Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (26): 4216-4223.doi: 10.12307/2022.827
Previous Articles Next Articles
Effects of aerobic exercise intervention on synaptic plasticity in Alzheimer’s disease patients
Yang Jinxin1, Wang Kun1, Zhao Jing1, Chen Peijie1, Zhang Tingran1, Lu Wenyun2, Luo Jiong1
- 1Sports Detoxification Research Center, School of Physical Education, Southwest University, Chongqing 400715, China; 2School of Leisure, Shanghai University of Sport, Shanghai 200438, China
-
Received:2021-10-09Accepted:2021-11-19Online:2022-09-18Published:2022-03-09 -
Contact:Luo Jiong, PhD, Professor, Doctoral supervisor, Sports Detoxification Research Center, School of Physical Education, Southwest University, Chongqing 400715, China -
About author:Yang Jinxin, Master, Sports Detoxification Research Center, School of Physical Education, Southwest University, Chongqing 400715, China -
Supported by:the National Social Science Major Program of China, No. 19ZDA352 (to CPJ)
CLC Number:
Cite this article
Yang Jinxin, Wang Kun, Zhao Jing, Chen Peijie, Zhang Tingran, Lu Wenyun, Luo Jiong. Effects of aerobic exercise intervention on synaptic plasticity in Alzheimer’s disease patients[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(26): 4216-4223.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 突触可塑性研究概述 突触是大脑神经元细胞功能联系和信息传递的结构,而突触可塑性是指突触发育形态和信息传递功能上发生较为持久的改变。并且突触可塑性在神经系统发育、成熟及学习记忆中有重要作用。突触丢失及功能紊乱将更为直接地导致阿尔茨海默病患者大脑认知功能下降[8]。因此,突触的生成、数目以及功能对于维持大脑的功能至关重要。有研究表明突触相关蛋白也参与突触可塑性的调节[9],在阿尔茨海默病患者早期病理过程中就会出现突触相关蛋白如轴突膜蛋白(growth associated protein-43,GAP-43)、突触素水平的下降导致大脑突触丢失及功能受损。刘慧莉等[10]研究表明修复受损突触结构、提高突触传递可塑性,能够积极延缓阿尔茨海默病患者认知功能下降水平。因此探寻一种非药物、对于诱导激活突触可塑性具有良好效果的运动干预手段显得极为重要。 2.2 有氧运动改善突触可塑性改善认知的可能机制 "

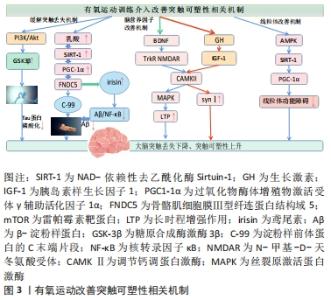
2.2.1 抑制淀粉样β蛋白沉积和Tau毒性蛋白磷酸化保护认知 β淀粉样肽生成和清除之间失衡沉积产生的老年斑诱发神经毒性加重突触丢失、并且发生突触功能障碍,从而造成大脑认知损伤[11]。LAMBERT等[12]研究证实可溶性和可扩散的淀粉样β蛋白寡聚体能够结合神经元并触发神经毒性信号,导致突触丢失并损害突触可塑性[13]。在阿尔茨海默病病理中,微管相关Tau蛋白异常高度磷酸化,致使其与微管结合能力降低,从微管脱位造成细胞内神经纤维缠结[14],且Tau蛋白识别错误与树突相关突触蛋白相互作用而传递神经毒性, 导致突触损伤和功能障碍,从而致使大脑认知功能和记忆水平下降[15]。有氧运动可以诱导一种重要的调节因子NAD-依赖性去乙酰化酶Sirtuin-1(SIRT-1)水平激活,通过PGC1-1α/FNDC5途径介导,可以降低BACE-1和APP的C末端片段C-99水平,导致淀粉样蛋白生成通路的抑制,从而抑制Aβ生成[16-17]。并且运动诱导过氧化物酶体增殖物激活受体γ共激活因子1α(Peroxisome proliferator-activated receptor-γ-coactivator-1α,PGC-1α)刺激纤维连结蛋白Ⅲ型域包含蛋白5(FibronectinType Ⅲ Domain-Containing protein 5,FNDC5)表达增加[18],并且纤维连结蛋白FNDC5可以调控鸢尾素(irisin)水平,irisin表达提高可以降低淀粉样β蛋白沉积与神经元的结合,防止淀粉样β蛋白诱导的eIF2α-P和蛋白质合成的抑制,缓解淀粉样β蛋白造成的突触功能障碍[19]。此外邓雨婷等[20]研究证实运动可以刺激上调irisin表达水平抑制大脑中淀粉样β蛋白生成聚集,从而缓解突触损伤改善大脑认知水平。并且有氧运动可以通过提高大鼠海马组织PI3K/Akt信号通路活性,抑制GSK3β的激酶活性,从而降低了Tau蛋白的磷酸化水平,缓解突触损伤,提高其学习记忆能力[21-22]。 由上可知,积极的有氧运动会促进释放乳酸,调节SIRT-1,通过PGC1-1α/FNDC5途径抑制淀粉样β蛋白生成通路,上调鸢尾素水平缓解大脑突触丢失,并且通过提高PI3K/Akt信号通路活性,抑制GSK3β激酶活性和Tau蛋白异常高度磷酸化,从而抑制细胞内神经纤维缠结生成,从而缓解阿尔茨海默病患者大脑突触丢失,延缓认知能力下降。 2.2.2 提升脑内神经营养因子表达,调控突触持续可塑性 脑源性神经营养因子是一种促进神经元存活和突触完整性的神经营养因子,参与一系列细胞内信号传导过程,对大脑的突触可塑性和记忆功能的调节至关重要[23-24]。脑源性神经营养因子可以调控神经生成、学习和记忆以及神经元生存的相关蛋白质,对突触结构和功能以及神经生成具有重要作用[25],其表达水平与认知功能呈正相关,并且早期阿尔茨海默病患者认知衰退的同时脑源性神经营养因子表达水平较低[26]。付燕等[27]研究发现运动组大鼠脑源性神经营养因子蛋白表达水平及认知能力显著高于对照组,表明有氧运动可积极调控海马脑源性神经营养因子的表达,从而延缓衰老改善学习记忆能力。此外,另一项研究也证实有氧运动可提升老年人外周血液中脑源性神经营养因子水平,并且认知能力随脑源性神经营养因子水平提升而得到相应改善[28]。 运动训练提高了脑内兴奋性突触传递的关键介质 N-甲基-D-天冬氨酸受体(N-methyl-D-aspartate receptor,NMDAR)通道的开放电导水平、开放时间和开放概率,加速了对侧海马CA3区学习依赖型长时程增强的形成[29-30]。而运动使记忆和LTP相关信号分子钙调蛋白激酶(calcium/calmodulin protein kinaseⅡ,CAMKⅡ)和脑源性神经营养因子的表达水平正常化[31]。