Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (21): 3361-3367.doi: 10.12307/2023.136
Previous Articles Next Articles
Role of chitosan and its derivatives in the field of oral antibacterial
Liu Zhuoran1, Li Yumei2, Liu Junyan2, Yin Tong1, Jiang Ming3, Li Yourui1
- 1Department of Prosthodontics, Affiliated Hospital of Binzhou Medical College, Binzhou 256600, Shandong Province, China; 2Binzhou Medical College, Yantai 264000, Shandong Province, China; 3First Department of Endodontic Disease, Jinan Stomatological Hospital, Jinan 250000, Shandong Province, China
-
Received:2022-04-18Accepted:2022-05-18Online:2023-07-28Published:2022-11-24 -
Contact:Li Yourui, MD, Associate chief physician, Department of Prosthodontics, Affiliated Hospital of Binzhou Medical College, Binzhou 256600, Shandong Province, China Jiang Ming, MD, Associate chief physician, First Department of Endodontic Disease, Jinan Stomatological Hospital, Jinan 250000, Shandong Province, China -
About author:Liu Zhuoran, Master candidate, Department of Prosthodontics, Affiliated Hospital of Binzhou Medical College, Binzhou 256600, Shandong Province, China -
Supported by:Shandong Medical and Health Science and Technology Development Plan Project, No. 2017WS753 (to LYR); Scientific Research Program and Research Start-up Fund Project of Binzhou Medical College, No. BY2016KJ06 (to LYR)
CLC Number:
Cite this article
Liu Zhuoran, Li Yumei, Liu Junyan, Yin Tong, Jiang Ming, Li Yourui. Role of chitosan and its derivatives in the field of oral antibacterial[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3361-3367.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

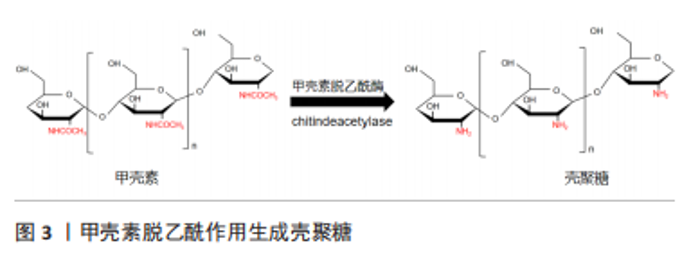
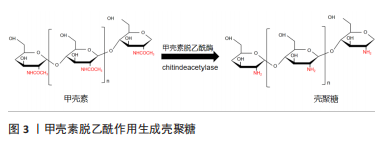
工业上通常采用浓碱煮沸加热除去甲壳素中大部分的乙酰基团来制备壳聚糖。壳聚糖的脱乙酰度是用D-氨基与D-氨基和N-乙酰D-氨基之和的比值来计算的。一般来说,脱乙酰度达到55%的甲壳素被称为壳聚糖,而脱乙酰度超过70%的壳聚糖是有使用价值的工业品[10]。壳聚糖随着脱乙酰度的增加,其溶解性得到改善,但其分子内与分子间较强的氢键使壳聚糖溶液的黏度增加,从而导致壳聚糖只能溶于各类有机酸和稀的无机酸,不易溶解在中性或碱性溶剂中,这极大限制了壳聚糖的应用[11]。 2.1.2 壳聚糖衍生物的分类 壳聚糖具有的官能团[氨基(-NH2)和羟基(-OH)]具有一定的化学活性,可对其进行改性或通过与其他高聚物进行共聚等方法改善其水溶性、机械强度以获得更加广泛的抗菌应用[12]。 酰化改性是壳聚糖最常见的改性方法。酰化反应破坏了壳聚糖分子内和分子间的氢键,使其结晶度减弱,水溶性增强。O-酰化壳聚糖通常用于纤维或聚合材料的薄膜中,以增强材料的疏水性和抗菌性[13]。N-酰化壳聚糖可用作药物输送中的载体或缓释剂,也可用作生物支架中的材料添加剂[14]。将烷基引入壳聚糖中,使其具有显著减弱的分子间氢键,从而改善其溶解性。N-烷基化壳聚糖可以由卤代烷制备,烷基化壳聚糖因其凝结和抗菌性能可用于制备医用材料[15]。羧化壳聚糖具有良好的水溶性,还具有比壳聚糖更好的增稠、成膜等性能。因此羧化壳聚糖在生物医学和制药领域非常活跃,具有促进伤口愈合的抗菌作用,以及抗病毒、抗肿瘤和抗凝血等作用[16]。季铵盐基是亲水基团,带正电荷,该基团的引入不仅改善了溶解性,还使季铵盐壳聚糖具有良好的抗菌性、生物相容性和无毒性,以及天然的黏附性和穿透黏液层并与上皮表面结合的能力,因此其可用于抗炎药物或用作伤口敷料材料的填充纤维[17]。 2.2 壳聚糖及其衍生物的抗菌机制 壳聚糖及其衍生物具有较好的抗菌活性,可抑制真菌、细菌、病毒等微生物的繁殖,其抗菌性能和抗菌机制一直是研究热点,但到目前为止具体的抗菌机制尚不清楚。文章对近几年来国内外壳聚糖抗菌机制研究方面的文章进行梳理,归纳了3种可能的抗菌机制[18-27],见表2。"

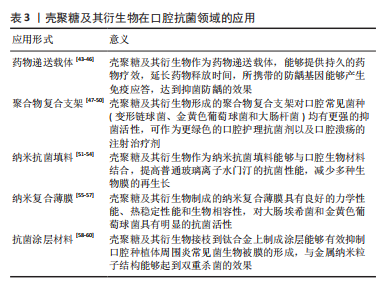
