Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (7): 1105-1111.doi: 10.12307/2023.745
Previous Articles Next Articles
Mechanism of satellite cell regulation and its role in ecological niche signaling during skeletal muscle regeneration
Kong Jianda1, Mu Yujing2, Zhu Lei1, Li Zhilin1, Chen Shijuan3
- 1School of Physical Education Science, Qufu Normal University, Jining 273100, Shandong Province, China; 2Dongzhimen Hospital of Beijing University of Chinese Medicine, Beijing 100010, China; 3College of Physical Education Science, Tianjin Normal University, Tianjin 300380, China
-
Received:2022-10-20Accepted:2022-12-12Online:2024-03-08Published:2023-07-17 -
Contact:Zhu Lei, School of Physical Education Science, Qufu Normal University, Jining 273100, Shandong Province, China -
About author:Kong Jianda, Master candidate, School of Physical Education Science, Qufu Normal University, Jining 273100, Shandong Province, China -
Supported by:Shandong Province Professional Degree Postgraduate Teaching Case Library Project, No. SDYAL20103 (to ZL)
CLC Number:
Cite this article
Kong Jianda, Mu Yujing, Zhu Lei, Li Zhilin, Chen Shijuan. Mechanism of satellite cell regulation and its role in ecological niche signaling during skeletal muscle regeneration[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1105-1111.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
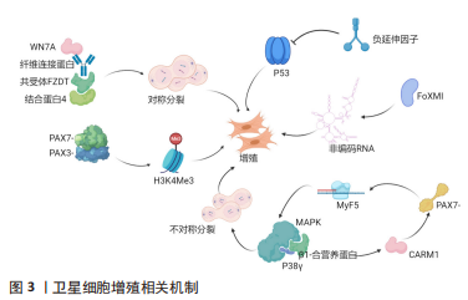
2.1 SCs概述 SCs是存在于骨骼肌中的一种特定成体干细胞群,通常为静止状态,在稳态环境中主要进行不对称的细胞分裂来修复受损的肌纤维并维持干细胞库的稳定,且只进行极少部分的增殖[1]。SCs亦可以在组织受损时激活并进行对称性分裂,进而生成新的干细胞并促进肌细胞激活和增殖,新的干细胞亦分化为新的肌细胞,以达到修复骨骼肌纤维的作用,进而促进骨骼肌的再生[1]。SCs可通过自身的调控作用或与所处生态位信号相互作用等方面实现自身的激活、增殖和分化,进而促进骨骼肌再生过程的有效进行[1]。 2.2 骨骼肌再生过程中SCs调控作用的机制 2.2.1 SCs的增殖 SCs分裂模式分为对称分裂和不对称分裂,其中对称分裂可扩大干细胞库,而不对称分裂可产生分化的子细胞和干细胞来补充干细胞库。SCs分裂增殖的相关机制详情见图3。"
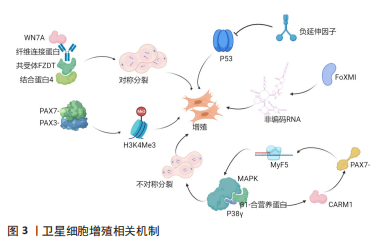
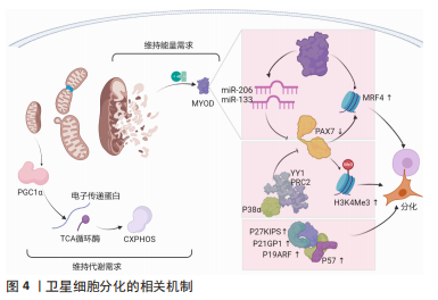
SCs不对称分裂填充干细胞库,并随后稳态环境进行对称分裂并提供足够的成肌细胞,进而形成新的肌纤维,但目前未直接研究对机体内细胞分裂的模式。SCs能够在细胞的对称分裂和不对称分裂之间交替进行,即SCs的激活和增殖都有一种计数机制(Notch通路)来进行自我调节或调节每种细胞分裂模式相关的信号以及其姊妹细胞之间的外部通信[1]。另外,SCs会残余在受损的肌纤维和细胞外基质中,并决定SCs的分裂模式和趋势[2]。因此,改变SCs对骨骼肌损伤的反应会导致阻碍骨骼肌再生并衰竭干细胞库。另外,SCs与局部环境的通信对SCs破坏后的分裂模式和促进扩张具有重要意义。环境诱导SCs产生WNT7A,与自身产生的纤维连接蛋白相结合,刺激共受体FZD7与蛋白质4结合,进而激活SCs对称分裂[3]。在传统上与肌纤维相关的抗肌萎缩蛋白糖蛋白的复合物亦能够维持细胞和干细胞后代的平衡。骨骼肌营养不良使糖蛋白β1与营养蛋白相结合调控p38γ MAPK和CARM1,进而不对称分裂阻止子代SCs核易位反应[4]。相反,在作用于分化的子细胞中,CARM1诱导PAX7甲基化,后诱导MYF5转录,而p38α/βMAPKs不对称分裂,Notch通路中NUMB和DNA链可产生定型和自我更新的后代[5]。SCs的负延伸因子在骨骼肌损伤后可通过下调p53活性抑制其作为细胞周期检查点蛋白的作用并维持SCs增殖[6]。FOXMI可以促进SCs两种长链非编码RNA的表达,进而促进核糖体蛋白的转录,并减少细胞凋亡[7]。另外,PAX7-和PAX3-PAXBP1(促进组蛋白甲基转移酶复合物的募集)相结合,并在PAX7启动子上激活H3K4me3,从而介导SCs相关位点的染色质开放[8]。另外,H3K4me3作用于SCs的增殖。SCs从增殖到分化会抑制PAX7基因,进而从H3K4me3相关的染色质介导状态过渡到H3K27me3介导的抑制状态。另外,磷酸化p38γ可增加MYOD与KMT1A的相互作用,进而阻止MYOD的表达。MYOD与HDAC1的相互作用,后者可抑制p21Cip1对MYOD转录,E2F家族募集p300/CBP组蛋白乙酰转移酶和PCAF/GCN5组蛋白乙酰化酶到细胞周期蛋白基因,这说明其他控制SCs增殖的表观遗传机制涉及积极压制成肌分化基因的表达和激活特定增殖相关基因的表达[9]。综上而言,增殖期SCs的组蛋白乙酰化水平是各时期SCs中最高的,其可能的机制包括两点:①SCs分泌乳酸的糖酵解反应[10];②SIRT1脱乙酰酶活性降低后上调表达乙酰辅酶A[11]。 SCs还发现了激活和增殖的新机制:①CHD抑制受体交互蛋白激酶34(RIPK3)依赖性坏死,然后确保SCs肌源性系在增殖过程中的保真度,但CHD缺乏条件会减少神经或免疫基因,破坏干细胞增殖和骨骼肌再生[12];②解旋酶DHX36调节转录因子Gnai表达Gnai2 mRNA[13]。这说明多种机制控制SCs增殖以确保细胞周期进展和子代SCs激活和增殖,同时避免SCs过早肌源性分化或朝着非肌源性趋势发展。另外,CHEN等[14]发现骨骼肌损伤时增殖的SCs可从基板迁移到受损纤维处并与其融合。可以发现,目前的研究主要集中于SCs的激活、增殖、分化和自我更新,缺少调控SCs转移相关机制的研究。 2.2.2 SCs的分化 子代SCs增殖、分化和融合形成新的肌纤维才可完整重建骨骼肌完全重建,而SCs库的恢复至稳态则需要SCs恢复到静止状态。SCs的分化机制可从表观遗传学水平进行综述(图4)。"