脑源性神经营养因子可激活突触受体NMDAR,并且通过CAMKⅡ调控机体钙反应,随后激活丝裂原激活蛋白激酶(mitogen-activated protein kinase,MAPK)参与NMDA向细胞核的信号转导过程,进而促进大脑突触可塑性积极反应[32]。此外,海马体脑源性神经营养因子水平提高进而促进突触素Ⅰ水平提高和CREB的磷酸化调节突触可塑性[33-34]。 胰岛素样生长因子是一种依赖于生长激素调节能刺激细胞分裂的调控因子,能够调控大脑突触可塑性和功能障碍,促进神经元的分化与存活修复神经元损伤[35-37]。而运动后胰岛素样生长因子1的升高可能是由于运动后血乳酸浓度升高,可以通过刺激下丘脑释放血液中的生长激素,从而诱导胰岛素样生长因子1分泌[38-40]。有研究表明,体育锻炼能促进血液循环中生长激素的分泌,进而刺激胰岛素样生长因子1的产生,进而刺激大脑,并且增强脑源性神经营养因子的产生 [41]。多项动物实验也证实,有氧运动可增强海马齿状回脑源性神经营养因子和胰岛素样生长因子1的表达水平提高,从而促进成年小鼠海马的神经发生,改善空间学习和记忆[42-44]。此外胰岛素样生长因子1可通过调节相关突触激酶CaMKⅡ表达水平和MAPK磷酸化,促进突触相关蛋白突触素Ⅰ的释放,改善突触可塑性[45]。 基于上述观察,适宜的有氧运动可以调节生长激素水平,提高胰岛素样生长因子1表达水平,激活CaMKⅡ/MAPK信号通路,调节突触蛋白突触素Ⅰ,改善突触可塑性,从而有利于提高认知功能。 2.2.3 促进线粒体生物发生及代谢,改善突触可塑性 线粒体通常存在于突触终末[46],并且突触末梢的线粒体在产生ATP、调节细胞钙平衡和代谢需求上发挥重要作用,线粒体机能障碍将导致突触信息传递受阻、突触功能和结构可塑性紊乱变性[47]。PGC1α可以激活许多代谢相关转录因子,是线粒体生物发生顺利进行的主调控因子[48]。并且沉默信息调节因子1(silent information regulator 1,SIRT1)可诱导PGC1-α去乙酰化和激活,从而增强线粒体生物合成功能[49]。研究表明,SIRT1、PGC1a以及AMP依赖性蛋白激酶(adenosine monophosphate activated protein kinase,AMPK)3者之间的功能调控相互联系[50],AMPK激活调控SIRT1的表达水平。 多项动物实验证实 ,通过有氧跑轮运动对衰老小鼠进行为期2周的运动干预,发现运动显著激活小鼠SIRT1信号通路,提示增加SIRT1表达提高PGC1-α水平,降低了β-分泌酶1(BACE-1)和C-99水平,减少β淀粉样蛋白沉积,促进线粒体生物发生、改善线粒体功能和学习记忆缺,缓解大脑损伤[51-53]。而BRIONES等[54]通过对大鼠进行运动训练后,发现运动训练使大鼠大脑突触以及突触前末梢线粒体数量明显增加。这表明运动可以通过刺激突触前轴突末梢的线粒体的数量增加从而促进突触可塑性改变。有氧运动可通过AMPK-SIRT1-PGC-1α信号通路增加脑线粒体生物发生的调控因子,促进突触末梢线粒体的生物发生,减轻线粒体功能障碍,从而改善突触信息传递效率提高认知能力。 综上所述,有氧运动干预诱导突触可塑性机制呈现一系列蛋白分子调控的连锁反应,阿尔茨海默病患者突触可塑性改善从而能够逆转大脑认知能力水平下降,这与有氧运动介入后的突触丢失缓解、脑营养因子以及突触部位线粒体功能改善等有关,见图3。"
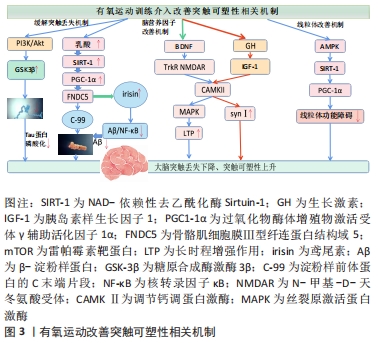

虽然现在对于有氧运动调控突触可塑性的分子作用机制还处于研究阶段,但总体上有氧运动通过不同强度、不同运动频率及运动持续时间等外界应激手段,达到对身体内部突触可塑性蛋白表达水平和突触形态结构刺激调控的作用,在一定程度改善和保护患者的认知功能。 有氧运动诱导突触可塑性的大脑信号机制。在运动时:①运动释放乳酸,调节SIRT-1,通过PGC1-1α/FNDC5途径抑制淀粉样β蛋白生成通路,上调鸢尾素水平缓解大脑突触丢失,并且通过提高PI3K/Akt信号通路活性,抑制GSK3β激酶活性,调控Tau蛋白过度磷酸化抑制神经元纤维缠结,从而缓解阿尔茨海默病患者大脑突触丢失;②有氧运动能提高大脑内脑源性脑源性神经营养因子水平,脑源性神经营养因子通过NMDAR激活CAMKⅡ/MAPK级联反应,并且通过其受体TrkB激活MAPK,上调突触相关蛋白突触素上升,改善大脑突触可塑性;③有氧运动能调节生长激素水平、提高胰岛素样生长因子1表达,激活PI3K/Akt-CREB及CaMKⅡ/MAPK信号通路、调节突触素SynⅠ、改善突触可塑性;④有氧运动通过AMPK-SIRT1-PGC-1α信号通路促进阿尔茨海默病脑线粒体的生物发生,减轻线粒体功能障碍。 2.3 有氧运动干预对阿尔兹海默病突触可塑性的影响研究 "

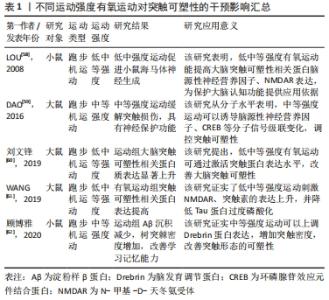
2.3.1 不同运动强度刺激突触可塑性影响 低强度有氧运动对于空间感知以及注意力具有积极效果,中等强度有氧运动对于认知的积极影响则主要集中于改善工作记忆、言语记忆以及注意力,而高强度有氧运动对于改善认知功能并不明显,甚至可能导致负面效果[55];原因在于高强度运动方案可能会增加皮质酮水平,从而降低脑源性神经营养因子表达水平以及抑制神经发生[56-57]。基于动物模型的分子水平研究发现,低中等强度可诱导突触相关蛋白分子表达。对大鼠进行1周的低、中、高等强度有氧运动干预,发现低或中等强度有氧运动组大鼠海马齿状回中的神经发生增强,而高强度运动组大鼠未有神经发生变化,这可能跟低中强度运动显著增加脑源性神经营养因子、NMDAR等突触蛋白水平有关[58]。在对阿尔茨海默病模型大鼠进行4周中等强度跑台运动干预研究后,进行免疫组织化学检测发现运动组大鼠海马齿状回和CA1区环磷腺苷效应元件结合蛋白(cAMP-response element binding protein,CREB)、脑源性神经营养因子和CaMKIV表达水平显著增加,这可能是因为有氧运动诱导分子信号级联变化,继而逆转阿尔茨海默病大鼠的认知缺陷及记忆损伤[59]。有学者发现10周低中等强度有氧运动干预后,进行免疫组织化学检测显示运动组大鼠突触相关因子脑源性神经营养因子、突触素表达水平显著上调,并且脑源性神经营养因子水平随年龄变化而变化,进而促进中年和老年运动大鼠脑皮质CaMKⅡα表达水平上调,激活AMPK信号通路和IP3R/AKT1/mTOR信号通路,改善突触可塑性[60]。在另外一项为期4周的有氧低强度到中等强度递增负荷运动干预中,研究者发现运动组糖尿病大鼠突触可塑性相关蛋白NMDAR和突触素的表达水平显著高于对照组,这证实了对低中等强度有氧运动显著提高小鼠大脑突触可塑性相关蛋白分子表达的猜测[61]。进行12周中等强度的有氧跑台运动干预后,阿尔茨海默病小鼠行为表现得到改善,通过免疫组织化学法以及Golgi染色法检测发现运动组小鼠大脑海马Aβ沉积减少、突触数量增加;这促进了大脑突触可塑性积极变化,延缓认知水平下降[62]。此外,多项研究发现中等强度运动比高强度运动对海马结构和功能的有益影响更大,中等强度有氧运动干预后突触可塑性相关蛋白水平显著提高,神经细胞凋亡减少,可改善衰老导致的认知水平下降[63-65]。 总的来看,在有氧运动干预阿尔茨海默病中,中低等强度的有氧运动比高强度有氧运动产生更积极的效果;中低等强度有氧运动能够增加突触密度及提升突触可塑性相关蛋白表达;减少突触丢失,延缓阿尔茨海默病患者的认知下降;并且阿尔茨海默病患者坚持低中等有氧运动方案能起到更理想的康复效果。 不同强度有氧运动干预诱导阿尔茨海默病突触可塑性的作用效果差异显著,部分研究信息见表1。"
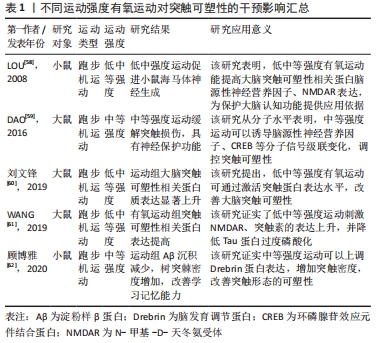
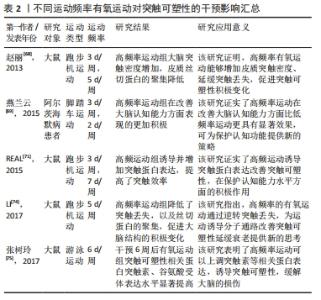
