Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (35): 5583-5588.doi: 10.12307/2022.924
Previous Articles Next Articles
Changes in irisin, leptin, adiponectin and visceral fat in obese rats undergoing high-intensity intermittent exercise
Lin Jiayu, Huang Huibin, Liang Bo, Chen Lijun
- Department of Endocrinology, the Second Affiliated Hospital of Fujian Medical University, Quanzhou 362000, Fujian Province, China
-
Received:2021-11-26Accepted:2022-01-12Online:2022-12-18Published:2022-05-17 -
Contact:Lin Jiayu, Department of Endocrinology, the Second Affiliated Hospital of Fujian Medical University, Quanzhou 362000, Fujian Province, China -
About author:Lin Jiayu, MD, Associate chief physician, Department of Endocrinology, the Second Affiliated Hospital of Fujian Medical University, Quanzhou 362000, Fujian Province, China -
Supported by:the Education and Research Project for Young and Middle-aged Teachers in Fujian Province, No. JT180197 (to LJY)
CLC Number:
Cite this article
Lin Jiayu, Huang Huibin, Liang Bo, Chen Lijun. Changes in irisin, leptin, adiponectin and visceral fat in obese rats undergoing high-intensity intermittent exercise[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(35): 5583-5588.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
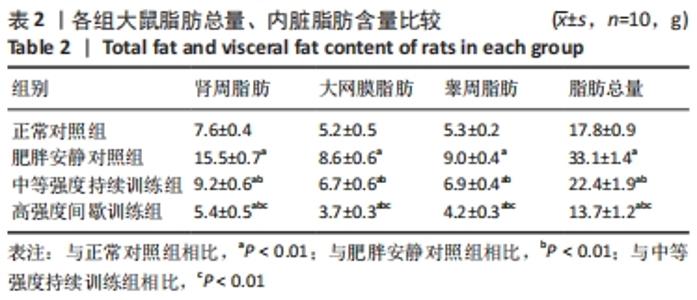
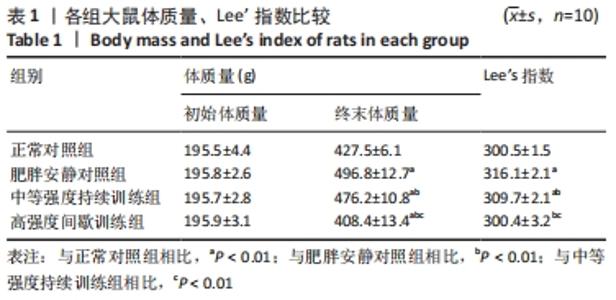
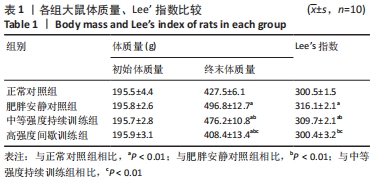
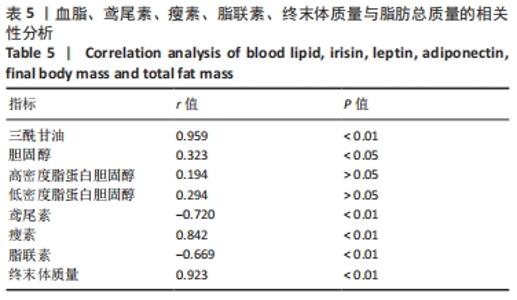
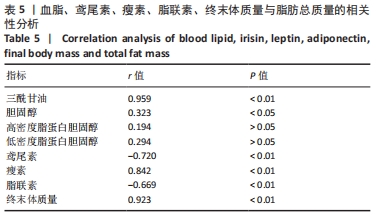
各组大鼠初始体质量处于同一水平(P > 0.05)。实验结束后,与正常对照组相比,肥胖安静对照组大鼠、中等强度持续训练组终末体质量明显增加,高强度间歇训练组终末体质量减低,差异有显著性意义(P < 0.01)。高脂喂养和运动干预组中,中等强度持续训练组、高强度间歇训练组终末体质量显著低于肥胖安静对照组,高强度间歇训练组终末体质量显著低于中等强度持续训练组(P < 0.01) 。 肥胖安静对照组、中等强度持续训练组Lee’s 指数显著高于正常对照组,中等强度持续训练组Lee’s 指数明显低于肥胖安静对照组(P < 0.01) ,高强度间歇训练组与正常对照组间无明显差异(P > 0.05)。 2.3 各组大鼠脂肪总质量与内脏脂肪含量 肥胖安静对照组、中等强度持续训练组的内脏脂肪(肾周脂肪、大网膜脂肪、睾周脂肪)、脂肪总质量均显著高于正常对照组(均P < 0.01)。高脂饲料喂养组中,中等强度持续训练组和高强度间歇训练组内脏脂肪、脂肪总质量均显著低于肥胖安静对照组;高强度间歇训练组内脏脂肪、脂肪总质量较中等强度训练组、正常对照组明显减低(均P < 0.01),见表2。 "
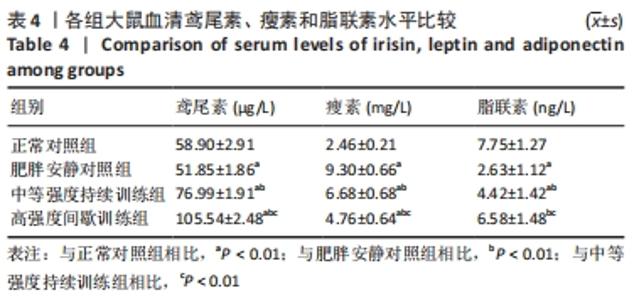
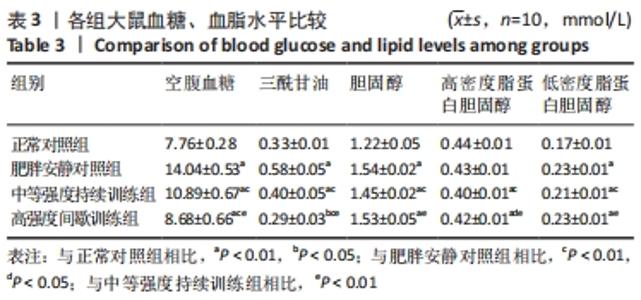
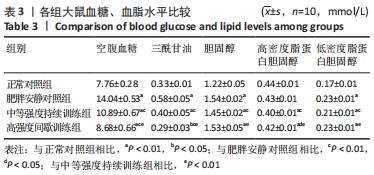
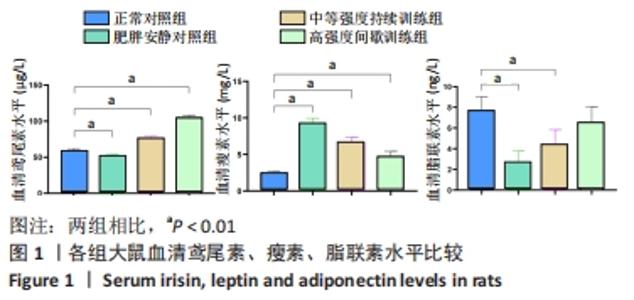
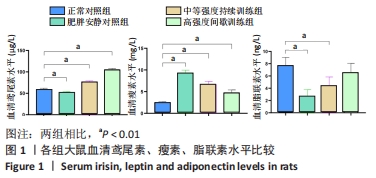
