Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (8): 1237-1244.doi: 10.12307/2023.055
Previous Articles Next Articles
Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats
Zhang Yan1, He Ruibo2, Wang Qingbo1, Pi Yihua1, Lu Chunmin1, Xu Chuanyi1, Ma Gang2, Peng Peng2
- 1Department of Physical Education, Guangxi University of Chinese Medicine, Nanning 530021, Guangxi Zhuang Autonomous Region, China; 2Department of Health Service, Logistics University of Chinese People’s Armed Police Forces, Tianjin 300309, China
-
Received:2022-02-07Accepted:2022-03-12Online:2023-03-18Published:2022-07-28 -
Contact:Peng Peng, MD, Lecturer, Department of Health Service, Logistics University of Chinese People’s Armed Police Forces, Tianjin 300309, China -
About author:Zhang Yan, Master, Associate professor, Department of Physical Education, Guangxi University of Chinese Medicine, Nanning 530021, Guangxi Zhuang Autonomous Region, China -
Supported by:the Natural Science Foundation of Tianjin, No. 17JCYBJC27400 (to PP); the Basic Research Project of Logistics University of Chinese People’s Armed Police Forces, No. WHY202107 (to PP)
CLC Number:
Cite this article
Zhang Yan, He Ruibo, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Ma Gang, Peng Peng. Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1237-1244.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
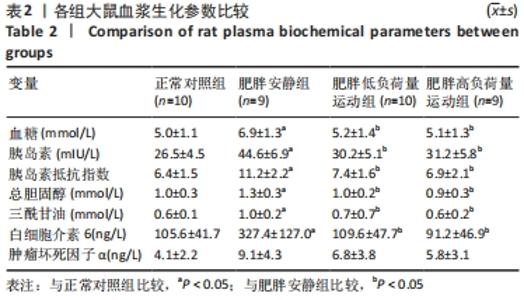
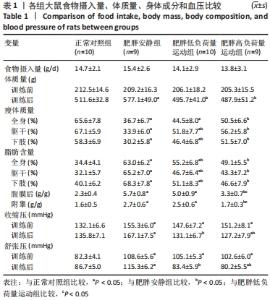
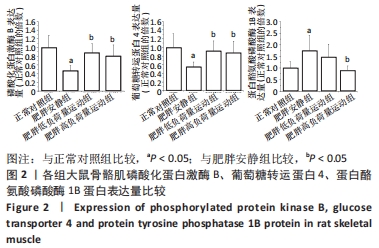
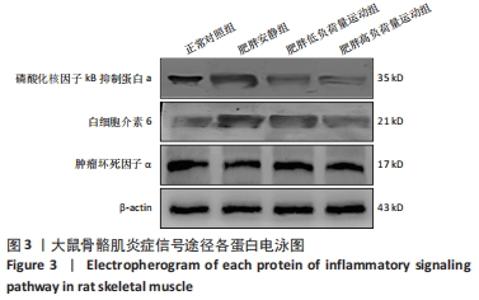
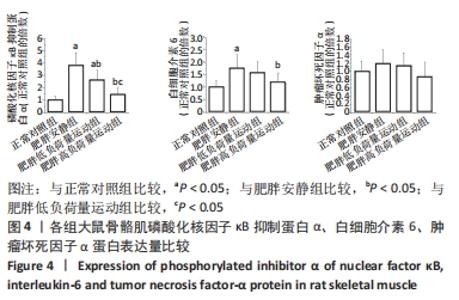
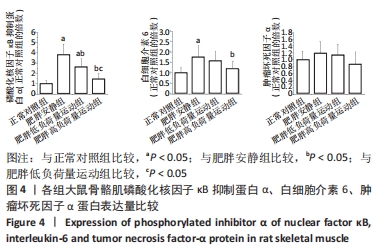
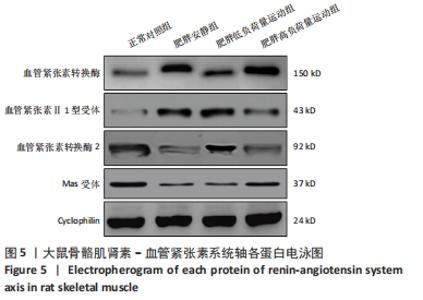
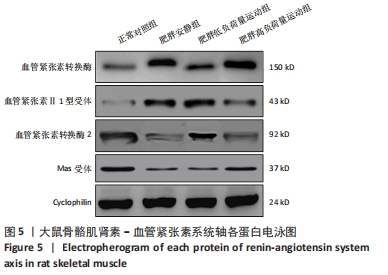
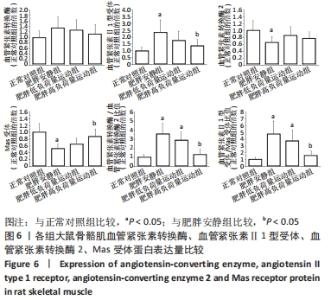
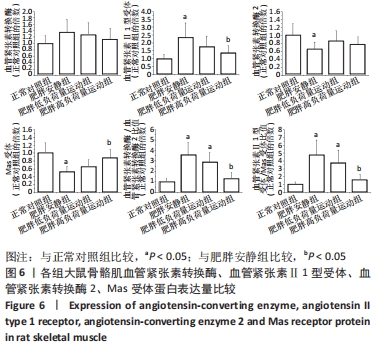
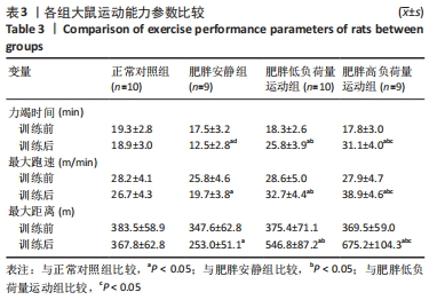
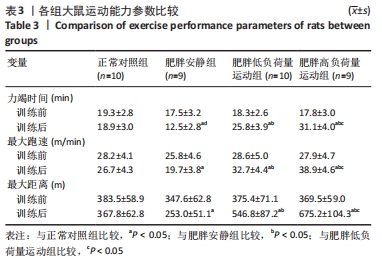
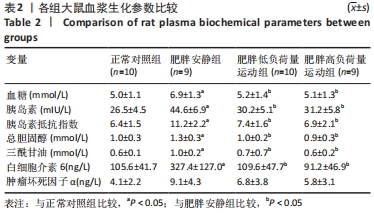
食物摄入量:各组食物摄入量比较差异无显著性意义(P > 0.05)。 体质量:训练前,各组体质量比较无显著性意义(P > 0.05);训练后,各组体质量均较训练前增加(P < 0.05),肥胖安静组体质量高于正常对照组(P < 0.05),2个运动组体质量低于肥胖安静组(P < 0.05)。 瘦体质量:与正常对照组比较,肥胖安静组和肥胖低负荷量运动组全身、躯干和下肢瘦体质量下降(P < 0.05);与肥胖安静组比较,肥胖低负荷量运动组躯干和下肢瘦体质量增加(P < 0.05),肥胖高负荷量运动组全身、躯干和下肢瘦体质量增加(P < 0.05)。 脂肪含量:与正常对照组比较,肥胖安静组和肥胖低负荷量运动组全身、躯干、下肢、腹膜后和附睾脂肪含量升高(P < 0.05);与肥胖安静组比较,肥胖低负荷量运动组全身、躯干和下肢脂肪含量下降(P < 0.05),肥胖高负荷量运动组全身以及各部位脂肪含量均降低(P < 0.05);与肥胖低负荷量运动组比较,肥胖高负荷量运动组腹膜后和附睾脂肪含量下降(P < 0.05)。 血压:训练前组间比较,肥胖安静组、肥胖低负荷量运动组和肥胖高负荷量运动组收缩压、舒张压均高于正常对照组(P < 0.05);训练后组内比较,肥胖安静组收缩压、舒张压高于训练前(P < 0.05),2个运动组收缩压、舒张压均低于训练前(P < 0.05);训练后组间比较,肥胖安静组收缩压、舒张压高于正常对照组(P < 0.05),2个运动组收缩压、舒张压低于肥胖安静组(P < 0.05),肥胖高负荷量运动组收缩压、舒张压低于正常对照组(P < 0.05)。 2.3 血浆生化参数 与正常对照组比较,肥胖安静组血糖、胰岛素、胰岛素抵抗指数、总胆固醇、三酰甘油和白细胞介素6水平升高(P < 0.05);与肥胖安静组比较,2个运动组血糖、胰岛素、胰岛素抵抗指数、三酰甘油、总胆固醇和白细胞介素6水平下降(P < 0.05),2个运动组间各生化参数比较差异无显著性意义(P > 0.05);肿瘤坏死因子α表达在各组间比较差异无显著性意义(P > 0.05),见表2。 "
