Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (31): 5062-5068.doi: 10.12307/2022.782
Previous Articles Next Articles
Biological role of exosomes in denervated muscle atrophy
Ye Hua1, Yang Jiaming1, Zhang Jiahong2, Niu Yanlong1, 3, Wang Maoyuan1, 2, 3
- 1Gannan Medical University, Ganzhou 341000, Jiangxi Province, China; 2Department of Rehabilitation Medicine, First Affiliated Hospital of Gannan Medical University, Ganzhou 341000, Jiangxi Province, China; 3Ganzhou Key Laboratory of Rehabilitation Medicine, Ganzhou 341000, Jiangxi Province, China
-
Received:2021-12-02Accepted:2021-12-28Online:2022-11-08Published:2022-04-25 -
Contact:Wang Maoyuan, MD, Professor, Gannan Medical University, Ganzhou 341000, Jiangxi Province, China; Department of Rehabilitation Medicine, First Affiliated Hospital of Gannan Medical University, Ganzhou 341000, Jiangxi Province, China; Ganzhou Key Laboratory of Rehabilitation Medicine, Ganzhou 341000, Jiangxi Province, China -
About author:Ye Hua, Master candidate, Gannan Medical University, Ganzhou 341000, Jiangxi Province, China -
Supported by:National Natural Science Foundation of China, No. 82060420 (to WMY)
CLC Number:
Cite this article
Ye Hua, Yang Jiaming, Zhang Jiahong, Niu Yanlong, Wang Maoyuan. Biological role of exosomes in denervated muscle atrophy[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 5062-5068.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
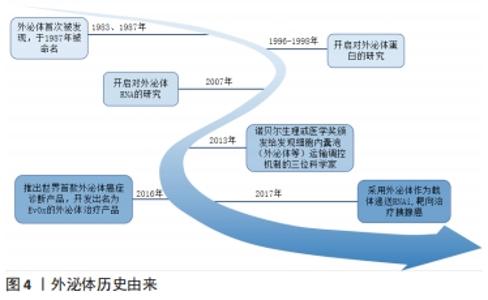
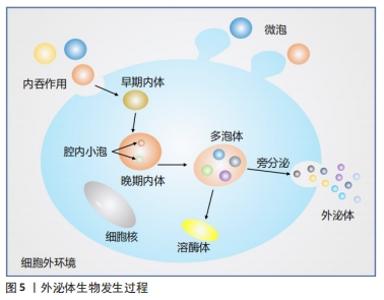
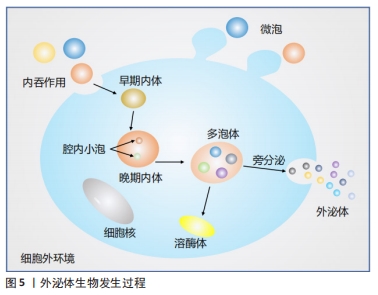
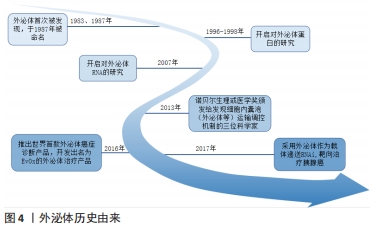
外泌体是晚期内体或多泡体(multivesicular bodies,MVBs)与质膜融合后释放到细胞外环境的囊泡,1983年首次在大鼠网织红细胞中观察到[9],该领域的先驱ROSE JOHNSTONE 在1987年以“外泌体”一词来命名,近年来对这一过程有了进一步了解[10]。内体分选复合体是外泌体合成释放的重要机制,由内体分选复合体0、内体分选复合体Ⅰ、内体分选复合体Ⅱ、内体分选复合体Ⅲ,以及相关的ATP酶、Vps4复合物组成[11],内体分选复合体0识别泛素化物质,内体分选复合体Ⅰ、Ⅱ、Ⅲ参与腔内小泡出芽,Vps4复合物确保最终的膜断裂和(或)内体分选复合体的回收。然而STUFFERS等[12]证明多泡体的生物发生可以在没内体分选复合体的情况下发生。关于外泌体的历史由来,见图4;外泌体生物发生过程,见图5。 "
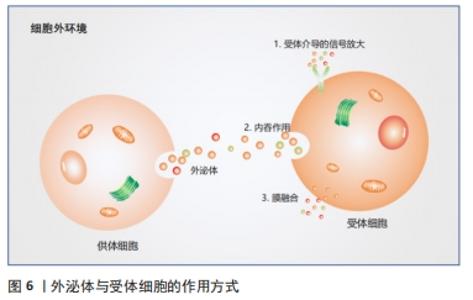
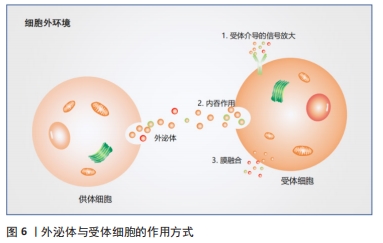
2.1.2 外泌体的内容物 外泌体含有多种内容物,包括蛋白质、脂质、mRNA、线粒体DNA、microRNA(miRNA)和其他非编码RNA[20]。根据蛋白质组学分析,外泌体不仅含有分泌细胞种类的特异性蛋白质,而且还有与分泌细胞无关的固定蛋白质集合,这些蛋白质参与细胞发育的一些基本过程,如细胞的黏附、膜相互融合、代谢和信号转导[21]。另外还含有细胞因子、转录因子受体、生长因子受体和其他生物活性分子[22]。蛋白质分选为外泌体内容物的机制尚未得到广泛研究。但该过程有2种主要途径:一种是依赖于内体分选复合体[23],另一种是独立于内体分选复合体[24]。2007年,第1次发现细胞分泌的外泌体含有mRNA和miRNA[25],RNA与蛋白质一样,有一部分为分泌细胞类型的特异性RNA,而有一部分RNA无论细胞来源如何都是固定的[26]。研究人员发现具有3’端非翻译区(3’ -UTR)的mRNA可以通过特定的RNA片段被包装到外泌体中[27]。同样,有1个四核苷酸序列可以诱导miRNA包装到外泌体中,并被核糖核蛋白识别[28]。另一种类似的RNA结合蛋白SYNCRIP可以控制mRNA分选到外泌体中[29],这种蛋白质通过识别特定的序列来结合特定的RNA,并将它们加载到外泌体中。研究表明,外泌体发挥功能的一个重要途径就是运输miRNA,从而对其靶细胞mRNA的3’端非翻译区进行功能调节,其核心是将初级转录产物加工成活性的miRNAs,这些miRNAs可以直接在受体细胞中发挥作用(只发生在哺乳动物细胞中),miRNA加工的关键成分Disher和Ago2存在于外泌体中[30]。这些发现表明外泌体不仅是一个被动的加工载体,更是一个miRNA工厂。与外泌体来源细胞相比,外泌体富含胆固醇、膜磷脂和鞘磷脂[31],这表明外泌体膜可能含有脂筏、富含胆固醇的膜亚结构域和在信号传递和分选中起重要作用的鞘磷脂[32-34]。 2.1.3 细胞间通讯 外泌体是一个天然的载体系统,能够以自分泌、旁分泌和内分泌的方式作用于受体细胞,转移供体细胞的核酸、蛋白质和脂质,受体细胞和外泌体之间的信号传递主要通过3种方式来进行:与膜上的受体蛋白相互识别、直接膜融合和内吞作用[10],见图6。此外还有对外泌体和受体细胞之间跨膜信号转导的途径进行研究[35],如外泌体通过受体-配体相互作用促进神经干细胞或前体细胞与微环境之间的交流[36]。首先,游离干扰素γ与细胞外囊泡相关的干扰素γ受体1结合形成干扰素γ/干扰素γ受体1复合物;然后,细胞外囊泡相关复合物通过靶细胞中的信号转导与转录激活因子1 (signal transducerand activator of transcription 1,STAT1)通路激活信号转导。外泌体的内化取决于细胞类型和外泌体表面蛋白[37]。已经发现许多促进外泌体摄取的蛋白质,例如四跨膜蛋白CD9 和CD81 以及细胞间黏附分子1[38-39],硫酸乙酰肝素蛋白聚糖被癌细胞衍生的外泌体用作内化受体[40]。外泌体的大小分布也会影响它们内化,受体细胞优先摄取较小的外泌体[41]。"
