Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (21): 3368-3374.doi: 10.12307/2023.110
Previous Articles Next Articles
Application of inorganic nonmetal biomaterials in the treatment of osteosarcoma and the regeneration of tumor-related bone defects
Niu Lin1, 2, 3, Mei Yukun1, 2, Zou Rui1, 2, Zhang Yuwei1, 2, Zhang Yifei1, 2, Hao Yaqi1, 2, 3, Dong Shaojie1, 2, 3
- 1Key Laboratory of Shaanxi Province for Craniofacial Precision Medicine Research, Hospital of Stomatology, Xi’an Jiaotong University, Xi’an 710004, Shaanxi Province, China; 2Clinical Research Center of Shaanxi Province for Dental and Maxillofacial Diseases, Xi’an 710004, Shaanxi Province, China; 3Department of Prosthodontics, Hospital of Stomatology, Xi’an Jiaotong University, Xi’an 710004, Shaanxi Province, China
-
Received:2022-04-13Accepted:2022-05-11Online:2023-07-28Published:2022-11-24 -
Contact:Dong Shaojie, MD, Assistant researcher, Key Laboratory of Shaanxi Province for Craniofacial Precision Medicine Research, Hospital of Stomatology, Xi’an Jiaotong University, Xi’an 710004, Shaanxi Province, China; Clinical Research Center of Shaanxi Province for Dental and Maxillofacial Diseases, Xi’an 710004, Shaanxi Province, China; Department of Prosthodontics, Hospital of Stomatology, Xi’an Jiaotong University, Xi’an 710004, Shaanxi Province, China -
About author:Niu Lin, MD, Chief physician, Key Laboratory of Shaanxi Province for Craniofacial Precision Medicine Research, Hospital of Stomatology, Xi’an Jiaotong University, Xi’an 710004, Shaanxi Province, China; Clinical Research Center of Shaanxi Province for Dental and Maxillofacial Diseases, Xi’an 710004, Shaanxi Province, China; Department of Prosthodontics, Hospital of Stomatology, Xi’an Jiaotong University, Xi’an 710004, Shaanxi Province, China Mei Yukun, Master candidate, Key Laboratory of Shaanxi Province for Craniofacial Precision Medicine Research, Hospital of Stomatology, Xi’an Jiaotong University, Xi’an 710004, Shaanxi Province, China; Clinical Research Center of Shaanxi Province for Dental and Maxillofacial Diseases, Xi’an 710004, Shaanxi Province, China -
Supported by:National Natural Science Foundation of China (Youth Project), No. 82102221 (to DSJ); Basic Research Funds Free Exploration and Innovation-Teacher Project of Xi’an Jiaotong University, No. xzy012021069 (to DSJ); Xi’an Innovation Capability Strong Foundation Program - Medical Research Project, No. 21YXYJ0123 (to DSJ)
CLC Number:
Cite this article
Niu Lin, Mei Yukun, Zou Rui, Zhang Yuwei, Zhang Yifei, Hao Yaqi, Dong Shaojie. Application of inorganic nonmetal biomaterials in the treatment of osteosarcoma and the regeneration of tumor-related bone defects[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3368-3374.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
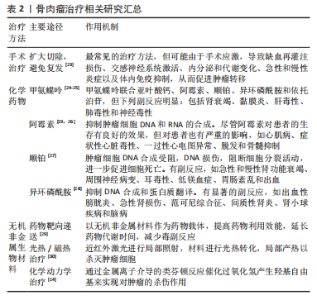
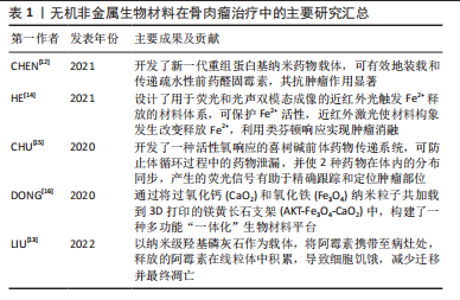
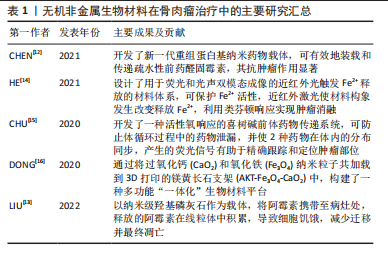
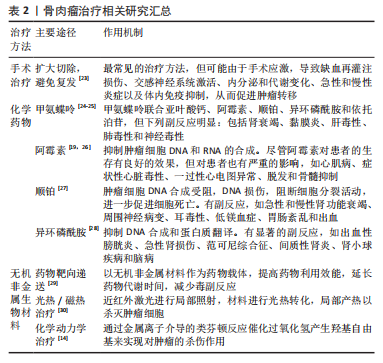