2.3.2 不同运动频率刺激突触可塑性影响 在研究有氧运动对阿尔茨海默病大脑突触可塑性水平影响的文献中,阿尔茨海默病患者的认知水平与运动频率有密切关系。运动频率不同,导致得出的结果也不相同。LUCIA等[66]认为通过每周5 d的30 min中等强度有氧运动可以有效预防认知恶化;而一项综述研究也证实高频率运动比低频率运动对阿尔茨海默病患者认知具有更加显著的积极影响[67]。在一项为期12周的有氧运动干预实验中,发现运动组的大鼠海马突触数目增加且高频运动组的大鼠突触数目增加更为显著,这表明高频率运动可诱导突触可塑性延缓认知衰退[68]。在另外一项为期6个月的有氧运动干预中,将运动组阿尔茨海默病受试者以每周3次和每周2次的锻炼次数为标准分为2组,干预完成后测试结果也显示高频率有氧运动组阿尔茨海默病患者在认知功能、生活能力、神经精神症状以及对照顾者的困扰评分方面获益更多[69]。其他研究指出为期3个月,每周3次的中等强度有氧训练可以改善轻中度阿尔茨海默病患者的认知功能,延缓认知衰退[70]。 此外,基于动物模型的实验证明,高频率运动可刺激突触结构及相关蛋白有益性变化。REAL等[71]对实验大鼠进行为期4周的有氧干预后,进行免疫组织化学检测发现间歇运动组(每周3次)和持续运动组(每周7次)大鼠突触蛋白和谷氨酸受体表达增加,且间歇运动组突触蛋白(神经元突触前终末)增加明显,持续运动组谷氨酸受体表达(神经元突触后部位)水平上升;这强调了高频率有氧运动诱导大脑突触可塑性分子机制。有学者也指出每周5次、持续8周的有氧运动干预可以通过激活分子信号通路明显逆转大鼠脑源性神经营养因子和突触素水平降低[72],促进大鼠海马神经营养因子脑源性神经营养因子和突触蛋白突触素的生成。有研究发现在每周3次、持续6周的50%负荷有氧间歇训练的小鼠脑片中,大脑CA1区记录的突触长时程增强有所增加,对于突触可塑性具有积极正向影响[73]。而在另外一项为期12周有氧干预活动中,将实验大鼠分为对照组、久坐组、低频(每周3次)和高频(每周5次)运动组,经过对大鼠大脑免疫组织化学染色检测,结果显示高频运动组对比其他实验组大鼠大脑突触密度增加更为显著、突触丢失下降明显;这证实了之前的研究发现,高频运动可以在一定程度上阻止衰老大脑的皮质突触素减少,改善伴随突触丢失而导致的认知水平下降[74]。此外,对阿尔茨海默病模型大鼠进行为期6周的有氧运动干预后,大鼠免疫组织化学检测证实高频次有氧运动组大脑突触可塑性相关蛋白Syp以及神经递质受体mGluR1表达水平显著高于对照组,这表明高频次有氧运动可促进突触可塑性积极变化,进而延缓大脑衰老[75]。 已有多数研究认为,高频次有氧运动可以提高大脑突触可塑性,改善阿尔茨海默病患者认知功能,更有利于预防和改善阿尔茨海默病患者的认知衰退,其起作用机制可能因为高频次运动更容易提高患者大脑突触相关蛋白表达,对身体产生积极效益,具体研究见表2。"
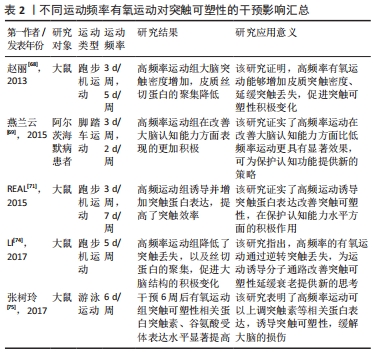
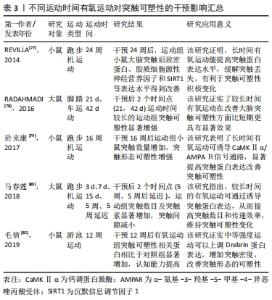
2.3.3 不同运动持续时间刺激突触可塑性影响 无论是短期还是长期有氧运动都可以增加成年齿状回的细胞增殖;然而,短期的有氧运动并不能有效促进成年动物齿状回的长时程增强,只有在跑步56 d时才能观察到长时程增强的可靠增加[76]。REVILLA等[77]发现,6个月有氧运动提高了运动组小鼠海马内SIRT1、神经胶质源性营养因子、突触相关蛋白的表达,从而增强了海马突触可塑性。而相关研究发现,短时间有氧锻炼提高了运动组大鼠海马长时程增强,但是这些运动相关的海马变化是可逆的;只有在持续锻炼(42 d)运动组大鼠大脑才能观察到细胞反应性和长时程增强显著增强。因此,持续有氧运动是改善海马齿状回可塑性的有效方案[78]。在一项为期16周有氧运动干预研究中,发现运动组小鼠海马区突触数目显著增加且突触相关蛋白CaMKⅡα表达水平上升明显,这说明有氧运动可通过调控分子级联信号CaMKⅡα/AMPAR,诱导突触可塑性积极变化[79]。在5周中等强度游泳运动干预后,也观察到持续5周运动组大鼠脑部线粒体分布上升、突触密度增加,海马突触蛋白突触素表达增加;并且运动诱导的海马突触可塑性变化具有运动时间依赖性,短期有氧运动不容易使突触数量发生显著性增加的[80]。对阿尔茨海默病模型大鼠进行为期8周的有氧跑台运动干预后,发现运动组大鼠神经元树突密度显著增加,突触可塑性相关蛋白突触素、突触后致密区蛋白95、脑源性神经营养因子等蛋白水平升高,延缓了认知功能衰退速度[81]。此外,在对阿尔茨海默病模型小鼠进行12周有氧跑台运动干预后,免疫组织化学染色检测发现运动组小鼠淀粉样β蛋白生成减少、海马突触数量显著增加,突触可塑性相关蛋白MAP2、NCAM、神经递质受体(glutamate receptor,GluR1)及下游蛋白分子CaMKⅡ表达水平显著提高,这证实了长期有氧运动可以诱导小鼠突触可塑性,改善记忆和认知能力[82]。 从已有研究来看,短期和中长期有氧运动都可以促进突触可塑性相关蛋白表达上升,但是短期有氧运动诱导的突触相关蛋白变化并不能保持,不能维持成年动物大脑齿状区的长时程增强。中长期有氧运动可以通过诱导分子级联信号上调大脑突触可塑性相关蛋白分子的积极表现,增强大脑突触可塑性,产生良好的缓解认知衰退的效果。中长期规律有氧运动能有效诱导突触相关蛋白分子表达水平,提高阿尔茨海默病大脑的突触可塑性,改善其认知功能下降,部分研究见表3。"
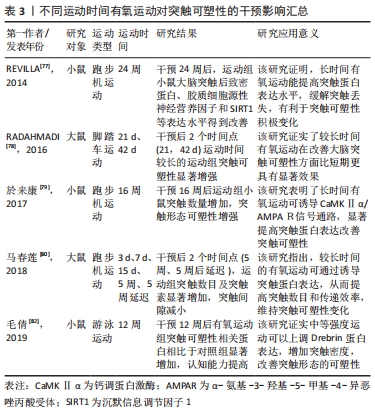
| [1] SERRANO-POZO A, FROSCH MP, MASLIAH E, et al. Neuropathological alterations in Alzheimer disease. Cold Spring Harb Perspect Med. 2011; 1(1):a006189. [2] Lu Y, Dong Y, Tucker D, et al. Treadmill exercise exerts neuroprotection and regulates microglial polarization and oxidative stress in a streptozotocin-induced rat model of sporadic alzheimer’s disease. J Alzheimer’s Dis. 2017;56(4):1469-1484. [3] MORENO-JIMÉNEZ EP, FLOR-GARCÍA M, TERREROS-RONCAL J, et al. Adult hippocampal neurogenesis is abundant in neurologically healthy subjects and drops sharply in patients with Alzheimer’s disease. Nat Med. 2019;25(4):554-560. [4] WANG H, XU X, GAO J, et al. Enriched environment and so‐cial isolation affect cognition ability via altering excitatory and in‐hibitory synaptic density in mice hippocampus. Neurochem Res. 2020. doi: 10.1007/s11064-020-03102-2. [5] LEE SH, LUTZ D, MOSSALAM M, et al. Presenilins regulate synaptic plasticity and mitochondrial calcium homeostasis in the hippocampal mossy fiberpathway. Mol Neurodegener. 