2.2.1 静电作用 目前最认同的抗菌模型是壳聚糖C-2位上的氨基,在pH < 6时带正电荷,而细菌的细胞表面带负电荷,正负电荷的相互作用,发生静电作用,改变了细胞的通透性,使壳聚糖在细菌细胞表面形成一层复合物屏障膜,从而阻碍细菌生长所必需的营养物质的吸收[18]。 一些研究表明,增加壳聚糖上-NH3+的正电荷数量会使其更强地结合到细菌的细胞壁上,从而显示出更高的活性。这种相互作用可能会在细胞周围形成一层致密膜,封闭了菌体表面的营养物质或代谢废物交换的通道,进而抑制了菌体生长[19]。SAITO等[20]研究了壳聚糖偶联物对铜绿假单胞菌和耐甲氧西林金黄色葡萄球菌等菌体的抑菌活性,结果表明分子质量较低的壳聚糖可以通过静电作用穿过细胞膜,进入病原体细胞的内部增加细菌外膜和质膜的通透性,导致膜破裂和细胞内内容物的释放,从而导致细菌细胞死亡。 2.2.2 抑制RNA和蛋白质的合成 有研究表明,低分子质量的壳聚糖可以进入病原体细胞内部,可能与病原体细胞内部的DNA或RNA结合,从而起到抑制病原体生长或杀死病原菌的作用[21]。 AMIDI等[22]总结了壳聚糖蛋白质治疗以及抗原输送系统的机制,发现高分子质量壳聚糖分子解离后的溶液可以与细胞膜结合,改变其通透性,低分子质量壳聚糖分子的解离溶液在穿透细胞核的同时可以与DNA结合,可能会阻碍其转录,从而影响DNA向大肠杆菌的转化过程。SHIH等[23]通过筛选突变库来揭示壳聚糖对于白色念珠菌的抗真菌机制,结果表明壳聚糖分子能够穿过由多层胞壁质组成的细胞壁到达质膜,抑制细胞壁相关基因表达(如ALS2,PGA45和ACE2)以及转运蛋白基因的表达,阻碍蛋白质的合成,从而降低细胞表面对于壳聚糖的防御作用,起到一定的抑菌作用。 2.2.3 金属离子螯合作用 壳聚糖及其衍生物分子中有-OH和-NH2,从构象上看,它们都是平伏键,但在一定的pH值条件下,这种特殊结构使它们对一定离子半径的金属离子具有螯合作 用[24]。Ca2+,Mg2+和Zn2+等金属离子是细胞代谢所需的重要部分,壳聚糖及其衍生物能够借此干扰细胞代谢(如毒素的合成),从而达到抑菌的目的[25]。 WEIPFLOG等[26]研究了脱乙酰度在80%的壳聚糖对于重金属离子的螯合作用,结果发现壳聚糖对于微量金属元素(包括:Cu2+,Fe2+,Zn2+,Mg2+等)有均匀的吸附作用,分析壳聚糖可以与病原体细胞中的金属离子以及营养物质中的微量元素螯合,从而抑制细胞的生长繁殖。壳聚糖结合金属离子的能力主要与分子结构中未质子化的氨基有关,羧酸甲基化增强了壳聚糖对金属离子的吸附能力。同时,一些研究成果表明,当壳聚糖溶解于酸性溶液中时,绝大部分氨基都发生质子化,壳聚糖的抑菌效果是降低的,此时螯合作用不再是抑菌的主导机制[27]。 2.3 影响壳聚糖抗菌性能的主要因素 ARDEAN等[28]认为壳聚糖的抗菌活性和作用模式的差异与多种因素相关,这些因素主要包括:微生物来源和壳聚糖的内在因素(如:脱乙酰度及浓度、分子质量和聚合度等);此外,不同来源的壳聚糖和不固定的特定环境因素(如温度和pH值等)在很大程度上也会影响壳聚糖的抗菌能力。因此,下文将重点讨论各种因素对壳聚糖抗菌活性的影响。 2.3.1 壳聚糖的来源 壳聚糖由甲壳素脱乙酰化得到,广泛存在于甲壳素类动物、昆虫、真菌和藻类等物质中,是地球上仅次于纤维素的最丰富的多糖类。Byun等[29]研究表明,蟹腿壳与蟹全壳制备的壳聚糖在理化和功能特性上存在较大差异,与蟹腿制备的壳聚糖相比,蟹壳制备的壳聚糖氮含量、脱乙酰度显著提高,抗菌活性提高。昆虫(六足类)也是甲壳素的一个很有前途的来源。由于昆虫的繁殖率、繁殖的简便性以及对生态系统变化的抵抗力使得来自昆虫的壳聚糖材料的产量也高于甲壳类动物。通过比较美洲大蠊为原料制备的壳聚糖膜和虾壳聚糖膜发现其具有更好的抗菌和抗氧化性能并显示出优异的物理性能[30]。 2.3.2 壳聚糖的相对分子质量 壳聚糖的抗菌活性随其相对分子质量的不同而对细菌的抑制活性机制不同[31]。在一些研究中提出,杀菌效果需要最小相对分子质量为10 000的壳聚糖,低分子质量(相对分子质量< 50 000)壳聚糖分子的解离溶液在穿透细胞核的同时可以与DNA结合,抑制mRNA的合成[32]。LI 等[33]研究了壳聚糖纤维对大肠杆菌、金黄色葡萄球菌和白色念珠菌的抑菌活性,结果表明,壳聚糖纤维对金黄色葡萄球菌的抑制率随分子质量的增加先升高后降低,在脱乙酰度相近的条件下,对大肠杆菌的杀菌活性随分子质量的增加而降低。这归因于壳聚糖颗粒的大小和构象,低分子质量壳聚糖小链的迁移率、引力和离子相互作用比大链的更容易[34]。 2.3.3 壳聚糖的脱乙酰度与浓度 随着壳聚糖溶液浓度的提高,溶液中游离氨基的含量也增加,其抗菌效果也增大。通常壳聚糖抑制细菌生长所需的浓度与壳聚糖的乙酰化程度有关;乙酰化度为7.5%的溶液比乙酰化度为15%的溶液更有效[35]。在较低的浓度下,壳聚糖才能结合到带负电荷的细胞表面,特别是革兰阴性菌,因此它会干扰细胞膜的渗透性;然而在较高的浓度下,由于氨基而带正电荷的壳聚糖可能覆盖在细胞表面,而细胞内的成分被堵塞在细胞内[36]。随着脱乙酰度的提高,壳聚糖对金黄色葡萄球菌(S.aureus)的抑制效果不断提高。BYUN等[29]研究发现脱乙酰度为81.56%的壳聚糖对金黄色葡萄球菌(S.aureus)的抑菌效果远远优于脱乙酰为62.71%的壳聚糖。 2.3.4 环境pH值及温度 许多研究表明壳聚糖的抗菌活性随溶液酸性的增加而增强[37]。壳聚糖在pH < 6时具有聚阳离子性质,随着pH的降低,壳聚糖在细菌表面的吸附量增加,而当溶液pH 6.5-7.0时,由于壳聚糖的氨基质子化程度极低因而几乎没有抑菌活性[38]。NO等[39]研究了不同pH值下壳聚糖对大肠杆菌(Escherichia Coli)、芽孢杆菌(Bacillus megaterium)和金黄色葡萄球菌(Staphylococcus aureus)的抑菌作用,结果发现:壳聚糖的抗菌活性受 pH 值的反向影响,随着pH值的降低(pH < 6)壳聚糖的抑菌效果逐渐提高。温度对壳聚糖的抗菌活性有很大影响,ARDILA等[40]研究了在37 ℃、20 ℃和7 ℃下壳聚糖溶液对鼠伤寒沙门氏菌(Salmonella typhimurium)的抑菌效果,结果为37 ℃时抑菌效果最好,20 ℃次之,这主要是因为温度会影响壳聚糖与细胞的反应速率和细胞表面可结合位点数目,较低的温度会改变细胞的表面结构、减少壳聚糖的表面结合位点。 2.3.5 其他因素 壳聚糖本身的抗菌活性全部归于氨基,而氨基直接受到脱乙酰度的影响。由于革兰阳性和革兰阴性细胞壁的组成不同,壳聚糖与这2种细菌的相互作用也不同。在一些研究中,研究人员发现,氨基对细胞壁中阴离子自由基的亲和力较高,因此对革兰阴性菌的杀菌效果比对革兰阳性菌的杀菌效果更好[41]。但在其他研究中,由于革兰阴性外膜屏障的存在,认为革兰阳性菌对壳聚糖的抗菌活性更为敏感。CHUNG等[42]通过研究发现短碳链长度的有机酸溶剂增加壳聚糖的抗菌活性比无机酸溶剂好,同时添加EDTA能增强对大肠杆菌的抗菌活性,但降低了对金黄色葡萄球菌的抗菌活性。 2.4 壳聚糖及其衍生物在口腔抗菌领域的应用 口腔是一个复杂的微生物环境,其中微生物生态失调会引起不同的口腔常见疾病,如口腔溃疡、牙周炎、牙龈炎以及种植体周围炎等。在这些口腔疾病的治疗过程中,抑制相关细菌的生长是非常重要的一个环节。 2.4.1 药物递送载体 近年来,随着纳米技术的发展,壳聚糖衍生物已被制备成纳米材料,并将其广泛应用于医学材料和生物医学领域[43]。壳聚糖及其衍生物既可作为药物的靶向给药载体,也可作为疫苗的佐剂使用。