Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (22): 3464-3471.doi: 10.12307/2024.486
Previous Articles Next Articles
Antibacterial properties of silver nanoparticle-coated stainless steel prepared via active screen plasma surface modification in vitro
Wang Zhaofei1, He Guoyun1, Tian Fangcan2, Li Guangfeng1, Cao Zhonghua1, Liu Xiangfei1
- 1Department of Orthopedic Surgery, Shanghai ZhongYe Hospital, Genertec Universal Medical Group, Shanghai 200941, China; 2Department of Orthopedic Surgery, Xuanwei Second People’s Hospital, Xuanwei 655099, Yunnan Province, China
-
Received:2023-08-02Accepted:2023-09-28Online:2024-08-08Published:2024-01-20 -
Contact:Liu Xiangfei, Associate chief physician, Department of Orthopedic Surgery, Shanghai ZhongYe Hospital, Genertec Universal Medical Group, Shanghai 200941, China -
About author:Wang Zhaofei, Doctoral candidate, Attending physician, Department of Orthopedic Surgery, Shanghai ZhongYe Hospital, Genertec Universal Medical Group, Shanghai 200941, China -
Supported by:Science and Technology Commission of Baoshan District of Shanghai, No. 20-E-38 and 2023-E-27 (to WZF); Baoshan District Health Commission Excellent Youth (Yucai) Program, No. BSWSYC-2023-15 (to WZF)
CLC Number:
Cite this article
Wang Zhaofei, He Guoyun, Tian Fangcan, Li Guangfeng, Cao Zhonghua, Liu Xiangfei. Antibacterial properties of silver nanoparticle-coated stainless steel prepared via active screen plasma surface modification in vitro[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3464-3471.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
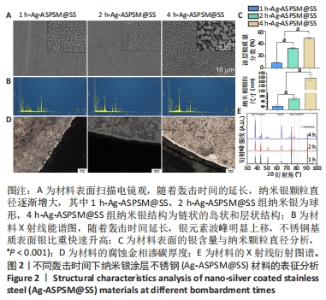
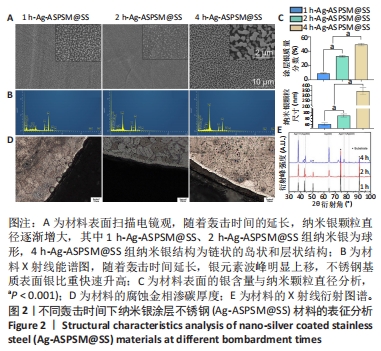
2.1 各组Ag-ASPSM@SS的表征结果 扫描电镜下观察3组样品的表面形貌,见图2A。在1 h-Ag-ASPSM@SS和2 h-Ag-ASPSM@SS样品中,纳米银颗粒几乎是球形的,颗粒分散良好;在4 h-Ag-ASPSM@SS样品中,纳米银颗粒聚集形成了一个链状的岛状和层状结构,大小不一,呈弥散分布。从X射线能谱图来看,随着攻击时间的延长Ag-ASPSM@SS表面的银含量增加,1 h-Ag-ASPSM@SS、2 h-Ag-ASPSM@SS、4 h-Ag-ASPSM@SS分别约含有8.90%,32.98%与49.28%的银,组间比较差异有显著性意义(P < 0.01),见图2B。在1 h-Ag-ASPSM@SS样品中大多数纳米银颗粒的直径约为60 nm,然而随着Ag-ASPSM轰击时间增加到2,4 h,纳米银颗粒的直径逐渐增大到约80 nm和400 nm,组间比较差异有显著性意义(P < 0.001),见图2C。 样品内部的腐蚀区域是不锈钢基体,样品边缘的白色光亮部分是渗碳层,腐蚀金相检测显示,1 h-Ag-ASPSM@SS和2 h-Ag-ASPSM@SS样品的平均渗碳厚度分别为5 μm和6 μm,4 h-Ag-ASPSM@SS样品的渗碳层的平均厚度为6 μm,与2 h轰击的样品接近,见图2D。 X射线衍射分析显示,除不锈钢外,3组Ag-ASPSM@SS样品基本上都没有杂乱的峰,这些峰主要在2θ范围内,即38.11°(111),44.28°(200),64.43°(220),77.47°(311)和81.54°(222)。结果还显示随着轰击时间的增加,银涂层的厚度增加,不锈钢衍射峰强度开始下降。然而随着沉积时间的增加,银涂层的(111)取向趋于减弱,取向系数从66.5%下降到40.0%,其他取向(200)(220)(311)和(222)的取向系数逐渐提高,有上升趋势,见图2E。"
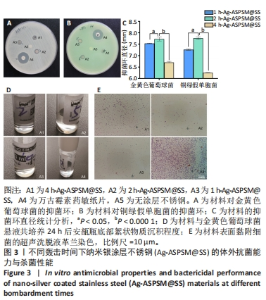
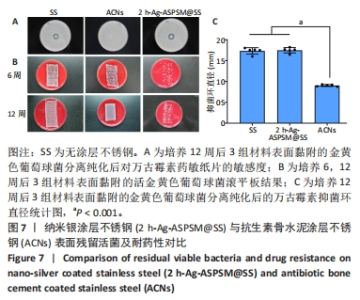
2.2 各组Ag-ASPSM@SS体外抗菌能力与杀菌性能 抗菌能力:对于金黄色葡萄球菌ATCC25923菌株与铜绿假单胞菌ATCC27853菌株,材料A5(空白对照不锈钢)没有显示出明显的抑菌环;对于金黄色葡萄球菌ATCC25923菌株,材料A1、A2、A3和A4的抑菌环直径分别为(6.70±0.02),(7.73±0.12),(7.54±0.01),(17.56±0.76) mm,材料组间比较差异有显著性意义(P <0.05,P < 0.000 1);对于铜绿假单胞菌ATCC27853菌株,材料A1、A2、A3和A4的抑菌环直径分别为(6.25±0.02),(7.76±0.03),(7.26±0.02),(17.24±1.70) mm,材料组间比较差异有显著性意义(P < 0.05,P < 0.000 1),见图3A-C。 综合来看,材料A2(2 h-Ag-ASPSM@SS)对金黄色葡萄球菌ATCC25923和铜绿假单胞菌ATCC27853呈现出更好的抑制效果,比万古霉素和多黏菌素药敏纸片的效果略差。 杀菌性能:将各组样品浸入金黄色葡萄球菌悬液中,观察细菌悬浮液底部的絮状物质沉积程度,可见A2组底部有明显的絮状物沉积,这代表了死亡细菌的沉积;A4组底部有明亮和清晰的物质,表明A2组材料在24 h内具有更好的有效杀菌效果;此外,革兰染色结果与上述现象一致,见图3D,E。"

