Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (3): 453-460.doi: 10.12307/2023.048
Previous Articles Next Articles
Influencing factors and mechanism of nanoparticle renal targeting
Li Zhen1, 2, Liu Hongbao2
- 1Shaanxi University of Traditional Chinese Medicine, Xianyang 712046, Shaanxi Province, China; 2Department of Nephrology, Tang Du Hospital of Air Force Military Medical University, Xi’an 710038, Shaanxi Province, China
-
Received:2021-06-28Accepted:2021-08-04Online:2023-01-28Published:2022-06-01 -
Contact:Liu Hongbao, Associate chief physician, Associate professor, Master’s supervisor, Department of Nephrology, Tang Du Hospital of Air Force Military Medical University, Xi’an 710038, Shaanxi Province, China -
About author:Li Zhen, Master candidate, Shaanxi University of Traditional Chinese Medicine, Xianyang 712046, Shaanxi Province, China; Department of Nephrology, Tang Du Hospital of Air Force Military Medical University, Xi’an 710038, Shaanxi Province, China -
Supported by:Innovation and Development Plan of Tang Du Hospital, No. 2021QYJC-001 (to LHB); Innovation and Development Fund of Tang Du Hospital, No. 2019QYTS003 (to LHB)
CLC Number:
Cite this article
Li Zhen, Liu Hongbao. Influencing factors and mechanism of nanoparticle renal targeting[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 453-460.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

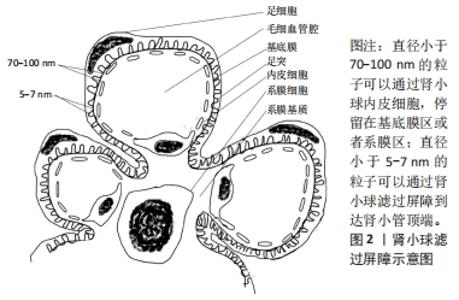
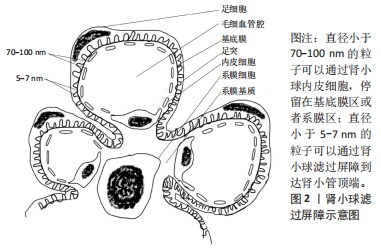
