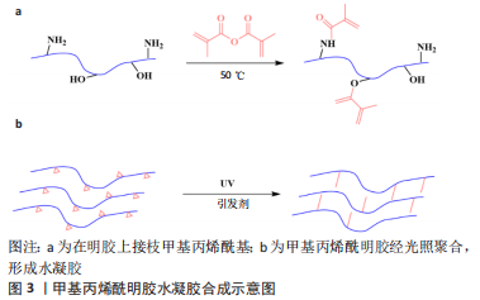
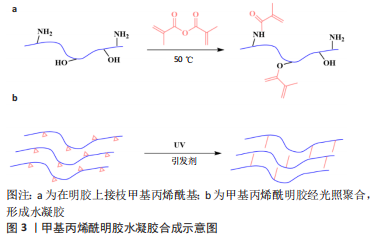
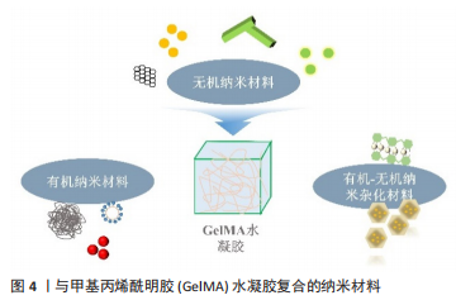
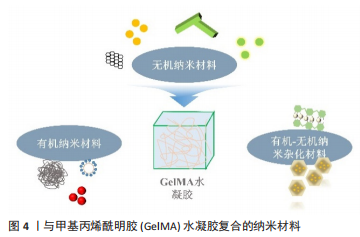
Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (5): 753-758.doi: 10.12307/2024.315
Previous Articles Next Articles
Application value of nanocomposite gelatin methacryloyl hydrogels in different bone defect environments
Zhu Liwei, Wang Jiangyue, Bai Ding
- National Clinical Medical Research Center for Oral Diseases, State Key Laboratory of Oral Diseases, Department of Orthodontics, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China
-
Received:2023-03-27Accepted:2023-05-15Online:2024-02-18Published:2023-08-17 -
Contact:Bai Ding, PhD, Professor, Chief physician, Doctoral supervisor, National Clinical Medical Research Center for Oral Diseases, State Key Laboratory of Oral Diseases, Department of Orthodontics, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China -
About author:Zhu Liwei, Master candidate, National Clinical Medical Research Center for Oral Diseases, State Key Laboratory of Oral Diseases, Department of Orthodontics, West China Hospital of Stomatology, Sichuan University, Chengdu 610041, Sichuan Province, China -
Supported by:National Natural Science Foundation of China, No. 82071146 (to BD); Exploration and Research & Development Interdisciplinary Innovation Project of West China Hospital of Stomatology of Sichuan University, No. RD-03-202108 (to BD)
CLC Number:
Cite this article
Zhu Liwei, Wang Jiangyue, Bai Ding. Application value of nanocomposite gelatin methacryloyl hydrogels in different bone defect environments[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 753-758.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
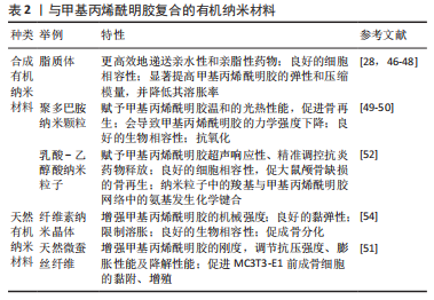
其中,最常见的是磷酸钙基纳米材料和硅基纳米材料,如:纳米级羟基磷灰石、二氧化硅纳米颗粒、生物玻璃等,这些刚性成分分散于GelMA基质中,可有效增强其力学强度。WANG等[22]通过简单物理混合的方式加入纳米级羟基磷灰石纳米粒子,压力测试显示当纳米级羟基磷灰石添加量为5%时,水凝胶可承受最大压应力为(278.62±7.49) kPa,较未添加纳米材料的GelMA水凝胶提高了3 倍以上,且水凝胶的孔径没有明显变化。作者认为这主要是因为纳米级羟基磷灰石的加入在水凝胶内部形成了[HO]-Ca2+-[OH]桥结构,使内部结构更加稳定,从而改善了水凝胶的理化性能。然而也有研究显示,过量的无机纳米颗粒会导致水凝胶透光性能的降低,从而在GelMA发生光交联时降低水凝胶的交联度[14]。同时,过量的无机纳米粒子对接种于水凝胶的细胞有一定的毒性,并且导致水凝胶支架孔径的减小,从而影响氧气扩散、营养物质的运输,进一步影响细胞活力[28]。目前大多数无机纳米材料复合GelMA的研究多是将纳米粒子直接与GelMA进行物理共混后进行光照,但是由于颗粒间的静电相互作用,无机纳米颗粒易聚集,难以保证其均匀分散[36,43]。 基于此,有学者采用GelMA水凝胶光固化后连续矿化的方法引入纳米级羟基磷灰石无机相[23],或在无机纳米粒子上接枝有机基团[14,43-44],以促进纳米粒子在GelMA中均匀分布,进而提高支架材料的机械强度。MIAO等[23]的研究显示,经连续矿化方式引入的纳米级羟基磷灰石颗粒均匀分布在水凝胶微孔表面,复合材料的矿物质含量约为40%,抗压强度较未添加纳米材料的GelMA水凝胶提高了5 倍以上。 除了增强GelMA水凝胶的力学强度,无机纳米材料还可增强GelMA水凝胶的触变性能。层状双氢氧化物在聚合物网络中会形成具有剪切稀化特性的机械稳定网络,可有效改善GelMA作为生物墨水时的可印刷性,从而实现更好的结构保真度。由于强烈的界面相互作用,层状双氢氧化物的添加使水凝胶抗压强度从652 kPa显著提高到1 168 kPa,打印的纳米复合水凝胶支架无需外加成骨诱导性因子即可支持封装的成骨细胞存活、扩散和增殖[13]。为实现相关材料的临床应用,研究者尚需进一步探究材料诱导体外成骨分化的效果并进行相应动物实验的验证。 此外,无机纳米粒子还可以促进原位骨矿化。