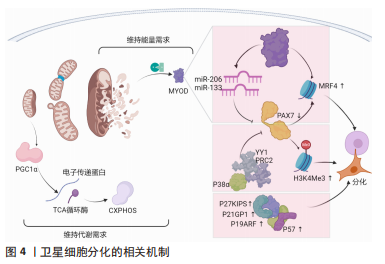
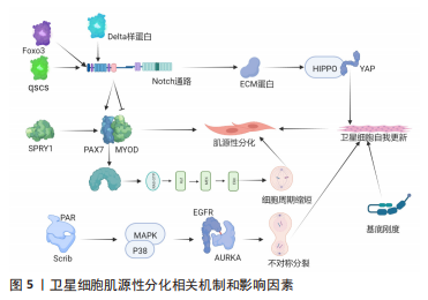
有3种途径可实现SCs的肌源性分化:①SCs恢复静止时上调p27KIP1,分化时上调p21CIP1、p19ARF和p57,MASSENET等[9]发现p38α和YY1、PRC2相互作用在表观遗传水平上抑制PAX7表达,即SCs增殖到分化的转变需要p38α/βMAPK信号通路的调控作用;②SCs抑制剂101的表达诱导细胞的细胞周期退出,并激活MYOD和MEF2转录活性以及促进染色质重塑复合物进入肌原性启动子来诱导SCs的肌原性分化[15];③SCs染色质上的组蛋白和分化过程中的增殖SCs与乙酰化程度普遍降低相比,乙酰化标记通过MYOD特异性基因从干性基因和细胞周期重定向到肌肉分化[4],而MYOD则加强了细胞周期的退出,诱导miR-206和miR-133表达,下调PAX7[16],并通过不同的表观遗传、转录和转录后机制激活分化肌细胞生成素的主要调节因子,最终MYOD和肌原蛋白协同作用激活MRF4等终末分化基因[17]。另外,SCs分化后主要转向OXPHOS,分化的SCs具有较高质量的线粒体,并通过2种途径维持成熟肌肉组织的能量需求:①使电子传递链蛋白表达水平增加[18];②促进TCA循环酶表达水平上调[19]。在SCs分化的早期阶段会发生线粒体分裂和线粒体吞噬导致线粒体清除,后过氧化物酶体增殖活化受体γ辅助活化因子1α和线粒体融合扩增线粒体生物发生,进而转向OXPHOS以支持活性肌管的代谢需求[20],而SCs分化时线粒体清除和葡萄糖对线粒体呼吸的利用增加都能够导致干细胞基因周围这种表观遗传标记的水平降低[21]。然而,过氧化物酶影响SCs的分化而不影响SCs增殖,说明细胞器对诱导SCs再生有不同的作用。 Shintaku全基因组染色质免疫沉淀后的测序显示MYOD与线粒体生物合成、脂肪酸氧化和电子传递链相互作用[22]。这说明MYOD控制终末肌原分化所需的线粒体相关代谢功能。SCs代谢重编程发生在再生阶段,分化的成肌细胞通过MYMK重建肌纤维结构,例如:①Myomaker蛋白激活肌细胞融合和骨骼肌形成[23];②微蛋白Minion控制细胞融合和骨骼肌形成[24];③Myomerger蛋白诱导非致密细胞的融合促进骨骼肌发育[25]。 2.2.3 SCs分化后对骨骼肌的重建 SCs分化后需要对骨骼肌修复重建(图5),最重要的一步为肌源性分化。骨骼肌损伤后SCs恢复静止,可实现完全的干细胞稳态,增殖的SCs亚群需3种通路调控:①p38MAPK通路[26];②FGF-ERK MAPK通路[27];③JAK-STAT3通路[28]。"
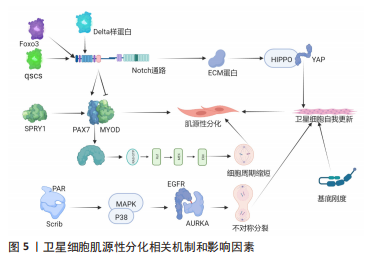
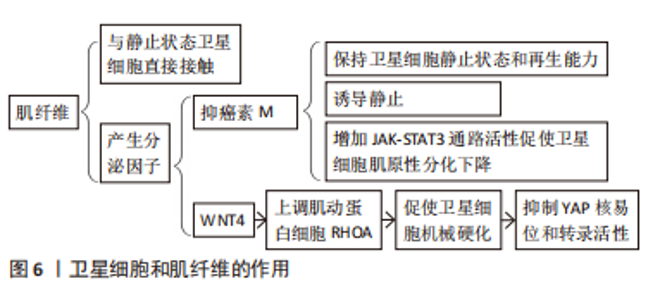
SPRY1在SCs通过对慢循环亚群中特异性表达的反应成纤维细胞生长因子抑制信号传输ERK促进细胞周期退出的信号传导途径,SPRY1缺失导致骨骼肌再生后PAX7和MYOD自我更新群体下降50%[29]。FOXO3诱导Notch信号通路抑制SCs肌原性分化和恢复静止[30]。与此相一致的是,Notch中Delta样蛋白1致力于分化的子细胞表达,子代静止卫星细胞表达NOTCH3受体激活Notch信号传导并促进其恢复静止,后抑制MYOD表达并上调PAX7,以诱导维持SCs的状态[31]。UMB和p38α/βMAPK接受子代细胞并获得更定向的分化状态NUMB和模板DNA链(非p38α/βAPK调节)子代细胞来自我更新和补充SCs库,PAR和Scrib细胞可能是p38α/βMAPK驱动因素不对称分布模式[32]。另外,SCs若未表达MYF5,则SCs群体不对称分裂,通过诱导MYF受体(由p38γMAPK调节)表达,表皮生长因子受体和AURKA刺激信号传输SCs沿顶基轴向有丝分裂中心体分裂,促进SCs不对称分裂[33]。这说明SCs对生物化学信号敏感。另外,基底板的刚度亦影响SCs库的自我更新能力[34],即SCs亦对生物物理信号敏感。另外,若Notch信号通路缺失,SCs不能产生细胞外基质蛋白,故SCs无法集中于正常的亚层静止位置[35],细胞外基质蛋白诱导SCs通过Hippo通路的YAP信号实现自我更新能力[36]。 然而,目前SCs在体内分化或自我更新的时间尚不清楚,仅可知:①SCs自我更新时交替进行对称裂和不对称分裂,损伤后3 d主要是对称分裂,损伤后5 d两种分裂均进行[6];②只有SCs增殖才可补充SCs库,循环后的SCs可以恢复到静止状态[37]。这说明在SCs分化中并非所有增殖的SCs都会发生细胞分化,且SCs的不对称分裂可产生一个分化的细胞和一个自我更新的细胞。 2.3 骨骼肌再生过程中SCs与其生态位信号的相互作用 2.3.1 SCs和肌纤维 主要通过静止状态的SCs与肌纤维产生的分泌因子来发挥作用(图6)。SCs的接触位点在基底板和肌纤维质膜之间,其通过极化的黏附作用接触(如钙黏蛋白),可与其所处生态位信号中直接相关的生态位信号相连[1]。驱动SCs从静止到激活的基本机制为骨骼肌损伤诱导的干细胞-细胞的相互作用,而SCs中N-钙黏蛋白和M-钙黏蛋白的基因则被去除,并定位于SCs和肌纤维之间的直接接触位点,诱导SCs从静止卫星细胞向脂肪干细胞过渡,模拟损伤后早期的静止卫星细胞激活阶段[38]。"
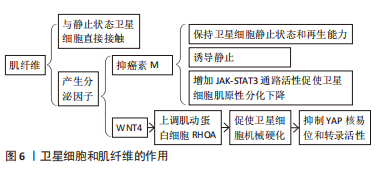
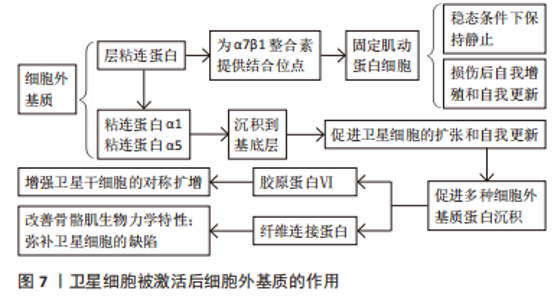
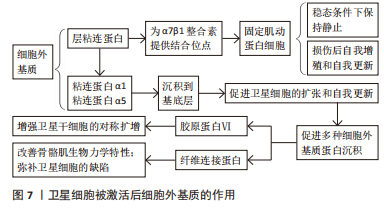