肥胖安静对照组空腹血糖水平显著高于正常对照组 (P < 0.01)。在高脂喂养组中,中等强度持续训练组、高强度间歇训练组血糖水平显著低于肥胖安静对照组(均P < 0.01) ,高强度间歇训练组血糖水平明显低于中等强度持续训练组(P < 0.01)。 与正常对照组相比,肥胖安静对照组、中等强度持续训练组的三酰甘油、胆固醇和 LDL-C水平明显升高(均P < 0.01),高强度间歇训练组的三酰甘油水平降低(P < 0.05)、胆固醇和LDL-C水平明显升高(均P < 0.01),中等强度训练组、高强度间歇训练组的HDL-C水平减低(P < 0.01),肥胖安静对照组HDL-C水平无显著变化(P > 0.05)。 高脂喂养和运动干预组中,与肥胖安静对照组相比,中等强度持续训练组三酰甘油、胆固醇、LDL-C、HDL-C水平降低(均P < 0.01);高强度间歇训练组三酰甘油(P < 0.01)和HDL-C(P < 0.05)水平降低,胆固醇和LDL-C水平无显著变化(P > 0.05)。与中等强度持续训练组相比,高强度间歇训练组三酰甘油水平降低(P < 0.01),胆固醇、LDL-C、HDL-C水平升高(均P < 0.01)。 2.5 运动干预对肥胖大鼠血清Irisin、瘦素和脂联素水平的影响 结果显示,与正常对照组相比,肥胖安静对照组血清Irisin和脂联素水平明显降低、瘦素水平明显升高(均P < 0.01),中等强度持续训练组、高强度间歇训练组大鼠血清Irisin和瘦素水平显著上升(均P < 0.01),中等强度持续训练组脂联素水平降低(P < 0.01),高强度间歇训练组脂联素水平无显著变化(P > 0.05)。高脂喂养和运动干预组中,与中等强度持续训练组相比,高强度间歇训练组大鼠血清Irisin水平上升更明显 (P < 0.01),见表4及图1。 "
| [1] MENDIS S. Global status report on noncommunicable diseases 2014.Geneva: World Health Organisation; 2014. [2] BOULÉ NG, HADDAD E, KENNY GP, et al. Effects of exerciseon glycemic control and body mass in type 2 diabetes mellitus: a meta-analysis of controlled clinical trials. JAMA. 2001;286:1218-1227. [3] SIGAL RJ, KENNY GP, WASSERMAN DH, et al. Physical activity/ exercise and type 2 diabetes. Diabetes Care. 2004;27:2518-2539. [4] TERADA T,FRIESEN A,CHAHAL BS, et al. Feasibility and preliminary efficacy of high intensity interval training in type 2 diabetes. Diabetes Res Clin Pract. 2013;99(2):120-129. [5] FRANCOIS ME, LITTLE JP. Effectiveness and safety of high-intensity interval training in patients with type 2 diabetes. Diabetes Spectr. 2015; 28(1):39-44. [6] BOSTROM P, WU J, JEDRY TC, et al. A PGC1-alpha-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012;481(7382):463-468. [7] WU J, BOSTROM P, SPARKS LM, et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell. 2012;150(2): 366-376. [8] GUO Q, WEI X, HU H, et al. The saturated fatty acid palmitate induces insulin resistance through Smad3-mediated down-regulation of FNDC5 in myotubes. Biochem Biophys Res Commun. 2019;520(3):619-626. [9] BEDFORD TG, TIPTON CM, WILSON NC, et al. Maximum oxygen consumption of rats and its changes with various experimental procedures. J Appl Physiol. 1979;47(6):1278-1283. [10] ZHANG YJ, LI J, HUANG W, et al. Effect of electroacupuncture combined with treadmill exercise on body weight and expression of PGC-1α, Irisin and AMPK in skeletal muscle of diet-induced obesity rats. Zhen Ci Yan Jiu. 2019;44(7):476-480. [11] 实况报道.肥胖和超重[ER/OL].[2016-6].http//www.who.int/mediacentre/factsheets/fs311/zh/. [12] KAHN SE, HULL RL, UTZSCHNEIDER KM. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature. 