这些研究也为解决口腔细菌引发的问题提供了新思路。 口腔黏膜给药系统由于唾液、口腔运动和非自主吞咽的不断变化阻碍了药物的牢固黏附,因此药物在口腔黏膜上的滞留时间仍然有限。RYU等[44]开发了一种黏附性多糖口腔贴片,称为“Chittal”,它利用了耐湿贻贝黏附性的化学原理,壳聚糖作为贴片药物的载体,与唾液和嘴唇黏液层接触后立即与黏液界面的黏蛋白形成不溶的黏附层;而后,壳聚糖在共价交联和物理缠结的协同作用下转化为黏附性水凝胶,其黏附性为药物提供了持久的治疗效果,从而促进了口腔溃疡的愈合。 DUBASHYNSKAYA等[45]以透明质酸和水溶性阳离子壳聚糖衍生物二乙氨基乙基壳聚糖为载体,建立黏菌素给药系统。体外释放实验表明,与纯黏菌素相比,所开发的药物释放系统提供了一两小时的黏菌素缓释,提示该给药系统可用于治疗由革兰阴性多重耐药微生物引起的感染。杨梦璐[46]利用壳聚糖作为载体的主要成分,制备壳聚糖/海藻酸钠pH敏感型复合载体包覆防龋基因疫苗,体外实验结果表明该载体展现了对pH敏感的缓释效能,对基因疫苗在低pH值环境下展现了一定的保护性能,能携带防龋基因疫苗引起免疫应答,产生抑菌效果。 2.4.2 聚合物复合支架 口腔临床上现在常用的抗菌剂为洗必泰,但其容易使牙或者修复体上着色。PHUANGKAEW等[47]以N-[(2-羟基-3-三甲基铵)丙基]疏水壳聚糖、十二烷基壳聚糖和邻苯二甲酰化壳聚糖为原料,与缩水甘油三甲基氯化铵反应,合成了两亲性季铵盐壳聚糖衍生物并评价了它们对变形链球菌的抗菌效果,己基取代度为30%的季铵盐壳聚糖水溶性最好,其最低抑菌浓度和最低杀菌浓度均优于未改性壳聚糖,它的抗生物被膜效果与洗必泰相当,表明它可以作为一种更绿色的口腔护理制剂的抗菌剂。PIEGAT等[48]用微量稀释法证明了N,O-酰化壳聚糖衍生物显著减少了不同浓度的活细菌细胞(金黄色葡萄球菌、大肠杆菌和幽门螺杆菌)数量,与壳聚糖相比,N,O-酰化壳聚糖衍生物具有更强的抗菌活性。衍生物的两亲性导致了该衍生物与黏蛋白之间强烈的相互作用,提示了N,O-酰化壳聚糖衍生物在抗菌应用中的巨大潜力。 LUO等[49]研制出了一种可注射的热凝性壳聚糖接枝聚N-异丙基丙烯酰胺聚合物,在生理温度下可以可逆地形成半固体凝胶,便于口腔溃疡部位的注射治疗。同时实验结果证明,此类壳聚糖聚合物的质量分数为5.0%时对大肠杆菌和金黄色葡萄球菌的抑制率最高可达90%;除此之外,其能够缩短大鼠口腔溃疡的愈合时间,有望成为治疗口腔黏膜溃疡的注射治疗剂。70%的慢性感染涉及病原菌生物被膜,且其对抗生素的耐药性增加。WANG等[50]利用N-(2-羟基)丙基-3-三甲基壳聚糖氯化铵检测其对与根管感染相关的粪肠球菌的抗菌作用,结果表明,季铵盐壳聚糖在无菌双蒸水中对粪肠球菌生物被膜有显著的抑菌作用,提示其在根管消毒和根管充填方面具有潜在的应用前景。 2.4.3 纳米抗菌填料 牙髓感染是一种生物被膜疾病,目前的治疗方案很难根治,因此,持续的微生物可能导致持续或复发的疾病。使用增强型填充材料来修饰生物膜再生的潜力是一种很有前途的策略。ABUSREWIL等[51]评价了壳聚糖改性硅酸钙生物材料的抗生物被膜效果,探讨了ProRootMTA,Biodentine和牛牙本质盘上单菌种和多菌种生物膜的形成。实验结果表明只有与壳聚糖结合的生物牙本质显示出剂量依赖性地减少多物种生物膜的再生长,在壳聚糖存在的情况下,跨界生物膜显示出增强了细菌的耐受性。这项研究证明了提高生物牙本质抗菌性能的潜力。 在临床口腔修复中,义齿性口炎是最常见的疾病之一。WALCZAK等[52]通过壳聚糖-盐改性聚甲基丙烯酸甲酯[poly (methacrylate),PMMA],研制一种新型的抗真菌义齿基托材料,并对标本进行了白色念珠菌生物被膜实验。结果表明尽管在PMMA中加入壳聚糖-盐显著增加了样品的表面粗糙度,但用壳聚糖-盐对PMMA树脂进行改性并不能改善PMMA义齿基托材料的抗真菌性能。但DE MORI等[53]用银纳米线和甲基丙烯酰壳聚糖配制新型PMMA黏结剂,结果证明能够降低聚合温度和生物膜的形成。传统的玻璃离子水门汀是目前最常用的牙科修复材料之一,但其机械强度较低,限制了其应用。IBRAHIM等[54]用不同体积分数(5%,10%,25%,50%)的壳聚糖溶液加入玻璃离子水门汀溶液中,同时对其抗菌性能进行测定。结果显示在玻璃离子水门汀的聚丙烯酸溶液中以5%-10%的体积比加入壳聚糖酸性溶液,在不影响其与牙本质表面黏附性的情况下,提高了普通玻璃离子水门汀的抗变形链球菌的抗菌性能。 2.4.4 纳米复合薄膜 慢性牙周炎是牙菌斑引起的牙周支持组织的慢性炎症,也是成人失牙的主要原因之一。ZHANG等[55]用不同尺寸的氧化锌颗粒分别加入到壳聚糖复合膜中,制备了壳聚糖复合膜。根据壳聚糖膜和壳聚糖/纳米氧化锌复合膜对大肠杆菌和金黄色葡萄球菌的抗菌实验,壳聚糖复合膜具有较好的抗菌活性,含0.3%尺寸为50 nm的氧化锌粒子的薄膜具有最好的抑菌率,表明较小尺寸的纳米氧化锌粒子具有更好的抑菌活性,作为抗菌添加剂具有潜在的应用前景。 YIN等[56]以季铵盐改性羧甲基壳聚糖、聚乙烯醇和五水硫酸铜为原料,通过溶液浇铸和离子吸附工艺制备了抗菌季铵化羧甲基壳聚糖/聚乙烯醇/铜共混膜。检测结果表明负载铜离子后,材料的力学性能和热性能都有所提高;通过负载Cu2+,共混膜表现出良好的抗菌活性,约98.3%的金黄色葡萄球菌和99.9%的大肠杆菌可在60 min内灭活;对细胞毒性进行了研究,结果表明所制备的膜具有良好的细胞活性和生物相容性。ZHANG等[57]以羧甲基壳聚糖为载体,采用水乳液共聚法合成了一种新型的水性聚氨酯-明胶水解物水凝胶膜。一系列的测试表明该水凝胶膜具有良好的热稳定性、溶胀性和可控的生物降解性,纸片扩散试验表明,该水凝胶膜对大肠埃希菌和金黄色葡萄球菌具有明显的抗菌活性。 2.4.5 抗菌涂层材料 随着口腔种植技术的发展与普及,口腔种植体周围炎也时常发生。CAMACHO-ALONSO等[58]评价了利用光动力疗法和壳聚糖对种植体周围炎常见菌(如金黄色葡萄球菌、大肠埃希菌和铜绿假单胞菌)钛附着生物膜可能的协同抗菌作用,该研究通过测定钛板上细胞集落形成单位以确定抑菌效果,结果表明光动力疗法与壳聚糖联合应用对与种植体周围炎密切相关的细菌有协同抗菌作用。 LIU等[59]制备了聚乙二醇化壳聚糖修饰的银纳米粒子(PEG-g-CS@AgNPs),研究了该银纳米粒子对革兰阴性大肠埃希菌和革兰阳性金黄色葡萄球菌的抑菌活性,其最小抑菌浓度远低于壳聚糖和聚乙二醇接枝壳聚糖。聚乙二醇接枝壳聚糖@AgNPs涂层优异的抗菌性能归功于聚乙二醇化抗菌与放菌的结合,以及壳聚糖与银离子释放的双重杀菌作用。D’Almeida等[60]以丙基丁二酸三乙氧基硅酯为偶联剂,采用共价键两步法将壳聚糖接枝到钛合金上制成涂层。微生物学试验表明,该涂层对大肠埃希菌和金黄色葡萄球菌有较好的抗菌活性并有效地抑制了生物被膜的形成。 文章最后总结了壳聚糖及其衍生物作为新型纳米生物材料能够以不同方式应用于口腔抗菌领域的研究,见表3。 "
