Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (22): 3457-3463.doi: 10.12307/2024.489
Previous Articles Next Articles
Preparation and characterization of silk fibroin/bioactive glass composite fiber membrane
Wang Lu, Xu Jie, Xia Yijing, Zhang Xinsong, Zhao Bin
- School and Hospital of Stomatology of Shanxi Medical University, Shanxi Province Key Laboratory of Oral Diseases Prevention and New Materials, Taiyuan 030001, Shanxi Province, China
-
Received:2023-08-02Accepted:2023-09-25Online:2024-08-08Published:2024-01-19 -
Contact:Zhao Bin, Professor, Doctoral supervisor, School and Hospital of Stomatology of Shanxi Medical University, Shanxi Province Key Laboratory of Oral Diseases Prevention and New Materials, Taiyuan 030001, Shanxi Province, China -
About author:Wang Lu, MD, Lecturer, School and Hospital of Stomatology of Shanxi Medical University, Shanxi Province Key Laboratory of Oral Diseases Prevention and New Materials, Taiyuan 030001, Shanxi Province, China Xu Jie, Master candidate, School and Hospital of Stomatology of Shanxi Medical University, Shanxi Province Key Laboratory of Oral Diseases Prevention and New Materials, Taiyuan 030001, Shanxi Province, China -
Supported by:Shanxi Provincial Graduate Education Innovation Program (Graduate Research Innovation Project), No. 2023KY419 (to XJ); Basic Research Program of Shanxi Province (Free Exploration), No. 202203021211225 (to WL); Shanxi Province Medical Key Science and Technology Project Plan Guided Science and Technology Special Purpose, No. 2022XM56 (to WL)
CLC Number:
Cite this article
Wang Lu, Xu Jie, Xia Yijing, Zhang Xinsong, Zhao Bin. Preparation and characterization of silk fibroin/bioactive glass composite fiber membrane[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3457-3463.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
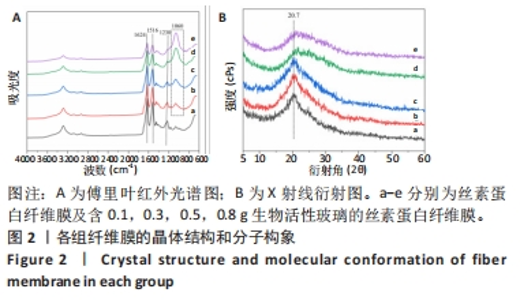
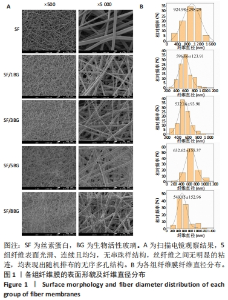
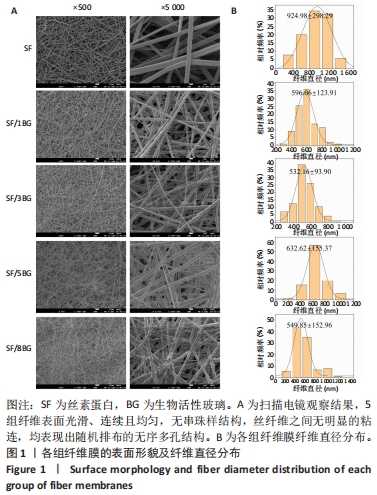
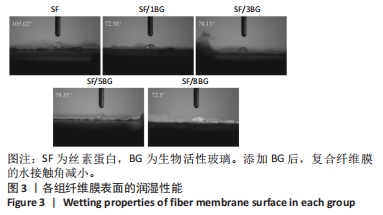
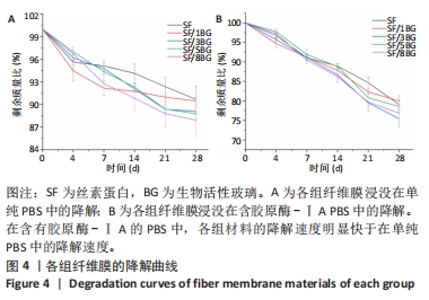
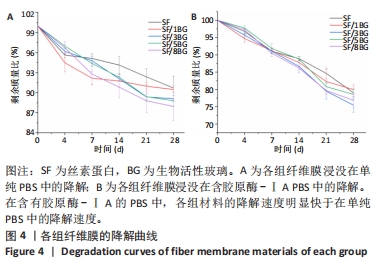
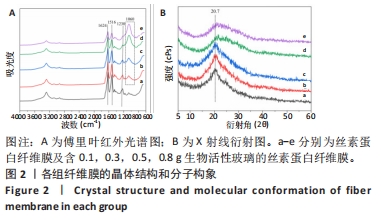
2.2 各组纤维膜结构表征结果 通过傅里叶红外光谱和X射线衍射分析纤维膜的晶体结构和分子构象,见图2。傅里叶红外光谱显示,5组纤维膜均在1 230 cm-1(酰胺Ⅲ)、1 516 cm-1(酰胺Ⅱ)、1 624 cm-1(酰胺Ⅰ)附近处显示出SF的特征峰,表明经过乙醇处理后的纤维膜表现出典型的Silk Ⅱ结构。添加BG后,可以观察到在1 060 cm-1处出现BG的特征峰(Si-O-Si振动),且随着BG含量的增加而不断增强。X射线衍射分析显示,5组纤维膜均在20.7°处出现明显的衍射峰,表现出典型的Silk Ⅱ结构,此结果与傅里叶红外光谱相一致;同时观察到随着BG含量的增加,纤维膜在15°-30°范围处出现明显的宽且弥散的无定形衍射峰,显示出典型的非晶态聚合物的衍射模式。"
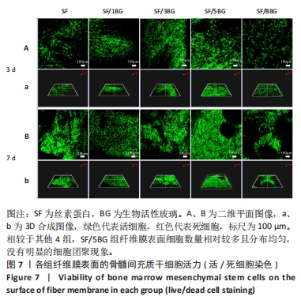
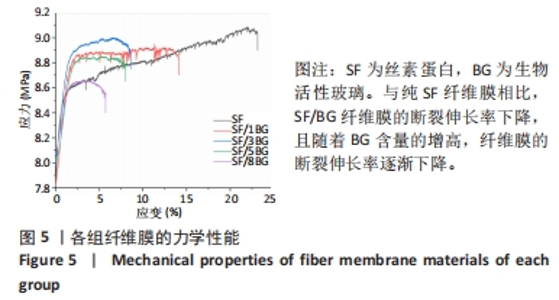
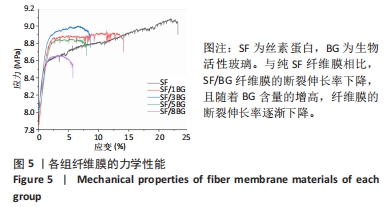
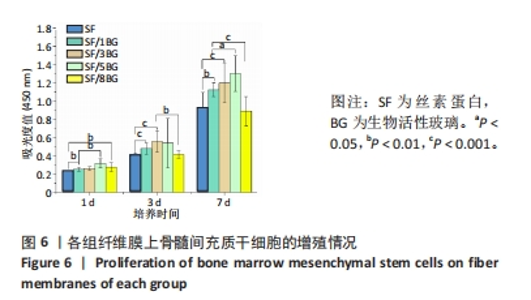
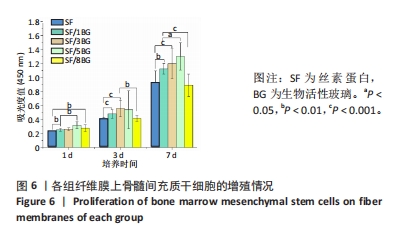
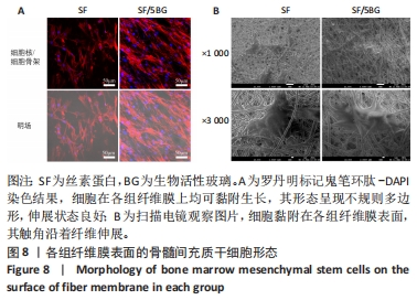
