Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (5): 789-794.doi: 10.12307/2024.245
Previous Articles Next Articles
Preparation and application of injectable sodium alginate hydrogels
Chen Xiaofang1, 2, Zheng Guoshuang2, Li Maoyuan1, 2, Yu Weiting1, 2
- 1Zhongshan Clinical College of Dalian University, Dalian 116001, Liaoning Province, China; 2Department of Orthopedics, Affiliated Zhongshan Hospital of Dalian University, Dalian 116001, Liaoning Province, China
-
Received:2022-12-08Accepted:2023-03-04Online:2024-02-18Published:2023-08-17 -
Contact:Yu Weiting, PhD, Researcher, Master’s supervisor, Zhongshan Clinical College of Dalian University, Dalian 116001, Liaoning Province, China; Department of Orthopedics, Affiliated Zhongshan Hospital of Dalian University, Dalian 116001, Liaoning Province, China -
About author:Chen Xiaofang, Master candidate, Zhongshan Clinical College of Dalian University, Dalian 116001, Liaoning Province, China; Department of Orthopedics, Affiliated Zhongshan Hospital of Dalian University, Dalian 116001, Liaoning Province, China -
Supported by:Key Research and Development Program of Liaoning Province, No. 2020JH2/10300093 (to YWT); Innovative Talents Support Program for Universities of Liaoning Province, No. Liaoning Jiaohan [2018]478 (to YWT); “Millions of Talents Project” of Liaoning Province, No. Liaoning People’s Press [2019]45 (to YWT)
CLC Number:
Cite this article
Chen Xiaofang, Zheng Guoshuang, Li Maoyuan, Yu Weiting. Preparation and application of injectable sodium alginate hydrogels[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 789-794.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
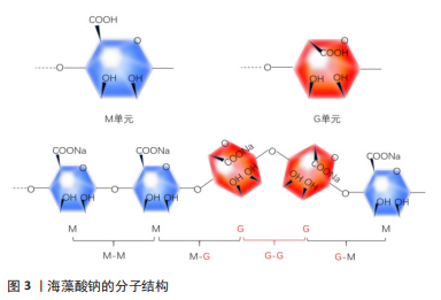
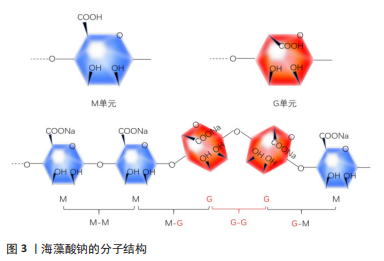
2.2 海藻酸钠的理化性质 2.2.1 生物相容性 海藻酸钠具有良好的抗肿瘤、抗氧化、抗菌等生物活性,因此,海藻酸钠良好的生物相容性对其作为人体可注射材料在组织工程上应用具有重要意义。 2.2.2 生物降解性 海藻酸钠的物理化学性质与其成分及配比的关系密切,海藻酸类物质在一定程度上会发生降解,其主要特点是黏度下降、分子质量发生改变,主要有2种:①氧化分解:在强酸、碱、高温、高压、物理射线等环境下,或在有氧化剂的情况下,海藻酸盐分子的糖苷键断裂、海藻酸盐大分子分解,从而导致海藻酸盐分子质量下降;②酶降解:海藻酸盐水解酶可特异性识别 GG、GM、MM片段,使其降解为含有特定 M、G单元的降解产物,为生产具有特殊结构和物理性能的海藻酸盐,以满足医药行业的性能需求提供原材料。 2.2.3 易加工修饰性 由于海藻酸钠能迅速与二价阳离子Ba2+、Ca2+在常温常压下形成凝胶,该工艺操作简单、容易成型,并且整个反应过程可在生理条件下进行,使得海藻酸盐材料在组织工程领域尤其在可注射水凝胶制备方面显示出独特的优势。LIU等[10]利用近红外光可以对肿瘤进行治疗,对海藻酸钠进行改性使其具有光敏性,从而实现了在治疗肿瘤的同时能达到原位成胶的效果。DANG等[11]通过在海藻酸钠上接枝温敏性基团,体内注射后在人体温度下缓慢原位成胶。 海藻酸盐是一种聚阴离子天然高分子材料,细胞难以黏附在海藻酸盐水凝胶上。研究者将生物活性物质修饰到海藻酸盐的羧基或羟基基团上,从而改善海藻酸盐基水凝胶的生物学性能,对支架上的细胞黏附、增殖及成骨相关基因的表达具有重要意义。将精氨酸-甘氨酸-天冬氨酸活性肽修饰到海藻酸盐水凝胶上,显著提升了细胞的黏附作用及增殖能力。利用RGD活性肽修饰海藻酸盐的研究报道颇多,ZHANG等[12]研究发现活性肽修饰的海藻酸盐水凝胶对人牙髓干细胞的黏附能力显著增强,并在一定程度上增加了成骨标志物基因的表达。 2.3 可注射的海藻酸钠水凝胶的制备方法 可注射海藻酸钠水凝胶的制备方法主要有2种:化学和物理交联,见表1。最常用的是物理离子交联法,其大部分交联剂无毒无害,有利于在人体内使用,而且基本不受自然环境限制,可以节约运输成本,同时离子交联法操作简单、方便快捷。但海藻酸钠水凝胶的性能相对单一,共价交联的海藻酸钠水凝胶增加了可注射水凝胶的可塑性,并可通过对其进行化学修饰改性来调控凝胶化时间、凝胶化方式赋予海藻酸钠凝胶更多功能。"
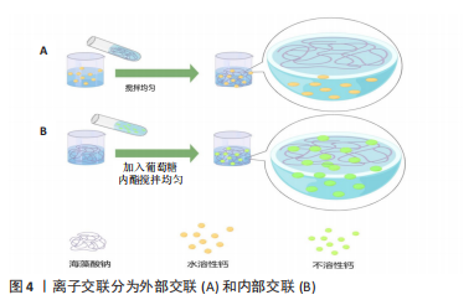
2.3.1 物理交联的可注射海藻酸钠水凝胶 (1)离子交联法:是制备海藻酸钠凝胶的最常用方法。结果表明,海藻酸钠的G单元容易与二价离子发生结合反应,而不同的二价金属离子对海藻酸钠的亲和性也有一定的差异,并按其键合力大小依次为:Pb2+ > Cu2+ > Cd2+ > Ba2+ > Sr2+ > Ca2+> Zn2+ > Mn2+[22]。另外,海藻酸还能与部分三价金属离子(如Fe3+[23-25])形成络合物,但因其上的氧原子会与三价金属离子相互吸引而使这种结构不稳[26]。而 Mn2+和Cu2+等离子对人体有一定的毒性[10-28],因此在生物医药领域应用时,常常选用 Ca2+ 作为交联剂[22]。 通过离子交联获得藻酸盐水凝胶可分为2种:内部凝胶化和外部凝胶化,如图4所示。外部凝胶化一般是向含有Ca2+的溶液中加入海藻酸钠溶液,多数可注射海藻酸钠水凝胶是与注射部位自身来源的钙离子交联而成的[13,29-34]。另外,最常用的外部钙离子来源的交联剂是氯化钙(CaCl2)溶液[12,35-48],由于CaCl2交联的海藻酸钠成胶速度很快,所以通常先制作成可通过注射器针孔大小的海藻酸钙微球[12,35,40,42,49],或将CaCl2溶液加入海藻酸钠溶液[36,38,41],从而形成水凝胶。ZHANG等[13]利用脊髓损伤的区域聚集大量Ca2+来原位交联海藻酸钠,设计了一种含碱性成纤维细胞生长因子的海藻酸钠缓释系统,在体内原位给药。XIONG等[32]制备了一种静脉注射多柔比星负载的聚乳酸-羟基乙酸共聚物纳米粒子涂层与细胞膜和肿瘤内注射淋巴趋化素负载钠海藻酸盐,在生理钙离子浓度下原位凝胶化来诱导肿瘤细胞的高度免疫原性死亡。ZHANG等[12]研制了可注射杂化活性肽-海藻酸盐/磷灰石水凝胶微球,包裹了人牙髓干细胞和血管内皮生长因子,结果表明这种水凝胶微球在血管牙髓再生方面具有巨大的潜力。FAN等[38]介绍了一种可注射、不透光的液态金属/海藻酸钙水凝胶,通过在同一段血管的一头注射CaCl2溶液、另一头注射以液态金属液滴作为不透光的单元制备成海藻酸钠溶液形成交联网络,作为血管内栓塞和肿瘤栓塞治疗的候选材料。"