2017;12(1):48. [6] ZHANG Y, LI P, FENG J, et al. Dysfunction of NMDA receptors in Alzheimer’s disease. Neurol Sci. 2016;37(7):1039-1047. [7] HOFFMANN K, SOBOL NA, FREDERIKSEN KS, et al. Moderate-to-high intensity physical exercise in patients with Alzheimer’s disease: a randomized controlled trial. J Alzheimers Dis. 2016;50(2):443-453. [8] RAJENDRAN L, PAOLICELLI RC. Microglia-mediated synapse loss in alzheimer’s disease. J Neurosci. 2018;38(12):2911-2919. [9] GOETZL EJ, KAPOGIANNIS D, SCHWARTZ JB, et al. Decreased synaptic proteins in neuronal exosomes of frontotemporal dementia and Alzheimer’s disease. FASEB J. 2016;30(12):4141-4148. [10] 刘慧莉,赵刚.运动、阿尔茨海默病与突触可塑性[J].中国康复理论与实践,2012,18(3):244-246. [11] KAMAT PK, KALANI A, RAI S, et al. Mechanism of oxidative stress and synapse dysfunction in the pathogenesis of alzheimer’s disease: understanding the therapeutics strategies. Mol Neurobiol. 2016;53(1): 648-661. [12] LAMBERT MP, BARLOW AK, CHROMY BA, et al. Diffusible, nonfibrillar ligands derived from Ab1–42 are potent central nervous system neurotoxins. Proc Natl Acad Sci U S A. 1998;95:6448-6453. [13] SHANKAR GM, BLOODGOOD BL, TOWNSEND M, et al. Natural oligomers of the Alzheimer amyloid-beta protein induce reversible synapse loss by modulating an NMDA-type glutamate receptor-dependent signaling pathway. J Neurosci. 2007;27(11):2866-2875. [14] TÖNNIES E, TRUSHINA E. Oxidative stress, synaptic dysfunction, and Alzheimer’s disease. J Alzheimers Dis. 2017;57(4):1105-1121. [15] ZEMPEL H, MANDELKOW E. Lost after translation: missorting of Tau protein and consequences for Alzheimer disease. Trends Neurosci. 2014;37(12):721-732. [16] KOO JH, KANG EB, OH YS, et al. Treadmill exercise decreases amyloid-β burden possibly via activation of SIRT-1 signaling in a mouse model of Alzheimer’s disease. Exp Neurol. 2017;288:142-152. [17] EL HAYEK L, KHALIFEH M, ZIBARA V, et al. Lactate mediates the effects of exercise on learning and memory through SIRT1-dependent activation of hippocampal brain-derived neurotrophic factor (BDNF). J Neurosci. 2019;39(13):2369-2382. [18] SANCHIS-GOMAR F, PEREZ-QUILIS C. The p38-PGC-1α-irisin-betatrophin axis: exploring new pathways in insulin resistance. Adipocyte. 2014; 3(1):67-68. [19] LOURENCO MV, FROZZA RL, DE FREITAS GB, et al. Exercise-linked FNDC5/irisin rescues synaptic plasticity and memory defects in Alzheimer’s models. Nat Med. 2019;25(1):165-175. [20] 邓雨婷,马春莲,杨翼.Irisin:运动改善突触可塑性缓解阿尔茨海默症认知下降的新靶点[J].中国体育科技,2020,56(9):30-36. [21] 房国梁,田野,赵杰修,等.有氧运动对肥胖大鼠海马组织tau蛋白磷酸化水平及PI3K/Akt信号通路的影响[J].中国运动医学杂志, 2017,36(5):375-382. [22] 韩鹏.有氧运动对APP/PS1小鼠中枢GSK3活性及Tau蛋白的影响[D].北京:北京体育大学,2018. [23] LOPRINZI PD, FRITH E. A brief primer on the mediational role of BDNF in the exercise-memory link. Clin Physiol Funct Imaging. 2019;39(1):9-14. [24] MOHAMMADI A, AMOOEIAN VG, RASHIDI E. Dysfunction in brain-derived neurotrophic factor signaling pathway and susceptibility to schizophrenia, parkinson’s and Alzheimer’s diseases. Curr Gene Ther. 2018;18(1):45-63. [25] ROTHMAN SM, MATTSON MP. Activity-dependent, stress-responsive BDNF signaling and the quest for optimal brain health and resilience throughout the lifespan. Neuroscience. 2013;239:228-240. [26] QIN XY, CAO C, CAWLEY NX, et al. Decreased peripheral brain-derived neurotrophic factor levels in Alzheimer’s disease: a meta-analysis study (N=7277). Mol Psychiatry. 