2.2 骨肉瘤临床治疗研究现状 骨肉瘤侵袭性强,界限不明确,易发生早期转移和扩散[17]。1970年前,骨肉瘤的治疗多以手术切除为主,复发率高,患者预后差。在1970-1980年,手术-化疗联合应用的治疗方案开始兴起,顺铂、阿霉素、甲氨蝶呤等治疗骨肉瘤的一线化疗药物的广泛应用,使骨肉瘤患者的5年生存率出现明显提升[8,18-19]。随着医学进步,骨肉瘤的治疗方式也在不断改进,局部根治性切除手术和辅助化学治疗相结合是现阶段治疗效果比较好的方案。具体方法包括前期化学治疗、中期局部外科切除病变和后期辅助化学治疗,其中化学治疗是重要一步。骨肉瘤细胞对化疗药物的敏感度也被证实为重要的预后影响因素。有研究发现,放疗与化疗联合应用,化疗药物可增强骨肉瘤的放疗敏感性,具有一定的协同效果。目前各种放射增敏疗法的临床应用进一步提高了放疗效果。OGAWA等[20]应用KORTUC I(照射部位覆盖一层H2O2纱布)治疗骨肉瘤,有效地缓解无法手术切除患者的临床症状。此外,质子治疗和重离子治疗也用于骨肉瘤的治疗。BLATTMANN等[21]联合质子治疗和重离子连续治疗,对正常组织损伤小,同时可获得较好的预后。由于骨肉瘤对射线不敏感,因此单纯的放疗仅应用于难以切除骨肉瘤的姑息控制、术后病灶的清除和缓解全身转移患者的痛苦,并非骨肉瘤治疗的常规方案。 由于骨肉瘤细胞对化疗药物存在耐药性,现阶段的化学治疗方法出现瓶颈,难以突破平台效应,存活率相比于20世纪90年代并没有显著提高。另外,全身化疗存在诸多问题,如药物靶向效率低,全身毒副反应明显等。骨肉瘤的化疗耐药性问题使化疗药物的有效性不断弱化,超过1/3的恶化与复发与耐药性有关。此外,当前的治疗方案要求完全彻底切除或扩大切除肿瘤区域,且仍有2%-7%的复发率;术后大段骨缺损的修复也是骨肉瘤治疗需要解决的问题。因此,新的治疗手段引起研究者重视,例如开发新型纳米药物递送系统,产生过高热、活性氧或免疫反应实现骨肉瘤治疗,构建兼具肿瘤治疗、防止复发和骨组织再生功能的多功能植入材料等,显示出良好的治疗效果和极大的应用潜力,其中,无机非金属材料被广泛用于构建新型纳米材料和多功能植入材料[22],在开发新型骨肉瘤治疗策略中扮演了重要角色。如何构建集肿瘤消融、防止复发及促进骨修复与一体治疗手段,将成为骨肉瘤治疗研究领域的热点之一[23-30]。目前骨肉瘤治疗相关研究成果见表2。"

2.3 无机非金属纳米材料在骨肉瘤治疗与修复的应用 2.3.1 在骨肉瘤早期诊断的应用 骨肉瘤是一类侵袭性强、易发生早期转移的恶性肿瘤,骨肉瘤患者发生早期转移后,5年生存率可降至20%以下[31-32]。骨肉瘤治疗的困境亟待新型诊疗策略的出现,而实现早期、准确的诊断,是提高其治愈率的重要途径之一。纳米材料技术的发展,为肿瘤治疗带来新的机遇,纳米材料的理化特性在药物靶向递送、光热/磁热治疗、光动力治疗、催化治疗和免疫治疗等方面具有独特的优势。常规的肿瘤影像检测技术中,核磁共振易受骨基质内钙元素干扰,正电子放射断层造影术分辨率低,不适于骨肉瘤的诊断[33]。目前,临床上对于骨肉瘤的诊断主要依靠CT进行,而CT技术仍存在易受高密度骨皮质干扰,难以区分肿瘤与正常组织的不足[34]。因此,开发新一代对比剂,提高CT显影分辨率,是提高骨肉瘤的影像检测能力乃至最终治疗效果的关键之一。 纳米肿瘤诊疗材料,是指针对特定肿瘤微环境或细胞特性而设计、构建的兼具诊断与治疗作用,并具有良好生物相容性的多功能纳米复合材料。理想的肿瘤诊疗材料应当能够精确、快速富集在目标区域,准确显示疾病的生化和形态特征,而且可在肿瘤组织达到有效浓度而不损害健康的器官,可被从体内清除或被清除生物降解为安全无毒性物质,可满足对人体安全的要求[35]。无机非金属纳米材料,生物安全性较好,可在体内降解,在骨肉瘤诊疗应用中具有较大潜力。另外,YANG等[36]发现,叶酸受体α在骨肉瘤细胞表面具有较高的表达水平,为骨肉瘤纳米诊疗材料的开发提供了新的靶点。 在此基础上,CIPRESTE等[37]通过掺杂钆元素,制备了具有超顺磁性质、并利用叶酸表面改性的羟基磷灰石纳米棒(hydroxyapatite gadolinium-nanorods,HA-Gd)。由于羟基磷灰石自身具有良好的骨亲和性和叶酸的靶向能力,HA-Gd可作为良好的骨肉瘤对比剂。此外,合成的HA-Gd具有多孔结构,在负载化疗药、放疗增敏剂或荧光标记材料等方面具有很好的潜力。该课题组进一步合成了掺杂放射性同位素64Cu的纳米羟基磷灰石复合材料,证实了其设想的可行性。LU等[38]利用介孔二氧化硅包裹硫化铋和阿霉素,并通过偶联骨肉瘤靶向多肽,构建了多功能复合材料体系,具有良好的CT影像增强和肿瘤治疗效果。IMANI等[39]构建了掺杂钛元素的TiO2纳米珠,获得了具有光学成像、核磁共振显影增强和活性氧产生能力,兼具肿瘤诊断与治疗功能的多功能纳米材料体系,并在骨肉瘤的检测和治疗中取得了良好的效果,为今后无机非金属纳米材料在骨肉瘤诊疗一体化的开发和应用提供了新的思路。 2.3.2 在骨肉瘤化疗药物递送的应用 骨肉瘤侵袭转移能力强,手术切除后易复发,因而临床上通常将化疗作为重要的术后辅助治疗手段之一。传统化疗药物治疗骨肉瘤,有全身毒性明显、肿瘤富集效果差、存在抗药性问题。口服的常规化疗药物主要是多柔比星、甲氨蝶呤、顺铂及异环磷酰胺等,但在实际应用过程中,这些药物可能存在体内利用效能低、药物代谢时间短、不可避免的毒副反应等问题[40-41]。 过去的研究中,主要将研究重点放在如何提高纳米材料的载药效率及纳米材料自身的生物相容性上。如CHEN等[29]曾利用聚乳酸-羟基乙酸共聚物-葡聚糖聚合物纳米颗粒来进行载药,材料自身具有良好的生物相容性、载药效率较高且血液中药物代谢时间较长。而目前研究则更偏向于增强药物靶向性,降低药物对于正常组织的毒副反应。利用肿瘤与正常组织之间pH值、谷胱甘肽浓度和活性氧水平等的差异,设计并利用不同的纳米材料载药,以达到加强药物靶向性的目的。WANG等[42]研发出高分子纳米药物用于骨肉瘤的化学药物治疗,以重组蛋白作为醛基阿霉素的有效载体,可降低药物的心脏毒性,提高药物的利用率。该重组药物半衰期明显长于游离药物单体,并表现出优异的长效抗肿瘤治疗效果,可作为一种积极的替代药物递送方法。 目前,除了全身应用化疗药物外,还有研究专注于如何在局部应用化疗药物杀灭肿瘤,并减少全身用药带来的副反应。GHOSH [43]等利用纳米羟基磷灰石作为药物的载体,并用抗癌药物如阿霉素和聚乙烯醇聚合物包覆,利用MTT法检测发现其对骨肉瘤细胞(MG 63)的显著细胞毒性,其对肿瘤的杀灭效果显著。LIU等[13]利用羟基磷灰石同阿霉素进行可逆性结合,制备出纳米级的功能性羟基磷灰石颗粒,其通过局部应用于病灶区域,进入肿瘤细胞的溶酶体中。在酸性环境下,羟基磷灰石与阿霉素的结合被破坏,释放的阿霉素在线粒体中积累,导致细胞饥饿,减少迁移和凋亡;其在骨肉瘤模型小鼠上进行进一步验证,发现通过羟基磷灰石颗粒局部传递阿霉素比传统应用阿霉素具有更强的肿瘤根除效果。即除了系统性化疗外,辅助羟基磷灰石可以作为细胞内传递阿霉素的载体,以预防骨肉瘤手术切除后的肿瘤复发。 