| [1] VALM AM. The structure of dental plaque microbial communities in the transition from health to dental caries and periodontal disease. J Mol Biol. 2019;431(16):2957-2969. [2] BIHAREE A, SHARMA A, KUMAR A, et al. Antimicrobial flavonoids as a potential substitute for overcoming antimicrobial resistance. Fitoterapia. 2020;146:104720. [3] KULKARNI N, SHINDE SD, JADHAV GS, et al. Peptide-chitosan engineered scaffolds for biomedical applications. Bioconjug Chem. 2021;32(3):448-465. [4] YU D, FENG J, YOU H, et al. The microstructure, antibacterial and antitumor activities of chitosan oligosaccharides and derivatives. Mar Drugs. 2022;20(1):69. [5] ALLAN C, ADNIGER LA. The fungicidal effect of chitosan on fungi of varying cell wall composition. Exp Mycol. 1979;3:285-287. [6] WU YB, YU SH, MI FL, et al. Preparation and characterization on mechanical and antibacterial properties of chitsoan/cellulose blends. Carbohydrate Polymers. 2004;57(4):435-440. [7] SENTHIL KUMAR R, RAVIKUMAR N, KAVITHA S, et al. Nanochitosan modified glass ionomer cement with enhanced mechanical properties and fluoride release. Int J Biol Macromol. 2017;104(Pt B):1860-1865. [8] LIN MH, WANG YH, KUO CH, et al. Hybrid ZnO/chitosan antimicrobial coatings with enhanced mechanical and bioactive properties for titanium implants. Carbohydr Polym. 2021;257:117639. [9] 崔升,袁美玉,付俊杰,等.抗菌用壳聚糖及其金属粒子复合材料研究进展[J].精细化工,2021,38(9):1757-1764, 1778. [10] LI J, TIAN X, HUA T, et al. Chitosan natural polymer material for improving antibacterial properties of textiles. ACS Appl Bio Mater. 2021;4(5):4014-4038. [11] BABU A, RAMESH R. Multifaceted applications of chitosan in cancer drug delivery and therapy. Mar Drugs. 2017;15(4):96. [12] RIAZ RAJOKA MS, MEHWISH HM, WU Y, et al. Chitin/chitosan derivatives and their interactions with microorganisms: a comprehensive review and future perspectives. Crit Rev Biotechnol. 2020;40(3):365-379. [13] PAVINATTO A, SOUZA A L, DELEZUK JA, et al. Interaction of O-acylated chitosans with biomembrane models: probing the effects from hydrophobic interactions and hydrogen bonding. Colloids Surf B Biointerfaces. 2014;114:53-59. [14] ABRICA-GONZÁLEZ P, ZAMORA-JUSTO JA, SOTELO-LÓPEZ A, et al. Gold nanoparticles with chitosan, N-acylated chitosan, and chitosan oligosaccharide as DNA carriers. Nanoscale Res Lett. 2019;14(1):258. [15] DU X, WU L, YAN H, et al. Microchannelled alkylated chitosan sponge to treat noncompressible hemorrhages and facilitate wound healing. Nat Commun. 2021;12(1):4733. [16] SHEN J, JIN B, QI YC, et al. Carboxylated chitosan/silver-hydroxyapatite hybrid microspheres with improved antibacterial activity and cytocompatibility. Mater Sci Eng C Mater Biol Appl. 2017;78:589-597. [17] YI Y, FEI J, XU Y X, et al. Preparation and characterization of a Schiff-based chitosan-fructose quaternary ammonium salt for medical applications. J Biomater Sci Polym Ed. 2020;31(6):804-815. [18] ISLAM S, ARNOLD L, PADHYE R. Comparison and characterisation of regenerated chitosan from 1-butyl-3-methylimidazolium chloride and chitosan from crab shells. Biomed Res Int. 2015;2015:874316. [19] KHATTAK S, WAHID F, LIU LP, et al. Applications of cellulose and chitin/chitosan derivatives and composites as antibacterial materials:current state and perspectives. Appl Microbiol Biotechnol. 