| [1] DONFRANCESCO C, PROFUMO E, LO NOCE C, et al. Trends of overweight, obesity and anthropometric measurements among the adult population in Italy: The CUORE Project health examination surveys 1998, 2008, and 2018. PLoS One. 2022;17(3):e0264778. [2] KACHUR S, LAVIE CJ, DE SCHUTTER A, et al. Obesity and cardiovascular diseases. Minerva Med. 2017;108(3):212-228. [3] WEI Y, SOWERS JR, CLARK SE, et al. Angiotensin II-induced skeletal muscle insulin resistance mediated by NF-kappaB activation via NADPH oxidase. Am J Physiol Endocrinol Metab. 2008;294(2):E345-E351. [4] DI MEO S, IOSSA S, VENDITTI P. Improvement of obesity-linked skeletal muscle insulin resistance by strength and endurance training. J Endocrinol. 2017;234(3):R159-R181. [5] PATEL S, RAUF A, KHAN H, et al. Renin-angiotensin-aldosterone (RAAS): the ubiquitous system for homeostasis and pathologies. Biomed Pharmacother. 2017;94:317-325. [6] POWERS SK, MORTON AB, HYATT H, et al. The renin-angiotensin system and skeletal muscle. Exerc Sport Sci Rev. 2018;46(4):205-214. [7] YANG T, XU C. Physiology and pathophysiology of the intrarenal renin-angiotensin system: an update. J Am Soc Nephrol. 2017;28(4):1040-1049. [8] PAZ OCARANZA M, RIQUELME JA, GARCÍA L, et al. Counter-regulatory renin-angiotensin system in cardiovascular disease. Nat Rev Cardiol. 2020;17(2):116-129. [9] YAMAMOTO K, TAKESHITA H, RAKUGI H. ACE2, angiotensin-(1-7) and skeletal muscle: review in the era of COVID-19. Clin Sci (Lond). 2020; 134(22):3047-3062. [10] SHEIKH AM, VISSING J. Exercise therapy for muscle and lower motor neuron diseases. Acta Myol. 2019;38(4):215-232. [11] SAKAMOTO M. Effects of physical agents on muscle healing with a focus on animal model research. Phys Ther Res. 2021;24(1):1-8. [12] GOMES-SANTOS IL, FERNANDES T, COUTO GK, et al. Effects of exercise training on circulating and skeletal muscle renin-angiotensin system in chronic heart failure rats. PLoS One. 2014;9(5):e98012. [13] THOMPSON PD, ARENA R, RIEBE D, et al. ACSM’s new preparticipation health screening recommendations from ACSM’s guidelines for exercise testing and prescription, ninth edition. Curr Sports Med Rep. 2013;12(4):215-217. [14] WEN CP, WAI JP, TSAI MK, et al. Minimum amount of physical activity for reduced mortality and extended life expectancy: a prospective cohort study. Lancet. 2011;378(9798):1244-1253. [15] FLACK KD, HAYS HM, MORELAND J, et al. Exercise for weight loss: further evaluating energy compensation with exercise. Med Sci Sports Exerc. 