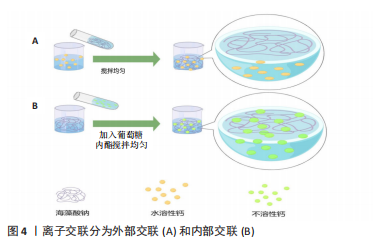

内部凝胶化则多数采用不溶性钙如碳酸钙(CaCO3)与葡萄糖酸内酯复合物作为钙离子源[50-55],还有其他少数钙离子来源如生物玻璃[56-58]、钙铝硅酸盐来制备水凝胶[59]。即时配制的葡萄糖酸内酯溶液缓释H+离子,从而使各种钙化合物释放出钙离子,形成均一的固态凝胶。ZHANG等[60]以海藻酸钠、CaCO3、葡萄糖酸内酯和右旋糖酐为载体,将血小板源性生长因子BB和骨髓间充质干细胞载入海藻酸钠中,注入伤口以达到治疗的目的。ZHU等[57]将含骨髓干细胞与关节软骨细胞共培养的可注射热敏海藻酸钠/琼脂糖/生物玻璃复合水凝胶进行关节软骨再生研究。 可注射海藻酸钠水凝胶也可以通过调整硫酸钙(CaSO4)[61-65]、葡萄糖酸钙等难溶性钙化合物水溶液的浓度和比例控制凝胶化速度来制备[66-71]。 WU等[69]以葡萄糖酸钙为交联剂研制出了一种用于治疗心肌梗死不同阶段的负载血管内皮活性因子和丝素蛋白可注射海藻酸盐复合水凝胶微球。RAIMONDO等[64]将2个注射器通过连接器连接,把体积比为3∶1组成的低分子海藻酸盐和高分子海藻酸盐负载血管内皮生长因子和胰岛素样生长因子与CaSO4水溶液按25∶1的比例来回快速挤压混合,形成可注射、可生物降解的水凝胶。CHOI等[61]通过调整CaSO4浓度对海藻酸钠/透明质酸水凝胶的凝胶化速度和均一度进行调控,实现较好的注射性能和原位凝胶。 外部凝胶化钙离子由外向内渗透,外部是交联海藻酸钙层,内部是非交联海藻酸钠区,凝胶粒子的外层交联密度较大,形成外密内疏的非均质凝胶结构。相较于外部凝胶化,内部凝胶化能够得到强度、时间可控的、由液态变为固态的均质海藻酸盐水凝胶,也更有利于药物缓慢释放以及对力学强度有一定要求的骨组织工程应用。 (2)聚电解质复合物:是利用聚阴离子体与聚阳离子体间的相反电荷之间相互作用产生的聚电解质物,又称聚离子复合物[72]。海藻酸钠是一种能与聚阳离子电解液产生聚电解质复合物的聚阴离子化电解液。NIE等[14]应用海藻酸钠与聚D-赖氨酸的即时胶凝反应形成多孔微球,通过腹腔注射包裹间充质干细胞的多孔微球治疗慢性系统性红斑狼疮。LV等[15]以羧甲基壳聚糖和海藻酸盐为基础,通过聚电解质络合形成凝胶,研制了可原位注射使伤口愈合的水凝胶。ALIZADEH等[73]制备的壳聚糖-苯胺五聚体/海藻酸盐/琼脂糖水凝胶,可促进成人嗅黏膜来源间充质干细胞向多巴胺能神经元分化,该可注射导电碳水化合物致冷剂水凝胶可用于帕金森病的治疗。 (3)氢键及疏水作用:采用降低pH值的方法制备海藻酸钠凝胶。通过缓慢或受控的方式释放出氢,可以获得到可控制成胶时间的海藻酸凝胶[74]。葡萄糖酸内酯在水中溶解后缓慢释放出H+,所以存在葡萄糖酸内酯的情况下能得到可注射海藻酸凝胶。LV等[15]以羧甲基壳聚糖和海藻酸盐为基础,加入葡萄糖酸内酯后通过聚电解质络合形成凝胶,葡萄糖酸内酯缓慢水解并释放质子,再一次加强凝胶强度,研制了可使伤口愈合的原位注射性水凝胶。 2.3.2 化学交联的可注射海藻酸钠水凝胶 共价交联型水凝胶是由高分子聚合物通过化学键结合而成的三维立体网状结构。海藻酸钠的糖醛酸单元含有羟基和羧基,在一定的条件下可以与一些交联剂上的基团反应交联,以获得到一种可注射、共价交联的海藻酸钠水溶性凝胶。 席夫碱反应:是目前应用最广泛的共价交联法。海藻酸钠分子的糖醛酸单元为顺二醇结构,用高碘酸钠(NaIO4)氧化顺二醇中的C-C键,形成2个醛基;氧化海藻酸钠的醛基可以与含氨基物质发生席夫碱交联反应[16,75-81],并且通过改变海藻酸钠的氧化程度及浓度控制交联反应速度来达到可注射的目的。FAN等[16]通过醛氧化藻酸盐与赖氨酸的游离胺或明胶的羟赖氨酸反应形成席夫碱,有望成为治疗食管内镜黏膜下剥离术的黏膜下注射制剂。BASU等[76]制备的水凝胶通过形成可逆亚胺键与氧化海藻酸盐进行交联,可用作小分子药物辛伐他汀的可注射载体,通过改变海藻酸盐的氧化程度和硅酸盐纳米粒子的浓度来调节水凝胶的流变性实现可注射。 羧基的交联:通常先用1-乙基-(3-二甲基氨基丙基)碳二亚胺/N-羟基琥珀酰亚胺来活化海藻酸钠的羧基,再与含有伯胺的分子发生缩合反应,例如多肽或者蛋白质可交联海藻酸钠的羧基[17,82]。FAN等[17]报告了一种新的细胞膜衍生水凝胶支架,用1-乙基-(3-二甲基氨基丙基)碳二亚胺/N-羟基琥珀酰亚胺活化羧基的海藻酸钠与红细胞膜囊泡溶液交联制作支架,这种支架具有大孔结构,可以包裹和持续释放疏水模型药物。 光交联:紫外光交联反应条件温和,副产物少,不需交联剂。大多数以甲基丙烯酸酐改性海藻酸钠为原料,用偶氮引发剂VA-086作为诱导剂,通过紫外光照射使其发生交联反应[82]。该工艺虽然能改善凝胶体的力学性能和稳定性,但同时也会使凝胶体的吸水性下降[18,83-84]。同时,由于光敏诱导剂在胶体凝固后很难从其网络体系中彻底清除,增加了凝胶细胞毒性,导致其生物相容性变差[85]。TAVAFOGHI等[18]设计了一种明胶甲基丙烯酰基与甲基丙烯酸改性海藻酸盐氢键结合的可光交联、可注射杂化水凝胶,形成了耐用、机械弹性和成本效益高的伤口敷料。HUANG等[82]将纤维连接蛋白偶联的甲基丙烯酰化海藻酸钠在光引发剂存在下紫外光照射下交联形成水凝胶,将骨髓间充质干细胞衍生的细胞外囊泡包封其中延长给药时间。HASANI-SADRABADI等[84]设计了一种可作为可注射间充质干细胞载体、可调节力学性能、可光交联和具有骨传导作用的海藻酸盐黏合剂,用于颅面部骨组织工程学的应用。 共价交联:共价交联的可注射海藻酸钠水凝胶通常是在海藻酸钠上接枝降冰片烯基团或四嗪基团,通过降冰片烯基团和四嗪基团之间的逆电子需求反应进行共价交联。TRUNG等[86]采用海藻酸盐-降冰片烯和水溶性聚乙二醇基二硫化物交联剂之间的逆电子需求反应,形成共价交联的海藻酸盐水凝胶网络。YANG等[19]利用逆电子需求和离子反应法合成了双马来酰亚胺-聚乙二醇/含铜生物玻璃陶瓷与呋喃-海藻酸钠双交联型可注射复合水凝胶微球。KOSHY等[87]制备了利用海藻酸盐降冰片烯和海藻酸盐四嗪共价交联的可注射的海藻酸钠复合水凝胶,能够持续地、生物活性地释放各种封装的蛋白质。其他的共价交联,如MOODY等[88]利用四氯氰化物共价交联叠氮修饰的海藻酸盐水凝胶,在组织工程学和体内药物输送中应用。 2.3.3 其他交联的可注射海藻酸钠水凝胶 酶交联:研究表明,采用酶交联法不仅能减少戊二醛、环氧氯丙烷等低分子质量引发剂的使用,还能提高可注射海藻酸钠水凝胶的强度和生物相容性。因为酶交联法具有较高的酶活性,需要温和的反应条件;还因为一种酶仅能催化一种或多种化学反应,所以不会产生副作用,因此,采用酶交联法制备可注射水凝胶更具有应用价值[20,89-90]。JIANG等[20]在活化的海藻酸钠上接枝盐酸酪胺制得酪胺修饰的海藻酸钠,然后在酪胺修饰的海藻酸盐/丝胶/石墨烯水溶液中加入过氧化物酶和过氧化氢(H2O2),即可得到交联的海藻酸钠水凝胶。ZHANG等[89]用激活的海藻酸盐接枝多巴胺、硫酸软骨素和再生丝素蛋白,利用过氧化物酶和H2O2为交联剂催化形成可注射海藻酸水凝胶,以促进软骨缺损的再生。 温敏性交联:在海藻酸钠上接枝温敏性基团,可以达到调节温度即可形成可注射海藻酸钠水凝胶。IATRIDI等[21]以疏水性N-叔丁基丙烯酰胺单体为富集单体,合成了一种热响应共聚物接枝海藻酸钠,用于干细胞移植和药物缓释。ZAKERIKHOOB等[91]合成了温敏性交联聚(N-异丙基丙烯酰胺)-海藻酸钠接枝共聚物,用于伤口姜黄素的缓释。KIM等[92]以温度响应性聚(e-己内酯-丙交酯)-b-聚(乙二醇)-b-聚(e-己内酯-丙交酯)和O-磷酸乙醇胺作为磷酸化官能团对海藻酸钠进行修饰,低温下自由流动的生物溶胶在生理温度(37 ℃)下可转变为稳定的黏弹性凝胶。LIU等[93]将泊洛沙姆接枝到海藻酸盐上合成的海藻酸盐-泊洛沙姆共聚物与丝素蛋白结合,制备了具有共价网络和非共价交联网络的温敏性海藻酸钠水凝胶。DANG等[11]提出了一种基于半胺功能化海藻酸钠-g-泊洛沙姆的新型热敏水凝胶,在海藻酸钠主链上引入半胱胺不仅增强了凝胶强度,而且还为所生成的水凝胶提供了内在的抗菌活性。 2.4 可注射的海藻酸钠水凝胶的应用 可注射海藻酸钠水凝胶在组织工程中的应用主要分为4大类:药物载体、细胞支架、创伤修复和缺损填充基质,如表2 所示。为达到缓释药物效果,或者保护药物效果以免被肠道或免疫系统所清除或减效,许多研究员将可注射海藻酸钠水凝胶作为药物载体;还有的科学家为保护细胞治疗法中的细胞在体内免受排斥免疫或给干细胞提供相对稳定的细胞外环境,设计合适的可注射海藻酸钠水凝胶作为细胞支架。创伤修复不仅需要保护创面免受二次伤害,还需要提供细胞修复所需的微环境,可注射海藻酸钠水凝胶可为创伤修复提供湿润合适的恢复环境。由于非锐器损伤所导致的缺损部位并不规则,所需的填充材质需要定制较麻烦,而可注射海藻酸钠水凝胶却不需要考虑这一问题,能及时快速地输注填充缺损。"