2017;22(2):312-320. [27] 付燕,谢攀,李雪,等.长期有氧运动对大鼠脑衰老过程中学习记忆与海马BDNF表达的影响[J].中国运动医学杂志,2015,34(8):750-756. [28] 刘瑾彦,刘向云,陈佩杰,等.易筋经对老年人认知功能和外周血BDNF水平的影响[J].上海体育学院学报,2018,42(2):109-112. [29] LEE HK, TAKAMIYA K, HAN JS, et al. Phosphorylation of the AMPA receptor GluR1 subunit is required for synaptic plasticity and retention of spatial memory. Cell. 2003;112(5):631-643. [30] YU Q, LI X, WANG J, et al. Effect of exercise training on long-term potentiation and NMDA receptor channels in rats with cerebral infarction. Exp Ther Med. 2013;6(6):1431-1436. [31] DAO AT, ZAGAAR MA, ALKADHI KA. Moderate treadmill exercise protects synaptic plasticity of the dentate gyrus and related signaling cascade in a rat model of Alzheimer’s disease. Mol Neurobiol. 2015; 52(3):1067-1076. [32] PLÁTENÍK J, KURAMOTO N, YONEDA Y. Molecular mechanisms associated with long-term consolidation of the NMDA signals. Life Sci. 2000;67(4):335-364. [33] HONG YP, LEE HC, KIM HT. Treadmill exercise after social isolation increases the levels of NGF, BDNF, and synapsin I to induce survival of neurons in the hippocampus, and improves depression-like behavior. J Exerc Nutrition Biochem. 2015;19(1):11-18. [34] MUTLU O, GUMUSLU E, KOKTURK S, et al. Effects of chronic administration of adipokinetic and hypertrehalosemic hormone on animal behavior, BDNF, and CREB expression in the hippocampus and neurogenesis in mice. Fund Clin Pharmacol. 2016;30(1):4-13. [35] STREET ME, SMERIERI A, MONTANINI L, et al. Interactions among pro-inflammatory cytokines, IGF system and thyroid function in pre-pubertal obese subjects. J Biol Regul Homeost Agents. 2013;27(1):259-266. [36] AYADI AE, ZIGMOND MJ, SMITH AD. IGF-1 protects dopamine neurons against oxidative stress: association with changes in phosphokinases. Exp Brain Res. 2016;234(7):1863-1873. [37] FENG X, HUANG D, LU X, et al. Insulin-like growth factor 1 can promote proliferation and osteogenic differentiation of human dental pulp stem cells via mTOR pathway. Dev Growth Differ. 2014;56(9):615-624. [38] FRYSTYK J. Exercise and the growth hormone-insulin-like growth factor axis. Med Sci Sports Exerc. 2010;42(1):58-66. [39] STEIN AM, SILVA TMV, COELHO FGM, et al. Physical exercise, IGF-1 and cognition A systematic review of experimental studies in the elderly. Dement Neuropsychol. 2018;12(2):114-122. [40] HÄKKINEN K, PAKARINEN A, KRAEMER WJ, et al. Selective muscle hypertrophy, changes in EMG and force, and serum hormones during strength training in older women. J Appl Physiol. 2001;91(2):569-580. [41] MAASS A, DÜZEL S, BRIGADSKI T, et al. Relationships of peripheral IGF-1, VEGF and BDNF levels to exercise-related changes in memory, hippocampal perfusion and volumes in older adults. Neuroimage. 2016;131:142-154. [42] 俞嘉玲,马黎,马兰,等. 2014.自主跑轮运动上调成年小鼠海马齿状回区细胞增殖及BDNF、IGF1和WNT4的表达水平[J].生理学报,2014,66(5):559-568. [43] VANZELLA C, NEVES JD, VIZUETE AF, et al. Treadmill running prevents age-related memory deficit and alters neurotrophic factors and oxidative damage in the hippocampus of Wistar rats. Behav Brain Res. 2017;334:78-85. [44] PLOUGHMAN M, GRANTER-BUTTON S, CHERNENKO G, et al. Endurance exercise regimens induce differential effects on brain-derived neurotrophic factor, synapsin-I and insulin-like growth factor I after focal ischemia. Neuroscience. 