2020;52(11):2466-2475. [16] NASCIMENTO AR, MACHADO M, DE JESUS N, et al. Structural and functional microvascular alterations in a rat model of metabolic syndrome induced by a high-fat diet. Obesity (Silver Spring). 2013; 21(10):2046-2054. [17] 范朋琦, 秦永生, 彭朋. 不同运动方式对自发性高血压大鼠心脏重塑和运动能力的影响[J]. 现代预防医学,2018,45(23):4341-4345. [18] 孟宪欣, 管泽毅, 葛吉生, 等. 间歇运动干预自发性高血压大鼠病理性心脏肥大:运动强度与健康效应的关系[J]. 体育科学,2019, 39(6):73-82. [19] MATTHEWS DR, HOSKER JP, RUDENSKI AS, et al. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985; 28(7):412-419. [20] SILVA DM, GOMES-FILHO A, OLIVON VC, et al. Swimming training improves the vasodilator effect of angiotensin-(1-7) in the aorta of spontaneously hypertensive rat. J Appl Physiol (1985). 2011;111(5): 1272-1277. [21] FILHO AG, FERREIRA AJ, SANTOS SH, et al. Selective increase of angiotensin(1-7) and its receptor in hearts of spontaneously hypertensive rats subjected to physical training. Exp Physiol. 2008;93(5):589-598. [22] CHASTON TB, DIXON JB. Factors associated with percent change in visceral versus subcutaneous abdominal fat during weight loss: findings from a systematic review. Int J Obes (Lond). 2008;32(4):619-628. [23] PETRIDOU A, SIOPI A, MOUGIOS V. Exercise in the management of obesity. Metabolism. 2019;92:163-169. [24] WESTERTERP KR. Exercise, energy balance and body composition. Eur J Clin Nutr. 2018;72(9):1246-1250. [25] WEWEGE MA, THOM JM, RYE KA, et al. Aerobic, resistance or combined training: A systematic review and meta-analysis of exercise to reduce cardiovascular risk in adults with metabolic syndrome. Atherosclerosis. 2018;274:162-171. [26] ZACHAREWICZ E, HESSELINK M, SCHRAUWEN P. Exercise counteracts lipotoxicity by improving lipid turnover and lipid droplet quality. J Intern Med. 2018;284(5):505-518. [27] SVEGLIATI-BARONI G, PIERANTONELLI I, TORQUATO P, et al. Lipidomic biomarkers and mechanisms of lipotoxicity in non-alcoholic fatty liver disease. Free Radic Biol Med. 2019;144:293-309. [28] Baek KW, Cha HJ, Ock MS, et al. Effects of regular-moderate exercise on high-fat diet-induced intramyocellular lipid accumulation in the soleus muscle of Sprague-Dawley rats. J Exerc Rehabil. 2018;14(1):32-38. [29] LI C, CULVER SA, QUADRI S, et al. High-fat diet amplifies renal renin angiotensin system expression, blood pressure elevation, and renal dysfunction caused by Ceacam1 null deletion. Am J Physiol Endocrinol Metab. 2015;309(9):E802-810. [30] SCHÜTTEN MT, HOUBEN AJ, DE LEEUW PW, et al. The link between adipose tissue renin-angiotensin-aldosterone system signaling and obesity-associated hypertension. Physiology (Bethesda). 2017;32(3): 197-209. [31] ALMEIDA JA, MOTTA-SANTOS D, PETRIZ BA, et al. High-intensity aerobic training lowers blood pressure and modulates the renal renin-angiotensin system in spontaneously hypertensive rats. Clin Exp Hypertens. 