在目前的研究中,肌纤维是唯一能与静止状态的SCs直接接触的细胞,基底层和间质细胞外基质封存了各种生长因子以及和静止卫星细胞激活和增殖有关的信号分子[39]。另外,肌纤维能够产生众多分泌因子,其中最具代表性的为抑癌素M和WNT4。SAMPATH等[40]的研究发现,抑癌素M可保持SCs的静止状态和再生能力,并能够通过一种未知的机械作用在移植时诱导静止。抑癌素M亦为白细胞介素6细胞因子家族成员,且该类细胞因子通过增加JAK-STAT3信号通路活性促使衰老过程中SCs的肌原性分化下降[28]。ELIAZER等[41]发现WNT4可上调肌动蛋白细胞骨架调控因子RHOA引起SCs的机械硬化,进而抑制关键的细胞机械传感器YAP的核易位和转录活性,YAP作为Hippo通路中的相关因子能够控制包括许多参与组织再生的过程来激活SCs所必需的各种细胞[36]。 2.3.2 SCs和细胞外基质 骨骼肌损伤可以诱导生态位信号变化,包括肌纤维损伤,这通常是基底层的损伤,从而导致SCs暴露在这个信号中,SCs基底板可以组成细胞外基质网络从肌纤维间质中分裂[42]。在基底板成分中,层粘连蛋白为SCs中表达的α7β1整合素提供结合位点,将其肌动蛋白细胞骨架固定在细胞外基质上[43]。该作用是维持SCs在稳态条件下保持静止状态以及维持损伤后自我增殖和自我更新的必要条件。 在SCs被激活后,SCs将层粘连蛋白α1和层粘连蛋白α5沉积到基底层,该过程亦可促进SCs的扩张和自我更新[44](图7)。TIERNEY等[45]进行了一项对发育肌发生连续阶段的SCs行为的分析研究,其结果表明SCs的扩张增加可改变生态位信号组成并促进多种细胞外基质蛋白沉沉积(包括腱蛋白C、纤维连接蛋白和胶原蛋白Ⅵ)。另外,纤维连接蛋白中的细胞外基质糖蛋白纤连蛋白可与SDC4结合刺激WNT7a诱导增强卫星干细胞的对称扩增的能力[3],胶原蛋白Ⅵ则能够改善Col6a1(-/-)小鼠骨骼肌的生物力学特性和弥补SCs的缺陷[46]。另外,SCs亦可使胶原蛋白Ⅵ和纤维连接蛋白表达,即细胞外基质的组成和信号通路由SCs来源的因子决定,并作用于其信号循环中,并通过机械输入在再生过程中控制干细胞的功能[47]。纤维连接蛋白诱导的WNT信号传导和机械转导诱导的YAP信号传导即是其中一条介导这些作用的途径[36,41]。"
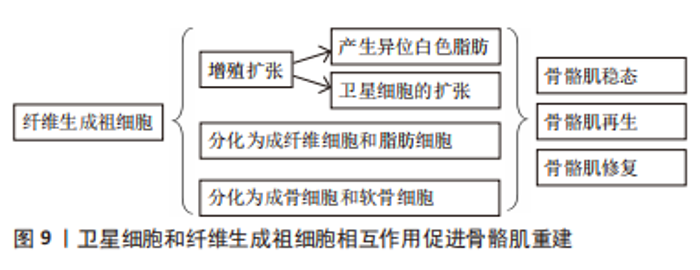
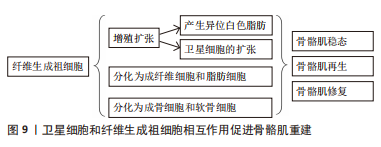
LIU等[48]认为骨骼肌交界位的完整性可促进转导启动骨骼肌收缩的运动神经元信号,且SCs和骨骼肌交界位可以互相作用,在SCs消耗和神经骨骼肌损伤后,再生骨骼肌交界位附近的静止卫星细胞激活能促进肌纤维神经再生,限制去神经支配引起的组织萎缩和纤维化。MADARO等[49]在脊髓损伤、脊髓性骨骼肌萎缩和肌萎缩性侧索硬化症的小鼠模型和肌萎缩性侧索硬化症患者的骨骼肌中也发现了由STAT3-IL-6信号异常激活的纤维生成祖细胞,激活纤维生成祖细胞中的STAT3-IL-6信号能有效地对抗急性去势和肌萎缩性侧索硬化症小鼠模型(SODG93A小鼠)中的骨骼肌萎缩和纤维化现象。神经骨骼肌接头完整性丧失后可使致病性纤维生成祖细胞激活,这进一步说明了纤维生成祖细胞在应对稳态扰动时的功能多样性,并表明它们对神经骨骼肌疾病的发病机制有潜在机制。 目前相关领域的研究仅发现神经细胞可作为骨骼肌再生过程中的生态位细胞,但其在骨骼肌再生过程中对SCs的调节作用尚不清楚。 2.3.4 SCs和纤维生成祖细胞 纤维生成祖细胞通常被定义为一种间质祖细胞,其驻留于肌肉组织中。在脂肪浸润的模型中移植纯化的纤维生成祖细胞可为皮下或骨骼肌内输送能够产生异位的白色脂肪,但将其移植于健康的骨骼肌中则不能够如此,纤维生成祖细胞在完整的肌肉组织中保持静止状态,但在受损后的肌肉组织中可进行有效的增殖[50]。而在肌肉组织来源的细胞群中,只有血小板衍生生长因子受体(platelet-derived growth factor receptor,PDGFR) alpha(+)细胞在体外和体内均显示有效的脂肪生成和分化,而通过SCs衍生的肌纤维则抑制PDGFR alpha(+)细胞的脂肪生成[50]。另外,纤维生成祖细胞在一定条件下可分化为成纤维细胞和脂肪细胞,甚至在某些条件下可分化为成骨细胞和软骨细胞[37]。然而,纤维生成祖细胞在静息状态的骨骼肌中不能够发生分化,并且能够在骨骼肌损伤后进行激活和增殖扩张,甚至调节SCs的扩张[50],造成纤维生成祖细胞扩张的原因为损伤严重的神经疾病导致骨骼肌交界位的完整性丧失。在上述情况下,功能失调的纤维生成祖细胞群体的积累能够直接导致骨骼肌萎缩[51]。然而,纤维生成祖细胞亦有助于长期维持骨骼肌的平衡,并在骨骼肌的再生和维持中起关键作用[52]。 骨骼肌再生与调控纤维生成祖细胞生成和清除的时间有关。其中,由PDGFRα表达的纤维生成祖细胞基因消融能够导致骨骼肌损伤后SCs丢失和免疫细胞的扩增,进而导致骨骼肌的再生亦受损[53]。纤维生成祖细胞亦可使骨骼肌再生缺陷发作或慢性退行性疾病后脂肪和纤维化积累。在众多纤维生成祖细胞中,PDGFRα(+)细胞在mdx小鼠的横膈膜纤维化区域中积累[54],骨骼肌组织损伤后纤维生成祖细胞增殖并产生脂肪细胞[55]。WOSCZYNA等[16]发现靶向microRNA介导的基因抑制限制了骨骼肌间充质基质细胞的脂肪形成转化。MALECOVA等[56]研究发现,scRNA-seq可揭示纤维生成祖细胞的异质性并对骨骼肌稳态和骨骼肌修复产生至关重要的作用,其发现:①骨骼肌急性损伤诱导血管细胞黏附分子1,属于纤维生成祖细胞亚群,其作用为促纤维化,这与细胞外基质短暂沉积促进SCs的不对称分裂相一致;②血管细胞黏附分子1仅在骨骼肌损伤时短暂形成,且纤维生成祖细胞在恢复稳态后被清除,但表达血管细胞黏附分子1的纤维生成祖细胞持续存在于从Duchenne肌肉萎缩症(一种慢性骨骼肌损伤的病理状态)小鼠模型中分裂出来的骨骼肌中。另外,免疫反应的稳定对于控制纤维生成祖细胞数量和防止骨骼肌再生过程中的细胞外基质过度沉积有着重要作用。LEMOS等[57]的研究发现,在骨骼肌损伤后浸润性炎症细胞可促进纤维生成祖细胞的扩张,随后炎症性巨噬细胞通过分泌肿瘤坏死因子直接诱导纤维生成祖细胞的凋亡和清除,但在慢性损伤状态下产生转化生长因子β1交替激活的巨噬细胞能够诱导纤维化,从而导致纤维化的发生[58]。 综上所述,在SCs和纤维生成祖细胞的相互作用使纤维生成祖细胞有3种途径促进骨骼肌重建:①增殖扩张;②分化为成纤维细胞和脂肪细胞;③分化为成骨细胞和软骨细胞(图9)。"
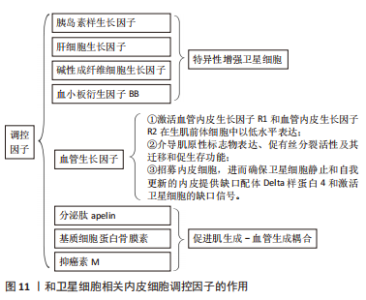
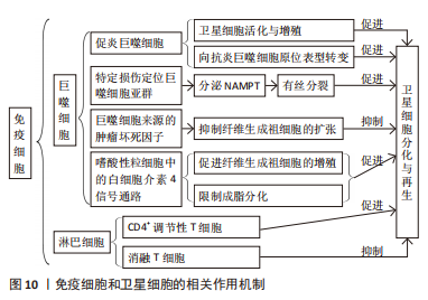
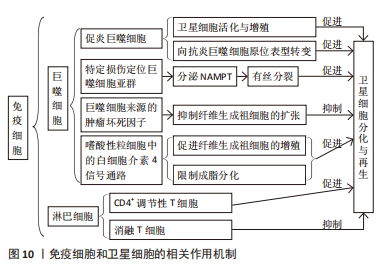
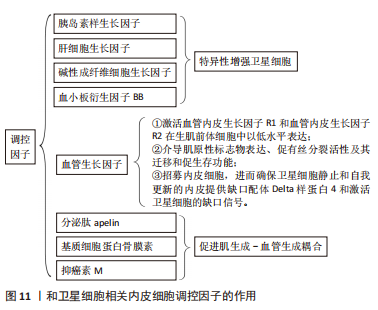