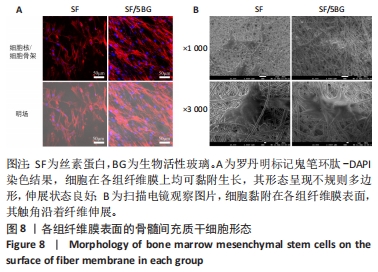
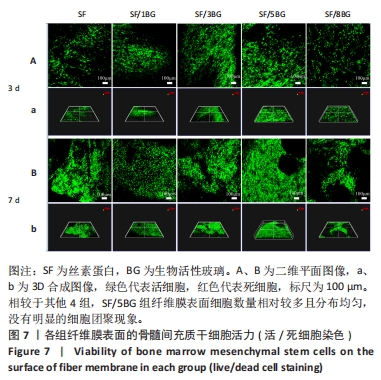
随着培养时间的延长,各组纤维膜上附着的细胞都在稳定增殖,没有表现出明显的细胞毒性。随着纤维膜中BG添加量的增加,细胞增殖吸光度值增加;然而当BG添加量过多时,细胞增殖吸光度值下降,表明添加适量BG的复合纤维膜可以促进骨髓间充质干细胞的增殖。此外还发现,相较于SF组、SF/8BG纤维膜,SF/1BG、SF/3BG、SF/5BG纤维膜均可有效促进骨髓间充质干细胞的生长,其中SF/5BG纤维膜的促细胞增殖能力最佳。 2.7 各组纤维膜对骨髓间充质干细胞活力的影响 培养3 d,各组纤维膜表面均有细胞生长,且未见明显的死细胞;培养7 d,各组纤维膜表面的细胞数量增多,表明材料对于细胞生长无明显毒性作用,具有良好的生物相容性,见图7。相较于其他4组,SF/5BG组纤维膜表面细胞数量相对较多且分布均匀,没有明显的细胞团聚现象,表明添加一定量的BG有利于细胞增殖,该结果与CCK-8检测结果一致。此外,还可见各组纤维膜上的细胞具有一定的形态,呈现多边形形状,且有明显的伪足生长,表明所制备的纤维膜可以有效促进细胞的黏附、生长和增殖。"
| [1] APRILE P, LETOURNEUR D, SIMON-YARZA T. Membranes for Guided Bone Regeneration: A Road from Bench to Bedside. Adv Healthc Mater. 2020;9(19):e2000707. [2] REN Y, FAN L, ALKILDANI S, et al. Barrier Membranes for Guided Bone Regeneration (GBR): A Focus on Recent Advances in Collagen Membranes. Int J Mol Sci. 2022;23(23):14987. [3] LIANG H, YIN J, MAN K, et al. A long-lasting guided bone regeneration membrane from sequentially functionalised photoactive atelocollagen. Acta Biomater. 2022;140:190-205. [4] LI Z, DU T, GAO C, et al. In-situmineralized homogeneous collagen-based scaffolds for potential guided bone regeneration. Biofabrication. 2022;14(4):10.1088/1758-5090/ac8dc7. [5] QIAN S, ZHAO B, MAO J, et al. Biomedical applications of Janus membrane. Biomed Technol. 2023;2:58-69. [6] 赵文俊,陈宇.引导组织/骨再生牙周功能梯度膜的研究进展[J].国际口腔医学杂志,2021,48(4):391-397. [7] LIN D, CHAI Y, MA Y, et al. Rapid initiation of guided bone regeneration driven by spatiotemporal delivery of IL-8 and BMP-2 from hierarchical MBG-based scaffold. Biomaterials. 2019;196:122-137. [8] ZHOU Z, CUI J, WU S, et al. Silk fibroin-based biomaterials for cartilage/osteochondral repair. Theranostics. 2022;12(11):5103-5124. [9] WANG Z, LIN M, XIE Q, et al. Electrospun silk fibroin/poly(lactide-co-ε-caprolactone) nanofibrous scaffolds for bone regeneration. Int J Nanomedicine. 2016;11:1483-1500. [10] BRAUER DS. Bioactive glasses-structure and properties. Angew Chem Int Ed Engl. 2015;54(14):4160-4181. [11] GUPTA S, MAJUMDAR S, KRISHNAMURTHY S. Bioactive glass: A multifunctional delivery system. J Control Release. 2021;335:481-497. [12] VERMEULEN S, TAHMASEBI BIRGANI Z, HABIBOVIC P. Biomaterial-induced pathway modulation for bone regeneration. Biomaterials. 2022;283:121431. [13] 吕孝帅,李正茂,杨雪超.生物活性玻璃58S/丝素蛋白膜促进人牙髓干细胞增殖与分化[J].牙体牙髓牙周病学杂志,2015,25(10): 577-583. [14] FENBO M, XINGYU X, BIN T. Strontium chondroitin sulfate/silk fibroin blend membrane containing microporous structure modulates macrophage responses for guided bone regeneration. Carbohydr Polym. 2019;213:266-275. [15] WUBNEH A, TSEKOURA EK, AYRANCI C, et al. Current state of fabrication technologies and materials for bone tissue engineering. Acta Biomater. 