2.2 外泌体与骨骼肌 2.2.1 外泌体与骨骼肌生成 外泌体被认为是在肌细胞和其他细胞之间传递细胞信号的载体,导致靶细胞表型发生变化[42],参与骨骼肌的生成过程,见表1。2010年的一项研究证明C2C12细胞可以释放携带线粒体DNA和一些功能相关蛋白的外泌体,推测线粒体DNA可以通过外泌体途径进入靶细胞的线粒体,将信号传递给靶细胞[43]。小鼠高脂肪饮食诱导骨骼肌释放外泌体,诱导成肌细胞增殖并参与改变体外肌细胞周期和分化调节基因的表达[44]。在去除外泌体的血清中培养C2C12细胞和成肌细胞形成的肌管少于在正常血清中培养形成的肌管,同时肌生长抑制素水平升高和肌球蛋白表达降低,表明骨骼肌分泌的外泌体可能包含肌生成的特定生化信号,参与肌肉生成过程[45]。"
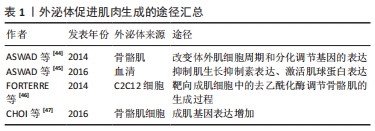
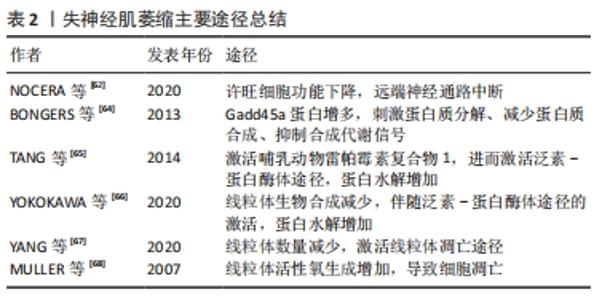
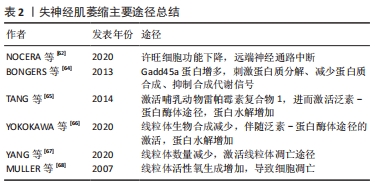
从C2C12成肌细胞和肌管释放的外泌体miRNA库显示出特异性的miRNAs子集,肌管分泌的外泌体miRNAs靶向成肌细胞中的组蛋白去乙酰化酶以调节骨骼肌的生成过程[46]。来自人类骨骼肌细胞的外泌体富含各种生肌因子显著诱导人脂肪源性干细胞的肌源性分化,成肌基因在人脂肪源性干细胞中表达增加[47]。C2C12细胞分泌的外泌体可促进人脐静脉内皮细胞的增殖[48],C2C12细胞来源外泌体促进运动神经元细胞系NSC-34的存活和突起生长[49],显示出肌肉特异性外泌体的旁分泌作用。 2.2.2 外泌体介导骨骼肌萎缩过程 肌纤维的大小取决于新旧蛋白的合成与降解之间的平衡。病理状态下,肌纤维蛋白降解加快,蛋白质合成受到抑制,导致肌肉无力、疲劳,甚至发生萎缩。肌肉中蛋白质的降解主要通过以下2种途径:①泛素-蛋白酶体途径,这一途径在肌肉萎缩过程中起主要作用[50],骨骼肌中最具有特征性的E3泛素连接酶:肌肉萎缩盒基因(MAFbx/Atrogin-1)和肌肉环状指基因1(MuRF1),它们导致蛋白质泛素化然后被蛋白酶体降解。②自噬-溶酶体途径,多种表现为萎缩和营养不良的骨骼肌疾病都与自噬活性降低有关[51-52]。叉头盒转录因子(forkhead box O,FoxO)可以通过调节肌肉萎缩盒基因和肌肉环状指基因1来提高泛素-蛋白酶体和自噬-溶酶体蛋白水解系统降解蛋白表达[53],HUDSON等[54]证明miR-182可阻碍叉头盒转录因子3a靶基因的表达来抑制肌肉萎缩的过程。同年,他们发现在肌肉萎缩条件下,miR-23a以外泌体为载体运输至细胞外,使得胞内对肌肉萎缩盒基因和肌肉环状指基因1的抑制作用减弱,进一步导致肌肉萎缩[55]。恶病质C26细胞分泌的外泌体在体外可使C2C12肌管直径减小,体内可降低小鼠肌力和胫前肌质量。经miRNA测序发现C26细胞分泌的外泌体中miR-195a-5p和miR-125b-1-3p丰富,miR-195a-5p和miR-125b-1-3p下调Bcl-2和激活凋亡信号通路,诱导C2C12成肌细胞萎缩[56]。肿瘤来源外泌体携带的多种miRNAs具有促进炎症、激活分解代谢甚至参与调节细胞降解途径的能力,这些miRNAs可以传递给肌肉细胞并对其发挥作用[57]。在肌肉萎缩疾病中观察到循环中某些miRNAs的升高[58],但这些miRNAs是否都以外泌体为载体参与循环还需进一步的研究证明。 2.3 失神经肌萎缩 2.3.1 失神经肌萎缩机制 神经系统通过神经肌肉接头与骨骼肌连接,其对骨骼肌的控制包括[59]:①神经运动控制。大脑皮质或脑干产生神经冲动并向下传导,肌膜去极化,肌电耦合启动,肌肉收缩。②神经营养控制。神经肌肉接头处的神经末梢释放可溶性因子,起到营养作用。周围神经的横断、压迫引起的骨骼肌萎缩比其他病因引起的萎缩快得多,如制动、恶病质、营养不良和衰老等。当肌肉失去神经支配时,会出现快速的肌肉萎缩,包括肌肉质量减少、肌肉力量下降和肌纤维直径缩小,肌细胞凋亡和肌细胞内的蛋白降解增加同时发生[60],这主要是由于泛素-蛋白酶体途径和凋亡途径的激活[61]。 远端长期失神经支配的许旺细胞会逐渐失去支持轴突再生的能力,最初增殖的许旺细胞维持再生轴突穿过基膜管,并分泌许多刺激和引导轴突再生的神经营养因子[62]。然而,如果长时间没有轴突相互作用,许旺细胞执行这些重要功能的能力会逐渐下降,一旦许旺细胞衰老,远端通路就不再支持轴突再生。因此骨骼肌因无法进行主动收缩和失去营养作用逐步发生萎缩,其内的蛋白质稳态发生变化,肌肉失神经可诱导肌肉环状指基因1和肌肉萎缩盒基因的表达[63]。组蛋白去乙酰化酶4诱导Gadd45a mRNA和蛋白增多[64],这是一种小肌核蛋白,是失神经诱导肌肉纤维萎缩所必需的。哺乳动物雷帕霉素复合物1靶分子的活性在失神经肌肉中显著增强,抑制蛋白激酶B(PKB/Akt)的活性,导致叉头盒转录因子激活,增加编码E3泛素连接酶肌肉萎缩盒基因和肌肉环状指基因1的表达[65],导致肌肉萎缩。失神经支配导致的线粒体生物合成减少,与小鼠骨骼肌线粒体翻译因子的变化相对应[66],肌肉失神经可降低小鼠比目鱼肌线粒体翻译因子、线粒体延长因子和线粒体蛋白的表达,并伴随着泛素-蛋白酶体途径的激活,但其上游触发机制尚不清楚。 失神经支配后的腓肠肌迅速萎缩,其内的线粒体数量明显减少[67],并且激活线粒体凋亡途径,凋亡途径相关通路蛋白发生变化,包括Bcl-2家族蛋白、caspase家族蛋白、凋亡相关的细胞调节因子,对失神经肌肉支配作用增加[61]。失神经7 d后,肌肉线粒体活性氧生成量增加近30倍[68],活性氧的增加会促进线粒体通透性过渡孔打开,线粒体通透性过渡孔开放的发生率越高,细胞色素和细胞凋亡诱导因子的释放就越多,导致细胞凋亡[69],见表2。 "
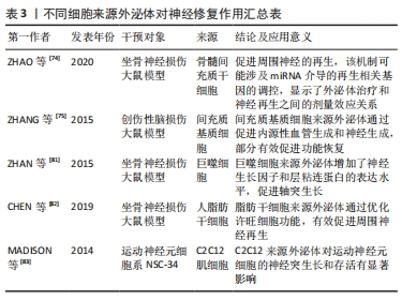