肾小球内皮细胞是肾小球滤过屏障的第一部分,位于肾小球毛细血管壁,它们的特征是有穿孔结构。这些穿孔具有70-100 nm较大直径[14-15],没有被横膈膜封闭,使得较大的分子能够相对容易地穿过肾小球内皮[16]。下一个屏障是肾小球基底膜,是肾小球的主要过滤屏障,是一种网状结缔组织,表面包含直径为4-10 nm的网格[17],由外层、致密层和内部层板组成。中层为致密层,富有带负电荷的涎酸蛋白,内外两层密度较稀,称疏松层,富含阴离子硫酸肝素,构成肾小球的电荷屏障,有利于正电荷载体系统穿过肾小球滤过屏障[18-19]。肾小球的最后一道屏障由足突细胞组成,足细胞是终末分化细胞,通过稀疏的足突附着于基底膜上,有多种裂隙膜蛋白相互插入构成了肾小球滤过屏障的分子筛[14],是防止中、大分子量蛋白质漏出的重要分子屏障。这些狭缝隔膜决定了肾小球过滤的大分子和颗粒的尺寸上限。在正常生理条件下,水动力直径高达5-7 nm的颗粒可以通过肾小球过滤,到达近端管状细胞的顶端。 肾小管由近端小管、细段、远端小管及连接小管组成,其中发挥重要功能的是近端小管,负责血液和尿液之间内源性和外源性物质的主动转运。在近端管状细胞的顶端侧主要表达巨蛋白和立方蛋白[20]。此外,叶酸受体和葡萄糖转运蛋白等也在近端管状细胞的顶膜上表达[21-22],这些转运蛋白主要参与葡萄糖、肽、神经递质和一些大蛋白质分子等小分子的摄取[23-24]。利用这种特性通过受体介导的胞吞作用向近端管状细胞输送药物似乎是一种有效的方法。最后在肾小球毛细血管间有系膜组织,包括系膜细胞和系膜基质。系膜细胞可以维持肾小球微血管床的结构完整性,主要负责生长和平衡肾小球基质,调节过滤表面积和清除免疫复合物[25]。因此,将药物输送到系膜细胞以治疗各种肾脏疾病是有用的[26]。直径大于基底膜孔径、小于肾小球内皮细胞孔径(70-100 nm)的纳米药物可以穿过内皮细胞,积聚在基底膜和系膜组织中[27]。 2.2 纳米药物的特点 纳米材料可以粗略地定义为在1-1 000 nm尺寸范围内的工程大分子[28],所涉及的材料类型很广泛,可包括脂质体、改性生物分子、聚合物、金属和半导体等。因为纳米材料灵活可变的结构和可修饰性,可作为载体通过局部组织渗透效应被动地靶向积聚在特定组织,或者通过与特定的受体、配体结合,主动靶向目标部位发挥作用。纳米药物不仅可以靶向积聚在目标部位,而且可以控制药物代谢动力学,使药物在目标部位缓慢释放。 虽然纳米药物在靶向肿瘤治疗中取得了可观的前景,但是在非肿瘤肾脏疾病方面还处于早期阶段。虽然已经有研究表明有些纳米载药系统可以靶向输送药物到肾脏,但只是在动物实验阶段,而且除过在肾脏,药物在其他器官分布也增多,观察短时间内没有毒副反应,不清楚是否更长时间有慢性毒副反应,所以研究更精准靶向肾脏,长期安全有效的肾靶向载药系统尤其重要。该篇文章综述了目前研究中有潜力的肾靶向纳米载药系统的影响因素及其可能机制和在非肿瘤肾脏疾病中的研究进展,为后面研究者合成有效可行靶向肾脏的纳米药物递送系统提供参考。 2.3 纳米药物肾靶向影响因素和机制 "


2.3.1 被动靶向 尺寸:一般认为直径大于1 000 nm的粒子主要累积在肺中[29],而大于100 nm小于1 000 nm的颗粒可以与血管上的内皮细胞相互作用,主要被肝脏和脾脏捕获,很少分布到肾脏[30-31];直径小于100 nm,大于5-7 nm的粒子一般停留在肾系膜区 [32]。 与前面的观点一致,GUO等[33]研究发现3种尺寸白蛋白纳米粒子(75,95,130 nm)的3种不同粒径都很容易通过内皮开窗进入小鼠肾脏的系膜空间,经过比较,95 nm显示出明显更高的系膜细胞定位。这可能是因为肾小球滤过随着纳米粒子大小的减小而增加,一定大小的纳米粒子和近端小管亲和力之间达到最佳平衡,表现为最大的肾靶向性。 表面电荷:因为肾小球的电荷屏障,正电荷更容易排出体外,而负电荷容易在肾脏中积累。LIANG等[34]采用带负电荷量子点(约等于3.7 nm)作为模型体系,发现在系膜细胞中积累,相反,类似尺寸(约等于5.67 nm)的阳离子量子点在注射后不久容易排泄到尿液中。同样BORGMAN等[35]比较了带负电荷生物素化甲基丙烯酰胺共聚物与非生物素化甲基丙烯酰胺共聚物在大鼠中的生物分布,生物素化甲基丙烯酰胺共聚物导致肾脏积累显著增加。研究发现生物素化负电荷侧基团的存在在肾脏积累中起着主要作用[36-37]。 2.3.2 主动靶向 通过在纳米载体表面修饰相应的适配体、抗体、或者肽,载药系统通过肾小球滤过屏障,与肾小管上的相应配体特异性结合,以内吞的方式靶向肾脏发挥作用。 