2018;80:1-30. [16] XING J, ZHANG M, LIU X, et al. Multi-material electrospinning: from methods to biomedical applications. Mater Today Bio. 2023;21:100710. [17] FENG X, LI J, ZHANG X, et al. Electrospun polymer micro/nanofibers as pharmaceutical repositories for healthcare. J Control Release. 2019; 302:19-41. [18] PENG S, JIN G, LI L, et al. Multi-functional electrospun nanofibres for advances in tissue regeneration, energy conversion & storage, and water treatment. Chem Soc Rev. 2016;45(5):1225-1241. [19] SANTOS MS, CARVALHO MS, SILVA JC. Recent Advances on Electrospun Nanofibers for Periodontal Regeneration. Nanomaterials (Basel). 2023; 13(8):1307. [20] DENCHAI A, TARTARIN D, MELE E. Cellular Response to Surface Morphology: Electrospinning and Computational Modeling. Front Bioeng Biotechnol. 2018;6:155. [21] CHI J, WANG M, CHEN J, et al. Topographic Orientation of Scaffolds for Tissue Regeneration: Recent Advances in Biomaterial Design and Applications. Biomimetics (Basel). 2022;7(3):131. [22] XUE J, WU T, DAI Y, et al. Electrospinning and Electrospun Nanofibers: Methods, Materials, and Applications. Chem Rev. 2019;119(8): 5298-5415. [23] ORASH MAHMOUD SALEHII A, NOURBAKHSH MS, RAFIENIA M, et al. Corneal stromal regeneration by hybrid oriented poly (ε-caprolactone)/lyophilized silk fibroin electrospun scaffold. Int J Biol Macromol. 2020; 161:377-388. [24] KAUSHIK S, THUNGON PD, GOSWAMI P. Silk Fibroin: An Emerging Biocompatible Material for Application of Enzymes and Whole Cells in Bioelectronics and Bioanalytical Sciences. ACS Biomater Sci Eng. 2020;6(8):4337-4355. [25] XU C, XIA Y, WANG L, et al. Polydopamine-assisted immobilization of silk fibroin and its derived peptide on chemically oxidized titanium to enhance biological activity in vitro. Int J Biol Macromol. 2021;185: 1022-1035. [26] SINGH BN, PRAMANIK K. Generation of bioactive nano-composite scaffold of nanobioglass/silk fibroin/carboxymethyl cellulose for bone tissue engineering. J Biomater Sci Polym Ed. 2018;29(16):2011-2034. [27] KIM OH, PARK JH, SON JI, et al. Bone Marrow Mesenchymal Stromal Cells on Silk Fibroin Scaffolds to Attenuate Polymicrobial Sepsis Induced by Cecal Ligation and Puncture. Polymers (Basel). 2021;13(9):1433. [28] BHARADWAZ A, JAYASURIYA AC. Recent trends in the application of widely used natural and synthetic polymer nanocomposites in bone tissue regeneration. Mater Sci Eng C Mater Biol Appl. 2020;110:110698. [29] BENTO R, GADDAM A, FERREIRA JMF. Sol-Gel Synthesis and Characterization of a Quaternary Bioglass for Bone Regeneration and Tissue Engineering. Materials (Basel). 