2006;444(7121):840-846. [13] KANG YS, KIM JC, KIM JS, et al. Effects of Swimming Exercise on Serum Irisin and Bone FNDC5 in Rat Models of High-Fat Diet-Induced Osteoporosis. Sports Sci Med. 2019;18(4):596-603. [14] 王宁宁,刘洋,丁宁,等.运动对棕色脂肪活性和白色脂肪棕色化的影响及其干预肥胖的作用研究进展[J].中国运动医学杂志,2017, 36(5):448-455. [15] MANDERS RJ, VAN DIJK JW, VAN LOON LJ. Low-intensity exercise reduces the prevalence of hyperglycemia in type 2 diabetes. Med Sci Sports Exerc. 2010;42:219-225. [16] DE MATTEIS R,LUCERTINI F,GUESCINI M,et al. Exercise as a new physiological stimulus for brown adipose tissue activity. Nutr Metab Cardiovasc Dis. 2013;23(6):582-590. [17] XU X, YING Z, CAI M, et al. Exercise ameliorates high-fat diet-induced metabolic and vascular dysfunction,and increases adipocyte progenitor cell population in brown adipose tissue. Am J Physiol Regul Integr Comp Physiol. 2011;300(5):R1115-1125. [18] WEWEGE M, VAN DEN BERG R, WARD RE, et al. The effects of high-intensity interval training vs. moderate-intensity continuous training on body composition in overweight and obese adults: a systematic review and meta-analysis. Obes Rev. 2017;18(6):635-646. [19] HUH JY, MOUGIOS V, SKRAPARLIS A, et al. Irisin in response to acute and chronic whole-body vibration exercise in humans. Metabolism. 2014;63(7):918-921. [20] ERICKSON HP. Irisin and FNDC5 in retrospect: An exercise hormone or a transmembrane receptor? Adipocyte. 2013;2(4):289-293. [21] JEDRYCHOWSKI MP, WRANN CD, PAULO JA, et al. Detection and quantitation of circulating human irisin by tandem mass spectrometry. Cell Metab. 2015;22(4):734-740. [22] STENGEL A, HOFMANN T, G OE BEL-STENGEL M, et al. Circulating levels of Irisin in patients with anorexia nervosa and different stages of obesity--correlation with body mass index. Peptides. 2013;39:125-130. [23] NORHEIM F, LANGLEITE TM, HJORTH M, et al. The effects of acute and chronic exercise on PGC-1α, irisin and browning of subcutaneous adipose tissue in humans. FEBS J. 2014;281(3):739-749. [24] TSUCHIYA Y, IJICHI T, GOTO K. Effect of sprint training on resting serum irisin concentration-sprint training once daily vs. twice every other day. Metabolism. 