纳米粒子本身的纳米形态即可在成骨早期促进细胞的黏附和增殖,其长期释放的硅、钙、磷、镁、锌、锂离子等亦可起到免疫调控、促血管生成、促成骨分化等功能[15,25,35]。此外,矿物基纳米材料还可通过共价连接[14]、静电相互作用及物理吸附等方式搭载蛋白质类药物[32] 。SHI等[16]利用硅酸盐纳米片与具有高表面积和带电特性的基质细胞衍生因子1α建立物理相互作用,可以递送和释放活性生长因子超过30 d,且不改变蛋白质因子的结构,释放的蛋白质能够在低浓度下促进骨髓间充质干细胞归巢和迁移。 近年来,碳基、磷基以及部分金属基纳米材料具有良好的光热效应,可增强GelMA的抗菌及抗肿瘤性能,在感染性骨缺损、骨肉瘤等模型中也有较多研究,将在后续进行举例。 2.1.2 有机纳米粒子 相比于无机纳米材料,有机纳米粒子的生物相容性及生物降解性相对更为优越[45]。不过目前关于有机纳米材料复合GelMA水凝胶在骨组织工程领域的相关报道还很有限,目前相对较为成熟的研究包括脂质体[46-48] 、聚多巴胺颗粒[49-50]、天然微蚕丝纤维[51] 、乳酸-乙醇酸纳米粒子[52] 、纤维素纳米晶体等天然或合成有机纳米材料[53] ,见表2。"
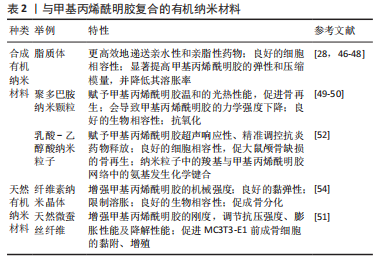
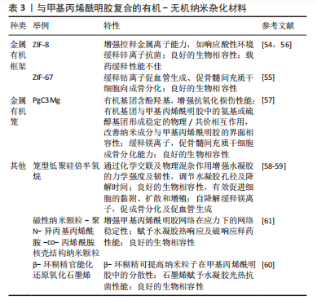
含醇脂质体是一种新型脂质体,具有高变形性和高客体包封率,能够更高效地递送亲水性和亲脂性药物[28]。将载有促血管和促成骨的小分子药物去铁胺的含醇脂质体与GelMA/结冷胶甲基丙烯酸酯水凝胶共混,可以更有效地控制局部药物递送浓度,维持去铁胺的生物活性,同时避免不必要的不良反应,体外实验表明,该水凝胶在第18 天的药物累积释放量为(67.04±0.71)%;大鼠颅骨缺损实验表明,持续释放的去铁胺通过缺氧诱导因子1α信号通路促进血管生成和骨再生[46]。此外,脂质体的掺入可显著提高GelMA的弹性和压缩模量,并降低其溶胀率[47]。 诸多研究显示,有机纳米粒子对GelMA水凝胶力学性能的增强可能与纳米粒子与GelMA中某些官能团的强烈相互作用有关[52-53]。GANGULY等[53]对纤维素纳米晶体复合GelMA水凝胶进行了X射线光电子能谱分析,发现加入纤维素纳米晶体后,GelMA水凝胶中C-O及C=O键的数量分别由10.57%,23.82%降至1.80%,8.60%,显示二者间存在强烈的化学相互作用,进而提高复合材料的机械强度及黏弹性。有机纳米粒子与GelMA的化学键合还可提升水凝胶的药物缓释能力,HAN等[52]采用碳化二亚胺法使乳酸-乙醇酸纳米粒子上的羧基与GelMA上的氨基发生化学键合,从而避免抗炎药物在早期的大量释放,进而增强GelMA的药物缓释性能。 有机纳米材料除了增强GelMA的药物运输能力和力学强度外,还可赋予其光热性能,提供适宜骨组织修复的温热环境。研究显示,温和的光热反应(40-43 ℃)可加速骨组织再生[49,52]。聚多巴胺纳米颗粒相比石墨烯等传统光热剂易于制备、光热反应温和、生物相容性佳。GelMA水凝胶与聚多巴胺纳米颗粒的组合可显著促进大鼠颅骨缺损的修复,但是,聚多巴胺纳米颗粒的引入会导致水凝胶的力学强度下降,因此研究者又同时加入聚甲基丙烯酸甲酯嵌段(一种广泛用于固定骨科金属植入物的骨水泥)来提升水凝胶的力学强度[49]。基于以上研究,可预见有机纳米颗粒复合GelMA水凝胶材料在骨组织工程领域具有良好的应用前景。 2.1.3 有机-无机纳米杂化材料 有机-无机纳米杂化材料是指有机和无机组分在纳米尺度杂化形成的一种新型材料,常见如金属有机框架[54-56]、金属有机笼[57]、笼型低聚硅倍半氧烷等[58-59],见表3。这些有机-无机纳米杂化材料结合了有机/无机纳米材料的优势,相较于传统的无机纳米材料具有金属离子缓释曲线可调、生物降解性好、毒性低等优点[55]。金属有机框架由金属离子和有机桥配体自组装而成,是一类具有可调设计、可降解、低毒等优点的多孔晶态材料,在金属有机框架的降解过程中,这些中心金属离子可以逐渐释放出来,因此金属有机框架作为金属离子控释剂具有巨大的研究潜力[54]。ZIF-67是金属有机框架的一个子类,与传统金属有机框架相比具有出色的水解稳定性,通过调整ZIF-67 纳米颗粒中有机组分2-甲基咪唑与2-乙基咪唑的比例,可将钴离子的释放总时间在7-21 d的范围内进行调节,从而与骨形成过程中的早期血管生成相匹配[55]。"
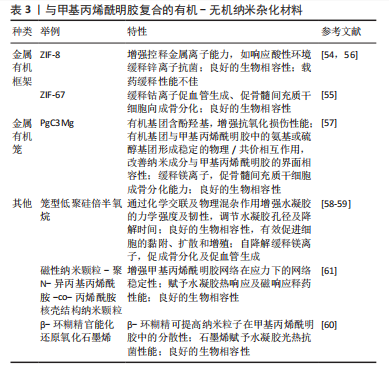
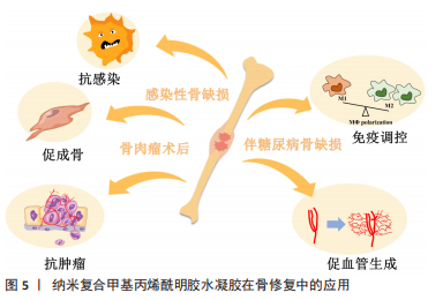
此外,有机官能团的引入也使得纳米粒子不仅具有简单的物理掺杂作用,还能与GelMA水凝胶进行化学交联,增强纳米相与有机聚合物相的相互作用,提高纳米粒子在GelMA中的分散性及GelMA网络在应力下的网络稳定性,并延长水凝胶的降解寿命[14,60-61]。笼型低聚硅倍半氧烷是由Si-O形成内部无机框架核心,外围由有机基团所包围形成的一种具有新型核-壳结构的无机-有机纳米杂化材料,笼型低聚硅倍半氧烷纳米粒子不仅可作为物理填料,其侧链中的双键也能够与GelMA聚合物反应形成聚合物水凝胶网络,增加各组分间的链缠结及组分间的相互作用力,从而增强水凝胶的力学强度[58] 。笼型低聚硅倍半氧烷与GelMA的结合有效促进骨髓间充质干细胞的成骨分化,而且在体内外促进血管生成,进而促进大鼠颅骨缺损的骨修复[59]。 2.2 纳米材料复合GelMA水凝胶在骨修复中的应用 骨组织工程的材料设计要考虑目标骨组织的潜在状况,不同的骨缺损环境,如长骨的创伤性大块骨缺损、颌面部的牙周炎、老年性骨质疏松的骨折等,对骨组织工程材料的生物学性能如抗感染、免疫调控、促成骨性能等提出了较高的要求[6-9] ,见图5。基于此,下文将以感染性骨缺损、伴糖尿病骨缺损、骨肉瘤切除术后3种骨缺损情况为例,介绍纳米复合GelMA水凝胶在骨修复中的应用。"
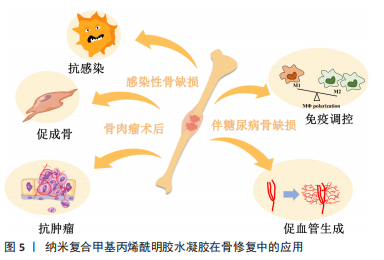