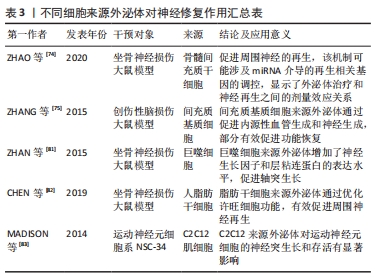
2.3.2 外泌体在失神经肌萎缩后肌肉再生中的作用 在发生外周神经损伤后为防止肌肉发生不可逆萎缩,修复神经损伤显得尤为重要。在神经损伤模型中,将脂肪源性干细胞注入腓肠肌,可减轻肌肉萎缩,对神经损伤有一定的疗效[70]。其他再生细胞如间充质干细胞[71]、肌卫星细胞等对失神经肌萎缩也存在一定作用[72]。虽然再生细胞在肌萎缩修复中起一定作用,但细胞移植存在很多问题,如免疫排斥反应、存活能力有限、再生能力减弱。最近的研究表明,干细胞通过旁分泌因子发挥治疗作用,而不是干细胞分化,外泌体在旁分泌机制中起关键作用[73]。外泌体已经在多种疾病模型中展开研究,由于其低免疫原性、安全、能够穿过血脑屏障,相比细胞移植更具优势,临床应用前景更为广阔。 不同细胞来源外泌体通过直接促进受损神经细胞的修复和再生,或者间接调节炎症反应以提供一个神经修复的最佳环境,缓解肌肉萎缩。骨髓间充质干细胞来源外泌体具有显著的神经保护和轴突形成作用[74];此外,初步证实外泌体可能以miRNA依赖的方式促进神经再生,其机制可能与miRNA介导再生相关基因的表达调控有关。间充质干细胞来源外泌体可以通过促进内源性血管生成来促进大鼠神经损伤后的功能恢复[75]。外泌体是间充质干细胞的主要旁分泌效应器[76],通过激活PKB/Akt、细胞外调节蛋白激酶(extracellular regulated protein kinases,ERK)和STAT3信号通路,诱导生长因子的表达[77]。从间充质干细胞中分离出来的外泌体miRNAs(如miR-199b、miR-218、miR-148a、miR-135b和miR-221)可以影响神经元的分化、增殖和轴突生长[78]。此外,间充质干细胞衍生的外泌体可以传递miRNA来调节心脏、创伤和骨修复中的炎症反应[79-80],以提供最佳的神经修复环境。巨噬细胞来源外泌体含有可以促进神经再生的miRNA,在体外和体内都参与这种再生过程[81],体外可以促进许旺细胞的迁移和增殖,促使神经生长因子和层粘连蛋白表达增多;在M2巨噬细胞来源外泌体中miR-223的表达增多,而miR-223抑制剂共同孵育的许旺细胞迁移和增殖能力受到抑制,并下调了神经生长因子和层粘连蛋白的表达水平。人脂肪干细胞来源外泌体可以优先被许旺细胞内化[82],脂肪干细胞来源外泌体刺激显著上调RAC家族的小GTPase 1(Rac1)、基质金属肽酶9和含胶原三螺旋重复蛋白1(Cthc1)的mRNA水平,说明脂肪干细胞来源外泌体促进许旺细胞迁移和髓鞘形成,见表3。脂肪干细胞来源外泌体刺激可提高许旺细胞中神经生长因子、脑源性神经营养因子和睫状神经营养因子的mRNA水平,形成有利于轴突生长的微环境。C2C12细胞衍生的外泌体可以提高运动神经元细胞系NSC-34的存活和神经突生长,这表明肌肉也可以影响运动神经元的存活和轴突生长[83]。另外,失神经肌肉纤维释放的外泌体miR-206对肌萎缩过程中的细胞间通讯有明显影响[84],有人认为miR-206还可能参与肌肉质量的调节和神经支配过程中突触的形成,如miR-206通过转化生长因子β1、Smad3和组蛋白去乙酰化酶4信号调节卫星细胞分化,减轻失神经诱导的大鼠骨骼肌萎缩[85],因此,失神经肌纤维来源外泌体可能通过以旁分泌的方式向邻近细胞提供miR-206来帮助局部肌肉修复。 "
| [1] NEUBRECH F, HEIDER S, HARHAUS L, et al. Chitosan nerve tube for primary repair of traumatic sensory nerve lesions of the hand without a gap: study protocol for a randomized controlled trial. Trials. 2016;17:48. [2] ROSÉN B, VIKSTRÖM P, TURNER S, et al. Enhanced early sensory outcome after nerve repair as a result of immediate post-operative re-learning: a randomized controlled trial. J Hand Surg Eur Vol. 2015; 40(6):598-606. [3] 钟声,章明星,刘建卫,等.电针治疗失神经肌萎缩机制研究进展[J].中国中医药现代远程教育,2018,16(19):146-147. [4] TIELAND M, TROUWBORST I, CLARK BC. Skeletal muscle performance and ageing. J Cachexia Sarcopenia Muscle. 2018;9(1):3-19. [5] VIJAYAVENKATARAMAN S. Nerve guide conduits for peripheral nerve injury repair: A review on design, materials and fabrication methods. Acta Biomater. 2020;106:54-69. [6] 杨东丽,杨琼,罗嘉,等.外泌体参与骨骼肌机能调控研究进展[J].动物医学进展,2018,39(1):103-108. [7] ZHANG Y, CHOPP M, LIU XS, et al. Exosomes Derived from Mesenchymal Stromal Cells Promote Axonal Growth of Cortical Neurons. Mol Neurobiol. 2017;54(4):2659-2673. [8] HE C, ZHENG S, LUO Y, et al. Exosome Theranostics: Biology and Translational Medicine. Theranostics. 2018;8(1):237-255. [9] HARDING C, HEUSER J, STAHL P. Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. J Cell Biol. 1983;97(2):329-339. [10] KALLURI R, LEBLEU VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367(6478):eaau6977. [11] HENNE WM, STENMARK H, EMR SD. Molecular mechanisms of the membrane sculpting ESCRT pathway. Cold Spring Harb Perspect Biol. 2013;5(9):a016766. [12] STUFFERS S, SEM WEGNER C, STENMARK H, et al. Multivesicular endosome biogenesis in the absence of ESCRTs. Traffic. 