巨噬细胞是骨骼肌再生过程中最具代表性的免疫细胞群,其中促炎巨噬细胞促进SCs的活化和增殖[59],并且向抗炎巨噬细胞的原位表型转变刺激了SCs的分化和再生过程的修复阶段[60],故早期髓细胞可使用消融作用抑制肌肉再生,而晚期该细胞的消融作用较温和[61]。该两种巨噬细胞的炎症反应对于SCs的正常功能和再生具有不可或缺的作用。另有研究发现,部分信号分子和代谢物可能作为巨噬细胞作用于SCs活性的介质[53]。RATNAYAKE等[47]的研究发现了斑马鱼肌肉中一个特定的损伤定位巨噬细胞亚群,其在促进再生方面起短暂作用,且这些巨噬细胞分泌的烟酰胺磷酸核糖基转移酶向SCs中的CCR5受体发出信号并作为有丝分裂刺激巨噬细胞。另外,LEMOS等[57]发现髓系细胞也通过影响其他直接调节SCs功能的生态位信号群来间接影响SCs,巨噬细胞来源的肿瘤坏死因子抑制可纤维生成祖细胞的扩张。另外,HEREDIA等[62]发现嗜酸性粒细胞来源的白细胞介素4信号通路促进纤维生成祖细胞的增殖,同时限制它们的成脂分化,从而支持肌生成。BAHT等[63]则发现信号分子METRNl诱导了一种从促炎到抗炎的巨噬细胞转变,从而影响SCs并促进肌纤维的形成。 淋巴细胞亦可作为骨骼肌再生过程中SCs的相关生态位信号,其典型代表为CD4+调节性T细胞,同时也影响SCs在再生过程中的功能,其动力学遵循抗炎巨噬细胞,消融T细胞则抑制骨骼肌的自我修复作用[64]。T细胞以直接的方式促进成肌细胞分化,由表皮生长因子家族生长因子双向调节蛋白介导,并以间接的方式通过促进抗炎巨噬细胞转化[64]。 2.3.6 SCs和内皮细胞 骨骼肌高度血管化,SCs靠近毛细血管。在骨骼肌损伤后,内皮细胞和SCs平行增殖,肌纤维恢复并伴有新生血管。内皮细胞来源的因子通过其对SCs的作用直接支持血管再生[65]:①内皮细胞培养物通过胰岛素样生长因子、肝细胞生长因子、碱性成纤维细胞生长因子、血小板衍生因子BB和血管内皮生长因子特异性增强SCs;②血管内皮生长因子受体1和血管内皮生长因子受体2在生肌前体细胞中以低水平表达,并在血管内皮生长因子刺激后被强烈激活,其表达在骨骼肌分化过程中受到协调调节,而且血管内皮生长因子介导的肌原性标志物表达、促有丝分裂活性及其迁移和促生存功能可能有助于增加肌肉组织的生成(图11)。"
| [1] FUCHS E, BLAU HM. Tissue Stem Cells: Architects of Their Niches. Cell Stem Cell. 2020;27(4):532-556. [2] WEBSTER MT, MANOR U, LIPPINCOTT-SCHWARTZ J, et al. Intravital Imaging Reveals Ghost Fibers as Architectural Units Guiding Myogenic Progenitors during Regeneration. Cell Stem Cell. 2016;18(2):243-252. [3] MOYLE LA, CHENG RY, LIU H, et al. Three-dimensional niche stiffness synergizes with Wnt7a to modulate the extent of satellite cell symmetric self-renewal divisions. Mol Biol Cell. 2020;31(16):1703-1713. [4] CHANG NC, SINCENNES MC, CHEVALIER FP, et al. The Dystrophin Glycoprotein Complex Regulates the Epigenetic Activation of Muscle Stem Cell Commitment. Cell Stem Cell. 2018;22(5):755-768.e6. [5] 王屿萌,廖苾芝,周达岸.大鼠骨骼肌挫伤修复过程中p38MAPK通路、炎症反应的作用[J].中国老年学杂志,2021,41(19):4340-4344. [6] ROBINSON DCL, RITSO M, NELSON GM, et al. Negative elongation factor regulates muscle progenitor expansion for efficient myofiber repair and stem cell pool repopulation. Dev Cell. 2021;56(7):1014-1029.e7. [7] CHEN Z, BU N, QIAO X, et al. Forkhead Box M1 Transcriptionally Regulates the Expression of Long Noncoding RNAs Snhg8 and Gm26917 to Promote Proliferation and Survival of Muscle Satellite Cells. Stem Cells. 2018;36(7):1097-1108. [8] DIAO Y, GUO X, LI Y, et al. Pax3/7BP is a Pax7- and Pax3-binding protein that regulates the proliferation of muscle precursor cells by an epigenetic mechanism. Cell Stem Cell. 2012;11(2):231-241. [9] MASSENET J, GARDNER E, CHAZAUD B, et al. Epigenetic regulation of satellite cell fate during skeletal muscle regeneration. Skelet Muscle. 2021; 11(1):4. [10] PALA F, DI GIROLAMO D, MELLA S, et al. Distinct metabolic states govern skeletal muscle stem cell fates during prenatal and postnatal myogenesis. J Cell Sci. 2018; 131(14):jcs212977. [11] RYALL JG, DELL’ORSO S, DERFOUL A, et al. The NAD(+)-dependent SIRT1 deacetylase translates a metabolic switch into regulatory epigenetics in skeletal muscle stem cells. Cell Stem Cell. 2015;16(2):171-183. [12] SREENIVASAN K, RODRÍGUEZ-DELAROSA A, KIM J, et al. CHD4 ensures stem cell lineage fidelity during skeletal muscle regeneration. Stem Cell Reports. 