2019;103(5): 1989-2006. [20] SAITO H, SAKAKIBARA Y, SAKATA A, et al. Antibacterial activity of lysozyme-chitosan oligosaccharide conjugates (LYZOX) against pseudomonas aeruginosa, acinetobacter baumannii and methicillin-resistant staphylococcus aureus. PLoS One. 2019;14(5):e0217504. [21] JAMSHIDI D, SAZEGAR MR. Antibacterial activity of a novel biocomposite chitosan/graphite based on zinc-grafted mesoporous silica nanoparticles. Int J Nanomedicine. 2020;15:871-883. [22] AMIDI M, MASTROBATTISTA E, JISKOOT W, et al. Chitosan-based delivery systems for protein therapeutics and antigens. Adv Drug Deliv Rev. 2010;62(1):59-82. [23] SHIH PY, LIAO YT, TSENG YK, et al. A potential antifungal effect of chitosan against candida albicans is mediated via the inhibition of SAGA complex component expression and the subsequent alteration of cell surface integrity. Front Microbiol. 2019;10:602. [24] XIAO X, YU Y, SUN Y, et al. Heavy metal removal from aqueous solutions by chitosan-based magnetic composite flocculants. J Environ Sci (China). 2021;108:22-32. [25] GRANDE-TOVAR CD, CHAVES-LOPEZ C, SERIO A, et al. Chitosan coatings enriched with essential oils: effects on fungi involved in fruit decay and mechanisms of action. Trends Food Sci Tech. 2018;78:61-71. [26] WEIßPFLOG J, GÜNDEL A, VEHLOW D, et al. Solubility and selectivity effects of the anion on the adsorption of different heavy metal ions onto chitosan. Molecules. 2020;25(11):2482. [27] CHUNG YC, YEH JY, TSAI CF. Antibacterial characteristics and activity of water-soluble chitosan derivatives prepared by the Maillard reaction. Molecules. 2011;16(10):8504-8514. [28] ARDEAN C, DAVIDESCU CM, NEMEŞ NS, et al. Factors influencing the antibacterial activity of chitosan and chitosan modified by functionalization. Int J Mol Sci. 2021;22(14):7449. [29] BYUN SM, NO HK, HONG JH, et al. Comparison of physicochemical, binding, antioxidant and antibacterial properties of chitosans prepared from ground and entire crab leg shells. Int J Food Sci Tech. 2013;48(1):136-142. [30] MA J, FAQIR Y, TAN C, et al. Terrestrial insects as a promising source of chitosan and recent developments in its application for various industries. Food Chem. 2022;373(Pt A):131407. [31] SRIMANEEPONG V, THANAMEE T, WATTANASIRMKIT K, et al. Efficacy of low-molecular weight chitosan against Candida albicans biofilm on polymethyl methacrylate resin. Aust Dent J. 2021;66(3):262-269. [32] KAŠPAROVÁ P, ZMUDA M, VAŇKOVÁ E, et al. Low-molecular weight chitosan enhances antibacterial effect of antibiotics and permeabilizes cytoplasmic membrane of Staphylococcus epidermidis biofilm cells. Folia Microbiol (Praha). 