综上,将化疗药物与纳米载体结合,特异性结合肿瘤细胞的抗体、多肽或肿瘤微环境特有靶向分子,或利用纳米材料的肿瘤富集(enhanced permeability and retention effect,EPR)效应、表面电荷吸附,或者局部应用缓释药物,能够实现化疗药物的高浓度富集,增强肿瘤细胞内吞、减少药物从细胞排出,降低全身不良反应,可期望获得更强的化疗效果。 2.3.3 在骨肉瘤新型疗法的应用 除了利用传统的化疗药物作为肿瘤治疗效应物之外,亦可通过金属离子介导的类芬顿反应催化过氧化氢产生羟基自由基来实现对肿瘤的杀伤作用,这一新兴的肿瘤治疗方法称为化学动力学疗法。HE等[14]设计和开发了一种用于双模成像同时兼具光热响应作用的近红外光触发纳米材料递送系统,通过在肿瘤部位外加近红外激光照射可引发纳米合成材料LET-6的构象发生改变以释放内部存储的Fe2+离子,在特定部位通过催化类芬顿反应产生羟基自由基杀灭骨肉瘤细胞,并在U87MG荷瘤小鼠模型中有显著的治疗作用。CHU等[15]利用喜树碱前体药物传递系统,将喜树碱和光敏剂邻苯二酚-a同时偶联到同一聚乙二醇甲基醚上,在局部进行近红外激光照射即可在局部区域可控、按需释放喜树碱介导的化疗反应和邻苯二酚-a分子介导的光动力反应,增强了对肿瘤生长的抑制。 此外光热疗法也被认为是治愈恶性肿瘤的一种有效策略,具有特异性高、侵袭性小及见效快等优点。WANG等[30]通过利用吲哚菁绿-L-精氨酸与介孔多巴胺和羟基磷灰石进行结合,得到具有核壳和介孔结构的多功能纳米颗粒,将其注射在肿瘤原位并通过近红外激光照射,即可同时产热并产生活性氧,并可进一步诱导L-精氨酸催化释放一氧化氮气体。该纳米复合材料具有较大的比表面积、良好的吸附性能、高负载效率和良好的一氧化氮催化释放能力,并通过整合光热疗法和气体疗法协同实现了MG-63骨肉瘤细胞的体外消融。 2.3.4 兼顾骨肉瘤治疗与缺损修复 由于骨肉瘤多伴有组织缺损且通常超出机体自愈范围,如何在消融肿瘤的过程中利用无机非金属材料的理化特性同步促进缺损区域的修复,实现“一材多用”,是目前的研究热点之一。LIU等[44]利用羟基磷灰石、牛血清白蛋白和紫杉醇合成出三元纳米生物材料-抗肿瘤药物复合物,可在在肿瘤原位进行注射,具有明显的抗癌作用和缓释作用,并能有效抑制肿瘤的增殖和转移,其中羟基磷灰石更可进一步诱导成骨分化。应用该药物可作为骨肉瘤术后辅助治疗与诱导骨组织再生的有效治疗方法。TAN等[45]设计了一种含有姜黄素微球的甲基纤维素水凝胶,姜黄素微球具有良好的光热转化效能,可用于光热治疗。在体外实验中,热疗和姜黄素对骨肉瘤细胞显示出明显的杀灭作用,同时姜黄素的缓释促进了碱性磷酸酶的表达和骨髓间充质干细胞的钙沉积;而在动物实验中,在肿瘤原位注射水凝胶后,808 nm激光照射下,在5 min内产生局部热疗(相当于51 ℃),以消融肿瘤;同时,热加速的姜黄素释放和热增加的细胞膜通透性导致肿瘤细胞的凋亡;光热共化疗组在治疗后第2天开始成功抑制肿瘤生长。之后,由于姜黄素的缓释作用,进一步促进了骨重建。 上述研究初步证实了无机非金属材料的多功能性,体现了其用于骨肉瘤同步治疗与修复的性能优势,为构建兼顾骨肉瘤治疗与缺损修复的新型材料体系的设计与应用提供了良好的基础。 2.4 无机非金属植入材料在骨肉瘤治疗与修复的应用 尽管上述无机非金属纳米材料在骨肉瘤的诊疗与修复的研究已经显示了良好的效果,然而有研究证实,进入血液的纳米材料除了小部分可以通过血管渗透作用进入肿瘤组织之外,大部分则进入机体主要器官并富集[46]。另外,其长期安全性尚需全面评估,以及较低的治疗效率、较多的全身毒性以及肿瘤坏死区域并不通畅的血运,限制了其作用效率[47-49]。因此,肿瘤的局部治疗越来越受到重视。无机非金属植入材料相容性好,机械强度较高,治疗位置精确、效率高,同时兼具促进细胞黏附、迁移、增殖、分化以及引导组织再生的能力。 通过在支架负载具有肿瘤治疗能力的药物或材料,可实现微观尺寸向宏观尺寸的转变,提高了治疗效率,避免了全身毒性,在肿瘤诊疗领域展现出极大潜力。 2.4.1 在药物局部递送与修复的应用 可植入式给药系统作为局部给药的一种方式,可在减少全身药物暴露的同时,在目标部位富集高浓度的药物,优于常规给药方法如口服、静脉给药等方法[50]。该策略允许抗癌药物在几周到几个月甚至几年的时间内持续在局部释放,从而最大限度地减少不良反应,提高药物的利用率[51]。WANG等[52]设计了一种聚L-乳酸植入物作为抗癌药物的特殊载体,其具有优异的载药效能、良好的生物降解性、可靠的生物安全性,发现在此聚L-乳酸植入物的辅助下的局部化疗显示出优于传统化疗的抗骨肉瘤功效。三维打印给药系统可以同时实现骨肉瘤治疗中的个体化局部化疗、多种药物给药、长期可持续药物释放,且不需要再次进行手术。LIU等[53]使用硫酸钙/羟基磷灰石生物材料作为阿霉素的局部给药载体,在局部、持续和受控递送阿霉素以辅助化疗骨肉瘤。值得注意的是,分别在体外环境中进行药物释放研究发现:在生理pH 7.4和酸性(pH 5)环境下,该系统分别有近28%和36%的负载药物在4周内释放,大约63%的药物在体内4周后释放;同时,该载体系统本身的羟基磷灰石将有助于外科手术切除后骨缺损的再生;通过在病灶局部释放阿霉素,可以增强原发性肿瘤对新辅助化疗的反应,且不会导致全身毒性增加。此外,利用可注射水凝胶载药进行局部治疗也成为可靠的治疗选项。YOON等[54]设计了将阿霉素和顺铂负载于可注射可见光固化水凝胶给药系统,在骨肉瘤治疗中显示出临床应用的良好潜力。 因此,在无机非金属材料的基础上结合高分子支架、水凝胶等复合材料体系,可实现长期、缓释局部给药,可避免多药耐药性和血液循环障碍,将抗癌药物输送到癌症组织,并通过联合化疗将耐药性降低、提高化疗效果。上述复合材料的设计开发与研究评价,可为改善无机非金属材料的药物负载、缓释能力提供新的思路。 2.4.2 在骨肉瘤新型消融疗法与修复策略中的应用 3D打印技术是一种新兴的增材制造技术,可通过该技术构建无机非金属支架等可植入材料。在3D打印技术不断发展的基础上,有学者尝试在3D打印生物玻璃支架表面负载二维黑磷纳米片,成功构建了兼具光热及骨缺损修复功能的材料体系,获得了良好的肿瘤光热消融和颅骨缺损修复效果。在β-磷酸三钙表面使用氧化石墨烯涂覆,也可以获得良好的肿瘤光热消融效果,并赋予材料更明显的促成骨能力。受此启发,该课题组曾在早期研究中探索应用磁热治疗骨肉瘤方案,通过将过CaO2和Fe3O4纳米材料同步负载到3D打印的镁黄长石支架中,构建了一种多功能“一体化”生物材料平台,利用Fe3O4超顺磁纳米材料在高频交变磁场内产生的磁热效应进行肿瘤治疗[16]。由于高频交变磁场具有较高的组织穿透能力、与生物组织不发生相互作用,使得其更适用于骨肿瘤等深部器官肿瘤的治疗。此外,Fe3O4能够在酸性环境下催化肿瘤内富集的过氧化氢,通过“类芬顿”反应产生活性氧,可被用于肿瘤的化学动力学治疗。为进一步克服肿瘤内低浓度H2O2难以产生足量活性氧、术后遗留超出自愈范围的骨缺损等问题,将过氧化氢的“储备库”CaO2在3D打印镁黄长石表面与Fe3O4共同负载,实现了良好的磁热性能和化学动力学性能,能够有效杀伤肿瘤细胞并抑制体内肿瘤的生长。CaO2同时释放钙元素,从而促进成骨细胞的增殖、成骨分化,并促进体内骨缺损的修复,证明功能改性的3D打印支架能够同步实现骨肉瘤的高效治疗与骨缺损的快速修复。此外,亦有研究将5-氟尿嘧啶均匀涂覆至磷酸钙骨水泥支架,改性后的支架材料显示出良好的骨肿瘤治疗效果,可用作个性化医疗的骨移植材料[55]。 通过在无机非金属植入材料表面涂覆药物、光热及磁热分子等方式,将其功能改性并用于肿瘤局部治疗时范围精确、效率较高,同时兼具促进成骨分化以及引导组织修复再生的能力。作为骨肉瘤术后缺损修复及辅助化疗的解决方案,在无机非金属支架负载具有肿瘤治疗能力的效应分子,实现微观向宏观的转变并进一步提高治疗效率,避免传统给药形式引起的全身副反应,将在骨肉瘤的诊疗中展现出极大价值。"