2016;65(4):492-495. [25] HECKSTEDEN A, WEGMANN M, STEFFEN A, et al. Irisin and exercise training in humans-results from a randomized controlled training trial. BMC Med. 2013;11:235. [26] KIM HJ, LEE HJ, SO B, et al. Effect of aerobic training and resistance training on circulating irisin level and their association with change of body composition in overweight/obese adults: a pilot study. Physiol Res. 2016;65(2):271. [27] TONG HV, LUU NK, SON HA, et al. Adiponectin and pro-inflammatory cytokines are modulated in Vietnamese patients with type 2 diabetes mellitus. J Diabetes Investig. 2017;8(3):295-305. [28] GHADGE AA, KHAIRE AA. Leptin as a predictive marker for metabolic syndrome. Cytokine. 2019;121:154735. [29] MARTÍNEZ-SÁNCHEZ N. There and Back Again: Leptin Actions in White Adipose Tissue. Int J Mol Sci. 2020;21(17):6039. [30] LARSEN MA, ISAKSEN VT, PAULSSEN EJ, et al. Postprandial Leptin and Adiponectin in response to sugar and fat in obese and normal weight individuals. Endocrine. 2019;66(3):517-525. [31] KELLY BM, XENOPHONTOS S, KING JA, et al. An evaluation of low volume high-intensity intermittent training (HIIT) for health risk reduction in overweight and obese men. BMC Obes. 2017;4:17. [32] BLUHER S, KAPPLINGER J, HERGET S, et al. Cardiometabolic risk markers, adipocyte fatty acid binding protein (aFABP) and the impact of high-intensity interval training (HIIT) in obese adolescents. Metabolism. 2017;68:77-87. [33] GEROSA-NETO J, ANTUNES BM, CAMPOS EZ, et al. Impact of long-term high-intensity interval and moderate-intensity continuous training on subclinical inflammation in overweight/obese adults. J Exerc Rehabil. 2016;12(6):575-580. [34] LEE SS, YOO JH, SO YS. Effect of the low-versus high-intensity exercise training on endoplasmic reticulum stress and GLP-1 in adolescents with type 2 diabetes mellitus. J Phys Ther Sci. 2015;27(10):3063-3068. |
| [1] | FAN Yaru, LI Ruixin , LI Fengji, LUO Rui, LIU Hao, YAN Yingbin. Characterization and photothermal effect of indocyanine green encapsulated poly lactic acid-co-glycolic acid microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-6. |
| [2] | Wang Jianping, Zhang Xiaohui, Yu Jinwei, Wei Shaoliang, Zhang Xinmin, Xu Xingxin, Qu Haijun. Application of knee joint motion analysis in machanism based on three-dimensional image registration and coordinate transformation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-5. |
| [3] | Bao Xianguo, Gao Zengxin, Wu Zhanpo, Chen Youmin, Cheng Qinghua, Lu Haitao, Guo Changzheng, Xu Shuai. Correlation between lumbar posterior muscle and local kyphosis in patients with degenerative thoracolumbar kyphosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1418-1423. |