2021;66(6):983-996. [33] LI J, FU J, TIAN X, et al. Characteristics of chitosan fiber and their effects towards improvement of antibacterial activity. Carbohydr Polym. 2022; 280:119031. [34] VISHU KUMAR AB, VARADARAJ MC, GOWDA LR, et al. Characterization of chito-oligosaccharides prepared by chitosanolysis with the aid of papain and Pronase, and their bactericidal action against Bacillus cereus and Escherichia coli. Biochem J. 2005;391(Pt 2):167-175. [35] HOSSEINNEJAD M, JAFARI SM. Evaluation of different factors affecting antimicrobial properties of chitosan. Int J Biol Macromol. 2016;85:467-475. [36] HASSAN MI, MOHAMED AF, TAHER FA, et al. Antimicrobial activities of chitosan nanoparticles prepared-from lucila cuprina maggots (diptera: calliphoridae). J Egypt Soc Parasitol. 2016;46(3):563-570. [37] RISTIC T, HRIBERNIK S, FRAS-ZEMLJIC L. Electrokinetic properties of fibres functionalised by chitosan and chitosan nanoparticles. Cellulose. 2015;22(6):3811-3823. [38] MENG X, XING R, LIU S, et al. Molecular weight and pH effects of aminoethyl modified chitosan on antibacterial activity in vitro. Int J Biol Macromol. 2012;50(4):918-924. [39] NO HK, PARK NY, LEE SH, et al. Antibacterial activity of chitosans and chitosan oligomers with different molecular weights. Int J Food Microbiol. 2002;74(1-2):65-72. [40] ARDILA N, DAIGLE F, HEUZEY MC, et al. Effect of chitosan physical form on its antibacterial activity against pathogenic bacteria. J Food Sci. 2017;82(3):679-686. [41] CHUNG YC, SU YP, CHEN CC, et al. Relationship between antibacterial activity of chitosan and surface characteristics of cell wall. Acta Pharmacol Sin. 2004;25(7):932-936. [42] CHUNG YC, WANG HL, CHEN YM, et al. Effect of abiotic factors on the antibacterial activity of chitosan against waterborne pathogens. Bioresour Technol. 2003;88(3):179-184. [43] CARACCIOLO G, VALI H, MOORE A, et al. Challenges in molecular diagnostic research in cancer nanotechnology. Nano Today. 2019;27: 6-10. [44] RYU JH, CHOI JS, PARK E, et al. Chitosan oral patches inspired by mussel adhesion. J Control Release. 2020;317:57-66. [45] DUBASHYNSKAYA NV, RAIK SV, DUBROVSKII YA, et al. Hyaluronan/diethylaminoethyl chitosan polyelectrolyte complexes as carriers for improved colistin delivery. Int J Mol Sci. 2021;22(16):8381. [46] 杨梦璐.壳聚糖基防龋基因疫苗载体的制备及性能研究[D].遵义:遵义医科大学,2021. [47] PHUANGKAEW T, BOORANABUNYAT N, KIATKAMJORNWONG S, et al. Amphiphilic quaternized chitosan: synthesis, characterization, and anti-cariogenic biofilm property. Carbohydr Polym. 2022;277:118882. [48] PIEGAT A, ŻYWICKA A, NIEMCZYK A, et al. Antibacterial activity of N,O-Acylated chitosan derivative. Polymers (Basel). 