2.2.1 感染性骨缺损 感染性骨缺损的治疗被认为是骨科、颌面外科和口腔种植学领域的一个棘手问题。骨缺损区一旦出现细菌感染,骨组织的自愈能力将严重受限[33]。因此,众多学者将兼具抗菌及成骨性能的纳米粒子引入GelMA水凝胶,取得了较好的结果。 JING等[15]通过将镁改性的黑磷掺入GelMA中来制备光敏导电水凝胶,黑磷优异的光热和光动力效应可赋予GelMA较强的抗菌活性,改善炎症微环境、减少细菌诱导的骨组织损伤。在颅骨缺损感染模型中,黑磷-GelMA水凝胶显示出良好的抗菌活性,并能高效促进骨、神经再生,为感染骨缺损修复治疗提供了新的思路。 除此之外,通过纳米粒子自身的电荷调节也可实现抗菌和成骨。GENG等[31]发现,带正电荷的碳量子点对多重耐药菌表现出优异的抗菌活性,而带负电荷的碳量子点可显著促进骨再生。为了避免支架中带有正负电荷的碳量子点的聚集,该作者首先通过将带正电荷的碳量子点沉积在带负电的二硫化钨纳米片上以形成近中性的杂化物,而后将带正电荷的碳量子点-二硫化钨杂化物与带负电荷的碳量子点共同加入GelMA水凝胶中,构建了一种多功能可生物降解的的生物材料支架。在感染性颅骨缺损模型中,对照组即使在第60 天仍未观察到明显的新骨形成,而植入该生物材料支架的小鼠颅骨缺损则接近完全愈合。得益于GelMA水凝胶对不规则骨缺损形状的良好适应能力,兼具抗菌性能的纳米复合GelMA水凝胶在牙周炎治疗领域也有良好的应用。LIU等[56]利用在体内外可持续性释放锌离子的沸石咪唑骨架-8(一种由锌离子和咪唑酸盐配体桥接的金属有机框架材料)增强复合GelMA水凝胶的抗菌及成骨能力,得到的复合材料在大鼠牙周炎模型中有效促进牙槽骨再生。 2.2.2 伴糖尿病骨缺损 糖尿病是一种以血糖升高和全身性慢性炎症为特征的代谢紊乱。临床上,糖尿病患者骨折后骨再生和血管生成能力受损,常导致不愈合或延迟愈合,目前仍缺乏有效的解决方法[24,62]。糖尿病患者的骨缺损微环境非常复杂,持续性高血糖会导致活性氧和促炎因子增加、M1型促炎巨噬细胞极化增加、M2型抗炎巨噬细胞极化减少,从而诱发慢性炎症[63]。高血糖及慢性炎症状态进一步抑制血管生成、诱发内皮功能障碍、扰乱钙磷代谢、抑制成骨分化,从而导致生物矿化不良、骨折风险增加、骨愈合受损等问题[26,63-64]。因此,糖尿病骨缺损修复材料应该针对以上病理性微环境的特点加以设计。 目前,在糖尿病骨缺损修复方面的纳米复合GelMA水凝胶研究相对较少,已有研究大多是通过无机纳米颗粒复合GelMA水凝胶搭载抗炎因子或自身释放抗炎金属离子的方式调控巨噬细胞极化。SUN等[6]在GelMA水凝胶中加载RAW264.7及骨髓间充质干细胞,并通过介孔二氧化硅纳米颗粒的开孔结构负载抗炎因子骨形态发生蛋白4,该复合水凝胶支架有效提高RAW264.7细胞向M2型巨噬细胞的极化及骨髓间充质干细胞的成骨分化水平,在糖尿病大鼠的颅骨临界尺寸缺损模型中表现出良好的促骨再生效果。WU等[26]将具有锂离子缓释能力的生物活性玻璃纳米颗粒与GelMA水凝胶结合,利用锂离子可直接促成骨分化及调控巨噬细胞向M2型极化的原理,有效减轻炎症反应、促血管生成及成骨分化。此外,该作者还发现生物活性玻璃本身可作为矿化前体调控离子成核,其与GelMA的结合可形成仿生骨再生微环境,进一步启动仿生生物矿化过程、调节离子代谢、靶向LOX基因并调节其下游的胶原纤维免受糖基化干扰,进而促进糖尿病微环境下的生物矿化。 2.2.3 骨肉瘤切除术后 骨肉瘤是骨骼中最常见的原发性实体恶性肿瘤,在青少年和儿童中高发,最常见于血供丰富的长骨干骺端[65-66]。手术切除是骨肉瘤的常见临床治疗方法之一,然而,骨肉瘤切除造成的骨缺损相对较大,超过人体自愈限度[67],且患者自身免疫能力较差,易发生感染、肿瘤复发等情况。因此,理想的肿瘤术后修复材料应该在满足骨缺损修复要求的同时,还能实现抗感染、杀伤癌细胞等多重功能。 如前所述,GelMA可以很好地模拟骨骼细胞外基质的环境,调控细胞的黏附、增殖、迁移及分化等行为,可作为支架为骨修复提供一个良好的局部微环境,但其缺乏抗感染及抗癌作用,且GelMA本身的药物控释性能不足,不能直接搭载相关化疗药物。因此,有研究者将GelMA与其他药物递送系统(如脂质体[48]、MXene[30]、磁性纳米颗粒[61]、金纳米棒等[37])集成,通过协同作用产生更适合骨肉瘤切除术后的骨修复材料。 另外,在骨肉瘤相关的骨组织工程领域,具有光热效应的纳米材料与GelMA的结合也受到较多关注。MXene纳米片是一种新兴的二维无机纳米材料,具有良好的生物相容性及光热转换效率,在肿瘤光热治疗方面有着巨大的潜力;同时,因其比表面积大,且具有负电荷基团(如?F、?OH和=O),可以通过强静电相互作用携带阳性抗菌药物(如妥布霉素)。将MXene/妥布霉素与GelMA结合可同时实现促骨修复、抗菌、抗肿瘤复发的作用[30]。LIAO等[37]将具有光热效应的金纳米棒及有促成骨性能的纳米羟基磷灰石引入甲基丙烯酸化明胶/甲基丙烯酸化硫酸软骨素水凝胶,得到的杂化水凝胶在胫骨骨肉瘤小鼠模型上实现了良好的肿瘤治疗和骨再生的双重功能。 不过,目前纳米复合GelMA水凝胶在骨肉瘤相关的骨组织工程中的研究大多局限于体外实验,在同一个动物模型上讨论抗肿瘤和促骨修复的文献相对较少,这可能与骨肉瘤术后骨缺损的动物模型相对难建立有关。此外,基于抗菌、抗肿瘤、促骨修复思路设计的纳米复合GelMA水凝胶材料,不仅适用于骨肉瘤,也会为口腔癌引起的颌面部缺损、恶性肿瘤骨转移等复杂疾病的治疗带来一定的希望。 "

| [1] 刘肇兴,吴兴,林欢欢,等.复合明胶甲基丙烯基水凝胶在骨组织工程中的应用及前景[J].中国组织工程研究,2018,22(10):1593-1598. [2] 张孝利,余优成,吴兴文,等.GelMA水凝胶在骨组织工程中的研究进展[J].复旦学报(医学版),2021,48(6):847-851. [3] DONG Z, YUAN Q, HUANG K, et al. Gelatin methacryloyl (GelMA)-based biomaterials for bone regeneration. RSC Adv. 2019;9(31):17737-17744. [4] XIAO S, ZHAO T, WANG J, et al. Gelatin Methacrylate (GelMA)-Based Hydrogels for Cell Transplantation: an Effective Strategy for Tissue Engineering. Stem Cell Rev Rep. 2019;15(5):664-679. [5] SUN M, SUN X, WANG Z, et al. Synthesis and Properties of Gelatin Methacryloyl (GelMA) Hydrogels and Their Recent Applications in Load-Bearing Tissue. Polymers (Basel). 