2.4.1 药物载体 可注射海藻酸钠水凝胶材料通常是液态或半固态的,将一些药物如抗癌药、胰岛素等,或者是生物制剂与水凝胶结合,可制成能在体内原位成胶的水凝胶载体。这种方式既可以避免口服药物所带来的药效减半弊端,又能在一定程度上进行药物的靶向递送,达到药物在病灶局部的控释或缓释,还能避免生物制剂经过胃肠道时失活或者自身免疫性失活。但单纯海藻酸钠的药物缓释能力往往无法满足实际研究需要,相关研究者们往往通过对海藻酸钠进行改性来达到良好的药物缓释能力。有研究者将海藻酸钠交联温敏性聚(N-异丙基丙烯酰胺)作为姜黄素药物的载体,缓释姜黄素用于伤口愈合[91]。KOSHY等[87]将可注射的海藻酸钠凝胶作为蛋白制剂的载体,持续、生物活性地释放各种封装的蛋白质。BASU等[76]将海藻酸钠水凝胶作为小分子药物辛伐他汀的可注射缓释载体,持续释放辛伐他汀促进骨组织修复。 2.4.2 细胞支架 鉴于海藻酸钠良好的生物相容性及生物可降解性,在组织工程研究中,科学家们尝试通过不同的方法对可注射海藻酸钠水凝胶支架进行改性来模拟体内微环境,使之能模拟细胞生长环境,这使得可注射海藻酸钠水凝胶成为组织工程中细胞培养和输送的重要支架和载体。NIE等[14] 利用海藻酸钠与聚D-赖氨酸形成多孔微球包裹间充质干细胞免受免疫细胞的侵袭,同时维持自身免疫调节功能,并在体内快速进行养分交换。ZHU等[57]将骨髓间充质干细胞与关节软骨细胞共培养的可注射热敏性海藻酸钠/琼脂糖复合水凝胶用于关节软骨再生。 2.4.3 创伤修复 可海藻酸钠水凝胶因其良好的抗菌性和止血活性而在创面修复及再生中得到了显著成果,已成为创伤修复材料的不二之选。由于海藻酸钠的可注射性,能在创面原位形成凝胶,与创面形状完美吻合。ZHENG等[94]制备的藻酸钠/生物玻璃复合水凝胶中载有包含吡非尼酮的聚乳酸-乙醇酸微球,这种多层可注射水凝胶体系能依次递送生物活性分子,以满足每个创面愈合阶段的生物活性需求和时间需求。创伤伤口通过缝合的方式会造成二次损伤,可通过注射具有黏合作用的水凝胶以达到更好的修复结果。有研究将氧化海藻酸钠与生物玻璃制成一种具有强黏合力、促进血管生成和组织再生的创伤修复敷料,这种敷料比缝合的方式更加方便快捷[95]。 2.4.4 缺损填充基质 由于缺损部位的形状不规则,难以及时得到匹配的填充物来修复支撑缺损部位,可注射水凝胶材料能简易快速地解决这一问题。可注射海藻酸钠水凝胶作为不规则缺损创伤修复领域的首选。LIU等[10]设计的可注射海藻酸盐水凝胶在治疗骨肿瘤的同时,还能提供促进骨缺损修复的支撑基质更加有利于骨再生修复。CHUYSINUAN等[48]将透明质酸、丝素蛋白、羟基磷灰石、海藻酸等混合作为拔牙后填充牙槽骨的缺损填充基质,为重建牙槽骨和后期种植牙提供了基础。"

| [1] MAHINROOSTA M, FARSANGI ZJ, ALLAHVERDI A, et al. Hydrogels as intelligent materials: A brief review of synthesis, properties and applications. Mater Today Chem. 2018;8:42-55. [2] PARK SH, JI YB, PARK JY, et al. Injectable In Situ-Forming Hydrogels for Protein and Peptide Delivery. Adv Exp Med Biol. 2020;1250:35-48. [3] LU L, YUAN S, WANG J, et al. The Formation Mechanism of Hydrogels. Curr Stem Cell Res Ther. 2018;13(7):490-496. [4] XU Z, LAM MT. Alginate Application for Heart and Cardiovascular Diseases//REHM BHA, MORADALI MF. Alginates and Their Biomedical Applications .Springer Singapore: Singapore, 2018:185-212. [5] HERNANDEZ-GONZALEZ AC, TELLEZ-JURADO L, RODRIGUEZ-LORENZO LM. Alginate hydrogels for bone tissue engineering from injectables to bioprinting: A review. Carbohydr Polym. 2020;229:115514. [6] CAO L, LU W, MATA A, et al. Egg-box model-based gelation of alginate and pectin: A review. Carbohydr Polym. 2020;242:116389. [7] XING M, CAO Q, WANG Y, et al. Advances in Research on the Bioactivity of Alginate Oligosaccharides. Mar Drugs. 2020;18(3):144. [8] RAVICHANDRAN V, JAYAKRISHNAN A. Synthesis and evaluation of anti-fungal activities of sodium alginate-amphotericin B conjugates. Int J Biol Macromol. 2018; 108:1101-1109. [9] 金依静,李蓝,刘秋平,等.超声-复合酶解法提取海带中海藻酸钠的工艺优化[J].食品工业科技,2021,42(5):132-137. [10] LIU B, GU X, SUN Q, et al. Injectable In Situ Induced Robust Hydrogel for Photothermal Therapy and Bone Fracture Repair. Adv Funct Mater. 2021;31(19): 2010779. [11] DANG LH, DOAN P, NHI TTY, et al. Multifunctional injectable pluronic-cystamine-alginate-based hydrogel as a novel cellular delivery system towards tissue regeneration. Int J Biol Macromol. 2021;185:592-603. [12] ZHANG R, XIE L, WU H, et al. Alginate/laponite hydrogel microspheres co-encapsulating dental pulp stem cells and VEGF for endodontic regeneration. Acta Biomater. 2020;113:305-316. [13] ZHANG R, XIE L, WU F, et al. ALG-bFGF Hydrogel Inhibiting Autophagy Contributes to Protection of Blood-Spinal Cord Barrier Integrity via PI3K/Akt/FOXO1/KLF4 Pathway After SCI. Front Pharmacol. 2022;13:828896. [14] NIE M, CHEN G, ZHAO C, et al. Bio-inspired adhesive porous particles with human MSCs encapsulation for systemic lupus erythematosus treatment. Bioact Mater. 