度以及pH值差异的重要性可能不如高血小板浓度。 另外,KEMMOCHI等[44]认为富血小板血浆凝块结构脆弱不够稳定,相较来说,富血小板纤维蛋白的基质可通过压缩变成强纤维蛋白结构,形成一层外膜,使得富血小板纤维蛋白成为一种更为稳固的胶状固体。富血小板纤维蛋白与富血小板血浆在生长因子释放峰值方面有着明显区分[45],相较于富血小板血浆生长因子的快速释放,富血小板纤维蛋白能够更加稳定、缓慢、持续地释放生长因子,并且由于其结构稳定,同时具有纤维支架的功能[46-47]。KEMMOCHI等[44]通过将富血小板纤维蛋白置于半月板损伤处并同时在关节腔内注射富血小板血浆,发现患者术后评分虽有所改善但与对照组(即单纯半月板修复)相比仍无显著性差异。但富血小板纤维蛋白的胶状性质和缓慢释放生长因子的特性使其相较于富血小板血浆存在明显优势,单独使用富血小板纤维蛋白能否起到帮助半月板损伤修复的作用是一个值得研究的话题。 总之,目前使用富血小板血浆进行联合修复的方法尚不完善。首先,部分研究者直接将富血小板血浆注射到半月板损伤位点,而其他的研究者则通过纤维蛋白凝块或血凝块将富血小板血浆嵌入半月板撕裂部,现尚无明确研究表明哪种方法有更好的修复效果,因此对于2种方式的优劣性仍有待进一步探究。其次,不同研究中富血小板血浆制剂的浓度及制备方法也存在各种差异。而在临床上,对于使用富血小板血浆是否起到了辅助半月板损伤修复的作用出现了两极分化的结果,这一原因可能是由于不同的制备方法和使用方法所导致,因此,对富血小板血浆的临床应用仍有待于进一步研究。 2.5 纤维蛋白凝块辅助半月板非血供区损伤修复 纤维蛋白凝块中富含有血小板衍生生长因子和纤维粘连蛋白等物质,这些物质是可以促进半月板愈合的趋化物和有丝分裂刺激物[52]。近年来,一些学者尝试将纤维蛋白凝块用于辅助半月板损伤的修复,文章选取了4篇与富血小板血浆辅助修复半月板损伤相关的实验性研究对该部分进行综述[53-56],见表3。 "


ARNOCZKY等[53]为评估纤维蛋白凝块能否辅助半月板的修复,将纤维蛋白凝块填充于成年狗的半月板非血供区撕裂中,经过6个月的影像学检查,发现被纤维蛋白凝块所填充的缺损处通过纤维结缔组织的增殖而愈合,此结果说明了纤维蛋白凝块对于半月板的愈合是有效的。CHAHLA等[54]设计了一种标准化的纤维蛋白凝块制备流程以便于纤维蛋白凝块辅助半月板损伤修复的研究。NAKAYAMA等[55]将纤维蛋白凝块应用于内侧半月板撕裂的患者,使用手术侧足部静脉中抽取的血液制备纤维蛋白凝块,使用编织的聚合缝线固定住纤维蛋白凝块的两端,待半月板撕裂缝合结束后,将纤维蛋白凝块缝合在修复部位附近,经过短期随访发现有75%的患者愈合效果良好,25%的患者愈合效果不佳,此原因是由于未愈合的患者均存在有不同程度的膝关节内翻畸形。KAMIMURA等[56]则提出了一种将自体半月板碎片植入纤维蛋白凝块中用以辅助修复的技术,首先使用不锈钢搅拌棒使纤维蛋白凝块以管状黏附在不锈钢棒上,随后将患者损伤的半月板组织切除,将取得的半月板组织碎片放置于管状的纤维蛋白凝块内,随后将这一纤维蛋白凝块缝合于半月板缺损处,通过术后6个月的随访发现患者半月板原有缺损处出现了组织再生和愈合。这一研究证明了纤维蛋白凝块在临床上使用辅助半月板损伤修复的可行性。 目前来看,对于纤维蛋白凝块辅助愈合的研究数量仍不足,是否存在术后并发症仍有待讨论,但临床上已有研究在使用纤维蛋白凝块并取得良好效果,进一步增加相应研究数量是有必要的,如果所有研究均可取得良好效果且方法可复制,将证明纤维蛋白凝块在辅助半月板损伤修复的优越性。 2.6 髌下脂肪垫粉碎物辅助半月板非血供区损伤修复 膝关节的内环境中各组织间的相互作用关系一直是一个热门话题,研究发现正常的滑膜组织和包膜组织会抑制软骨合成代谢并刺激软骨基质的分解[57-58],在此基础上,NISHIMUTA等[59]对膝关节内稳态环境中各组织间的相互作用进行了更加深入的研究,但与预期相反,相较于单独培养的半月板细胞活性来说,与脂肪组织同时培养的半月板细胞活性更强,脂肪组织分泌的因子可以增强半月板细胞的增殖能力。基于此,杨君君等[60]首次于临床上应用髌下脂肪垫粉碎物联合半月板损伤缝合以促进半月板修复,通过在手术中对患者行半月板成型缝合术后, 于髁间窝前方髌下脂肪垫处,用刨削刀将一小块脂肪垫(0.5 cm×0.5 cm) 充分反复打碎成1.0 mm×1.0 mm大小,通过释放脂肪组织中的干细胞以帮助半月板损伤的修复,发现手术预后效果基本上与常规的髁间窝微骨折术相当。但就临床经验而言,术中过度处理髌下脂肪垫易导致术后患者膝前疼痛,术后康复活动受限,增加患者康复周期。因此,该技术的基础研究和临床改进仍有很大空间。使用动力系统粉碎髌下脂肪示意图,见图4。 "

| [1] RAI MF, BROPHY RH, ROSEN V. Molecular biology of meniscus pathology: lessons learned from translational studies and mouse models. J Orthop Res. 2020;38(9):1895-1904. [2] ENGLUND M, GUERMAZI A, ROEMER FW, et al. Meniscal tear in knees without surgery and the development of radiographic osteoarthritis among middle-aged and elderly persons: the multicenter osteoarthritis study. Arthritis Rheum. 2009;60(3):831-839. [3] HIRANAKA T, FURUMATSU T, OKAZAKI Y, et al. Postoperative clinical outcomes of unicompartmental knee arthroplasty in patients with isolated medial compartmental osteoarthritis following medial meniscus posterior root tear. Asia Pac J Sports Med Arthrosc Rehabil Technol. 2021;26:15-20. [4] Bugbee W. What Role for Chondroplasty in the Knee? Commentary on an article by Leslie J.Bisson, MD, et al. “Observation Versus Debridement of Unstable Chondral Lesions During Partial Meniscectomy. Analysis of Patient Outcomes and Degenerative Joint Disease at 5 Years in the Chondral Lesions And Meniscus Procedures (ChAMP) Randomized Controlled Trial”. J Bone Joint Surg Am. 2021;103(17):e70. [5] BEDRIN MD, KARTALIAS K, YOW BG, et al. Degenerative joint disease After Meniscectomy. Sports Med Arthrosc Rev. 2021;29(3):e44-e50. [6] HUSSAIN ZB, CHAHLA J, MANDELBAUM BR, et al. The role of meniscal tears in spontaneous osteonecrosis of the knee: a systematic review of suspected etiology and a call to revisit nomenclature. Am J Sports Med. 2019;47(2):501-507. [7] PAXTON ES, STOCK MV, BROPHY RH. Meniscal repair versus partial meniscectomy: a systematic review comparing reoperation rates and clinical outcomes. Arthroscopy. 