2018;10(11):1290. [6] SUN X, MA Z, ZHAO X, et al. Three-dimensional bioprinting of multicell-laden scaffolds containing bone morphogenic protein-4 for promoting M2 macrophage polarization and accelerating bone defect repair in diabetes mellitus. Bioact Mater. 2021;6(3):757-769. [7] KOONS GL, DIBA M, MIKOS AG. Materials design for bone-tissue engineering. Nat Rev Mater. 2020;5(8):584-603. [8] CHEN X, BAI S, LI B, et al. Fabrication of gelatin methacrylate/nanohydroxyapatite microgel arrays for periodontal tissue regeneration. Int J Nanomedicine. 2016;11: 4707-4718. [9] TURNBULL G, CLARKE J, PICARD F, et al. 3D bioactive composite scaffolds for bone tissue engineering. Bioact Mater. 2018;3(3):278-314. [10] PIAO Y, YOU H, XU T, et al. Biomedical applications of gelatin methacryloyl hydrogels. Engineered Regeneration. 2021;2:47-56. [11] SHAO Y, YOU D, LOU Y, et al. Controlled Release of Naringin in GelMA-Incorporated Rutile Nanorod Films to Regulate Osteogenic Differentiation of Mesenchymal Stem Cells. ACS Omega. 2019;4(21):19350-19357. [12] SAKR MA, SAKTHIVEL K, HOSSAIN T, et al. Recent trends in gelatin methacryloyl nanocomposite hydrogels for tissue engineering. J Biomed Mater Res A. 2022; 110(3):708-724. [13] ALARçIN E, İZBUDAK B, YüCE ERARSLAN E, et al. Optimization of methacrylated gelatin /layered double hydroxides nanocomposite cell-laden hydrogel bioinks with high printability for 3D extrusion bioprinting. J Biomed Mater Res A. 2023; 111(2):209-223. [14] SHAO N, GUO J, GUAN Y, et al. Development of Organic/Inorganic Compatible and Sustainably Bioactive Composites for Effective Bone Regeneration. Biomacromolecules. 2018;19(9):3637-3648. [15] JING X, XU C, SU W, et al. Photosensitive and Conductive Hydrogel Induced Innerved Bone Regeneration for Infected Bone Defect Repair. Adv Healthc Mater. 2023;12(3):e2201349. [16] SHI Z, XU Y, MULATIBIEKE R, et al. Nano-Silicate-Reinforced and SDF-1α-Loaded Gelatin-Methacryloyl Hydrogel for Bone Tissue Engineering. Int J Nanomedicine. 2020;15:9337-9353. [17] KURIAN AG, SINGH RK, PATEL KD, et al. Multifunctional GelMA platforms with nanomaterials for advanced tissue therapeutics. Bioact Mater. 2022;8:267-295. [18] ZHANG Y, CHEN H, LI J. Recent advances on gelatin methacrylate hydrogels with controlled microstructures for tissue engineering. Int J Biol Macromol. 2022;221: 91-107. [19] LV B, LU L, HU L, et al. Recent advances in GelMA hydrogel transplantation for musculoskeletal disorders and related disease treatment. Theranostics. 2023; 13(6):2015-2039. [20] XIE M, YU K, SUN Y, et al. Protocols of 3D Bioprinting of Gelatin Methacryloyl Hydrogel Based Bioinks. J Vis Exp. 2019;(154).doi: 10.3791/60545. [21] HOU S, NIU X, LI L, et al. Simultaneous nano- and microscale structural control of injectable hydrogels via the assembly of nanofibrous protein microparticles for tissue regeneration. Biomaterials. 