2021;6(1):84-90. [15] LV X, LIU Y, SONG S, et al. Influence of chitosan oligosaccharide on the gelling and wound healing properties of injectable hydrogels based on carboxymethyl chitosan/alginate polyelectrolyte complexes. Carbohyd Polym. 2019;205:312-321. [16] FAN C, XU K, HUANG Y, et al. Viscosity and degradation controlled injectable hydrogel for esophageal endoscopic submucosal dissection. Bioact Mater. 2021; 6(4):1150-1162. [17] FAN Z, DENG J, LI PY, et al. A new class of biological materials: Cell membrane-derived hydrogel scaffolds. Biomaterials. 2019;197:244-254. [18] TAVAFOGHI M, SHEIKHI A, TUTAR R, et al. Engineering Tough, Injectable, Naturally Derived, Bioadhesive Composite Hydrogels. Adv Healthc Mater. 2020;9(10):e1901722. [19] YANG Z, ZHAO F, ZHANG W, et al. Degradable photothermal bioactive glass composite hydrogel for the sequential treatment of tumor-related bone defects: From anti-tumor to repairing bone defects. Chem Eng J. 2021;419. doi.org/10.1016/j.cej.2021.129520 [20] JIANG LB, DING SL, DING W, et al. Injectable sericin based nanocomposite hydrogel for multi-modal imaging-guided immunomodulatory bone regeneration. Chem Eng J. 2021;418(4):129323. [21] IATRIDI Z, SARAVANOU SF, TSITSILIANIS C. Injectable self-assembling hydrogel from alginate grafted by P(N-isopropylacrylamide-co-N-tert-butylacrylamide) random copolymers. Carbohyd Polym. 2019;219:344-352. [22] 袁晓露,李宝霞,黄雅燕,等.海藻酸钠微囊的制备及应用进展[J].化工进展, 2022,41(6):3103-3112. [23] SHUAI F, ZHANG Y, YIN Y, et al. Fabrication of an injectable iron (III) crosslinked alginate-hyaluronic acid hydrogel with shear-thinning and antimicrobial activities. Carbohydr Polym. 2021;260:117777. [24] ANAMIZU M, TABATA Y. Design of injectable hydrogels of gelatin and alginate with ferric ions for cell transplantation. Acta Biomater. 2019;100:184-190. [25] ZHANG GZ, LV L, DENG YH, et al. Self-Healing Gelatin Hydrogels Cross-Linked by Combining Multiple Hydrogen Bonding and Ionic Coordination. Macromol Rapid Commun. 2017;38(12). doi: 10.1002/marc.201700018. [26] RUSSO R, MALINCONICO M, SANTAGATA G. Effect of cross-linking with calcium ions on the physical properties of alginate films. Biomacromolecules. 2007;8(10):3193-3197. [27] KALKOWSKI L, GOLUBCZYK D, KWIATKOWSKA J, et al. Two in One: Use of Divalent Manganese Ions as Both Cross-Linking and MRI Contrast Agent for Intrathecal Injection of Hydrogel-Embedded Stem Cells. Pharmaceutics. 2021;13(7):1076. [28] XU Y, ZHAO S, WENG Z, et al. Jelly-Inspired Injectable Guided Tissue Regeneration Strategy with Shape Auto-Matched and Dual-Light-Defined Antibacterial/Osteogenic Pattern Switch Properties. ACS Appl Mater Interfaces. 2020;12(49):54497-54506. [29] SUN L, SHEN F, TIAN L, et al. ATP-Responsive Smart Hydrogel Releasing Immune Adjuvant Synchronized with Repeated Chemotherapy or Radiotherapy to Boost Antitumor Immunity. Adv Mater. 2021;33(18):e2007910. [30] ZHAO Z, LI Q, QIN X, et al. An Injectable Hydrogel Reshaping Adenosinergic Axis for Cancer Therapy. Adv Funct Mater. 2022;32(24):2200801.1-2200801.10 [31] LI Q, ZHAO Z, QIN X, et al. A Checkpoint-Regulatable Immune Niche Created by Injectable Hydrogel for Tumor Therapy. Adv Funct Mater. 2021;31(37):2104630. [32] XIONG X, ZHAO J, SU R, et al. Double enhancement of immunogenic cell death and antigen presentation for cancer immunotherapy. Nano Today. 2021;39:101225. [33] OUYANG B, LIU F, RUAN S, et al. Localized Free Radicals Burst Triggered by NIR-II Light for Augmented Low-Temperature Photothermal Therapy. ACS Appl Mater Interfaces. 2019;11(42):38555-38567. [34] CHAO Y, XU L, LIANG C, et al. Combined local immunostimulatory radioisotope therapy and systemic immune checkpoint blockade imparts potent antitumour responses. Nat Biomed Eng. 2018;2(8):611-621. [35] YANG IH, CHEN YS, LI JJ, et al. The development of laminin-alginate microspheres encapsulated with Ginsenoside Rg1 and ADSCs for breast reconstruction after lumpectomy. Bioact Mater. 