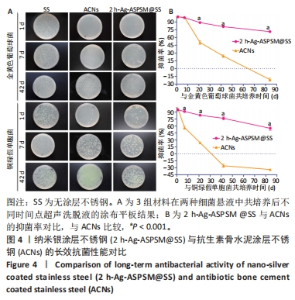
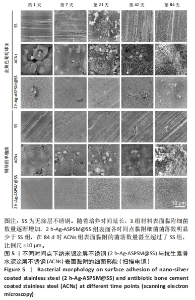
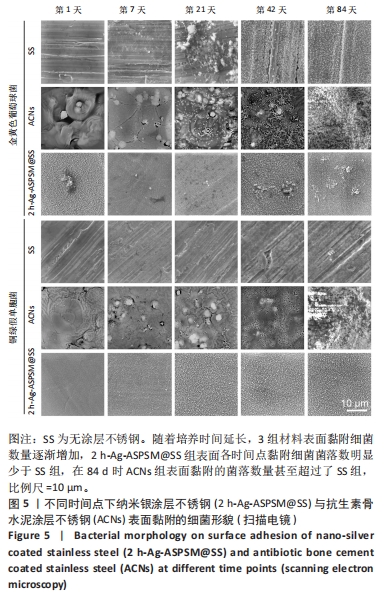
2.3 2 h-Ag-ASPSM@SS与ACNs的体外长效抗菌能力比较 图4展示了各组样品与细菌培养后特定时间点的涂布平板计数结果。在所有的时间节点(1,7,21,42,84 d),2 h-Ag-ASPSM@SS样品表面的金黄色葡萄球菌数量明显低于无涂层不锈钢组,表明2 h-Ag-ASPSM@SS样品对抑制金黄色葡萄球菌的定植有长期效果,其在84 d后仍能够显著减少金黄色葡萄球菌和铜绿假单胞菌的黏附。意外的是,培养84 d后,ACNs样品表面黏附的细菌菌落数量明显多于无涂层不锈钢。涂布平板计数定量分析结果显示,2 h-Ag-ASPSM@SS在1,7,21,42和84 d对金黄色葡萄球菌的抑菌率分别为100%,98.29%,88.81%,80.87%和71.39%;ACNs在1,7,21,42和84 d对金黄色葡萄球菌的抑菌率的抑菌率为100%,99.18%,50.47%,24.16%和-21.13%。与2 h-Ag-ASPSM@SS相比,ACNs呈现出强大的初始杀菌效果,然而随着时间的推移,ACNs的抑菌能力明显下降,甚至促进了细菌的黏附。"

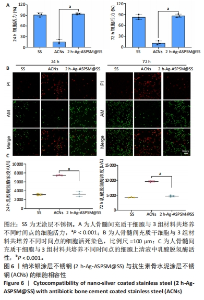
2.4 2 h-Ag-ASPSM@SS与ACNs的体外细胞相容性 如图6A所示,与无涂层不锈钢组比较,2 h-Ag-ASPSM@SS组培养24,72 h的细胞活力无明显变化(P > 0.05),ACNs组培养24,72 h的细胞活力降低(P < 0.001)。Calcein-AM/PI双重染色显示,与无涂层不锈钢组比较,2 h-Ag-ASPSM@SS组培养24,72 h的活细胞、死细胞数量无明显变化,ACNs组培养24,72 h的活细胞数量减少、死细胞数量增加,见图6B。如图6C所示,与无涂层不锈钢组比较,2 h-Ag-ASPSM@SS组培养24,72 h的细胞上清液内乳酸脱氢酶活性无明显变 (P > 0.05),ACNs组培养24,72 h的细胞上清液内乳酸脱氢酶活性升高(P < 0.001)。"

| [1] ZHANG L, YANG Y, XIONG YH, et al. Infection-responsive long-term antibacterial bone plates for open fracture therapy. Bioact Mater. 2023; 25:1-25. [2] TANWAR YS, FERREIRA N. The role of bioactive glass in the management of chronic osteomyelitis: a systematic review of literature and current evidence. Infect Dis (Lond). 2020;52(4):219-226. [3] LI G, LV K, CHENG Q, et al. Enhanced Bacterial-Infected Wound Healing by Nitric Oxide-Releasing Topological Supramolecular Nanocarriers with Self-Optimized Cooperative Multi-Point Anchoring. Adv Sci (Weinh). 2023;10(11):e2206959. [4] UDDIN TM, CHAKRABORTY AJ, KHUSRO A, et al. Antibiotic resistance in microbes: History, mechanisms, therapeutic strategies and future prospects. J Infect Public Health. 2021;14(12):1750-1766. [5] PULINGAM T, PARUMASIVAM T, GAZZALI AM, et al. Antimicrobial resistance: Prevalence, economic burden, mechanisms of resistance and strategies to overcome. Eur J Pharm Sci. 2022;170:106103. [6] MASTERS EA, RICCIARDI BF, BENTLEY KLDM, et al. Skeletal infections: microbial pathogenesis, immunity and clinical management. Nat Rev Microbiol. 2022;20(7):385-400. [7] CHENG X, PEI X, XIE W, et al. pH-Triggered Size-Tunable Silver Nanoparticles: Targeted Aggregation for Effective Bacterial Infection Therapy. Small. 2022;18(22):e2200915. [8] FERRERES G, IVANOVA K, TORRENT-BURGUéS J, et al. Multimodal silver-chitosan-acylase nanoparticles inhibit bacterial growth and biofilm formation by Gram-negative Pseudomonas aeruginosa bacterium. J Colloid Interface Sci. 2023;646:576-586. [9] WANG G, JIN W, QASIM AM, et al. Antibacterial effects of titanium embedded with silver nanoparticles based on electron-transfer-induced reactive oxygen species. Biomaterials. 