| [1] MARINA N, GEBHARDT M, TEOT L, et al. Biology and therapeutic advances for pediatric osteosarcoma. Oncologist. 2004;9(4):422-441. [2] PICCI P, Osteosarcoma (osteogenic sarcoma). Orphanet J Rare Dis. 2007;2:6. [3] HEARE T, HENSLEY MA, DELL’ORFANO S. Bone tumors: osteosarcoma and Ewing’s sarcoma. Curr Opin Pediatr. 2009;21(3):365-372. [4] GUILLON MA, MARY PM, BRUGIERE L, et al. Clinical characteristics and prognosis of osteosarcoma in young children: a retrospective series of 15 cases. BMC Cancer. 2011;11:407. [5] MISAGHI A, GOLDIN A, AWAD M, et al. Osteosarcoma: a comprehensive review. SICOT J. 2018;4:12. [6] LOH AH, NAVID F, WANG C, et al. Management of local recurrence of pediatric osteosarcoma following limb-sparing surgery. Ann Surg Oncol. 2014;21(6):1948-1955. [7] EBB D, MEYERS P, GRIER H, et al. Phase II trial of trastuzumab in combination with cytotoxic chemotherapy for treatment of metastatic osteosarcoma with human epidermal growth factor receptor 2 overexpression: a report from the children’s oncology group. J Clin Oncol. 2012;30(20):2545-2551. [8] AU KM, SATTERLEE A, MIN Y, et al. Folate-targeted pH-responsive calcium zoledronate nanoscale metal-organic frameworks: turning a bone antiresorptive agent into an anticancer therapeutic. Biomaterials. 2016;82:178-193. [9] WACHTEL M and SCHAFER BW, Targets for cancer therapy in childhood sarcomas. Cancer Treat Rev. 2010;36(4):318-327. [10] WU JJ, WILLIAMS GR, ZHU Y, et al. Ultrathin chalcogenide nanosheets for photoacoustic imaging-guided synergistic photothermal/gas therapy. Biomaterials. 2021;273:120807. [11] WANG J, HU X, XIANG D. Nanoparticle drug delivery systems: an excellent carrier for tumor peptide vaccines. Drug Deliv. 2018;25(1):1319-1327. [12] CHEN C, XIE L, REN T, et al. Immunotherapy for osteosarcoma: fundamental mechanism, rationale, and recent breakthroughs. Cancer Lett. 2021;500:1-10. [13] LIU Y, NADEEM A, SEBASTIAN S, et al. Bone mineral: a trojan horse for bone cancers. efficient mitochondria targeted delivery and tumor eradication with nano hydroxyapatite containing doxorubicin. Materials today Bio. 2022;14: 100227. [14] HE T, YUAN YY, JIANG C, et al. Light-triggered transformable ferrous ion delivery system for photothermal primed chemodynamic therapy. Angew Chem Int Edit. 2021;60(11): 6047-6054. [15] CHU BY, QU Y, HE XL, et al. ROS-responsive camptothecin prodrug nanoparticles for on-demand drug release and combination of chemotherapy and photodynamic therapy. Adv Funct Mater. 