Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (8): 1149-1155.doi: 10.12307/2022.1014
Correlation between exercise intensity and lipid metabolism disorder and oxidative stress in a high-diet rat model
Ruan Ling, Wang Guanghua, Wu Rongping, Jin Zhan, Lyu Zhenqing, Zhang Nan, Li Shoubang
- Department of Physical Education, Xi’an Shiyou University, Xi’an 710065, Shaanxi Province, China
-
Received:2021-12-13Accepted:2022-02-15Online:2023-03-18Published:2022-07-27 -
Contact:Li Shoubang, PhD, Professor, Master’s supervisor, Department of Physical Education, Xi’an Shiyou University, Xi’an 710065, Shaanxi Province, China -
About author:Ruan Ling, PhD, Associate professor, Department of Physical Education, Xi’an Shiyou University, Xi’an 710065, Shaanxi Province, China -
Supported by:Shaanxi Natural Science Basic Research Program, No. 2021JQ-586 (to RL)
CLC Number:
Cite this article
Ruan Ling, Wang Guanghua, Wu Rongping, Jin Zhan, Lyu Zhenqing, Zhang Nan, Li Shoubang. Correlation between exercise intensity and lipid metabolism disorder and oxidative stress in a high-diet rat model[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1149-1155.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
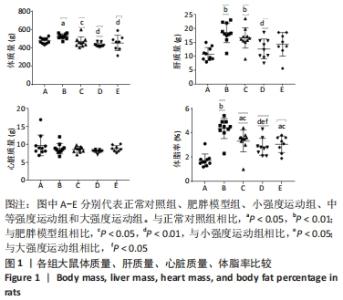
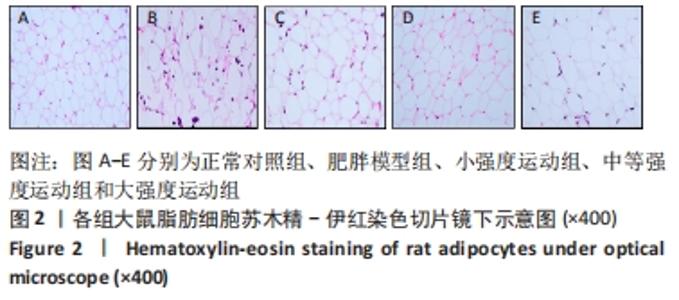
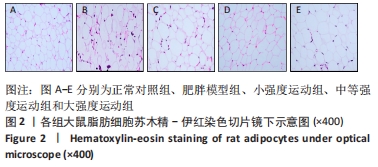
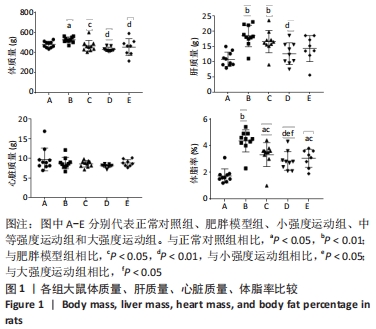
2.1 实验动物数量分析 共纳入大鼠50只,分为5组,后因肥胖大鼠模型构建,有3只大鼠未达到建模标准,故进入结果分析的实验动物数量共47只,其中正常对照组10只,肥胖模型组10只,小强度运动组10只,中等强度运动组9只,大强度运动组8只。 2.2 各组大鼠体质量、各器官质量和体脂率的变化 与正常对照组相比,肥胖模型组大鼠的体质量、体脂率和肝质量均显著增加(P < 0.05,P < 0.01,P < 0.01)。与肥胖模型组相比,小强度运动组、中等强度运动和大强度运动组的大鼠体质量显著降低(P < 0.05,P < 0.01,P < 0.01),中等强度运动组肝脏质量也显著下降(P < 0.01)。与正常对照组相比,肥胖模型组、小强度运动组和大强度运动组的体脂率均显著升高(P < 0.01,P < 0.05,P < 0.05);与肥胖模型组相比,小强度运动组、中等强度运动组和大强度运动组体脂率均显著降低(P < 0.05,P < 0.01,P < 0.05);与小强度运动组和大强度运动组相比,中等强度运动组体脂率显著降低(P < 0.05,P < 0.05),见图1。"
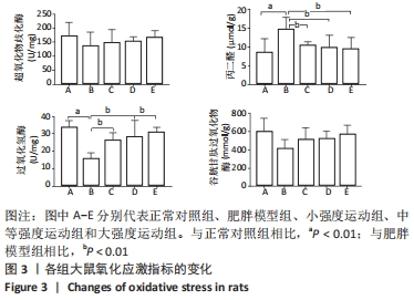
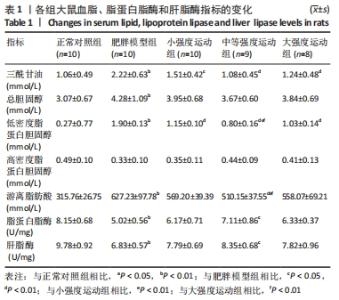
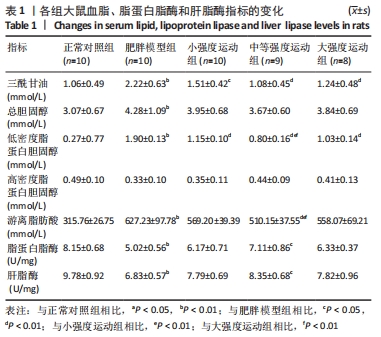
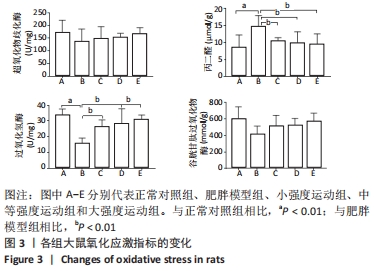
与正常对照组相比,肥胖模型组三酰甘油、总胆固醇、游离脂肪酸和低密度脂蛋白胆固醇水平明显升高(P < 0.01)。与肥胖模型组相比,小强度运动组、大强度运动和中等强度运动组三酰甘油水平显著降低(P < 0.05,P < 0.01,P < 0.01);与小强度运动组和大强度运动组相比,中等强度运动组游离脂肪酸水平显著降低(P < 0.01);与肥胖模型组相比,小强度运动组、中等强度运动组和大强度运动组低密度脂蛋白胆固醇水平均显著降低(P < 0.01);与小强度运动组和大强度运动组相比,中等强度运动组低密度脂蛋白胆固醇水平显著降低(P < 0.01);而总胆固醇和高密度脂蛋白水平,3个运动干预组之间差异无显著性意义(P > 0.05)。 与正常对照组相比,肥胖模型组脂蛋白脂酶和肝脂酶活性显著降低(P < 0.01),其他各运动组脂蛋白脂酶和肝脂酶活性均高于肥胖模型组,但仅有中等强度运动组有统计学差异(P < 0.05)。 2.5 各组大鼠氧化应激水平的变化 与正常对照组相比,肥胖模型组丙二醛水平显著升高(P < 0.01);小强度运动组、中等强度运动组和大强度运动组丙二醛水平均显著低于肥胖模型组(P < 0.01)。与正常对照组相比,肥胖模型组过氧化氢酶活性显著降低(P < 0.01);与肥胖模型组相比,各运动组过氧化氢酶活性均显著升高(P < 0.01)。但超氧化物歧化酶和谷胱甘肽过氧化物酶活性各组之间差异无显著性意义(P > 0.05)。见图3。 "