适配体:叶酸受体在肾近端管状上皮细胞中高水平表达,在这些结合物的肾摄取中起着关键作用[38],SANDOVAL等[39]合成叶酸偶联物(与叶酸受体结合),增加肾脏的吸收,在肾囊肿中的应用,还体现出显著降低的肾外效应。日本血凝病毒可以和肾小管周围毛细血管内皮细胞表面唾液酸受体结合,所以掺入紫外线灭活的日本血凝病毒的脂质体也被用于调节肾小球反应以治疗肾脏疾病,具有明显的肾脏靶向性。多黏菌素B对这些纳米颗粒进行表面修饰可以有效地靶向巨蛋白受体,所以多黏菌素B修饰的聚乙烯亚胺/核酸复合物,对肾小管有选择性,靶向肾脏[40]。 抗体片段:血管黏附分子1是一种在炎症激活的足细胞上表达的细胞表面受体[41],被证明是通过网格蛋白介导的内吞作用被细胞内化的,促进受体介导的纳米药物载体内吞作用[31]。VISWESWARAN等[42]使用脂质纳米载体与抗血管黏附分子1抗体结合,将有效的免疫抑制剂雷帕霉素传递给足细胞。与非靶向抗体修饰的雷帕霉素负载的脂质纳米载体相比,抗体修饰的雷帕霉素负载的脂质纳米载体细胞的进入更有效。肾脏病理状态下,表皮生长因子受体和转化生长因子β受体表达增加,它们定位于近端肾小管细胞的基底外侧膜[43],合成抗表皮生长因子受体、抗转化生长因子β受体修饰的载体系统不仅可以传递药物输送到近端肾小管细胞,而且针对参与疾病过程的生长因子受体产生抑制作用。 肽:已经发现的两种肽片段,分别为肾靶向肽和3K肽,被证明在肾小管中能特异性积累,其中肾靶向肽是一种理想的肾脏特异性载体,已被证明能有效识别和促进肾细胞的内吞作用[44-46],用于药物传递和肾脏疾病的治疗[47]。如WANG等[44]制备肾靶向肽修饰的脂质层的大黄酸载脂纳米粒子对链脲佐菌素诱导的糖尿病肾病小鼠具有良好的肾靶向分布和治疗作用。3K肽被研究在共轭环丙沙星,靶向用于泌尿道感染[48],减轻耐药,提高治疗效果。 2.3.3 增加循环半衰期 除过被动靶向和主动靶向外,延长药物在体内循环半衰期的方式也是增加药物在目标部位积聚的潜在策略。例如:形状、红细胞膜涂层、水溶性和脂溶性、韧性和刚度、密度等影响纳米粒子在体内循环半衰期的因素。 形状:ALIDORI等[49]研究了具有高纵横比的碳纳米管(10 nm×45 nm)比球形纳米粒子具有更长的循环半衰期及更高的给药效率。与此结论一致的还有GENG等[50]之前研究的丝状纳米胶束(直径20-60 nm)在循环中的持续时间比类似的球形聚合体长约10倍,长达1周。推测可能机制是较大纵横比形状的纳米粒子实现内小体/溶酶体逃逸,并通过圆柱形核孔,实现核靶向。最新JIANG等[51]研究发现一种DNA折纸纳米材料(长方形、三角形和管状排列),优先聚集在健康小鼠和横纹肌溶解诱发急性肾损伤的小鼠肾脏中,而长方形DNA折纸纳米材料其功效与抗氧剂相似,还具有肾保护作用,改善肌注甘油所致的横纹肌溶解急性肾损伤,具有良好的生物相容性和免疫原性。推测可能原因是紧凑折叠的结构暴露酶/蛋白减少,肝脏隔离,折纸纳米结构带负电形成蛋白质晕圈机会减少,减少在脾脏、肝脏的摄取,改善肾脏排泄并积累。 水溶性和脂溶性:PONNUSWAMY等[52]在2017年的研究显示,增加DNA纳米结构的脂质聚乙二醇涂层也可以提高药物生物利用度,从而为有效靶向提供更长的时间窗[53]。例如肾管靶向肽改性聚乙酸羟基乙酸共聚物经聚乙二醇修饰可以靶向输送积雪草酸至肾脏。还有HU等[54]研究显示聚乙二醇修饰的锡亚酸地塞米松系统更多地积累在小鼠急性肾损伤模型肾脏中,表现出更好的治疗效果。纳米粒子表面修饰聚乙二醇是最常见的亲水性,增加肾靶向的方式。聚乙二醇可以被共轭或吸收到纳米粒子表面,形成电晕,从而提供空间稳定,并具有防止蛋白质吸收等“隐身”特性,可避免网状系统、调理效应对纳米粒子的清除,增加在血液中的循环时间[55-56]。聚乙二醇在颗粒表面的链长、形状和密度是影响纳米粒子表面亲水性和吞噬性[57-58]。相反,原始的碳纳米管表现出明显的网状内皮系统摄取取向,这可归因于碳原子的亲脂性[59]。 