2011;27(9):1275-1288. [8] LUTZ C, DALMAY F, EHKIRCH FP, et al. Meniscectomy versus meniscal repair: 10 years radiological and clinical results in vertical lesions in stable knee. Orthop Traumatol Surg Res. 2015;101(8 Suppl):S327-S331. [9] STEIN T, MEHLING AP, WELSCH F, et al. Long-term outcome after arthroscopic meniscal repair versus arthroscopic partial meniscectomy for traumatic meniscal tears. Am J Sports Med. 2010;38(8):1542-1548. [10] WHITEHOUSE MR, HOWELLS NR, PARRY MC, et al. Repair of torn avascular meniscal cartilage using undifferentiated autologous mesenchymal stem cells: from in vitro optimization to a first-in-human study. Stem Cells Transl Med. 2017;6(4):1237-1248. [11] RIBITSCH I, PEHAM C, ADE N, et al. Structure-Function relationships of equine menisci. PLoS One. 2018;13(3):e0194052. [12] DANSO EK, OINAS JMT, SAARAKKALA S, et al. Structure-function relationships of human meniscus. J Mech Behav Biomed Mater. 2017;67:51-60. [13] BRINDLE T, NYLAND J, JOHNSON DL. The meniscus: review of basic principles with application to surgery and rehabilitation. J AthlTrain. 2001;36(2):160-169. [14] 李箭,蒋欣,裴福兴,等.膝关节半月板血供的应用解剖学研究[J].临床外科杂志,2003,11(5):335-336,364. [15] HORIBE S, SHINO K, NAKATA K, et al. Second-look arthroscopy after meniscal repair. Review of 132 menisci repaired by an arthroscopic inside-out technique. J Bone Joint Surg Br. 1995;77(2):245-249. [16] MURRELL GA, MADDALI S, HOROVITZ L, et al. The effects of time course after anterior cruciate ligament injury in correlation with meniscal and cartilage loss. Am J Sports Med. 2001;29(1):9-14. [17] Kang HJ, Chun CH, Kim KM, et al. The results of all-inside meniscus repair using the viper repair system simultaneously with anterior cruciate ligament reconstruction. Clin Orthop Surg. 2015;7(2):177-184. [18] Shelbourne KD, Carr DR. Meniscal repair compared with meniscectomy for bucket-handle medial meniscal tears in anterior cruciate ligament-reconstructed knees. Am J Sports Med. 2003;31(5):718-723. [19] Cannon WD Jr, Vittori JM. The incidence of healing in arthroscopic meniscal repairs in anterior cruciate ligament-reconstructed knees versus stable knees. Am J Sports Med. 1992;20(2):176-181. [20] 周钢,葛兴涛,林坚平,等.关节镜下Fast-Fix联合微骨折术修复水平状半月板撕裂的临床研究[J].重庆医学,2019,48(12):2027-2030. [21] 刘正鑫.关节镜下Fast-Fix联合微骨折术修复水平状半月板撕裂的临床效果观察[J].临床研究,2019,27(12):56-57. [22] DEAN CS, CHAHLA J, MATHENY LM, et al. Outcomes after biologically augmented isolated meniscal repair with marrow venting are comparable with those after meniscal repair with concomitant anterior cruciate ligament reconstruction. Am J Sports Med. 2017;45(6):1341-1348. [23] BARENIUS B, PONZER S, SHALABI A, et al. Increased risk of osteoarthritis after anterior cruciate ligament reconstruction: a 14-year follow-up study of a randomized controlled trial. Am J Sports Med. 2014;42(5):1049-1057. [24] BOSTAN B, GEVREK F, BALTA O, et al. Effects of different bone marrow stimulation techniques on avascular zone meniscal defects. Bratisl Lek Listy. 