2021;6(6):1699-1710. [36] SU X, CAO Y, LIU Y, et al. Localized disruption of redox homeostasis boosting ferroptosis of tumor by hydrogel delivery system. Mater Today Bio. 2021;12:100154. [37] CAO Y, ZHOU Y, PAN J, et al. A general strategy towards an injectable microwave-sensitive immune hydrogel for combined percutaneous microwave ablation and immunotherapy. Chem Eng J. 2021;422:130111. [38] FAN L, DUAN M, XIE Z, et al. Injectable and Radiopaque Liquid Metal/Calcium Alginate Hydrogels for Endovascular Embolization and Tumor Embolotherapy. Small. 2020;16(2):e1903421. [39] CHEN Y, HUANG J, CHEN R, et al. Sustained release of dermal papilla-derived extracellular vesicles from injectable microgel promotes hair growth. Theranostics. 2020;10(3):1454-1478. [40] LUDWINSKI FE, PATEL AS, DAMODARAN G, et al. Encapsulation of macrophages enhances their retention and angiogenic potential. NPJ Regen Med. 2019;4:6. [41] SUN SK, WU JC, WANG H, et al. Turning solid into gel for high-efficient persistent luminescence-sensitized photodynamic therapy. Biomaterials. 2019;218:119328. [42] MA Z, SONG W, HE Y, et al. Multilayer Injectable Hydrogel System Sequentially Delivers Bioactive Substances for Each Wound Healing Stage. ACS Appl Mater Interfaces. 2020;12(26):29787-29806. [43] WONG FSY, TSANG KK, CHU AMW, et al. Injectable cell-encapsulating composite alginate-collagen platform with inducible termination switch for safer ocular drug delivery. Biomaterials. 2019;201:53-67. [44] ZHAO J, LI J, ZHU C, et al. Design of Phase-Changeable and Injectable Alginate Hydrogel for Imaging-Guided Tumor Hyperthermia and Chemotherapy. ACS Appl Mater Interfaces. 2018;10(4):3392-3404. [45] DEEPTHI S, JAYAKUMAR R. Alginate nanobeads interspersed fibrin network as in situ forming hydrogel for soft tissue engineering. Bioact Mater. 2018;3(2):194-200. [46] JABBARI E, SEPAHVANDI A. Decellularized Articular Cartilage Microgels as Microcarriers for Expansion of Mesenchymal Stem Cells. Gels. 2022;8(3):148. [47] ZHANG F, XU M, SU X, et al. Afterglow Implant for Arterial Embolization and Intraoperative Imaging. Chemistry. 2022;28(2):e202103795. [48] CHUYSINUAN P, NOOEAID P, THANYACHAROEN T, et al. Injectable eggshell-derived hydroxyapatite-incorporated fibroin-alginate composite hydrogel for bone tissue engineering. Int J Biol Macromol. 2021;193:799-808. [49] PARK Y, JI ST, YONG U, et al. 3D bioprinted tissue-specific spheroidal multicellular microarchitectures for advanced cell therapy. Biofabrication. 2021;13(4).doi: 10.1088/1758-5090/ac212e. [50] MA S, ZHOU J, HUANG T, et al. Sodium alginate/collagen/stromal cell-derived factor-1 neural scaffold loaded with BMSCs promotes neurological function recovery after traumatic brain injury. Acta Biomater. 2021;131:185-197. [51] KIM H, SONG D, NGO HV, et al. Modulation of the clinically accessible gelation time using glucono-d-lactone and pyridoxal 5’-phosphate for long-acting alginate in situ forming gel injectable. Carbohydr Polym. 2021;272:118453. [52] BAO W, XIE L, ZENG X, et al. A Cocktail-Inspired Male Birth Control Strategy with Physical/Chemical Dual Contraceptive Effects and Remote Self-Cleared Properties. Acs Nano. 2019;13(2):1003-1011. [53] ZHANG Y, WANG T, ZHUANG Y, et al. Sodium Alginate Hydrogel-Mediated Cancer Immunotherapy for Postoperative In Situ Recurrence and Metastasis. ACS Biomater Sci Eng. 2021;7(12):5717-5726. [54] MOUSAVI A, MASHAYEKHAN S, BAHEIRAEI N, et al. Biohybrid oxidized alginate/myocardial extracellular matrix injectable hydrogels with improved electromechanical properties for cardiac tissue engineering. Int J Biol Macromol. 