2021;14(16):4515. [30] CRUSH J, HUSSAIN A, SEAH KTM, et al. Bioactive Glass: Methods for Assessing Angiogenesis and Osteogenesis. Front Cell Dev Biol. 2021;9: 643781. [31] El-RASHIDY AA, ROETHER JA, HARHAUS L, et al. Regenerating bone with bioactive glass scaffolds: A review of in vivo studies in bone defect models. Acta Biomater. 2017;62:1-28. [32] CHENG J, JUN Y, QIN J, et al. Electrospinning versus microfluidic spinning of functional fibers for biomedical applications. Biomaterials. 2017;114:121-143. [33] SUN W, GREGORY DA, TOMEH MA, et al. Silk Fibroin as a Functional Biomaterial for Tissue Engineering. Int J Mol Sci. 2021;22(3):1499. [34] WANG L, ZHANG Y, XIA Y, et al. Photocross-linked silk fibroin/hyaluronic acid hydrogel loaded with hDPSC for pulp regeneration. Int J Biol Macromol. 2022;215:155-168. [35] MORAN MC, RUANO G, CIRISANO F, et al. Mammalian cell viability on hydrophobic and superhydrophobic fabrics. Mater Sci Eng C Mater Biol Appl. 2019;99:241-247. [36] CIRALDO FE, BOCCARDI E, MELLI V, et al. Tackling bioactive glass excessive in vitro bioreactivity: Preconditioning approaches for cell culture tests. Acta Biomater. 2018;75:3-10. [37] RAHAMAN MN, DAY DE, BAL BS, et al. Bioactive glass in tissue engineering. Acta Biomater. 2011;7(6):2355-2373. [38] ZHAO T, ZHANG J, GAO X, et al. Electrospun nanofibers for bone regeneration: from biomimetic composition, structure to function. J Mater Chem B. 2022;10(32):6078-6106. [39] LUO X, ZHANG S, LUO B, et al. Engineering collagen fiber templates with oriented nanoarchitecture and concerns on osteoblast behaviors. Int J Biol Macromol. 2021;185:77-86. [40] WANG X, PENG Y, WU Y, et al. Chitosan/silk fibroin composite bilayer PCL nanofibrous mats for bone regeneration with enhanced antibacterial properties and improved osteogenic potential. Int J Biol Macromol. 2023;230:123265. |
| [1] | Wang Jianchun, Yang Shuqing, Su Xin, Wang Hongyuan. Different contents of B2O3 affect mechanical properties and bioactivity of bioactive glass scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 712-716. |
| [2] | Xu Yinghua, Liu Jing, You Quan, Wen Zhihao, Gao Lu. Effect of neodymium-doped:yttrium aluminum perovskite laser combined with two kinds of remineralizers on remineralization of early enamel caries [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 360-365. |
| [3] | Gu Mingxi, Wang Changcheng, Tian Fengde, An Ning, Hao Ruihu, Guo Lin. Preparation and in vitro evaluation of a three-dimensional porous cartilage scaffold made of silk fibroin/gelatin/chitosan [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 366-372. |
| [4] | Guo Qingxia, Wang Yue, Wu Tong. Silk fibroin methacryloyl hydrogel loaded with Scriptaid regulates polarization of microglia cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(29): 4628-4633. |