2009;10(7): 925-937. [13] HESSVIK NP, ØVERBYE A, BRECH A, et al. PIKfyve inhibition increases exosome release and induces secretory autophagy. Cell Mol Life Sci. 2016;73(24):4717-4737. [14] XU J, CAMFIELD R, GORSKI SM. The interplay between exosomes and autophagy - partners in crime. J Cell Sci. 2018;131(15):jcs215210. [15] KORKUT C, ATAMAN B, RAMACHANDRAN P, et al. Trans-synaptic transmission of vesicular Wnt signals through Evi/Wntless. Cell. 2009; 139(2):393-404. [16] ANEL A, GALLEGO-LLEYDA A, DE MIGUEL D, et al. Role of Exosomes in the Regulation of T-cell Mediated Immune Responses and in Autoimmune Disease. Cells. 2019;8(2):154. [17] ZHAO J, LI X, HU J, et al. Mesenchymal stromal cell-derived exosomes attenuate myocardial ischaemia-reperfusion injury through miR-182-regulated macrophage polarization. Cardiovasc Res. 2019;115(7):1205-1216. [18] WANG X, ZHOU Y, GAO Q, et al. The Role of Exosomal microRNAs and Oxidative Stress in Neurodegenerative Diseases. Oxid Med Cell Longev. 2020;2020:3232869. [19] ZHANG L, YU D. Exosomes in cancer development, metastasis, and immunity. Biochim Biophys Acta Rev Cancer. 2019;1871(2):455-468. [20] YANG L, PENG X, LI Y, et al. Long non-coding RNA HOTAIR promotes exosome secretion by regulating RAB35 and SNAP23 in hepatocellular carcinoma. Mol Cancer. 2019;18(1):78. [21] PEGTEL DM, GOULD SJ. Exosomes. Annu Rev Biochem. 2019;88:487-514. [22] SCHNEIDER A, SIMONS M. Exosomes: vesicular carriers for intercellular communication in neurodegenerative disorders. Cell Tissue Res. 2013; 352(1):33-47. [23] LARIOS J, MERCIER V, ROUX A, et al. ALIX- and ESCRT-III-dependent sorting of tetraspanins to exosomes. J Cell Biol. 2020;219(3): e201904113. [24] WEI D, ZHAN W, GAO Y, et al. RAB31 marks and controls an ESCRT-independent exosome pathway. Cell Res. 2021;31(2):157-177. [25] VALADI H, EKSTRÖM K, BOSSIOS A, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9(6):654-659. [26] GIBBINGS DJ, CIAUDO C, ERHARDT M, et al. Multivesicular bodies associate with components of miRNA effector complexes and modulate miRNA activity. Nat Cell Biol. 2009;11(9):1143-1149. [27] BATAGOV AO, KUROCHKIN IV. Exosomes secreted by human cells transport largely mRNA fragments that are enriched in the 3’-untranslated regions. Biol Direct. 2013;8:12. [28] VILLARROYA-BELTRI C, GUTIÉRREZ-VÁZQUEZ C, SÁNCHEZ-CABO F, et al. Sumoylated hnRNPA2B1 controls the sorting of miRNAs into exosomes through binding to specific motifs. Nat Commun. 2013;4:2980. [29] SANTANGELO L, GIURATO G, CICCHINI C, et al. The RNA-Binding Protein SYNCRIP Is a Component of the Hepatocyte Exosomal Machinery Controlling MicroRNA Sorting. Cell Rep. 2016;17(3):799-808. [30] TRAN N. Cancer Exosomes as miRNA Factories. Trends Cancer. 