2017;124:25-34. [10] QIN H, CAO H, ZHAO Y, et al. Antimicrobial and osteogenic properties of silver-ion-implanted stainless steel.ACS Appl Mater Interfaces. 2015;7(20):10785-10794. [11] MOUSAVI SM, HASHEMI SA, GHASEMI Y, et al. Green synthesis of silver nanoparticles toward bio and medical applications: review study. Artif Cells Nanomed Biotechnol. 2018;46(sup3):S855-S872. [12] YOUNAS W, KHAN FU, ZAMAN M, et al. Toxicity of synthesized silver nanoparticles in a widespread fish: A comparison between green and chemical. Sci Total Environ. 2022;845:157366. [13] JI X, LI X, DONG Y, et al. Synthesis and in-vitro antibacterial properties of a functionally graded Ag impregnated composite surface. Mater Sci Eng C Mater Biol Appl. 2019;99:150-158. [14] DONG Y, LI X, TIAN L, et al. Towards long-lasting antibacterial stainless steel surfaces by combining double glow plasma silvering with active screen plasma nitriding. Acta Biomater. 2011;7(1):447-457. [15] KVRYAN A, EFAW CM, HIGGINBOTHAM KA, et al. Corrosion Initiation and Propagation on Carburized Martensitic Stainless Steel Surfaces Studied via Advanced Scanning Probe Microscopy. Materials (Basel). 2019;12(6):940. [16] MENSAH LM, LOVE BJ. A meta-analysis of bone cement mediated antibiotic release: Overkill, but a viable approach to eradicate osteomyelitis and other infections tied to open procedures. Mater Sci Eng C Mater Biol Appl. 2021;123:111999. [17] STRACY M, SNITSER O, YELIN I, et al. Minimizing treatment-induced emergence of antibiotic resistance in bacterial infections. Science. 2022;375(6583):889-894. [18] SLANE J, GIETMAN B, SQUIRE M. Antibiotic elution from acrylic bone cement loaded with high doses of tobramycin and vancomycin. J Orthop Res. 2018;36(4):1078-1085. [19] JUNG KH, LEE CC, KIM TH, et al. Does spiked tibial cement spacer reduce spacer-related problems in two-stage revision total knee arthroplasty for infection? Int Orthop. 2022;46(9):2009-2017. [20] FEI Z, ZHANG Z, WANG Y, et al. Comparing the Efficacy of Articulating Spacers in Two-Stage Revision for Periprosthetic Joint Infection Following Total Knee Arthroplasty: All-Cement Spacers vs Sterilized Replanted Metal-Polyethylene Spacers. Int J Gen Med. 2022;15: 3293-3301. [21] WANG G, LUO W, ZHOU Y, et al. Custom-Made Antibiotic Cement-Coated Nail for the Treatment of Infected Bone Defect. Biomed Res Int. 2021;2021:6693906. [22] STREULI JC, EXNER GU, REIZE CL, et al. In vitro inhibition of coagulase-negative staphylococci by vancomycin/aminoglycoside-loaded cement spacers. Infection. 2006;34(2):81-86. [23] BOELCH SP, JORDAN MC, ARNHOLDT J, et al. Antibiotic elution and compressive strength of gentamicin/vancomycin loaded bone cements are considerably influenced by immersion fluid volume. J Mater Sci Mater Med. 2019;30(2):29. [24] BERBERICH C, SANZ-RUIZ P. Risk assessment of antibiotic resistance development by antibiotic-loaded bone cements: is it a clinical concern? EFORT Open Rev. 2019;4(10):576-584. [25] VAN VUGT TAG, ARTS JJ, GEURTS JAP. Antibiotic-Loaded Polymethylmethacrylate Beads and Spacers in Treatment of Orthopedic Infections and the Role of Biofilm Formation. Front Microbiol. 