2018;119(10):630-635. [25] SEGAWA Y, MUNETA T, MAKINO H, et al. Mesenchymal stem cells derived from synovium, meniscus, anterior cruciate ligament, and articular chondrocytes share similar gene expression profiles. J Orthop Res. 2009; 27(4):435-441. [26] NAKAGAWA Y, MUNETA T, KONDO S, et al. Synovialmesenchymal stem cells promote healing after meniscal repair in microminipigs. Osteoarthritis Cartilage. 2015;23(6):1007-1017. [27] SEKIYA I, KOGA H, OTABE K, et al. Additional use of synovial mesenchymal stem cell transplantation following surgical repair of a complex degenerative tear of the medial meniscus of the knee: a case report. Cell Transplant. 2019;28(11):1445-1454. [28] SUZUKI S, MUNETA T, TSUJI K, et al. Properties and usefulness of aggregates of synovial mesenchymal stem cells as a source for cartilage regeneration. Arthritis Res Ther. 2012;14(3):R136. [29] KONDO S, MUNETA T, NAKAGAWA Y, et al. Transplantation of autologous synovial mesenchymal stem cells promotes meniscus regeneration in aged primates. J Orthop Res. 2017;35(6):1274-1282. [30] YUN HW, SONG BR, SHIN DI, et al. Fabrication of decellularized meniscus extracellular matrix according to inner cartilaginous, middle transitional, and outer fibrous zones result in zone-specific protein expression useful for precise replication of meniscus zones. Mater Sci Eng C Mater Biol Appl. 2021;128:112312. [31] IANNUCCI LE, BOYS AJ, MCCORRY MC, et al. Cellular and chemical gradients to engineer the meniscus-to-bone insertion. Adv Healthc Mater. 2019;8(7):e1800806. [32] DE LUNA A, OTAHAL A, NEHRER S. Mesenchymal stromal cell-derived extracellular vesicles -silver linings for cartilage regeneration? Front Cell Dev Biol. 2020;8:593386. [33] BAO C, HE C. The role and therapeutic potential of MSC-derived exosomes in osteoarthritis. Arch BiochemBiophys. 2021;710:109002. [34] KAWATA K, KOGA H, TSUJI K, et al. Extracellular vesicles derived from mesenchymal stromal cells mediate endogenous cell growth and migration via the CXCL5 and CXCL6/CXCR2 axes and repair menisci. Stem Cell Res Ther. 2021;12(1):414. [35] ANDIA I, MAFFULLI N. Platelet-rich plasma for managing pain and inflammation in osteoarthritis. Nat Rev Rheumatol. 2013;9(12):721-730. [36] SAKATA R, REDDI AH. Platelet-rich plasma modulates actions on articular cartilage lubrication and regeneration. Tissue Eng Part B Rev. 2016;22(5):408-419. [37] CAVALLO C, FILARDO G, MARIANI E, et al. Comparison of platelet-rich plasma formulations for cartilage healing: an in vitro study.J Bone Joint Surg Am. 2014;96(5):423-429. [38] ANITUA E, ANDIA I, ARDANZA B, et al. Autologous platelets as a source of proteins for healing and tissue regeneration. Thromb Haemost. 2004;91(1):4-15. [39] BHARGAVA MM, ATTIA ET, MURRELL GA, et al. The effect of cytokines on the proliferation and migration of bovine meniscal cells. Am J Sports Med. 1999;27(5):636-643. [40] KAMINSKI R, KULINSKI K, KOZAR-KAMINSKA K, et al. A prospective, randomized, double-blind, parallel-group, placebo-controlled study evaluating meniscal healing, clinical outcomes, and safety in patients undergoing meniscal repair of unstable, complete vertical meniscal tears (bucket handle) augmented with platelet-rich plasma. Biomed Res Int. 2018; 2018:9315815. [41] DAI WL, ZHANG H, LIN ZM, et al. Efficacy of platelet-rich plasma in arthroscopic repair for discoid lateral meniscus tears. BMC Musculoskelet Disord. 