2019;223:119458. [22] WANG Y, CAO X, MA M, et al. A GelMA-PEGDA-nHA Composite Hydrogel for Bone Tissue Engineering. Materials (Basel). 2020;13(17):3735. [23] MIAO F, LIU T, ZHANG X, et al. Engineered bone tissues using biomineralized gelatin methacryloyl/sodium alginate hydrogels. J Biomater Sci Polym Ed. 2022; 33(2):137-154. [24] LI Y, LIU Z, ZHAO C, et al. A sustained-release PDGF-BB nanocomposite hydrogel for DM-associated bone regeneration. J Mater Chem B. 2023;11(5): 974-984. [25] CHENG H, YUE K, KAZEMZADEH-NARBAT M, et al. Mussel-Inspired Multifunctional Hydrogel Coating for Prevention of Infections and Enhanced Osteogenesis. ACS Appl Mater Interfaces. 2017;9(13):11428-11439. [26] WU Z, BAI J, GE G, et al. Regulating Macrophage Polarization in High Glucose Microenvironment Using Lithium-Modified Bioglass-Hydrogel for Diabetic Bone Regeneration. Adv Healthc Mater. 2022;11(13):e2200298. [27] XIN T, MAO J, LIU L, et al. Programmed Sustained Release of Recombinant Human Bone Morphogenetic Protein-2 and Inorganic Ion Composite Hydrogel as Artificial Periosteum. ACS Appl Mater Interfaces. 2020;12(6):6840-6851. [28] LI S, LI Z, YANG J, et al. Inhibition of Sympathetic Activation by Delivering Calcium Channel Blockers from a 3D Printed Scaffold to Promote Bone Defect Repair. Adv Healthc Mater. 2022;11(16):2200785. [29] 赵正宜.可塑形光固化GelMA/Laponite/nHAP凝胶支架在骨缺损修复中的应用研究[D].合肥:安徽医科大学,2022. [30] YIN J, HAN Q, ZHANG J, et al. MXene-Based Hydrogels Endow Polyetheretherketone with Effective Osteogenicity and Combined Treatment of Osteosarcoma and Bacterial Infection. ACS Appl Mater Interfaces. 2020;12(41): 45891-45903. [31] GENG B, LI P, FANG F, et al. Antibacterial and osteogenic carbon quantum dots for regeneration of bone defects infected with multidrug-resistant bacteria. Carbon. 2021;184:375-385. [32] PACELLI S, MALONEY R, CHAKRAVARTI AR, et al. Controlling Adult Stem Cell Behavior Using Nanodiamond-Reinforced Hydrogel: Implication in Bone Regeneration Therapy. Sci Rep. 2017;7(1):6577. [33] NIE R, SUN Y, LV H, et al. 3D printing of MXene composite hydrogel scaffolds for photothermal antibacterial activity and bone regeneration in infected bone defect models. Nanoscale. 2022;14(22):8112-8129. [34] 李家颖.骨损伤微环境响应性释放BMP-2多肽的MnO_2/GelMA复合水凝胶促进骨修复[D].苏州:苏州大学,2019. [35] PAN H, GAO H, LI Q, et al. Engineered macroporous hydrogel scaffolds via pickering emulsions stabilized by MgO nanoparticles promote bone regeneration. J Mater Chem B. 2020;8(28):6100-6114. [36] YAN J, WANG Y, RAN M, et al. Peritumoral Microgel Reservoir for Long-Term Light-Controlled Triple-Synergistic Treatment of Osteosarcoma with Single Ultra-Low Dose. Small. 2021;17(31):e2100479. [37] LIAO J, SHI K, JIA Y, et al. Gold nanorods and nanohydroxyapatite hybrid hydrogel for preventing bone tumor recurrence via postoperative photothermal therapy and bone regeneration promotion. Bioact Mater. 