2020;30(52): 2005918. [16] DONG SJ, CHEN Y, YU LD, et al. Magnetic hyperthermia-synergistic H2O2 self-sufficient catalytic suppression of osteosarcoma with enhanced bone-regeneration bioactivity by 3D-printing composite scaffolds. Adv Funct Mater. 2020;30(4):1907071. [17] BERHE S, DANZER E, MEYERS P, et al. Unusual abdominal metastases in osteosarcoma. J Pediatr Surg Case Rep. 2018;28:13-16. [18] RAINUSSO N, WANG LL, YUSTEIN JT. The adolescent and young adult with cancer: state of the art - bone tumors. Curr Oncol Rep. 2013;15(4):296-307. [19] 孙郁文,魏鹏,陈启旺,等.阿霉素结合骨修复材料应用于骨肉瘤治疗的研究进展[J].实用骨科杂志,2019,25(8):728-733. [20] OGAWA Y, UE H, TSUZUKI K, et al. New radiosensitization treatment (KORTUC I) using hydrogen peroxide solution-soaked gauze bolus for unresectable and superficially exposed neoplasms. Oncol Rep. 2008;19(6):1389-1394. [21] BLATTMANN C, OERTEL S, THIEMANN M, et al. Histone deacetylase inhibition sensitizes osteosarcoma to heavy ion radiotherapy. Radiat. Oncol. 2015;10:146. [22] 李耀明,姜宏,石永芳.壳聚糖/聚乳酸/羟基磷灰石/聚乙烯醇复合材料骨支架的制备及表征[J].中国组织工程研究,2022,26(18):2888-2893. [23] CHEN ZW, ZHANG PD, XU Y, et al. Surgical stress and cancer progression: the twisted tango. Mol Cancer. 2019;18(1):132. [24] HARRISON DJ, GELLER DS, GILL JD, et al. Current and future therapeutic approaches for osteosarcoma. Expert Rev Anticancer Ther. 2018;18(1):39-50. [25] XU M, XU F, YU C. Clinical analysis of osteosarcoma patients treated with high-dose methotrexate-free neoadjuvant chemotherapy. Curr Oncol. 2014; 21(5):E678-E684. [26] ANDO K, HEYMANN MF, STRESING V, et al. Current therapeutic strategies and novel approaches in osteosarcoma. Cancers. 2013;5(2):591-616. [27] TCHOUNWOU PB, DASARI S, NOUBISSI FK, et al. Advances in our understanding of the molecular mechanisms of action of cisplatin in cancer therapy. J Exp Pharmacol. 2021;13:303-328. [28] WITTIG JC, BICKELS J, PRIEBAT D, et al. Osteosarcoma: a multidisciplinary approach to diagnosis and treatment. Am Fam Physician. 2002;65(6): 1123-1132. [29] CHEN B, YANG JZ, WANG LF, et al. Ifosfamide-loaded poly (lactic-co-glycolic acid) PLGA-dextran polymeric nanoparticles to improve the antitumor efficacy in Osteosarcoma. BMC Cancer. 2015;15:752. [30] WANG YC, DAI HL, LI ZH, et al. Mesoporous polydopamine-coated hydroxyapatite nano-composites for ROS-triggered nitric oxide-enhanced photothermal therapy of osteosarcoma. J Mat Chem B. 2021;9(36): 7401-7408. [31] ISAKOFF MS, BIELACK SS, MELTZER P, et al. Osteosarcoma: current treatment and a collaborative pathway to success. J Clin Oncol. 2015;33(27):3029-3127. [32] WYCISLO KL, FAN TM. The Immunotherapy of canine osteosarcoma: a historical and systematic review. J Vet Intern Med. 2015;29(3):759-769. [33] HUANG R, WANG MW, ZHU YZ, et al. Development of PET probes for cancer imaging. Curr Top Med Chem. 2015;15(8):795-819. [34] JIN YY, NI DL, GAO L, et al. Harness the power of upconversion nanoparticles for spectral computed tomography diagnosis of osteosarcoma. Adv Funct Mater. 