2016; 2(7):329-331. [31] SKOTLAND T, HESSVIK NP, SANDVIG K, et al. Exosomal lipid composition and the role of ether lipids and phosphoinositides in exosome biology. J Lipid Res. 2019;60(1):9-18. [32] OUWENEEL AB, THOMAS MJ, SORCI-THOMAS MG. The ins and outs of lipid rafts: functions in intracellular cholesterol homeostasis, microparticles, and cell membranes: Thematic Review Series: Biology of Lipid Rafts. J Lipid Res. 2020;61(5):676-686. [33] PETERSEN SV, POULSEN NB, LINNEBERG MATTHIESEN C, et al. Novel and Converging Ways of NOX2 and SOD3 in Trafficking and Redox Signaling in Macrophages. Antioxidants (Basel). 2021;10(2):172. [34] BADAWY SMM, OKADA T, KAJIMOTO T, et al. Extracellular α-synuclein drives sphingosine 1-phosphate receptor subtype 1 out of lipid rafts, leading to impaired inhibitory G-protein signaling. J Biol Chem. 2018;293(21):8208-8216. [35] ZHANG B, WANG M, GONG A, et al. HucMSC-Exosome Mediated-Wnt4 Signaling Is Required for Cutaneous Wound Healing. Stem Cells. 2015;33(7):2158-2168. [36] COSSETTI C, IRACI N, MERCER TR, et al. Extracellular vesicles from neural stem cells transfer IFN-γ via Ifngr1 to activate Stat1 signaling in target cells. Mol Cell. 2014;56(2):193-204. [37] LÄSSER C. Exosomes in diagnostic and therapeutic applications: biomarker, vaccine and RNA interference delivery vehicle. Expert Opin Biol Ther. 2015;15(1):103-117. [38] VALCHEVA-KUZMANOVA S, ZHELYAZKOVA-SAVOVA M. Anxiolytic-like effect of Aronia melanocarpa fruit juice in rats. Methods Find Exp Clin Pharmacol. 2009;31(10):651-654. [39] YU B, KIM HW, GONG M, et al. Exosomes secreted from GATA-4 overexpressing mesenchymal stem cells serve as a reservoir of anti-apoptotic microRNAs for cardioprotection. Int J Cardiol. 2015;182:349-360. [40] CHRISTIANSON HC, SVENSSON KJ, VAN KUPPEVELT TH, et al. Cancer cell exosomes depend on cell-surface heparan sulfate proteoglycans for their internalization and functional activity. Proc Natl Acad Sci U S A. 2013;110(43):17380-17385. [41] CAPONNETTO F, MANINI I, SKRAP M, et al. Size-dependent cellular uptake of exosomes. Nanomedicine. 2017;13(3):1011-1020. [42] ROME S, FORTERRE A, MIZGIER ML, et al. Skeletal Muscle-Released Extracellular Vesicles: State of the Art. Front Physiol. 2019;10:929. [43] GUESCINI M, GUIDOLIN D, VALLORANI L, et al. C2C12 myoblasts release micro-vesicles containing mtDNA and proteins involved in signal transduction. Exp Cell Res. 2010;316(12):1977-1984. [44] ASWAD H, FORTERRE A, WIKLANDER OP, et al. Exosomes participate in the alteration of muscle homeostasis during lipid-induced insulin resistance in mice. Diabetologia. 2014;57(10):2155-2164. [45] ASWAD H, JALABERT A, ROME S. Depleting extracellular vesicles from fetal bovine serum alters proliferation and differentiation of skeletal muscle cells in vitro. BMC Biotechnol. 2016;16:32. [46] FORTERRE A, JALABERT A, CHIKH K, et al. Myotube-derived exosomal miRNAs downregulate Sirtuin1 in myoblasts during muscle cell differentiation. Cell Cycle. 