2021;180:692-708. [55] ZHANG Z, LI Z, WANG Y, et al. PDGF-BB/SA/Dex injectable hydrogels accelerate BMSC-mediated functional full thickness skin wound repair by promoting angiogenesis. J Mater Chem B. 2021;9(31):6176-6189. [56] ZHANG X, LI Y, MA Z, et al. Modulating degradation of sodium alginate/bioglass hydrogel for improving tissue infiltration and promoting wound healing. Bioact Mater. 2021;6(11):3692-3704. [57] ZHU Y, KONG L, FARHADI F, et al. An injectable continuous stratified structurally and functionally biomimetic construct for enhancing osteochondral regeneration. Biomaterials. 2019;192:149-158. [58] KONG L, WU Z, ZHAO H, et al. Bioactive Injectable Hydrogels Containing Desferrioxamine and Bioglass for Diabetic Wound Healing. ACS Appl Mater Interfaces. 2018;10(36):30103-30114. [59] WU B, PASCU EI, BRADY SA, et al. Fine property-tuning through Ca content control in a facile synthesis of glasses-based self-setting injectable hydrogel. Mater Design. 2021;211:110166. [60] ZHANG Z, LI Z, WANG Y, et al. PDGF-BB/SA/Dex injectable hydrogels accelerate BMSC-mediated functional full thickness skin wound repair by promoting angiogenesis. J Mater Chem B. 2021;9(31):6176-6189. [61] CHOI YH, KIM SH, KIM IG, et al. Injectable basic fibroblast growth factor-loaded alginate/hyaluronic acid hydrogel for rejuvenation of geriatric larynx. Acta Biomater. 2019;89:104-114. [62] MOODY CT, BROWN AE, MASSARO NP, et al. Restoring Carboxylates on Highly Modified Alginates Improves Gelation, Tissue Retention and Systemic Capture. Acta Biomater. 2022;138:208-217. [63] SUBBIAH R, CHENG A, RUEHLE MA, et al. Effects of controlled dual growth factor delivery on bone regeneration following composite bone-muscle injury. Acta Biomater. 2020;114:63-75. [64] RAIMONDO TM, LI H, KWEE BJ, et al. Combined delivery of VEGF and IGF-1 promotes functional innervation in mice and improves muscle transplantation in rabbits. Biomaterials. 2019;216:119246. [65] INGAVLE GC, GIONET-GONZALES M, VORWALD CE, et al. Injectable mineralized microsphere-loaded composite hydrogels for bone repair in a sheep bone defect model. Biomaterials. 2019;197:119-128. [66] JUNG SW, BYUN JH, OH SH, et al. Multivalent ion-based in situ gelling polysaccharide hydrogel as an injectable bone graft. Carbohyd Polym. 2018;180:216-225. [67] MOTEALLEH A, KART D, CZIEBOROWSKI M, et al. Functional Nanomaterials and 3D-Printable Nanocomposite Hydrogels for Enhanced Cell Proliferation and for the Reduction of Bacterial Biofilm Formation. ACS Appl Mater Interfaces. 2021; 13(36):43755-43768. [68] ZHOU J, LIU W, ZHAO X, et al. Natural Melanin/Alginate Hydrogels Achieve Cardiac Repair through ROS Scavenging and Macrophage Polarization. Adv Sci (Weinh). 2021;8(20):e2100505. [69] WU Y, CHANG T, CHEN W, et al. Release of VEGF and BMP9 from injectable alginate based composite hydrogel for treatment of myocardial infarction. Bioact Mater. 2021;6(2):520-528. [70] NAZEMI Z, NOURBAKHSH MS, KIANI S, et al. Co-delivery of minocycline and paclitaxel from injectable hydrogel for treatment of spinal cord injury. J Control Release. 2020;321:145-158. [71] HAO T, LI J, YAO F, et al. Injectable Fullerenol/Alginate Hydrogel for Suppression of Oxidative Stress Damage in Brown Adipose-Derived Stem Cells and Cardiac Repair. Acs Nano. 2017;11(6):5474-5488. [72] HAJIFATHALIHA F, MAHBOUBI A, NEMATOLLAHI L, et al. Comparison of different cationic polymers efficacy in fabrication of alginate multilayer microcapsules. Asian Pharm Sci. 2020;15(1):95-103. [73] ALIZADEH R, ZARRINTAJ P, KAMRAVA SK, et al. Conductive hydrogels based on agarose/alginate/chitosan for neural disorder therapy. Carbohyd Polym. 