2021;16(9):2089-2098. [13] SREENIVASAN K, IANNI A, KÜNNE C, et al. Attenuated Epigenetic Suppression of Muscle Stem Cell Necroptosis Is Required for Efficient Regeneration of Dystrophic Muscles. Cell Rep. 2020;31(7):107652. [14] CHEN X, YUAN J, XUE G, et al. Translational control by DHX36 binding to 5’UTR G-quadruplex is essential for muscle stem-cell regenerative functions. Nat Commun. 2021;12(1):5043. [15] TAYLOR MV, HUGHES SM. Mef2 and the skeletal muscle differentiation program. Semin Cell Dev Biol. 2017;72:33-44. [16] WOSCZYNA MN, PEREZ CARBAJAL EE, WAGNER MW, et al. Targeting microRNA-mediated gene repression limits adipogenic conversion of skeletal muscle mesenchymal stromal cells. Cell Stem Cell. 2021;28(7):1323-1334.e8. [17] 徐齐宇,王锋,张全兵,等.生肌调节因子在肌肉发育、发生和再生中的作用[J].中华物理医学与康复杂志,2021,43(6):571-576. [18] HORI S, HIRAMUKI Y, NISHIMURA D, et al. PDH-mediated metabolic flow is critical for skeletal muscle stem cell differentiation and myotube formation during regeneration in mice. FASEB J. 2019;33(7):8094-8109. [19] HOFFMANN C, HÖCKELE S, KAPPLER L, et al. The effect of differentiation and TGFβ on mitochondrial respiration and mitochondrial enzyme abundance in cultured primary human skeletal muscle cells. Sci Rep. 2018; 8(1):737. [20] LAUTAOJA JH, PEKKALA S, PASTERNACK A, et al. Differentiation of Murine C2C12 Myoblasts Strongly Reduces the Effects of Myostatin on Intracellular Signaling. Biomolecules. 2020;10(5):695. [21] YUCEL N, WANG YX, MAI T, et al. Glucose Metabolism Drives Histone Acetylation Landscape Transitions that Dictate Muscle Stem Cell Function. Cell Rep. 2019;27(13):3939-3955.e6. [22] SHINTAKU J, PETERSON JM, TALBERT EE, et al. MyoD Regulates Skeletal Muscle Oxidative Metabolism Cooperatively with Alternative NF-κB. Cell Rep. 2016;17(2):514-526. [23] MILLAY DP, O’ROURKE JR, SUTHERLAND LB, et al. Myomaker is a membrane activator of myoblast fusion and muscle formation. Nature. 2013;499(7458):301-305. [24] ZHANG Q, VASHISHT AA, O’ROURKE J, et al. The microprotein Minion controls cell fusion and muscle formation. Nat Commun. 2017;8:15664. [25] QUINN ME, GOH Q, KUROSAKA M, et al. Myomerger induces fusion of non-fusogenic cells and is required for skeletal muscle development. Nat Commun. 2017;8:15665. [26] TOMIDA T, ADACHI-AKAHANE S. Roles of p38 MAPK signaling in the skeletal muscle formation, regeneration, and pathology. Nihon Yakurigaku Zasshi. 2020;155(4):241-247. [27] 贺舟,常青,唐成林,等.大鼠骨骼肌急性损伤后早期运动训练和按摩对肌卫星细胞增殖相关因子的影响[J].中国康复理论与实践,2020, 26(1):49-54. [28] HU X, LI J, FU M, et al. The JAK/STAT signaling pathway: from bench to clinic. Signal Transduct Target Ther. 2021;6(1):402. [29] MANDL M, WAGNER SA, HATZMANN FM, et al. Sprouty1 Prevents Cellular Senescence Maintaining Proliferation and Differentiation Capacity of Human Adipose Stem/Progenitor Cells. J Gerontol A Biol Sci Med Sci. 2020; 75(12):2308-2319. [30] GARCÍA-PRAT L, PERDIGUERO E, ALONSO-MARTÍN S, et