2020;42(3):233-238. [32] ENDLICH PW, CLAUDIO ER, LIMA LC, et al. Exercise modulates the aortic renin-angiotensin system independently of estrogen therapy in ovariectomized hypertensive rats. Peptides. 2017;87:41-49. [33] WEI Y, SOWERS JR, NISTALA R, et al. Angiotensin II-induced NADPH oxidase activation impairs insulin signaling in skeletal muscle cells. J Biol Chem. 2006;281(46):35137-35146. [34] YOON KJ, ZHANG D, KIM SJ, et al. Exercise-induced AMPK activation is involved in delay of skeletal muscle senescence. Biochem Biophys Res Commun. 2019;512(3):604-610. [35] SYLOW L, KLEINERT M, RICHTER EA, et al. Exercise-stimulated glucose uptake - regulation and implications for glycaemic control. Nat Rev Endocrinol. 2017;13(3):133-148. [36] ELEFTHERIOU P, GERONIKAKI A, PETROU A. PTP1B inhibition, a promising approach for the treatment of diabetes type II. Curr Top Med Chem. 2019;19(4):246-263. [37] ROPELLE ER, PAULI JR, PRADA PO, et al. Reversal of diet-induced insulin resistance with a single bout of exercise in the rat: the role of PTP1B and IRS-1 serine phosphorylation. J Physiol. 2006;577(Pt 3):997-1007. [38] MEINEN S, LIN S, RUEGG MA. Angiotensin II type 1 receptor antagonists alleviate muscle pathology in the mouse model for laminin-α2-deficient congenital muscular dystrophy (MDC1A). Skelet Muscle. 2012;2(1):18-26. [39] KUNNUMAKKARA AB, SHABNAM B, GIRISA S, et al. Inflammation, NF-κB, and chronic diseases: how are they linked? Crit Rev Immunol. 2020; 40(1):1-39. [40] TIFTIK RN, TEMIZ-REŞITOĞLU M, GÜDEN DS, et al. Involvement of Rho-kinase/IκB-α/NF-κB activation in IL-1β-induced inflammatory response and oxidative stress in human chondrocytes. Can J Physiol Pharmacol. 2021;99(4):418-426. [41] MORALES MG, ABRIGO J, ACUÑA MJ, et al. Angiotensin-(1-7) attenuates disuse skeletal muscle atrophy in mice via its receptor, Mas. Dis Model Mech. 2016;9(4):441-449. |
| [1] | Ruan Ling, Wang Guanghua, Wu Rongping, Jin Zhan, Lyu Zhenqing, Zhang Nan, Li Shoubang. Correlation between exercise intensity and lipid metabolism disorder and oxidative stress in a high-diet rat model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1149-1155. |
| [2] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [3] | Liu Hongwen, Li Jiao, Xu Wenhao, Nie Hua, Liu Shaojiang, Xu Jie, Yin Li. Differential expression profiles of microRNAs in muscle tissue of denervated skeletal muscle atrophy rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 732-737. |
| [4] | Shui Xiaoping, Li Chunying, Li Shunchang, Sun Junzhi, Su Quansheng . Effects of aerobic and resistance exercises on brain-derived neurotrophic factor, nuclear factor-kappa B and inflammatory cytokines in skeletal muscle of type II diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 669-675. |