2020;13(1):107. [49] LUO Z, XUE K, ZHANG X, et al. Thermogelling chitosan-based polymers for the treatment of oral mucosa ulcers. Biomater Sci. 2020;8(5): 1364-1379. [50] WANG N, JI Y, ZHU Y, et al. Antibacterial effect of chitosan and its derivative on enterococcus faecalis associated with endodontic infection. Exp Ther Med. 2020;19(6):3805-3813. [51] ABUSREWIL S, BROWN JL, DELANEY C, et al. Chitosan enhances the anti-biofilm activity of biodentine against an interkingdom biofilm model. Antibiotics (Basel). 2021;10(11):1317. [52] WALCZAK K, SCHIERZ G, BASCHE S, et al. Antifungal and surface properties of chitosan-salts modified PMMA denture base material. Molecules. 2020;25(24):5899. [53] DE MORI A, DI GREGORIO E, KAO AP, et al. Antibacterial PMMA composite cements with tunable thermal and mechanical properties. ACS Omega. 2019;4(22):19664-19675. [54] IBRAHIM MA, NEO J, ESGUERRA RJ, et al. Characterization of antibacterial and adhesion properties of chitosan-modified glass ionomer cement. J Biomater Appl. 2015;30(4):409-419. [55] ZHANG X, ZHANG Z, WU W, et al. Preparation and characterization of chitosan/Nano-ZnO composite film with antimicrobial activity. Bioprocess Biosyst Eng. 2021;44(6):1193-1199. [56] YIN M, LIN X, REN T, et al. Cytocompatible quaternized carboxymethyl chitosan/poly (vinyl alcohol) blend film loaded copper for antibacterial application. Int J Biol Macromol. 2018;120(Pt A):992-998. [57] ZHANG M, YANG M, WOO MW, et al. High-mechanical strength carboxymethyl chitosan-based hydrogel film for antibacterial wound dressing. Carbohydr Polym. 2021;256:117590. [58] CAMACHO-ALONSO F, SALINAS J, SÁNCHEZ-SILES M, et al. Synergistic antimicrobial effect of photodynamic therapy and chitosan on the titanium-adherent biofilms of staphylococcus aureus, escherichia coli, and pseudomonas aeruginosa: an in vitro study. J Periodontol. 2021. doi: 10.1002/JPER.21-0306. [59] LIU G, LI K, LUO Q, et al. PEGylated chitosan protected silver nanoparticles as water-borne coating for leather with antibacterial property. J Colloid Interface Sci. 2017;490:642-651. [60] D’ALMEIDA M, ATTIK N, AMALRIC J, et al. Chitosan coating as an antibacterial surface for biomedical applications. PLoS One. 2017; 12(12):e0189537. |
| [1] | Li Rui, Liu Zhen, Guo Zige, Lu Ruijie, Wang Chen. Aspirin-loaded chitosan nanoparticles and polydopamine modified titanium sheets improve osteogenic differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 374-379. |
| [2] | Wu Lihao, Shao Anliang, Xu Lin, Ren Kang, Wang Hongjian, Chen Liang, Xu Ling. Evaluation of immunotoxicity of the absorbable macroporous polysaccharides composite hemostatic material [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 329-334. |
| [3] | Zong Mingrui, Liu Haiyan, Li Bing, Wu Xiuping. Application of carboxymethyl chitosan in tissue engineering of stomatology [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 447-452. |
| [4] | Qi Junqiang, Guo Chao, Niu Dongyang, Wang Haotian, Xiao Bing, Xu Guohua. Characteristics and application of bone repair materials of metal ion doped hydroxyapatite [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3415-3422. |