2019; 10:1626. [26] MENDEZ-PFEIFFER P, BALLESTEROS-MONRREAL MG, GAONA-OCHOA J, et al. Biosynthesis of Silver Nanoparticles Using Seasonal Samples of Sonoran Desert Propolis: Evaluation of Its Antibacterial Activity against Clinical Isolates of Multi-Drug Resistant Bacteria. Pharmaceutics. 2022; 14(9):1853. [27] MENICHETTI A, MAVRIDI-PRINTEZI A, MORDINI D, et al. Effect of Size, Shape and Surface Functionalization on the Antibacterial Activity of Silver Nanoparticles. J Funct Biomater. 2023;14(5):244. [28] WAKSHLAK RB, PEDAHZUR R, AVNIR D. Antibacterial activity of silver-killed bacteria: the “zombies” effect. Sci Rep. 2015;5:9555. [29] BOONRUANG C, SANUMANG W. Effect of nano-grain carbide formation on electrochemical behavior of 316L stainless steel. Sci Rep. 2021;11(1):12602. [30] CAO H, QIAO Y, LIU X, et al. Electron storage mediated dark antibacterial action of bound silver nanoparticles: smaller is not always better. Acta Biomater. 2013;9(2):5100-5110. [31] CALABRESE G, PETRALIA S, FRANCO D, et al. A new Ag-nanostructured hydroxyapatite porous scaffold: Antibacterial effect and cytotoxicity study. Mater Sci Eng C Mater Biol Appl. 2021;118:111394. [32] LASHIN I, FOUDA A, GOBOURI A A, et al. Antimicrobial and In Vitro Cytotoxic Efficacy of Biogenic Silver Nanoparticles (Ag-NPs) Fabricated by Callus Extract of Solanum incanum L. Biomolecules. 2021;11(3):341. [33] SOARES T, RIBEIRO D, PROENçA C, et al. Size-dependent cytotoxicity of silver nanoparticles in human neutrophils assessed by multiple analytical approaches. Life Sci. 2016;145:247-254. [34] PARK MV, NEIGH AM, VERMEULEN JP, et al. The effect of particle size on the cytotoxicity, inflammation, developmental toxicity and genotoxicity of silver nanoparticles. Biomaterials. 2011;32(36):9810-9817. [35] KOSE O, MANTECCA P, COSTA A, et al. Putative adverse outcome pathways for silver nanoparticle toxicity on mammalian male reproductive system: a literature review. Part Fibre Toxicol. 2023;20(1):1. [36] QIN H, CAO H, ZHAO Y, et al. In vitro and in vivo anti-biofilm effects of silver nanoparticles immobilized on titanium. Biomaterials. 2014;35(33): 9114-9125. |
| [1] | Shi Liu, Liang Pengchen, Chang Qing, Song Erhong. Application of machine learning in key properties of medical metal materials [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2766-2773. |
| [2] | Wang Jinlei, Li Ke, Zhao Liang. Platelet-camouflaged silver nanoparticle hydrogel accelerates wound healing in type 1 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2659-2666. |
| [3] | Ai Fangfang, Xiao Hongyan, Wang Fang, Zhu Yongzhao, Ma Lijun. Reversal effect of Lycium barbarum polysaccharide in combination with oxaliplatin on drug resistance of colon cancer stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 74-79. |