2018;28(33):1802656. [35] JOKERST JV, GAMBHIR SS. Molecular imaging with theranostic nanoparticles. Accounts Chem Res. 2011;44(10):1050-1060. [36] YANG R, KOLB EA, QIN J, et al. The folate receptor alpha is frequently overexpressed in osteosarcoma samples and plays a role in the uptake of the physiologic substrate 5-methyltetrahydrofolate. Clin Cancer Res. 2007; 13(9):2557-2567. [37] CIPRESTE MF, PERES AM, COTTA AAC, et al. Synthesis and characterization of Gd-159-doped hydroxyapatite nanorods for bioapplications as theranostic systems. Mater Chem Phys. 2016;181:301-311. [38] LU Y, LI LH, LIN ZF, et al. Enhancing Osteosarcoma Killing and CT imaging using ultrahigh drug loading and NIR-responsive bismuth sulfide@mesoporous silica nanoparticles. Adv Healthc Mater. 2018;7(19):1800602. [39] IMANI R, DILLERT R, BAHNEMANN DW, et al. Multifunctional gadolinium-doped mesoporous TiO2 nanobeads: photoluminescence, enhanced spin relaxation, and reactive oxygen species photogeneration, beneficial for cancer diagnosis and treatment. SMALL. 2017;13(20):1700349. [40] KOPP LM, WOMER RB, SCHWARTZ CL, et al. Effects of dexrazoxane on doxorubicin-related cardiotoxicity and second malignant neoplasms in children with osteosarcoma: a report from the Children’s Oncology Group. Cardiooncology. 2019;5(1):15. [41] CHAMBERLAIN FE, JONES RL, CHAWLA SP. Aldoxorubicin in soft tissue sarcomas. Future Oncol. 2019;15(13):1429-1435. [42] WANG SD, LI B, ZHANG HL, et al. Improving bioavailability of hydrophobic prodrugs through supramolecular nanocarriers based on recombinant proteins for osteosarcoma treatment. Angew Chem Int Edit. 2021;60(20):11252-11256. [43] GHOSH S, RAJU RSK, GHOSH N, et al. Development and physicochemical characterization of doxorubicin-encapsulated hydroxyapatite-polyvinyl alcohol nanocomposite for repair of osteosarcoma-affected bone tissues. C R Chim. 2019;22(1):46-57. [44] LIU YJ, QIAO ZG, GAO J, et al. Hydroxyapatite-bovine serum albumin-paclitaxel nanoparticles for locoregional treatment of osteosarcoma. Adv Healthc Mater. 2021;10(2):2000573. [45] TAN BW, WU YT, WU YZ, et al. Curcumin-microsphere/IR820 hybrid bifunctional hydrogels for in situ osteosarcoma chemo-co-thermal therapy and bone reconstruction. ACS Appl Mater Interfaces. 2021;13(27):31542-31553. [46] YOKOI K, KOJIC M, MILOSEVIC M, et al. Capillary-wall collagen as a biophysical marker of nanotherapeutic permeability into the tumor microenvironment. Cancer Res. 2014;74(16):4239-4246. [47] CHENG YH, HE CL, RIVIERE JE, et al. Meta-analysis of nanoparticle delivery to tumors using a physiologically based pharmacokinetic modeling and simulation approach. ACS Nano. 2020;14(3):3075-3095. [48] MICHIELS C, TELLIER C, FERON O. Cycling hypoxia: a key feature of the tumor microenvironment. Biochim Biophys Acta. 2016;1866(1):76-86. [49] MAEDA H. Toward a full understanding of the EPR effect in primary and metastatic tumors as well as issues related to its heterogeneity. Adv Drug Deliver Rev. 2015;91:3-6. [50] ZHOU HY, HERNANDEZ C, GOSS M, et al. Biomedical imaging in implantable drug delivery systems. Curr. Drug Targets. 