2014;13(1):78-89. [47] CHOI JS, YOON HI, LEE KS, et al. Exosomes from differentiating human skeletal muscle cells trigger myogenesis of stem cells and provide biochemical cues for skeletal muscle regeneration. J Control Release. 2016;222:107-115. [48] NIE Y, SATO Y, GARNER RT, et al. Skeletal muscle-derived exosomes regulate endothelial cell functions via reactive oxygen species-activated nuclear factor-κB signalling. Exp Physiol. 2019;104(8):1262-1273. [49] MADISON RD, MCGEE C, RAWSON R, et al. Extracellular vesicles from a muscle cell line (C2C12) enhance cell survival and neurite outgrowth of a motor neuron cell line (NSC-34). J Extracell Vesicles. 2014;3(1): 22865. [50] CID-DÍAZ T, SANTOS-ZAS I, GONZÁLEZ-SÁNCHEZ J, et al. Obestatin controls the ubiquitin-proteasome and autophagy-lysosome systems in glucocorticoid-induced muscle cell atrophy. J Cachexia Sarcopenia Muscle. 2017;8(6):974-990. [51] LI S, HAO M, LI B, et al. CACNA1H downregulation induces skeletal muscle atrophy involving endoplasmic reticulum stress activation and autophagy flux blockade. Cell Death Dis. 2020;11(4):279. [52] WANG M, HU R, WANG Y, et al. Atractylenolide III Attenuates Muscle Wasting in Chronic Kidney Disease via the Oxidative Stress-Mediated PI3K/AKT/mTOR Pathway. Oxid Med Cell Longev. 2019;2019:1875471. [53] O’NEILL BT, BHARDWAJ G, PENNIMAN CM, et al. FoxO Transcription Factors Are Critical Regulators of Diabetes-Related Muscle Atrophy. Diabetes. 2019;68(3):556-570. [54] HUDSON MB, RAHNERT JA, ZHENG B, et al. miR-182 attenuates atrophy-related gene expression by targeting FoxO3 in skeletal muscle. Am J Physiol Cell Physiol. 2014;307(4):C314-C319. [55] HUDSON MB, WOODWORTH-HOBBS ME, ZHENG B, et al. miR-23a is decreased during muscle atrophy by a mechanism that includes calcineurin signaling and exosome-mediated export. Am J Physiol Cell Physiol. 2014;306(6):C551-C558. [56] MIAO C, ZHANG W, FENG L, et al. Cancer-derived exosome miRNAs induce skeletal muscle wasting by Bcl-2-mediated apoptosis in colon cancer cachexia. Mol Ther Nucleic Acids. 2021;24:923-938. [57] LI C, WU Q, LI Z, et al. Exosomal microRNAs in cancer-related sarcopenia: Tumor-derived exosomal microRNAs in muscle atrophy. Exp Biol Med (Maywood). 2021;246(10):1156-1166. [58] COENEN-STASS AML, WOOD MJA, ROBERTS TC. Biomarker Potential of Extracellular miRNAs in Duchenne Muscular Dystrophy. Trends Mol Med. 2017;23(11):989-1001. [59] CISTERNA BA, CARDOZO C, SÁEZ JC. Neuronal involvement in muscular atrophy. Front Cell Neurosci. 2014;8:405. [60] PAUDYAL A, SLEVIN M, MAAS H, et al. Time course of denervation-induced changes in gastrocnemius muscles of adult and old rats. Exp Gerontol. 2018;106:165-172. [61] SIU PM, ALWAY SE. Mitochondria-associated apoptotic signalling in denervated rat skeletal muscle. J Physiol. 