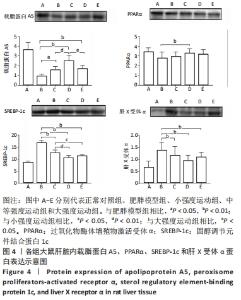
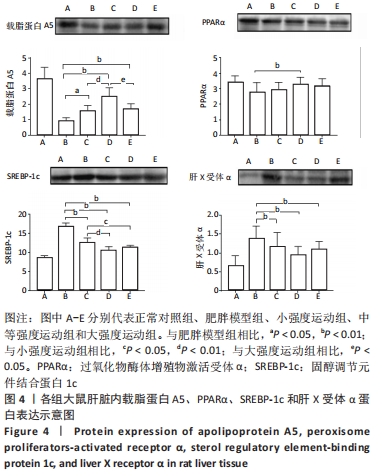
2.6 各组大鼠肝组织中载脂蛋白A5、PPARα、SREBP-1c和肝X受体α蛋白含量的变化 与肥胖模型组相比,小强度运动组、中等强度运动组和大强度运动组中的SREBP-1c和肝X受体α水平均显著降低(P < 0.01),载脂蛋白A5水平3个运动组均显著升高(P < 0.05,P < 0.01,P < 0.01),PPARα水平仅中等强度运动组表达显著升高(P < 0.01)。与小强度运动组相比,中等强度运动组的载脂蛋白A5水平表达显著增高(P < 0.01),SREBP-1c的表达在中等强度运动组和大强度运动组显著降低(P < 0.01,P < 0.05)。与大强度运动组相比,中等强度运动组的载脂蛋白A5表达显著升高(P < 0.05),见图4。"
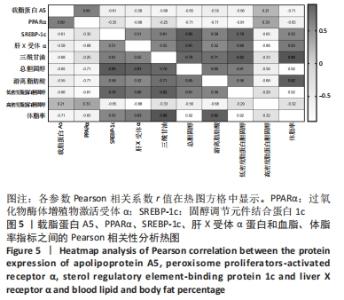
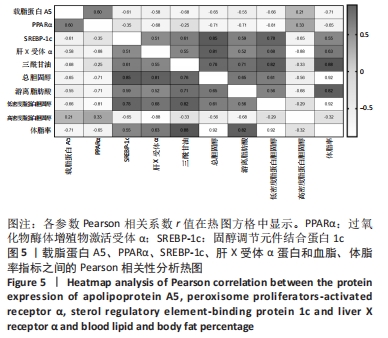
2.7 载脂蛋白A5、PPARα、SREBP-1c、肝X受体α蛋白和血脂、体脂率指标的相关性分析 载脂蛋白A5、PPARα、高密度脂蛋白胆固醇分别与肝X受体α、SREBP-1c、三酰甘油、总胆固醇、游离脂肪酸、低密度脂蛋白胆固醇和体脂率呈负相关;载脂蛋白A5与PPARα和高密度脂蛋白胆固醇呈正相关;肝X受体α 和SREBP-1c与三酰甘油、总胆固醇、游离脂肪酸、低密度脂蛋白胆固醇和体脂率呈正相关;载脂蛋白A5与体脂率呈正相关(P < 0.05),PPARα与游离脂肪酸(P < 0.01)和低密度脂蛋白胆固醇(P < 0.05)呈负相关,SREBP-1c与总胆固醇和低密度脂蛋白胆固醇(P < 0.01, P < 0.05)呈正相关;肝X受体α 与总胆固醇呈正相关(P < 0.01),肝X受体α与高密度脂蛋白胆固醇呈负相关(P < 0.01);体脂率与三酰甘油、总胆固醇、游离脂肪酸和低密度脂蛋白胆固醇呈正相关(P < 0.01),见图5。"
| [1] PANDA V, MISTRY K, SUDHAMANI S, et al. Amelioration of Abnormalities Associated with the Metabolic Syndrome by Spinacia oleracea (Spinach) Consumption and Aerobic Exercise in Rats. Oxid Med Cell Longev. 2017; 2017:2359389. [2] PANDA V, SHINDE P, DANDE P. Consumption of Spinacia Oleracea (spinach) and aerobic exercise controls obesity in rats by an inhibitory action on pancreatic lipase. Arch Physiol Biochem. 2020;126(3):187-195. [3] MANN S, BEEDIE C, JIMENEZ A. Differential effects of aerobic exercise, resistance training and combined exercise modalities on cholesterol and the lipid profile: review, synthesis and recommendations. Sports Med. 2014;44(2):211-221. [4] HARRISON M, MOYNA NM, ZDERIC TW, et al. Lipoprotein particle distribution and skeletal muscle lipoprotein lipase activity after acute exercise. Lipids Health Dis. 2012;11:64. [5] WANG Y, NAKAJIMA T, GONZALEZ FJ, et al. PPARs as Metabolic Regulators in the Liver: Lessons from Liver-Specific PPAR-Null Mice. Int J Mol Sci. 2020;21(6):2061. [6] KAZEMINASAB F, MARANDI M, GHAEDI K, et al. Effects of A 4-Week Aerobic Exercise on Lipid Profile and Expression of LXRalpha in Rat Liver. Cell J. 2017;19(1):45-49. [7] BEYER TP, SCHMIDT RJ, FOXWORTHY P, et al. Coadministration of a liver X receptor agonist and a peroxisome proliferator activator receptor-alpha agonist in Mice: effects of nuclear receptor interplay on high-density lipoprotein and triglyceride metabolism in vivo. J Pharmacol Exp Ther. 2004;309(3):861-868. [8] JAKEL H, NOWAK M, MOITROT E, et al. The liver X receptor ligand T0901317 down-regulates APOA5 gene expression through activation of SREBP-1c. J Biol Chem. 2004;279(44):45462-45469. [9] SUCHANEK P, LORENZOVA A, POLEDNE R, et al. Changes of plasma lipids during weight reduction in females depends on APOA5 variants. Ann Nutr Metab. 