2005;136(4):991-1001. [45] OGUNDELE OM, PARDO J, FRANCIS J, et al. A Putative mechanism of age-related synaptic dysfunction based on the impact of IGF-1 receptor signaling on synaptic CaMKⅡα phosphorylation. Front Neuroanat. 2018;12:35. [46] SHEPHERD GM, HARRIS KM. Three-dimensional structure and composition of CA3-->CA1 axons in rat hippocampal slices: implications for presynaptic connectivity and compartmentalization. J Neurosci. 1998;18(20):8300-8310. [47] MATTSON MP, LIU D. Mitochondrial potassium channels and uncoupling proteins in synaptic plasticity and neuronal cell death. Biochem Biophys Res Commun. 2003;304(3):539-549. [48] ONYANGO IG, LU J, RODOVA M, et al. Regulation of neuron mitochondrial biogenesis and relevance to brain health. Biochim Biophys Acta. 2010; 1802(1):228-234. [49] FERNANDEZ-MARCOS PJ, AUWERX J. Regulation of PGC-1α, a nodal regulator of mitochondrial biogenesis. Am J Clin Nutr. 2011;93(4): 884S-890S. [50] BULER M, AATSINKI SM, IZZI V, et al. SIRT5 is under the control of PGC-1α and AMPK and is involved in regulation of mitochondrial energy metabolism. FASEB J. 2014;28(7):3225-3237. [51] BAYOD S, GUZMÁN-BRAMBILA C, SANCHEZ-ROIGE S, et al. Voluntary exercise promotes beneficial anti-aging mechanisms in SAMP8 female brain. J Mol Neurosci. 2015;55(2):525-532. [52] KOO JH, KANG EB, OH YS, et al. Treadmill exercise decreases amyloid-β burden possibly via activation of SIRT-1 signaling in a mouse model of Alzheimer’s disease. Exp Neurol. 2017;288:142-152. [53] AZIMI M, GHARAKHANLOU R, NAGHDI N, et al. Moderate treadmill exercise ameliorates amyloid-β-induced learning and memory impairment, possibly via increasing AMPK activity and up-regulation of the PGC-1α/FNDC5/BDNF pathway. Peptides. 2018;102:78-88. [54] BRIONES TL, SUH E, JOZSA L, et al. Changes in number of synapses and mitochondria in presynaptic terminals in the dentate gyrus following cerebral ischemia and rehabilitation training. Brain Res. 2005; 1033(1):51-57. [55] KOŠČAK TIVADAR B. Physical activity improves cognition: possible explanations. Biogerontology. 2017;18(4):477-483. [56] GHODRATI-JALDBAKHAN S, AHMADALIPOUR A, RASHIDY-POUR A, et al. Low- and high-intensity treadmill exercise attenuates chronic morphine-induced anxiogenesis and memory impairment but not reductions in hippocampal BDNF in female rats. Brain Res. 2017;1663:20-28. [57] ORMEROD BK, LEE TT, GALEA LA. Estradiol initially enhances but subsequently suppresses (via adrenal steroids) granule cell proliferation in the dentate gyrus of adult female rats. J Neurobiol. 2003;55(2):247-260. [58] LOU SJ, LIU JY, CHANG H, et al. Hippocampal neurogenesis and gene expression depend on exercise intensity in juvenile rats. Brain Res. 2008;1210:48-55. [59] DAO AT, ZAGAAR MA, LEVINE AT, et al. Comparison of the effect of exercise on late-phase LTP of the dentate gyrus and CA1 of Alzheimer’s disease model. Mol Neurobiol. 2016;53(10):6859-6868. [60] 刘文锋,刘少鹏,傅让,等.耐力运动对增龄大鼠脑皮层突触可塑性的影响及相关调控机制[J].中国应用生理学杂志,2019,35(4):8. [61] WANG Q, HU J, LIU Y, et al. Aerobic exercise improves synaptic-related proteins of diabetic rats by inhibiting FOXO1/NF-κB/NLRP3 inflammatory signaling pathway and ameliorating PI3K/Akt insulin signaling pathway. J Mol Neurosci. 2019;69(1):28-38. [62] 顾博雅,高姗姗,赵丽.有氧运动对调节AD模型小鼠海马Ras/Drebrin增加树突棘可塑性分析[J].