2015;16(6):672-682. [51] MENG E, HOANG T, Micro- and nano-fabricated implantable drug-delivery systems. Ther Deliv. 2012;3(12):1457-1467. [52] WANG YH, SUN L, MEI ZG, et al. 3D printed biodegradable implants as an individualized drug delivery system for local chemotherapy of osteosarcoma. Mater Des. 2020;186:108336. [53] LIU Y, RAINA DB, SEBASTIAN S, et al. Sustained and controlled delivery of doxorubicin from an in-situ setting biphasic hydroxyapatite carrier for local treatment of a highly proliferative human osteosarcoma. Acta Biomater. 2021;131:555-571. [54] YOON SJ, MOON YJ, CHUN HJ, et al. Doxorubicin center dot hydrochloride/cisplatin-loaded hydrogel/nanosized (2-hydroxypropyl)-beta-cyclodextrin local drug-delivery system for osteosarcoma treatment in vivo. Nanomaterials. 2019;9(12):1652. [55] WU Y, WOODBINE L, CARR AM, et al. 3D printed calcium phosphate cement (CPC) scaffolds for anti-cancer drug delivery. Pharmaceutics. 2020;12(11):1077. [56] 吕奇,洪嵩.恶性骨肿瘤切除后用于骨缺损修复的生物材料的研究进展[J].生物骨科材料与临床研究,2022,19(1):75-79. [57] WILHELM S, TAVARES AJ, DAI Q, et al. Analysis of nanoparticle delivery to tumours. Nat Rev Mater. 2016;1(5):16014. [58] MATSUMURA Y, MAEDA H. A new concept for macromolecular therapeutics in cancer-chemotherapy - mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 1986;46(12):6387-6392. [59] FANG J, NAKAMURA H, MAEDA H. The EPR effect: unique features of tumor blood vessels for drug delivery, factors involved, and limitations and augmentation of the effect. Adv Drug Deliv Rev. 2011;63(3):136-151. [60] LAMMERS T, KIESSLING F, HENNINK WE, et al. Drug targeting to tumors: principles, pitfalls and (pre-) clinical progress. J Control Release. 2012; 161(2):175-187. [61] NICHOLS JW, BAE YH. Odyssey of a cancer nanoparticle: from injection site to site of action. Nano Today. 2012;7(6):606-618. [62] GILLIES RJ, SCHORNACK PA, SECOMB TW, et al. Causes and effects of heterogeneous perfusion in tumors. Neoplasia (New York). 1999;1(3):197-207. [63] NICHOLS JW, BAE YH. EPR: Evidence and fallacy. J Control Release. 2014;190:451-464. [64] FLYNN T, WEI CM. The pathway to commercialization for nanomedicine. Nanomed Nanotechnol Biol Med. 2005;1(1):47-51. [65] WANG SY, HU HZ, QING XC, et al. Recent advances of drug delivery nanocarriers in osteosarcoma treatment. J Cancer. 2020;11(1):69-82. [66] XU SF, LU HZ, ZHENG XW, et al. Stimuli-responsive molecularly imprinted polymers: versatile functional materials. J Mater Chem C. 2013;1(29): 4406-4422. [67] PENG HL, DONG RC, WANG SQ, et al. A pH-responsive nano-carrier with mesoporous silica nanoparticles cores and poly(acrylic acid) shell-layers: fabrication, characterization and properties for controlled release of salidroside. Int J Pharm. 2013;446(1-2):153-159. [68] SENAPATI S, MAHANTA AK, KUMAR S, et al. Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct Target Ther. 2018;3:7. [69] WANG CF, MA ZJ, YUAN KM, et al. Using scaffolds as drug delivery systems to treat bone tumor. Nanotechnology. 2022;33(21):212002. |