2021;6(8):2221-2230. [38] 潘昊田.负载Mg元素的GelMA大孔水凝胶支架构建及其骨组织工程的应用研究[D].广州:华南理工大学,2020. [39] QU L, DUBEY N, RIBEIRO JS, et al. Metformin-loaded nanospheres-laden photocrosslinkable gelatin hydrogel for bone tissue engineering. J Mech Behav Biomed Mater. 2021;116:104293. [40] HUANG K, WU J, GU Z. Black Phosphorus Hydrogel Scaffolds Enhance Bone Regeneration via a Sustained Supply of Calcium-Free Phosphorus. ACS Appl Mater Interfaces. 2019;11(3):2908-2916. [41] OU Q, HUANG K, FU C, et al. Nanosilver-incorporated halloysite nanotubes/gelatin methacrylate hybrid hydrogel with osteoimmunomodulatory and antibacterial activity for bone regeneration . Chem Eng J. 2020;382:123019. [42] KURIAN AG, SINGH RK, LEE JH, et al. Surface-Engineered Hybrid Gelatin Methacryloyl with Nanoceria as Reactive Oxygen Species Responsive Matrixes for Bone Therapeutics. ACS Appl Bio Mater. 2022;5(3):1130-1138. [43] YANG Y, XU T, ZHANG Q, et al. Biomimetic, Stiff, and Adhesive Periosteum with Osteogenic-Angiogenic Coupling Effect for Bone Regeneration. Small. 2021; 17(14):e2006598. [44] 杨开明.仿贻贝磁性羟基磷灰石复合明胶水凝胶应用于骨修复研究[D].成都:西南交通大学,2021. [45] 余幸鸽,林开利.纳米复合水凝胶在骨组织工程中的应用[J].中国组织工程研究,2020,24(34):5441-5446. [46] LI Z, LI S, YANG J, et al. 3D bioprinted gelatin/gellan gum-based scaffold with double-crosslinking network for vascularized bone regeneration. Carbohydr Polym. 2022;290:119469. [47] ELKHOURY K, SANCHEZ-GONZALEZ L, LAVRADOR P, et al. Gelatin Methacryloyl (GelMA) Nanocomposite Hydrogels Embedding Bioactive Naringin Liposomes. Polymers (Basel). 2020;12(12):2944. [48] WU W, DAI Y, LIU H, et al. Local release of gemcitabine via in situ UV-crosslinked lipid-strengthened hydrogel for inhibiting osteosarcoma. Drug Deliv. 2018;25(1): 1642-1651. [49] WU Y, ZHANG X, TAN B, et al. Near-infrared light control of GelMA/PMMA/PDA hydrogel with mild photothermal therapy for skull regeneration. Biomater Adv. 2022;133: 112641. [50] WU Y, LI X, SUN Y, et al. Multiscale design of stiffening and ROS scavenging hydrogels for the augmentation of mandibular bone regeneration. Bioact Mater. 2023;20:111-125. [51] XIAO W, TAN Y, LI J, et al. Fabrication and characterization of silk microfiber-reinforced methacrylated gelatin hydrogel with turnable properties. J Biomater Sci Polym Ed, 2018;29(17):2068-2082. [52] HAN X, SHEN J, CHEN S, et al. Ultrasonic-controlled “explosive” hydrogels to precisely regulate spatiotemporal osteoimmune disturbance. Biomaterials. 2023;295:122057. [53] GANGULY K, DUTTA SD, RANDHAWA A, et al. Transcriptomic Changes toward Osteogenic Differentiation of Mesenchymal Stem Cells on 3D-Printed GelMA/CNC Hydrogel under Pulsatile Pressure Environment. Adv Healthc Mater. 