北京体育大学学报,2020,43(1): 126-133. [63] KIM HJ, LEE HJ, SO B, et al. Effect of aerobic training and resistance training on circulating irisin level and their association with change of body composition in overweight/obese adults: a pilot study. Physiol Res. 2016;65(2):271-279. [64] LI J, LIU Y, LIU B, et al. Mechanisms of aerobic exercise upregulating the expression of hippocampal synaptic plasticity-associated proteins in diabetic rats. Neural Plast. 2019;2019:7920540. [65] INOUE K, HANAOKA Y, NISHIJIMA T, et al. Long-term mild exercise training enhances hippocampus-dependent memory in rats. Int J Sports Med. 2015;36(4):280-285. [66] LUCIA A, RUIZ JR. Exercise is beneficial for patients with Alzheimer’s disease: a call for action. Br J Sports Med. 2011;45(6):468-469. [67] 刘军,刘仁仪,叶星.运动对阿尔茨海默病患者认知功能干预效果的meta分析[J].上海体育学院学报,2020,44(10):58-67. [68] 赵丽,李岩,顾博雅.有氧运动调节Rho/cofilin信号改善衰老大鼠皮质突触丢失[J].北京体育大学学报, 2013,36(11):61-65, 69. [69] 燕兰云,王蔚,沈飞飞,等.不同训练时间的有氧运动干预轻中度阿尔茨海默病的临床研究[J].中国康复医学杂志,2015,30(8):771-776. [70] 刘银,王彤,朱奕,等.中等强度有氧运动改善阿尔茨海默病患者认知功能的研究[J].中国康复,2017,32(5):386-389. [71] REAL CC, GARCIA PC, BRITTO LRG, et al. Different protocols of treadmill exercise induce distinct neuroplastic effects in rat brain motor areas. Brain Res. 2015;1624:188-198. [72] CAI M, WANG H, LI JJ, et al. The signaling mechanisms of hippocampal endoplasmic reticulum stress affecting neuronal plasticity-related protein levels in high fat diet-induced obese rats and the regulation of aerobic exercise. Brain Behav Immun. 2016;57:347-359. [73] D’ARCANGELO G, TRIOSSI T, BUGLIONE A, et al. Modulation of synaptic plasticity by short-term aerobic exercise in adult mice. Behav Brain Res. 2017;332:59-63. [74] LI Y, ZHAO L, GU B, et al. Aerobic exercise regulates Rho/cofilin pathways to rescue synaptic loss in aged rats. PLoS One. 2017;12(2):e0171491. [75] 张树玲,李雪,袁琼嘉,等.衰老过程中有氧运动干预对海马突触可塑性及PDE-4基因表达的影响[J].中国运动医学杂志,2017, 36(10):875-881. [76] PATTEN AR, SICKMANN H, HRYCIW BN, et al. Long-term exercise is needed to enhance synaptic plasticity in the hippocampus. Learn Mem. 2013;20(11):642-647. [77] REVILLA S, SUÑOL C, GARCÍA-MESA Y, et al. Physical exercise improves synaptic dysfunction and recovers the loss of survival factors in 3xTg-AD mouse brain. Neuropharmacology. 2014;81:55-63. [78] RADAHMADI M, HOSSEINI N, ALAEI H. Effect of exercise, exercise withdrawal, and continued regular exercise on excitability and long-term potentiation in the dentate gyrus of hippocampus. Brain Res. 2016;1653:8-13. [79] 於来康,顾博雅,李岩,等.有氧运动调节APP/PS1小鼠海马CaMKⅡα, AMPAR活性,增加突触可塑性[J].北京体育大学学报, 2017,40(1):41-45, 89. [80] 马春莲,丁海超,梅志强,等.中等强度游泳运动对海马突触可塑性的调节[J].体育科学,2018,38(3):34-39, 47. [81] 刘涛,白石,黄悦.跑台训练对阿尔茨海默病模型大鼠记忆能力和突触可塑性的影响[J].中国康复医学杂志,2016,31(12):1301-1306. [82] 毛倩.跑台运动对TgAPP/PS1小鼠海马离子型谷氨酸受体介导的突触可塑性的影响研究[D].上海:华东师范大学,2019. |
| [1] | Gu Zhengqiu, Xu Fei, Wei Jia, Zou Yongdi, Wang Xiaolu, Li Yongming. Exploratory study on talk test as a measure of intensity in blood flow restriction training [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1154-1159. |
| [2] | Wen Xiaoyu, Sun Yuhao, Xia Meng. Effects of serum containing Wuzang Wenyang Huayu Decoction on phosphorylated-tau protein expression in Alzheimer’s disease cell model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1068-1073. |
| [3] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [4] | Shui Xiaoping, Li Chunying, Li Shunchang, Sun Junzhi, Su Quansheng . Effects of aerobic and resistance exercises on brain-derived neurotrophic factor, nuclear factor-kappa B and inflammatory cytokines in skeletal muscle of type II diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 669-675. |