al. FoxO maintains a genuine muscle stem-cell quiescent state until geriatric age. Nat Cell Biol. 2020; 22(11):1307-1318. [31] ZHOU B, LIN W, LONG Y, et al. Notch signaling pathway: architecture, disease, and therapeutics. Signal Transduct Target Ther. 2022;7(1):95. [32] TROY A, CADWALLADER AB, FEDOROV Y, et al. Coordination of satellite cell activation and self-renewal by Par-complex-dependent asymmetric activation of p38α/β MAPK. Cell Stem Cell. 2012;11(4):541-553. [33] GUREVICH DB, NGUYEN PD, SIEGEL AL, et al. Asymmetric division of clonal muscle stem cells coordinates muscle regeneration in vivo. Science. 2016;353(6295):aad9969. [34] COSGROVE BD, GILBERT PM, PORPIGLIA E, et al. Rejuvenation of the muscle stem cell population restores strength to injured aged muscles. Nat Med. 2014; 20(3):255-264. [35] CHOI IY, LIM HT, CHE YH, et al. Inhibition of the Combinatorial Signaling of Transforming Growth Factor-Beta and NOTCH Promotes Myotube Formation of Human Pluripotent Stem Cell-Derived Skeletal Muscle Progenitor Cells. Cells. 2021;10(7):1649. [36] 赵莉娟,张宏,张国辉,等.Hippo信号通路在骨骼肌损伤修复中作用的研究进展[J].山东医药,2020,60(1):109-112. [37] EVANO B, KHALILIAN S, LE CARROU G, et al. Dynamics of Asymmetric and Symmetric Divisions of Muscle Stem Cells In Vivo and on Artificial Niches. Cell Rep. 2020;30(10):3195-3206.e7. [38] 陈炜,陈雨诗,刘禹熙,等.自噬对再生中肌肉干细胞的作用[J].中国细胞生物学学报,2022,44(3):484-491. [39] LI EW, MCKEE-MUIR OC, GILBERT PM. Cellular Biomechanics in Skeletal Muscle Regeneration. Curr Top Dev Biol. 2018;126:125-176. [40] SAMPATH SC, SAMPATH SC, HO ATV, et al. Induction of muscle stem cell quiescence by the secreted niche factor Oncostatin M. Nat Commun. 2018;9(1):1531. [41] ELIAZER S, MUNCIE JM, CHRISTENSEN J, et al. Wnt4 from the Niche Controls the Mechano-Properties and Quiescent State of Muscle Stem Cells. Cell Stem Cell. 2019;25(5):654-665.e4. [42] BRIGHTWELL CR, LATHAM CM, THOMAS NT, et al. A glitch in the matrix: the pivotal role for extracellular matrix remodeling during muscle hypertrophy. Am J Physiol Cell Physiol. 2022;323(3):C763-C771. [43] ROZO M, LI L, FAN CM. Targeting β1-integrin signaling enhances regeneration in aged and dystrophic muscle in mice. Nat Med. 2016;22(8):889-896. [44] 李伦宇,靳松林,黄增浩,等.肌卫星细胞自我更新及其信号调控的应用热点及问题[J].中国组织工程研究,2021,25(2):304-310. [45] TIERNEY MT, GROMOVA A, SESILLO FB, et al. Autonomous Extracellular Matrix Remodeling Controls a Progressive Adaptation in Muscle Stem Cell Regenerative Capacity during Development. Cell Rep. 2016;14(8):1940-1952. [46] TAKENAKA-NINAGAWA N, KIM J, ZHAO M, et al. Collagen-VI supplementation by cell transplantation improves muscle regeneration in Ullrich congenital muscular dystrophy model mice. Stem Cell Res Ther. 2021;12(1):446. [47] RATNAYAKE D, NGUYEN PD, ROSSELLO FJ, et al. Macrophages provide a transient muscle stem cell niche via NAMPT secretion. Nature. 