2023;12(11): 2202163. [54] LI N, XIE L, WU Y, et al. Dexamethasone-loaded zeolitic imidazolate frameworks nanocomposite hydrogel with antibacterial and anti-inflammatory effects for periodontitis treatment. Mater Today Bio. 2022;16:100360. [55] SUN Y, LIU X, ZHU Y, et al. Tunable and Controlled Release of Cobalt Ions from Metal-Organic Framework Hydrogel Nanocomposites Enhances Bone Regeneration. ACS Appl Mater Interfaces. 2021;13(49):59051-59066. [56] LIU Y, ZHU Z, PEI X, et al. ZIF-8-Modified Multifunctional Bone-Adhesive Hydrogels Promoting Angiogenesis and Osteogenesis for Bone Regeneration. ACS Appl Mater Interfaces. 2020;12(33):36978-36995. [57] TAN X, WU J, WANG R, et al. PgC(3)Mg metal-organic cages functionalized hydrogels with enhanced bioactive and ROS scavenging capabilities for accelerated bone regeneration. J Mater Chem B. 2022;10(28): 5375-5387. [58] ZHANG X, HUANG P, JIANG G, et al. A novel magnesium ion-incorporating dual-crosslinked hydrogel to improve bone scaffold-mediated osteogenesis and angiogenesis. Mater Sci Eng C Mater Biol Appl. 2021;121:111868. [59] ZHANG Y, CHEN M, TIAN J, et al. In situ bone regeneration enabled by a biodegradable hybrid double-network hydrogel. Biomater Sci. 2019;7(8):3266-3276. [60] LI Y, HE J, ZHOU J, et al. A conductive photothermal non-swelling nanocomposite hydrogel patch accelerating bone defect repair. Biomater Sci. 2022;10(5):1326-1341. [61] JALILI NA, JAISWAL MK, PEAK CW, et al. Injectable nanoengineered stimuli-responsive hydrogels for on-demand and localized therapeutic delivery. Nanoscale. 2017;9(40):15379-15389. [62] YANG C, ZHENG Z, YOUNIS MR, et al. 3D Printed Enzyme-Functionalized Scaffold Facilitates Diabetic Bone Regeneration. Adv Funct Mater. 2021;31(20):2101372. [63] XU Z, QI X, BAO M, et al. Biomineralization inspired 3D printed bioactive glass nanocomposite scaffolds orchestrate diabetic bone regeneration by remodeling micromilieu. Bioact Mater. 2023;25:239-255. [64] DE OLIVEIRA P, BONFANTE EA, BERGAMO ETP, et al. Obesity/Metabolic Syndrome and Diabetes Mellitus on Peri-implantitis. Trends Endocrinol Metab. 2020;31(8): 596-610. [65] HUANG Y, ZHAI X, MA T, et al. Unified Therapeutic-Prophylactic Tissue Engineering Scaffold Demonstrated to Prevent Tumor Recurrence and Overcoming Infection toward Bone Remodeling. Adv Mater. 2023: e2300313.doi: 10.1002/adma.202300313. [66] TIAN H, WU R, FENG N, et al. Recent advances in hydrogels-based osteosarcoma therapy. Front Bioeng Biotechnol. 2022;10:1042625. [67] LIU X, ZHANG Y, WU H, et al. A conductive gelatin methacrylamide hydrogel for synergistic therapy of osteosarcoma and potential bone regeneration. Int J Biol Macromol. 2023;228:111-122. |
| [1] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [2] | Wang Shanshan, Shu Qing, Tian Jun. Physical factors promote osteogenic differentiation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1083-1090. |