| [5] | Wei Xing, Liu Shufang, Mao Ning. Roles and values of blood flow restriction training in the rehabilitation of knee joint diseases [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 774-779. |
| [6] | Tang Jiping, Zhang Yeting. Exercise regulates adult hippocampal neurogenesis in Alzheimer’s disease: mechanism and role [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 798-803. |
| [7] | Huang Chuanjun, Zou Yu, Zhou Xiaoting, Zhu Yangqing, Qian Wei, Zhang Wei, Liu Xing. Transplantation of umbilical cord mesenchymal stem cells encapsulated in RADA16-BDNF hydrogel promotes neurological recovery in an intracerebral hemorrhage rat model [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 510-515. |
| [8] | Liu Ya, Liu Xia, Deng Penghui, Ji Wei, Li Jianping. Exercise effects on myocardial type I, III collagen and angiotensin II/transforming growth factor beta1/Smad2 pathway in diabetic myocardial fibrosis rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(26): 4173-4179. |
| [9] | Wang Yuyin, Wei Wenyue, Guo Minfang, Li Hongxia, Zhang Jing, Gu Qingfang, Liu Xiaoqin, Guo Xiaoping, Song Lijuan, Chai Zhi, Ma Cungen, Wei Jiezhong. Bushen Yizhi Anti-aging Prescription improves cognitive function of APP/PS1 mice by regulating microglia and macrophage polarization [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(26): 4166-4172. |
| [10] | Ao Pian, Zhao Xin, Yu Hongrong, Zhang Siqin, Zhang Xinyue, Gu Weili, Wei Li. Laminaria japonica polysaccharide protects radiation-induced brain injury by regulating the blood-brain barrier in mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(20): 3158-3163. |
| [11] | Liu Yulu, Jia Weiwei, Dai Yaling, Xu Wenshan, Ding Yanyi, Liang Shengxiang, Liu Weilin, Chen Lidian. Effects of time-specific AMP-activated protein kinase alpha1/2 gene knockout on hippocampal energy metabolism and synaptic plasticity in mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(20): 3230-3235. |
| [12] | Wei Wenyue, Wang Yuyin, Guo Minfang, Zhang Jing, Gu Qingfang, Song Lijuan, Chai Zhi, Yu Jiezhong, Ma Cungen. Fasudil inhibits neuronal apoptosis via regulating mitochondrial dynamics in APP/PS1 mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 232-238. |
| [13] | Gu Qingfang, Guo Minfang, Liu Xiaoqin, Mu Bingtao, Li Weimei, Song Lijuan, Chai Zhi, Ma Cungen, Yu Jiezhong. Mechanism by which bone marrow mesenchymal stem cells improve cognitive function in APP/PS1 double transgenic mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 2964-2969. |
| [14] | Gong Chao, Zhi Xiaodong, Zhang Yuqiang, Wang Chenliang, Wang Kang, Wang Wei. Ciliary neurotrophic factor combined with forskolin and 3-isobutyl-1-methylxanthine induced muscle derived stem cells to differentiate into Schwann cells phenotype through cyclic adenosine monophosphate signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 2970-2977. |
| [15] | Zhang Shuling, Li Junhan, Wang Jiaqian, Li Yalong, Wang Chun. Effects of aerobic exercises on JAK2/STAT5 signal pathway in the liver of mice with non-alcoholic fatty liver disease [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(17): 2690-2695. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||