2021; 591(7849):281-287. [48] LIU W, CHAKKALAKAL JV. The Composition, Development, and Regeneration of Neuromuscular Junctions. Curr Top Dev Biol. 2018;126:99-124. [49] MADARO L, PASSAFARO M, SALA D, et al. Denervation-activated STAT3-IL-6 signalling in fibro-adipogenic progenitors promotes myofibres atrophy and fibrosis. Nat Cell Biol. 2018;20(8):917-927. [50] LIU L, YUE X, SUN Z, et al. Reduction of senescent fibro-adipogenic progenitors in progeria-aged muscle by senolytics rescues the function of muscle stem cells. J Cachexia Sarcopenia Muscle. 2022;13(6):3137-3148. [51] BRITTO FA, GNIMASSOU O, DE GROOTE E, et al. Acute environmental hypoxia potentiates satellite cell-dependent myogenesis in response to resistance exercise through the inflammation pathway in human. FASEB J. 2020;34(1):1885-1900. [52] ARCHACKA K, GRABOWSKA I, MIERZEJEWSKI B, et al. Hypoxia preconditioned bone marrow-derived mesenchymal stromal/stem cells enhance myoblast fusion and skeletal muscle regeneration. Stem Cell Res Ther. 2021;12(1):448.dPMC8351116. [53] SHANG M, CAPPELLESSO F, AMORIM R, et al. Macrophage-derived glutamine boosts satellite cells and muscle regeneration. Nature. 2020; 587(7835):626-631. [54] CHEN A, TANG S, HE J, et al. Small extracellular vesicles from human adipose-derived mesenchymal stromal cells: a potential promoter of fat graft survival. Stem Cell Res Ther. 2021;12(1):263. [55] KOPINKE D, ROBERSON EC, REITER JF. Ciliary Hedgehog Signaling Restricts Injury-Induced Adipogenesis. Cell. 2017;170(2):340-351.e12. [56] MALECOVA B, GATTO S, ETXANIZ U, et al. Dynamics of cellular states of fibro-adipogenic progenitors during myogenesis and muscular dystrophy. Nat Commun. 2018;9(1):3670. [57] LEMOS DR, BABAEIJANDAGHI F, LOW M, et al. Nilotinib reduces muscle fibrosis in chronic muscle injury by promoting TNF-mediated apoptosis of fibro/adipogenic progenitors. Nat Med. 2015;21(7):786-794. [58] DE OLIVEIRA RC, WILSON SE. Fibrocytes, Wound Healing, and Corneal Fibrosis. Invest Ophthalmol Vis Sci. 2020;61(2):28. [59] 王伦平,陈蕊,严金川,等.巨噬细胞在不同组织再生中的研究进展[J].中国病理生理杂志,2021,37(3):565-570. [60] 张子涵,杨欢,黄庆生,等.细胞因子介导的骨骼肌系统与免疫系统间调控网络[J].医用生物力学,2022,37(2):374-378. [61] 程建红,洪莉,洪莎莎,等.巨噬细胞在骨骼肌损伤再生中的研究进展[J].医学综述,2021,27(16):3125-3129. [62] HEREDIA JE, MUKUNDAN L, CHEN FM, et al. Type 2 innate signals stimulate fibro/adipogenic progenitors to facilitate muscle regeneration. Cell. 2013; 153(2):376-388. [63] BAHT GS, BAREJA A, LEE DE, et al. Meteorin-like facilitates skeletal muscle repair through a Stat3/IGF-1 mechanism. Nat Metab. 2020;2(3):278-289. [64] BURZYN D, KUSWANTO W, KOLODIN D, et al. A special population of regulatory T cells potentiates muscle repair. Cell. 2013;155(6):1282-1295. [65] 彭勇,孙飙,戴剑松.调节性T细胞影响骨骼肌损伤再生能力机制的研究进展[J].生命的化学,2020,40(11):2053-2061. [66] KOIKE H, MANABE I, OISHI Y. Mechanisms of cooperative cell-cell interactions in skeletal muscle regeneration. Inflamm Regen. 2022;42(1):48. |
| [1] | Yang Yifeng, Huang Jian, Ye Nan, Wang Lin. Ischemia-reperfusion injury in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 955-960. |