| [4] | Zhang Haobo, Zhao Yunan, Yang Xuejun. Role and therapeutic implications of pyroptosis in intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1445-1451. |
| [5] | Jing Jinpeng, Zhang Yue, Liu Xiaomin, Liu Yi. Traditional Chinese medicine injection for promoting blood circulation in prevention of deep vein thrombosis after orthopedic surgery: network meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1467-1476. |
| [6] | Li Huo, Wang Peng, Gao Jianming, Jiang Haoran, Lu Xiaobo, Peng Jiang. Relationship between revascularization and internal microstructure changes in osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1323-1328. |
| [7] | Gu Zhengqiu, Xu Fei, Wei Jia, Zou Yongdi, Wang Xiaolu, Li Yongming. Exploratory study on talk test as a measure of intensity in blood flow restriction training [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1154-1159. |
| [8] | Kong Yamin, Yan Juntao, Ma Bingxiang, Li Huawei. Massage vibration intervenes with MyoD expression and proliferation and differentiation of muscle satellite cells in rats with sciatic nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1160-1166. |
| [9] | Wu Cong, Jia Quanzhong, Liu Lun. Relationship between transforming growth factor beta1 expression and chondrocyte migration in adult articular cartilage after fragmentation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1167-1172. |
| [10] | Li Zhiyi, He Pengcheng, Bian Tianyue, Xiao Yuxia, Gao Lu, Liu Huasheng. Bibliometric and visualized analysis of ferroptosis mechanism research [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1202-1209. |
| [11] | Xiang Xinjian, Liu Fang, Wu Liangliang, Jia Daping, Tao Yue, Zhao Zhengnan, Zhao Yu. High-dose vitamin C promotes the survival of autologous fat transplantation in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1242-1246. |
| [12] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [13] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [14] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [15] | Tian Chuan, Zhu Xiangqing, Yang Zailing, Yan Donghai, Li Ye, Wang Yanying, Yang Yukun, He Jie, Lü Guanke, Cai Xuemin, Shu Liping, He Zhixu, Pan Xinghua. Bone marrow mesenchymal stem cells regulate ovarian aging in macaques [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 985-991. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||