| [3] | Wang Jianchun, Yang Shuqing, Su Xin, Wang Hongyuan. Different contents of B2O3 affect mechanical properties and bioactivity of bioactive glass scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 712-716. |
| [4] | Tian Xin, Liu Tao, Yang Huilin, He Fan. In vitro evaluation of sustained release Kartogenin by gelatin methacryloyl microspheres for repairing nucleus pulposus degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 724-730. |
| [5] | Yin Tong, Yang Jilei, Li Yourui, Liu Zhuoran, Jiang Ming. Application of core-shell structured nanofibers in oral tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 766-770. |
| [6] | Chen Xiaofang, Zheng Guoshuang, Li Maoyuan, Yu Weiting. Preparation and application of injectable sodium alginate hydrogels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 789-794. |
| [7] | Wang Jiani, Chen Junyu. Angiogenesis mechanism of metal ions and their application in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 804-812. |
| [8] | Wang Wu, Fan Xiaolei, Xie Jie, Hu Yihe, Zeng Min. Hydroxyapatite-polyvinyl alcohol/collagen-chitosan-gelatin composite hydrogel for repairing rabbit osteochondral defect [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 682-689. |
| [9] | Zhang Ya, Mu Qiuju, Wang Zilin, Liu Hongjie, Zhu Lili. Hydrogel loaded with platelet-rich plasma promotes wound healing in diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 690-696. |
| [10] | Shen Ziqing, Xia Tian, Shan Yibo, Zhu Ruijun, Wan Haoxin, Ding Hao, Pan Shu, Zhao Jun. Vascularized tracheal substitutes constructed by exosome-load hydrogel-modified 3D printed scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 697-705. |
| [11] | Yang Yuqing, Chen Zhiyu. Role and application of early transient presence of M1 macrophages in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 594-601. |
| [12] | Long Zhirui, Huang Lei, Xiao Fang, Wang Lin, Wang Xiaobei. Characteristics of hydrogel microspheres in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 472-478. |
| [13] | Kong Xiangyu, Wang Xing, Pei Zhiwei, Chang Jiale, Li Siqin, Hao Ting, He Wanxiong, Zhang Baoxin, Jia Yanfei. Biological scaffold materials and printing technology for repairing bone defects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 479-485. |
| [14] | Xu Jing, Lyu Huixin, Bao Xin, Zhang Yi, Wang Yihan, Zhou Yanmin. Application of near infrared responsive hydrogels in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 486-492. |
| [15] | Dai Jing, Liu Shasha, Shen Mingjing. Exosome-loaded injectable hydrogel for repairing bone defects around implants [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 347-354. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||