2019;20(1):113. [42] DEGEN RM, BERNARD JA, OLIVER KS, et al. Commercial separation systems designed for preparation of platelet-rich plasma yield differences in cellular composition. HSS J. 2017;13(1):75-80. [43] EVERHART JS, CAVENDISH PA, EIKENBERRY A, et al. Platelet-rich plasma reduces failure risk for isolated meniscal repairs but provides no benefit for meniscal repairs with anterior cruciate ligament reconstruction. Am J Sports Med. 2019;47(8):1789-1796. [44] KEMMOCHI M, SASAKI S, TAKAHASHI M, et al. The use of platelet-rich fibrin with platelet-rich plasma support meniscal repair surgery. J Orthop. 2018;15(2):711-720. [45] DOHANEHRENFEST DM, DE PEPPO GM, DOGLIOLI P, et al. Slow release of growth factors and thrombospondin-1 in Choukroun’s platelet-rich fibrin (PRF): a gold standard to achieve for all surgical platelet concentrates technologies. Growth Factors. 2009;27(1):63-69. [46] WONG CC, KUO TF, YANG TL, et al. Platelet-rich fibrin facilitates rabbit meniscal repair by promoting meniscocytes proliferation, migration, and extracellular matrix synthesis. Int J Mol Sci. 2017;18(8):1722. [47] KOBAYASHI E, FLÜCKIGER L, FUJIOKA-KOBAYASHI M, et al. Comparative release of growth factors from PRP, PRF, and advanced-PRF. Clin Oral Investig. 2016;20(9):2353-2360. [48] GRIFFIN JW, HADEED MM, WERNER BC, et al. Platelet-rich plasma in meniscal repair: does augmentation improve surgical outcomes? Clin Orthop Relat Res. 2015;473(5):1665-1672. [49] BURCHARD R, HUFLAGE H, SOOST C, et al. Efficiency of platelet-rich plasma therapy in knee osteoarthritis does not depend on level of cartilage damage. J Orthop Surg Res. 2019;14(1):153. [50] GUENOUN D, MAGALON J, DE TORQUEMADA I, et al. Treatment of degenerative meniscal tear with intrameniscal injection of platelets rich plasma. Diagn Interv Imaging. 2020;101(3):169-176. [51] YANG CP, HUNG KT, WENG CJ, et al. Clinical outcomes of meniscus repair with or without multiple intra-articular injections of platelet rich plasma after surgery. J Clin Med. 2021;10(12):2546. [52] PORT J, JACKSON DW, LEE TQ, et al. Meniscal repair supplemented with exogenous fibrin clot and autogenous cultured marrow cells in the goat model.Am J Sports Med. 1996;24(4):547-555. [53] ARNOCZKY SP, WARREN RF, SPIVAK JM. Meniscal repair using an exogenous fibrin clot.An experimental study in dogs. J Bone Joint Surg Am. 1988;70(8):1209-1217. [54] CHAHLA J, KENNEDY NI, GEESLIN AG, et al. Meniscal repair with fibrin clot augmentation. Arthrosc Tech. 2017;6(6):e2065-e2069. [55] NAKAYAMA H, KANTO R, KAMBARA S, et al. Successful treatment of degenerative medial meniscal tears in well-aligned knees with fibrin clot implantation. Knee Surg Sports Traumatol Arthrosc. 2020;28(11):3466-3473. [56] KAMIMURA T, KIMURA M. Repair of a chronic large meniscal defect with implantation of autogenous meniscal fragments using a tubular-shaped fibrin clot. Arthrosc Tech. 2018;7(3):e257-e263. [57] PATWARI P, LIN SN, KURZ B, et al. Potent inhibition of cartilage biosynthesis by coincubation with joint capsule through an IL-1-independent pathway. Scand J Med Sci Sports. 2009;19(4):528-535. [58] VANKEMMELBEKE MN, ILIC MZ, HANDLEY CJ, et al. Coincubation of bovine synovial or capsular tissue with cartilage generates a soluble “Aggrecanase” activity. Biochem Biophys Res Commun. 1999;255(3):686-691. [59] NISHIMUTA JF, BENDERNAGEL MF, LEVENSTON ME. Co-culture with infrapatellar fat pad differentially stimulates proteoglycan synthesis and accumulation in cartilage and meniscus tissues. Connect Tissue Res. 2017; 58(5):447-455. [60] 杨君君,周益昭,黄术,等.关节镜下髁间窝微骨折术与髌下脂肪垫粉碎术在半月板损伤中的应用研究[J].中国内镜杂志,2018,24(1):22-28. |