| [2] | Liu Zhiyang, Fu Zeting, Xia Yu, Ding Haili. The role of BMAL1 and MyoD in exercise-induced skeletal muscle damage [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 510-515. |
| [3] | Zhu Xiaofeng, Chen Weiwei, Huang Jian. Effects of maternal high-fat diet and exercise intervention on insulin sensitivity and the hypothalamic arcuate nucleus in male offspring mice [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 556-561. |
| [4] | Xie Peng, Zhang Jiang, Deng Xiaolei, Wei Bo, Hou Decai. A systematic review of mouse model construction for sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 263-266. |
| [5] | Wang Jingfeng, Wen Dengtai, Wang Shijie, Gao Yinghui. Atg-mediated autophagy, exercise and skeletal muscle aging [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 295-301. |
| [6] | Long Yi, Yang Jiaming, Ye Hua, Zhong Yanbiao, Wang Maoyuan. Extracellular vesicles in sarcopenic obesity: roles and mechanisms [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 315-320. |
| [7] | Zhang Yan, He Ruibo, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Ma Gang, Peng Peng. Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1237-1244. |
| [8] | Zhang Qijian, Xu Ximing. Acquisition and application of ectodermal mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 928-934. |
| [9] | Liu Hongwen, Li Jiao, Xu Wenhao, Nie Hua, Liu Shaojiang, Xu Jie, Yin Li. Differential expression profiles of microRNAs in muscle tissue of denervated skeletal muscle atrophy rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 732-737. |
| [10] | Li Yutong, Liu Jingshu, Li Zhen. Immunoregulatory mechanism of choline acetyltransferase/alpha 7 nicotinic acetylcholine receptor/nuclear factor-kappa B signaling pathway in the pathogenesis of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(35): 5596-5602. |
| [11] | Meng Meng, Hu Guanyu, Wu Xingquan, Cong Deyu. Effects of skeletal muscle massage on skeletal muscle function and conversion of skeletal muscle fiber types in type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(35): 5628-5633. |
| [12] | Meng Xiangli, Lyu Xiaohong. Effects of eccentric exercise patterns on exercise-induced muscle damage [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(32): 5132-5136. |
| [13] | Zhao Zheng, Ding Wenfei, Shao Shuxin, Wang Jinyan, Jian Xigao. Degradation and drug release behavior of sirolimus-poly(trimethylene carbonate) modified magnesium alloy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(30): 4836-4843. |
| [14] | Chen Chao, Wang Xuenan, Zhan Enyu, Lyu Zhengpin, Zhang Fan. Mechanism underlying the mutual regulation between circadian rhythm and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(29): 4696-4703. |
| [15] | Deng Moyuan, Peng Kun. Role and regulation of macrophages in biomaterial-mediated fibrosis formation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(25): 4085-4092. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||