2005;565(Pt 1):309-323. [62] NOCERA G, JACOB C. Mechanisms of Schwann cell plasticity involved in peripheral nerve repair after injury. Cell Mol Life Sci. 2020;77(20): 3977-3989. [63] SACHECK JM, HYATT JP, RAFFAELLO A, et al. Rapid disuse and denervation atrophy involve transcriptional changes similar to those of muscle wasting during systemic diseases. FASEB J. 2007;21(1):140-155. [64] BONGERS KS, FOX DK, EBERT SM, et al. Skeletal muscle denervation causes skeletal muscle atrophy through a pathway that involves both Gadd45a and HDAC4. Am J Physiol Endocrinol Metab. 2013;305(7): E907-915. [65] TANG H, INOKI K, LEE M, et al. mTORC1 promotes denervation-induced muscle atrophy through a mechanism involving the activation of FoxO and E3 ubiquitin ligases. Sci Signal. 2014;7(314):ra18. [66] YOKOKAWA T, MORI R, SUGA T, et al. Muscle denervation reduces mitochondrial biogenesis and mitochondrial translation factor expression in mice. Biochem Biophys Res Commun. 2020;527(1):146-152. [67] YANG X, XUE P, CHEN H, et al. Denervation drives skeletal muscle atrophy and induces mitochondrial dysfunction, mitophagy and apoptosis via miR-142a-5p/MFN1 axis. Theranostics. 2020;10(3):1415-1432. [68] MULLER FL, SONG W, JANG YC, et al. Denervation-induced skeletal muscle atrophy is associated with increased mitochondrial ROS production. Am J Physiol Regul Integr Comp Physiol. 2007;293(3): R1159-R1168. [69] ADHIHETTY PJ, O’LEARY MF, CHABI B, et al. Effect of denervation on mitochondrially mediated apoptosis in skeletal muscle. J Appl Physiol (1985). 2007;102(3):1143-1151. [70] SCHAAKXS D, KALBERMATTEN DF, RAFFOUL W, et al. Regenerative cell injection in denervated muscle reduces atrophy and enhances recovery following nerve repair. Muscle Nerve. 2013;47(5):691-701. [71] 刘晓光,肖卫华,赵淋淋,等.间充质干细胞移植治疗骨骼肌损伤的研究进展[J].中国康复医学杂志,2015,30(12):1313-1317. [72] JOANISSE S, NEDERVEEN JP, SNIJDERS T, et al. Skeletal Muscle Regeneration, Repair and Remodelling in Aging: The Importance of Muscle Stem Cells and Vascularization. Gerontology. 2017;63(1):91-100. [73] PHINNEY DG, PITTENGER MF. Concise Review: MSC-Derived Exosomes for Cell-Free Therapy. Stem Cells. 2017;35(4):851-858. [74] ZHAO J, DING Y, HE R, et al. Dose-effect relationship and molecular mechanism by which BMSC-derived exosomes promote peripheral nerve regeneration after crush injury. Stem Cell Res Ther. 2020;11(1): 360. [75] ZHANG Y, CHOPP M, MENG Y, et al. Effect of exosomes derived from multipluripotent mesenchymal stromal cells on functional recovery and neurovascular plasticity in rats after traumatic brain injury. J Neurosurg. 2015;122(4):856-867. [76] WEN D, PENG Y, LIU D, et al. Mesenchymal stem cell and derived exosome as small RNA carrier and Immunomodulator to improve islet transplantation. J Control Release. 