| [1] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [2] | Chen Kaijia, Liu Jingyun, Cao Ning, Sun Jianbo, Zhou Yan, Mei Jianguo, Ren Qiang. Application and prospect of tissue engineering in treatment of osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1450-1456. |
| [3] | Yang Junliang, Lu Tan, Xu Biao, Jiang Yaqiong, Wang Fucheng. Three-dimensional finite element analysis of effects of partial anterior cruciate ligament rupture on knee joint stress [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1347-1353. |
| [4] | Feng Ruiqin, Han Na, Zhang Meng, Gu Xinyi, Zhang Fengshi. Combination of 1% platelet-rich plasma and bone marrow mesenchymal stem cells improves the recovery of peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 985-992. |
| [5] | Qiu Xiaoyan, Li Bixin, Li Jingdi, Fan Chuiqin, Ma Lian, Wang Hongwu. Differentiation of insulin-producing cells from human umbilical cord mesenchymal stem cells infected by MAFA-PDX1 overexpressed lentivirus [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1000-1006. |
| [6] | Liu Qiwei, Zhang Junhui, Yang Yuan, Wang Jinjuan. Role and mechanism of umbilical cord mesenchymal stem cells on polycystic ovary syndrome [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1015-1020. |
| [7] | Mei Jingyi, Liu Jiang, Xiao Cong, Liu Peng, Zhou Haohao, Lin Zhanyi. Proliferation and metabolic patterns of smooth muscle cells during construction of tissue-engineered blood vessels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1043-1049. |
| [8] | Wang Shanshan, Shu Qing, Tian Jun. Physical factors promote osteogenic differentiation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1083-1090. |
| [9] | Pan Xiaolong, Fan Feiyan, Ying Chunmiao, Liu Feixiang, Zhang Yunke. Effect and mechanism of traditional Chinese medicine on inhibiting the aging of mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1091-1098. |
| [10] | Kong Jianda, Mu Yujing, Zhu Lei, Li Zhilin, Chen Shijuan. Mechanism of satellite cell regulation and its role in ecological niche signaling during skeletal muscle regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1105-1111. |
| [11] | Huang Yuxin, Liang Wenzi, Chen Xiuwen, Ni Na, Zhao Yinglin, Lin Changmin. Role of autophagy in hair regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1112-1117. |
| [12] | Ma Shuwei, He Sheng, Han Bing, Zhang Liaoyun. Exosomes derived from mesenchymal stem cells in treatment of animals with acute liver failure: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1137-1142. |
| [13] | Zhang Kefan, Shi Hui. Research status and application prospect of cytokine therapy for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 961-967. |
| [14] | Yin Tong, Yang Jilei, Li Yourui, Liu Zhuoran, Jiang Ming. Application of core-shell structured nanofibers in oral tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 766-770. |
| [15] | Chen Xiaofang, Zheng Guoshuang, Li Maoyuan, Yu Weiting. Preparation and application of injectable sodium alginate hydrogels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 789-794. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||