| [5] | Gao Hua, Che Hui, Hu Dan, Hao Yuefeng. Bioactive glass: different application forms and functions by adjusting preparation process and doping elements [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(29): 4726-4733. |
| [6] | Zhou Zhi, Chen Zhijie, Huo Shicheng, Li Zhanchun. Strontium-containing mesoporous bioactive glass nanoparticles loaded with bisphosphonates ameliorate bone loss in ovariectomized mice [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2653-2658. |
| [7] | Li Xiaoyin, Yang Xiaoqing, Chen Shulian, Li Zhengchao, Wang Ziqi, Song Zhen, Zhu Daren, Chen Xuyi. Collagen/silk fibroin scaffold combined with neural stem cells in the treatment of traumatic spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 890-896. |
| [8] | Wang Ziqi, Li Xiaoyin, Jiang Jipeng, Song Zhen, Li Zhengchao, Chen Shulian, Chen Xuyi. Collagen/silk fibroin scaffold combined with human umbilical cord-derived mesenchymal stem cells in the treatment of traumatic brain injury in dogs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(34): 5483-5490. |
| [9] | He Fan, Shan Xianfeng, Xiong Xiuli, Wang Xuejin. Biocompatibility and in vitro osteogenic differentiation of alginates/chitosan/58S bioglass composite membrane for guided bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(25): 3984-3991. |
| [10] | Yu Lanning, Wang Qian, Jin Youshi, Fei Xiaowen, Wang Qingshan. Sealing effect of nano hydroxyapatite on dentinal tubules [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(25): 3964-3970. |
| [11] | Liu Jichao, Zhao Jinlong, Yu Yang. Silk fibroin collagen composite scaffold combined with platelet-rich plasma for repairing skin injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(25): 3971-3976. |
| [12] | Chen Jiana, Nie Minhai, Hu Xinyue, Liu Xuqian. Platelet-derived growth factor BB promotes proliferation and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(10): 1534-1540. |
| [13] | Tan Guozhong, Tu Xinran, Guo Liyang, Zhong Jialin, Zhang Yang, Jiang Qianzhou. Biosafety evaluation of three-dimensional printed gelatin/sodium alginate/58S bioactive glass scaffolds for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 521-527. |
| [14] | He Guanyu, Xu Baoshan, Du Lilong, Zhang Tongxing, Huo Zhenxin, Shen Li. Biomimetic orientated microchannel annulus fibrosus scaffold constructed by silk fibroin [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 560-566. |
| [15] | Kang Kunlong, Wang Xintao. Research hotspot of biological scaffold materials promoting osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 597-603. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||