| [4] | Zhao Wei, Feng Wei, Yang Tieyi, Ren Wei, Wang Yuxin, Lyu Huicheng, Chang Zhiqiang, Feng Xiaodong, Wang Ziheng, Guo Shibing. Antibiotic bone cement intramedullary nail prepared using 3D printed mold for the treatment of long bone infection in lower limbs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1023-1030. |
| [5] | Han Tao, Hao Jianqiang, Li Wenbo, Shi Jie, Gao Qiuming. Advantages and problems of antibiotic-loaded bone cements for bone and joint infections [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 470-477. |
| [6] | Jiang Xu, Cao Fuyang, Xiong Ao, Yang Meng, Tan Jun, Yu Yang, Zhang Shaokun, Xu Jianzhong. Distribution, drug resistance, and clinical characteristics of pathogenic bacteria in 102 cases of periprosthetic joint infection [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(36): 5837-5843. |
| [7] | Cao Fei, Hui Min, Dong Xiling, Wang Le, Wang Zuxu, Zhang Min, Zhang Xiaoming, Liu Tongbin. Preparation of silver-loaded nanohydroxyapatite/polycaprolactone composite nanofiber scaffold and its osteogenic and antibacterial properties [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5461-5467. |
| [8] | Ma Rui, Wang Jialin, Wu Mengjun, Ge Ying, Wang Wei, Wang Kunzheng. Relationship of pathogenic bacteria distribution with drug resistance and treatment cycle for periprosthetic joint infection after total joint arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 380-385. |
| [9] | Guo Juan, Zheng Shan, Xie Hui, Hu Yahui. An analysis of pathogenic bacteria infection in 422 kidney transplant recipients [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(32): 5198-5202. |
| [10] | Li Shibin, Zhang Xiaoyun, Zhang Xuan. Mechanism of autophagy in osteosarcoma [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(21): 3409-3415. |
| [11] | Zhang Jiamo, Zhang Xuan, Luo Huaming, Zhao Tao, Chen Jiangchuan, Liu Juan, Wang Ke. Pathogenic distribution and drug resistance of bacterial biofilm on ureteral stent [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(16): 2556-2560. |
| [12] | Tao Hai, Guo Weichun. Regulation of osteosarcoma and stem cell-related signaling pathways [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(13): 2102-2107. |
| [13] | Lü Guodong. Effects of mixed use of different materials of implants on fracture healing [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(18): 2812-2816. |
| [14] | Bao Qiu-ping, Li Hui-min, Li Xiao-jin, Yan Yan-hong. Biological characteristics of side population cells in multiple myeloma U266 cell line in relation to drug resistance [J]. Chinese Journal of Tissue Engineering Research, 2017, 21(29): 4648-4653. |
| [15] |
Hu Xin, Wang Lei, Zhang Qian, Ning Jia, Wu Wen-meng.
Cyclic fatigue performance of different types of stainless steel root canal files
[J]. Chinese Journal of Tissue Engineering Research, 2017, 21(26): 4125-4130.
|
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||