2019; 224:115161. [74] ZHANG X, LI Y, MA Z, et al. Modulating degradation of sodium alginate/bioglass hydrogel for improving tissue infiltration and promoting wound healing. Bioact Mater. 2021;6(11):3692-3704. [75] WANG P, PU Y, REN Y, et al. Dynamic regulable sodium alginate/poly(gamma-glutamic acid) hybrid hydrogels promoted chondrogenic differentiation of stem cells. Carbohyd Polym. 2022;275:118692. [76] BASU S, PACELLI S, PAUL A. Self-healing DNA-based injectable hydrogels with reversible covalent linkages for controlled drug delivery. Acta Biomater. 2020; 105:159-169. [77] RESMI R, PARVATHY J, JOHN A, et al. Injectable self-crosslinking hydrogels for meniscal repair: A study with oxidized alginate and gelatin. Carbohyd Polym. 2020; 234:115902. [78] LIANG W, CHEN J, LI L, et al. Conductive Hydrogen Sulfide-Releasing Hydrogel Encapsulating ADSCs for Myocardial Infarction Treatment. ACS Appl Mater Interfaces. 2019;11(16):14619-14629. [79] WANG L, DENG F, WANG W, et al. Construction of Injectable Self-Healing Macroporous Hydrogels via a Template-Free Method for Tissue Engineering and Drug Delivery. ACS Appl Mater Interfaces. 2018;10(43):36721-36732. [80] XING Y, QING X, XIA H, et al. Injectable Hydrogel Based on Modified Gelatin and Sodium Alginate for Soft-Tissue Adhesive. Front Chem. 2021;9:744099. [81] XUAN H, WU S, FEI S, et al. Injectable nanofiber-polysaccharide self-healing hydrogels for wound healing. Mater Sci Eng C Mater Biol Appl. 2021;128:112264. [82] HUANG CC, KANG M, SHIRAZI S, et al. 3D Encapsulation and tethering of functionally engineered extracellular vesicles to hydrogels. Acta Biomater. 2021;126:199-210. [83] DASH L, MAHANWAR PA. A Review on Organic Phase Change Materials and Their Applications. Biomaterials. 2021;5(9):268-284. [84] HASANI-SADRABADI MM, SARRION P, POURAGHAEI S, et al. An engineered cell-laden adhesive hydrogel promotes craniofacial bone tissue regeneration in rats. Sci Transl Med. 2020;12(534):eaay6853. [85] ZHAO D, TIE C, CHENG B, et al. Effect of altering photocrosslinking conditions on the physical properties of alginate gels and the survival of photoencapsulated cells. Polym Degrad Stab. 2020;179:109297. [86] TRUNG THANG V, GULFAM M, JO SH, et al. Injectable and biocompatible alginate-derived porous hydrogels cross-linked by IEDDA click chemistry for reduction-responsive drug release application. Carbohydr Polym. 2022;278:118964. [87] KOSHY ST, ZHANG DKY, GROLMAN JM, et al. Injectable nanocomposite cryogels for versatile protein drug delivery. Acta Biomater. 2018;65:36-43. [88] MOODY CT, PALVAI S, BRUDNO Y. Click cross-linking improves retention and targeting of refillable alginate depots. Acta Biomater. 2020;112:112-121. [89] ZHANG FX, LIU P, DING W, et al. Injectable Mussel-Inspired highly adhesive hydrogel with exosomes for endogenous cell recruitment and cartilage defect regeneration. Biomaterials. 2021;278:121169. [90] JI YR, YOUNG TH, TSAI TY, et al. Dopamine-Modified Alginate Hydrogel with Effectiveness and Safety for Preoperative Localization of Lung Nodules. ACS Biomater Sci Eng. 2021;7(9):4637-4644. [91] ZAKERIKHOOB M, ABBASI S, YOUSEFI G, et al. Curcumin-incorporated crosslinked sodium alginate-g-poly (N-isopropyl acrylamide) thermo-responsive hydrogel as an in-situ forming injectable dressing for wound healing: In vitro characterization and in vivo evaluation. Carbohyd Polym. 2021;271:118434. [92] KIM SH, THAMBI T, PHAN VHG, et al. Modularly engineered alginate bioconjugate hydrogel as biocompatible injectable scaffold for in situ biomineralization. Carbohyd Polym. 2020;233:115832. [93] LIU J, FANG Q, LIN H, et al. Alginate-poloxamer/silk fibroin hydrogels with covalently and physically cross-linked networks for cartilage tissue engineering. Carbohyd Polym. 2020;247:116593. [94] ZHENG H, WANG S, CHENG F, et al. Bioactive anti-inflammatory, antibacterial, conductive multifunctional scaffold based on MXene@CeO2 nanocomposites for infection-impaired skin multimodal therapy. Chem Eng J. 2021;424:130148. [95] GAO L, ZHOU Y, PENG J, et al. A novel dual-adhesive and bioactive hydrogel activated by bioglass for wound healing. Npg Asia Mater. 2019;11(1):66. |