| [5] | Zhu Hong, Lin Ziheng, He Rouye, Pan Jinbin, Liu Xiaochuan, He Xiaoling, Zhang Jingying. Antibacterial and hemostatic properties of chitosan collagen sponge [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(16): 2525-2533. |
| [6] | Zhong Wenjing, Zhou Zihan, Wang Haoyu, Li Shangyong, Xia Yujun. Preparation and in vitro evaluation of sustained-release nanoparticles loaded with sulfasalazine [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(16): 2461-2466. |
| [7] | Ren Wenyan, Liu Xue, Wang Yiyu. Grophene-family nanomaterials in the treatment of periodontal disease: beneficial for osteogenic differentiation and reconstruction of periodontal support tissues [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1954-1960. |
| [8] | Liu Siqi, Wu Mingrui, Qiao Lingran, Xie Liying, Chen Siyu, Han Zhibo, Zuo Lin. Effects of hydrogel loaded with human umbilical cord mesenchymal stem cells on diabetic wound repair in mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 21-27. |
| [9] | Le Guoping, Zhang Ming, Xi Licheng, Luo Hanwen. Preparation and in vitro evaluation of vancomycin hydrochloride@polylactic acid-glycolic acid copolymer-chitosan-hyaluronic acid composite sustained-release microspheres [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 528-534. |
| [10] | Long Zhisheng, Xiong Long, Gong Feipeng, Li Jingtang, Zeng Jianhua, Deng Ying, Lan Min, Kong Weihao, Chen Gang. Effect of artificial bone with multi-scale hydroxyapatite/chitosan microtubule structure on rabbit bone defect repair and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5436-5441. |
| [11] | Sun Xirao, Bao Jiaxin, Wang Chengyue. Construction of chitosan/mineralized collagen porous scaffold, osteogenic differentiation in vitro and biocompatibility [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5498-5503. |
| [12] | Xu Lin, Cheng Gangyi, Li Hongwei, Wang Hongjian, Lin Minghui, Guo Hongwei, Lin Lianfeng, Xu Ling. Hemostatic effect of macroporous polysaccharides composite hemostatic materials on small-artery severed injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(34): 5442-5447. |
| [13] | Sheng Weibei, Xiong Ao, Liu Su, Deng Jiapeng, Weng Jian, Yu Fei, Chen Yingqi, Zeng Hui. Effect of phosphocreatine modified chitosan hydrogel on polarization and inflammatory factor expression in rat bone marrow-derived macrophages [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(31): 5040-5046. |
| [14] | Lu Yunan, Zhang Xinzhao, Lin Binbin, Xu Gan, Chen Jingdi, Chen Shunyou. Naringin-chitosan/hydroxyapatite composite scaffold in repair of rat skull defect [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4441-4445. |
| [15] | Liu Ming, Wang Kai. Chitosan/alginate composite scaffold combined with hawthorn leaf flavonoids for spinal cord injury repair [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(28): 4466-4471. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||