2008;53(2):104-108. [10] SERRANO M, GRASA MDEL M, JANER G, et al. Oleoyl-estrone affects lipid metabolism in adrenalectomized rats treated with corticosterone through modulation of SREBP1c expression. J Steroid Biochem Mol Biol. 2009;117(1-3):15-22. [11] COTE I, NGO SOCK ET, LEVY E, et al. An atherogenic diet decreases liver FXR gene expression and causes severe hepatic steatosis and hepatic cholesterol accumulation: effect of endurance training. Eur J Nutr. 2013;52(5):1523-1532. [12] ZHANG R, YU Y, HU S, et al. Sesamin ameliorates hepatic steatosis and inflammation in rats on a high-fat diet via LXRalpha and PPARalpha. Nutr Res. 2016;36(9):1022-1030. [13] 朱天逸, 张蕴琨. 运动对大鼠肝脏FFA、PPARα和CPT-1的影响[J]. 南京体育学院学报(自然科学版),2017,16(4):39-43. [14] MACHADO MV, VIEIRA AB, NASCIMENTO AR, et al. Physical exercise restores microvascular function in obese rats with metabolic syndrome. Metab Syndr Relat Disord. 2014;12(9):484-492. [15] MACHADO MV, MARTINS RL, BORGES J, et al. Exercise Training Reverses Structural Microvascular Rarefaction and Improves Endothelium-Dependent Microvascular Reactivity in Rats with Diabetes. Metab Syndr Relat Disord. 2016;14(6):298-304. [16] BAETGE C, EARNEST CP, LOCKARD B, et al. Efficacy of a randomized trial examining commercial weight loss programs and exercise on metabolic syndrome in overweight and obese women. Appl Physiol Nutr Metab. 2017;42(2):216-227. [17] 汪军, 姚珂, 王瑞元. 8周跑台运动对肥胖大鼠血浆和下丘脑神经肽Y蛋白及其基因表达的影响[J]. 中国运动医学杂志,2008,27(6): 707-710. [18] BEDFORD TG, TIPTON CM, WILSON NC, et al. Maximum oxygen consumption of rats and its changes with various experimental procedures. J Appl Physiol Respir Environ Exerc Physiol. 1979;47(6): 1278-1283. [19] HAFSTAD AD, LUND J, HADLER-OLSEN E, et al. High- and moderate-intensity training normalizes ventricular function and mechanoenergetics in mice with diet-induced obesity. Diabetes. 2013; 62(7):2287-2294. [20] 刘海涛, 江大雷. 中等强度运动对去卵巢大鼠肝脏SREBP-1c表达的影响[J]. 天津体育学院学报,2016,31(3):259-263. [21] 李玉辉. 中等强度运动促进脑缺血再灌注大鼠脑功能修复[J]. 基因组学与应用生物学,2020,39(10):4761-4766. [22] 朱小烽, 王茹, 杨钦, 等. 不同强度急性有氧运动对肥胖小鼠PGC-1α及其下游因子的调控影响[J]. 体育科学,2017,37(3):44-50. [23] GREMEAUX V, DRIGNY J, NIGAM A, et al. Long-term lifestyle intervention with optimized high-intensity interval training improves body composition, cardiometabolic risk, and exercise parameters in patients with abdominal obesity. Am J Phys Med Rehabil. 