| [1] | Wen Xinghua, Ding Huanwen, Cheng Kai, Yan Xiaonan, Peng Yuanhao, Wang Yuning, Liu Kang, Zhang Huiwu. Three-dimensional finite element model analysis of intramedullary nailing fixation design for large femoral defects in Beagle dogs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1371-1376. |
| [2] | Tang Liang, Li Xiheng, Niu Ruijuan, Li Xinyue, Zou Xinying, Mao Tianjiao, Li Jiang. Naringin regulates the function of RAW264.7 macrophages to affect the osteogenic differentiation of MC-3T3-E1 cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1205-1210. |
| [3] | Zhang Tingting, Liu Juan, Zhang Xu. Bioactivity of phase-transition lysozyme for surface modification of zirconia all-ceramic implant material mediating hydroxyapatite coating [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1043-1049. |
| [4] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [5] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [6] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [7] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| [8] | Xu Yan, Li Ping, Lai Chunhua, Zhu Peijun, Yang Shuo, Xu Shulan. Piezoelectric materials for vascularized bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1126-1132. |
| [9] | Zhang Min, Zhang Xiaoming, Liu Tongbin. Application potential of naringin in bone tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 787-792. |
| [10] | Zhang Lichen, Chen Liang, Gu Yong. Inorganic ion bionic periosteum regulates immune microenvironment to promote bone repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 346-353. |
| [11] | Li Rui, Liu Zhen, Guo Zige, Lu Ruijie, Wang Chen. Aspirin-loaded chitosan nanoparticles and polydopamine modified titanium sheets improve osteogenic differentiation [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 374-379. |
| [12] | Han Tao, Hao Jianqiang, Li Wenbo, Shi Jie, Gao Qiuming. Advantages and problems of antibiotic-loaded bone cements for bone and joint infections [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 470-477. |
| [13] | Cheng Kang, Wang Bin, Tu Zhenxing, Wang Zixin, Zheng Yongxin, Tian Yuqing, Yang Xiao. Relationship of the morphology of three types of callus in the extension area after tibial bone transfer with bone healing [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(27): 4373-4378. |
| [14] | Zhou Hongxing, Zhang Baojian, Yuan Xiangsheng. Recent evaluation of individual biological acetabular cup application in hip revision [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(22): 3521-3525. |
| [15] | Qi Junqiang, Guo Chao, Niu Dongyang, Wang Haotian, Xiao Bing, Xu Guohua. Characteristics and application of bone repair materials of metal ion doped hydroxyapatite [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(21): 3415-3422. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||