| [1] | Yu Weijie, Liu Aifeng, Chen Jixin, Guo Tianci, Jia Yizhen, Feng Huichuan, Yang Jialin. Advantages and application strategies of machine learning in diagnosis and treatment of lumbar disc herniation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1426-1435. |
| [2] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [3] | Chen Kaijia, Liu Jingyun, Cao Ning, Sun Jianbo, Zhou Yan, Mei Jianguo, Ren Qiang. Application and prospect of tissue engineering in treatment of osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1450-1456. |
| [4] | Lin Zeyu, Xu Lin. Research progress in gout-induced bone destruction mechanism [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1295-1300. |
| [5] | Mei Jingyi, Liu Jiang, Xiao Cong, Liu Peng, Zhou Haohao, Lin Zhanyi. Proliferation and metabolic patterns of smooth muscle cells during construction of tissue-engineered blood vessels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1043-1049. |
| [6] | Wang Shanshan, Shu Qing, Tian Jun. Physical factors promote osteogenic differentiation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1083-1090. |
| [7] | Ma Shuwei, He Sheng, Han Bing, Zhang Liaoyun. Exosomes derived from mesenchymal stem cells in treatment of animals with acute liver failure: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1137-1142. |
| [8] | Zhang Kefan, Shi Hui. Research status and application prospect of cytokine therapy for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 961-967. |
| [9] | Shen Ziqing, Xia Tian, Shan Yibo, Zhu Ruijun, Wan Haoxin, Ding Hao, Pan Shu, Zhao Jun. Vascularized tracheal substitutes constructed by exosome-load hydrogel-modified 3D printed scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 697-705. |
| [10] | Li Jiaqi, Huang Yuanli, Li Yan, Wang Chunren, Han Qianqian. Mechanism and influencing factors in molecular weight degradation of non-cross-linked hyaluronic acid [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 747-752. |
| [11] | Zhu Liwei, Wang Jiangyue, Bai Ding. Application value of nanocomposite gelatin methacryloyl hydrogels in different bone defect environments [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 753-758. |
| [12] | Xu Rong, Wang Haojie, Geng Mengxiang, Meng Kai, Wang Hui, Zhang Keqin, Zhao Huijing. Research advance in preparation and functional modification of porous polytetrafluoroethylene artificial blood vessels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 759-765. |
| [13] | Liu Chuang, Shan Shuo, Yu Tengbo, Zhou Huan, Yang Lei. Advantages, discomfort and challenges of clinical application of orthopedic hemostatic materials [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 795-803. |
| [14] | Wang Jiani, Chen Junyu. Angiogenesis mechanism of metal ions and their application in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 804-812. |
| [15] | Zhang Ming, Wang Bin, Jia Fan, Chen Jie, Tang Wei. Application of brain-computer interface technology based on electroencephalogram in upper limb motor function rehabilitation of stroke patients [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 581-586. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||