| [5] | Lin Jiayu, Huang Huibin, Liang Bo, Chen Lijun. Changes in irisin, leptin, adiponectin and visceral fat in obese rats undergoing high-intensity intermittent exercise [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(35): 5583-5588. |
| [6] | Xiao Youding, Gao Qianjin, Wang Erli. Skeletal muscle function and exercise regulating cognitive function of the older adults [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(33): 5400-5406. |
| [7] | Sun Ying, Xiang Guangda, Xu Xiaoli. Effects of myeloid-derived growth factor on ventricular remodeling in aging mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 5020-5025. |
| [8] | Ye Hua, Yang Jiaming, Zhang Jiahong, Niu Yanlong, Wang Maoyuan. Biological role of exosomes in denervated muscle atrophy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 5062-5068. |
| [9] | Deng Xiaohui, Zhang Zengzeng, Zhang Zhihua, Zhu Lingyan. Mechanism and application prospects of mesenchymal stem cell exosomes gene-modified microRNA in the treatment of diabetic foot [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 5076-5084. |
| [10] | Wu Juhua, Yang Yanan, Weng Xiquan, Zhao Fangfang, Xu Guoqin, Lin Wenta. Effects of hypoxic exercise on skeletal muscle energy metabolism of rats with alimentary obesity [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(29): 4598-4604. |
| [11] | Fu Yu, Shang Huayu, Li Shunchang. Aerobic and resistance exercises can alleviate liver inflammation in type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(29): 4666-4671. |
| [12] | Yang Yi, Wang Kun, Liu Hengxu, Zhang Tingran, Lu Wenyun, Chen Peijie, Luo Jiong. Aerobic exercise improves the cognitive function of elderly patients with mild cognitive impairment [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(29): 4716-4722. |
| [13] | Liu Ya, Liu Xia, Deng Penghui, Ji Wei, Li Jianping. Exercise effects on myocardial type I, III collagen and angiotensin II/transforming growth factor beta1/Smad2 pathway in diabetic myocardial fibrosis rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(26): 4173-4179. |
| [14] | Yang Jinxin, Wang Kun, Zhao Jing, Chen Peijie, Zhang Tingran, Lu Wenyun, Luo Jiong. Effects of aerobic exercise intervention on synaptic plasticity in Alzheimer’s disease patients [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(26): 4216-4223. |
| [15] | Chen Chichi, Zhang Yu, He Jiachen, Shi Qin. Osteogenic differentiation of bone marrow mesenchymal stem cells in obese mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3846-3851. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||