2016;238:166-175. [77] SHABBIR A, COX A, RODRIGUEZ-MENOCAL L, et al. Mesenchymal Stem Cell Exosomes Induce Proliferation and Migration of Normal and Chronic Wound Fibroblasts, and Enhance Angiogenesis In Vitro. Stem Cells Dev. 2015;24(14):1635-1647. [78] XU JF, YANG GH, PAN XH, et al. Altered microRNA expression profile in exosomes during osteogenic differentiation of human bone marrow-derived mesenchymal stem cells. PLoS One. 2014;9(12):e114627. [79] SHAO L, ZHANG Y, LAN B, et al. MiRNA-Sequence Indicates That Mesenchymal Stem Cells and Exosomes Have Similar Mechanism to Enhance Cardiac Repair. Biomed Res Int. 2017;2017:4150705. [80] FURUTA T, MIYAKI S, ISHITOBI H, et al. Mesenchymal Stem Cell-Derived Exosomes Promote Fracture Healing in a Mouse Model. Stem Cells Transl Med. 2016;5(12):1620-1630. [81] ZHAN C, MA CB, YUAN HM, et al. Macrophage-derived microvesicles promote proliferation and migration of Schwann cell on peripheral nerve repair. Biochem Biophys Res Commun. 2015;468(1-2):343-348. [82] CHEN J, REN S, DUSCHER D, et al. Exosomes from human adipose-derived stem cells promote sciatic nerve regeneration via optimizing Schwann cell function. J Cell Physiol. 2019;234(12):23097-23110. [83] MADISON RD, MCGEE C, RAWSON R, et al. Extracellular vesicles from a muscle cell line (C2C12) enhance cell survival and neurite outgrowth of a motor neuron cell line (NSC-34). J Extracell Vesicles. 2014;3. [84] DE GASPERI R, HAMIDI S, HARLOW LM, et al. Denervation-related alterations and biological activity of miRNAs contained in exosomes released by skeletal muscle fibers. Sci Rep. 2017;7(1):12888. [85] HUANG QK, QIAO HY, FU MH, et al. MiR-206 Attenuates Denervation-Induced Skeletal Muscle Atrophy in Rats Through Regulation of Satellite Cell Differentiation via TGF-β1, Smad3, and HDAC4 Signaling. Med Sci Monit. 2016;22:1161-1170. |
| [1] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [2] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [3] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [4] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [5] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [6] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [7] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [8] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [9] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [10] | Guo Jia, Ding Qionghua, Liu Ze, Lü Siyi, Zhou Quancheng, Gao Yuhua, Bai Chunyu. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1093-1101. |
| [11] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [12] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [13] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [14] | Hu Wei, Xie Xingqi, Tu Guanjun. Exosomes derived from bone marrow mesenchymal stem cells improve the integrity of the blood-spinal cord barrier after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 992-998. |
| [15] | Xu Lei, Han Xiaoqiang, Zhang Jintao, Sun Haibiao. Hyaluronic acid around articular chondrocytes: production, transformation and function characteristics [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 768-773. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||