2012; 91(11):941-950. [24] DUVAL C, THISSEN U, KESHTKAR S, et al. Adipose tissue dysfunction signals progression of hepatic steatosis towards nonalcoholic steatohepatitis in C57BL/6 mice. Diabetes. 2010;59(12):3181-3191. [25] VAN DER HEIJDEN RA, SHEEDFAR F, MORRISON MC, et al. High-fat diet induced obesity primes inflammation in adipose tissue prior to liver in C57BL/6j mice. Aging (Albany NY). 2015;7(4):256-268. [26] DU PLESSIS J, VAN PELT J, KORF H, et al. Association of Adipose Tissue Inflammation With Histologic Severity of Nonalcoholic Fatty Liver Disease. Gastroenterology. 2015;149(3):635-648 e14. [27] 张崇林, 王前进, 刘绍生, 等. 有氧运动对代谢综合征大鼠氧化应激及心肌PPARα表达的影响[J]. 卫生研究,2018,47(5):789-793+797. [28] SAKURAI T, IZAWA T, KIZAKI T, et al. Exercise training decreases expression of inflammation-related adipokines through reduction of oxidative stress in rat white adipose tissue. Biochem Biophys Res Commun. 2009;379(2):605-609. [29] SILVEIRA LS, BIONDO LA, DE SOUZA TEIXEIRA AA, et al. Macrophage immunophenotype but not anti-inflammatory profile is modulated by peroxisome proliferator-activated receptor gamma (PPARgamma) in exercised obese mice. Exerc Immunol Rev. 2020;26:10-22. [30] LIRA FS, ROSA JC, YAMASHITA AS, et al. Endurance training induces depot-specific changes in IL-10/TNF-alpha ratio in rat adipose tissue. Cytokine. 2009;45(2):80-85. [31] Alborn WE, Johnson MG, Prince MJ, et al. Definitive N-terminal protein sequence and further characterization of the novel apolipoprotein A5 in human serum. Clin Chem. 2006;52(3):514-517. [32] REY M, KRUSE MS, MAGRINI-HUAMAN RN, et al. High-Fat Diets and LXRs Expression in Rat Liver and Hypothalamus. Cell Mol Neurobiol. 2019;39(7):963-974. [33] OKAZAKI H, GOLDSTEIN JL, BROWN MS, et al. LXR-SREBP-1c-phospholipid transfer protein axis controls very low density lipoprotein (VLDL) particle size. J Biol Chem. 2010;285(9):6801-6810. [34] LA FUENTE FP, QUEZADA L, SEPULVEDA C, et al. Exercise regulates lipid droplet dynamics in normal and fatty liver. Biochim Biophys Acta Mol Cell Biol Lipids. 2019;1864(12):158519. [35] SANTAMARINA-FOJO S, GONZALEZ-NAVARRO H, FREEMAN L, et al. Hepatic lipase, lipoprotein metabolism, and atherogenesis. Arterioscler Thromb Vasc Biol. 2004;24(10):1750-1754. [36] 秦英楠, 李向平. 载脂蛋白A5对甘油三酯代谢的影响及其机制[J]. 中国动脉硬化杂志,2010,18(6):495-498. |
| [1] | Fang Xingyan, Tian Zhenli, Zhao Zheyi, Wen Ping, Xie Tingting. Effects of sodium arsenite on human umbilical vein endothelial cell injury and sphingosine kinases 1/sphingosine 1-phosphate signaling axis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [2] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [3] | Guo Shuhui, Yang Ye, Jiang Yangyang, Xu Jianwen. Screening and validation of neurogenic bladder miRNA-mRNA regulatory network [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-8. |
| [4] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [5] | Wu Dongzhe, Gao Xiaolin, Li Chuangtao, Wang Hao. Constructing the prediction model of maximal oxygen uptake by back-propagation neural network based on the cardiorespiratory optimal point [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1224-1231. |
| [6] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [7] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [8] | Liang Jiaqi, Liu Hengxu, Yang Jinxin, Yang Yi, Deng Xuhui, Tan Mingjian, Luo Jiong. Health benefit relationship between exercise and intestinal bacteria [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1292-1299. |
| [9] | Huang Linke, Wei Linhua, Jiang Jie, Liu Qian, Chen Weiwei. Effects of estrogen combined with treadmill exercise on bone mass and articular cartilage in ovariectomized mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1166-1171. |
| [10] | Zhang Yan, He Ruibo, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Ma Gang, Peng Peng. Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1237-1244. |
| [11] | Tian Qinyu, Tian Xinggui, Tian Zhuang, Sui Xiang, Liu Shuyun, Lu Xiaobo, Guo Quanyi. Protection of manganese oxide nanoparticles for bone marrow mesenchymal stem cell spreading against oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 821-826. |
| [12] | Li Xiaoyin, Yang Xiaoqing, Chen Shulian, Li Zhengchao, Wang Ziqi, Song Zhen, Zhu Daren, Chen Xuyi. Collagen/silk fibroin scaffold combined with neural stem cells in the treatment of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 890-896. |
| [13] | Huang Guijiang, Ji Yuwei, Zhao Xin, Yang Yi, Zhao Yulan, Wang Peijin, Tang Wei, Jiao Jianlin. Effect and mechanism of different administration routes of placenta-derived mesenchymal stem cells in the treatment of tree shrews with osteoporotic fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 909-914. |
| [14] | Li Yujiao, Su Kunxia. High-intensity endurance exercise influences browning of white adipose tissue in a mouse model of high-fat diet induced obesity [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 707-713. |
| [15] | Li Zhichao, Tan Guoqing, Su Hui, Xu Zhanwang, Xue Haipeng. Regulatory role of non-coding RNAs as potential therapeutic targets in spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 758-764. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||