柔韧性与刚性:OLTRA等[60]比较柔性纤丝细胞和聚乙二醇化聚合物的体内留存时间,研究表明纤丝细胞能够循环长达1周,这比聚乙二醇化聚合物能够循环的时间长几倍。在体外将荧光标记的胶束与人源巨噬细胞一起孵育,显微镜图像显示,只有短于2.5 μm的丝状细胞被细胞内化,长丝状细胞没有检测到荧光。推测柔性长丝状体细胞逃避巨噬细胞的吞噬机制可能是,丝状细胞与流体对齐,柔性尾部逃避巨噬细胞的接触,并被水动力剪切力所吸引[60],这样流体动力剪切可以将它们从吞噬细胞上拉下来,从而逃脱内化,增加在目标器官的积累。所以柔性的长丝状细胞因为尾部逃逸有更长的血液循环时间,在目标部位有更多的积累。 红细胞膜涂层:PIAO等[61]将天然红细胞膜包覆在金纳米颗粒上,与聚乙烯基吡咯烷酮包覆的金纳米颗粒相比,其体内血液循环寿命增加了近10倍,显著改善不良治疗效果。有研究表明红细胞膜涂层聚乙酸羟基乙酸共聚物纳米颗粒材料显示出比涂有聚乙二醇的对应纳米颗粒更长的循环半衰期[62-63]。可能机制是完整的红细胞膜可以被涂层在纳米粒子表面,实现仿生功能化模拟,逃避免疫吞噬,延长循环时间。 密度:SLACK等[64]发现纳米粒子的密度也被证明会影响生物分布。TANG等[65]发现注射纳米粒子后2 h,肾脏清除效率随颗粒密度的降低呈指数增长。这种行为可能起源于密度依赖的层流血流中明显边缘的边缘作用所致,其中较高密度的纳米粒子循环更慢,导致较慢的肾清除,在靶部位停留时间更长。 被动靶向主要是通过改变纳米载体的自身物理化学性质,主要通过孔径屏障或电荷屏障,被动地运输到肾脏,主要是肾小球系膜区。主动靶向通过纳米粒子与相应的受体,抗体、配体、蛋白质片段相连通过肾小球滤过屏障,在近端肾小管与相应受体特异性结合,以内吞的方式运输到肾脏。除过主动靶向和被动靶向,其他如形状、水溶性和脂溶性、柔韧性与刚性、红细胞膜涂层等影响药物在体内的循环半衰期的因素也间接影响药物在肾脏的积聚。 2.3.4 其他因素 除过以上叙述的影响因素外,剂量和给药途径、其他修饰如丝氨酸功能化修饰也影响在肾脏部位的积聚。WILLIAMS等[66]发现分别给小鼠注射3个剂量(5,25,50 mg/kg)的中尺度纳米粒子,5 mg/kg剂量在肾脏蓄积比例比50 mg/kg剂量高,但不如25 mg/kg的肾脏蓄积量高,25 mg/kg的剂量肾脏的蓄积量比其他器官的大26-94倍。猜测不同剂量导致肾靶向摄取不同的因素可能与密度影响肾靶向的机制相似。同时他们还发现不同给药途径,灌胃、腹腔内给药、眶后静脉、尾静脉给药、皮下给药途径,最有效的肾定位发生在静脉给药途径,因为其他给药途径可能会因颗粒相对较大而阻碍进入血管[67]。这项研究主要针对的是直径300-400 nm的中尺度纳米粒子,所以是不是在所有尺寸纳米粒子中最有效的给药方式都是静脉给药还有待进一步研究。MATSUURA等[68]合成了用丝氨酸功能化的聚酰胺-胺树枝状大分子,他们报告表明,静脉注射丝氨酸修饰后的树枝状大分子,这些纳米载体的积累量比注射没有修饰的或用其他氨基酸修饰树枝状大分子多约3倍,所以丝氨酸修饰的聚酰胺-胺树枝状大分子可以作为治疗肾脏疾病的可能纳米载体。其靶向肾脏的机制是经过丝氨酸修饰后,肝脾内皮系统对药物载体的摄取减少,从而增加了药物在肾脏的靶向积聚。 因为肾小球的孔径屏障和电荷屏障,影响被动靶向因素主要是尺寸和电荷;主动靶向在肾脏靶向方式中占主要地位,与特异性性受体结合是主动靶向的基础;除过主动靶向和被动靶向外,影响循环半衰期长短的因素也是影响靶向的因素之一,见表1。 "


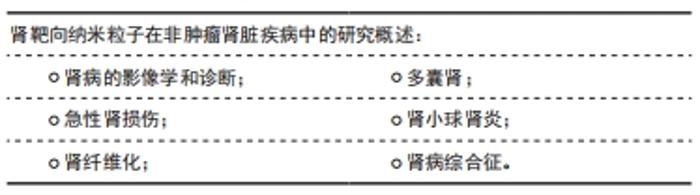
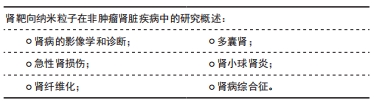
2.4.1 肾病的影像学和诊断 用于磁共振的树突状纳米材料已被证明在小鼠模型中肌酐升高之前可以检测到与急性肾损伤一致的变化,所以在早期诊断急性肾损伤有很大的潜力。HAUGER等[69]研究表明超小超顺磁氧化铁纳米粒子被用于磁共振成像,区分肾脏疾病和阻塞性肾病,以及识别肾排斥的迹象,鉴别各种肾病。SHIRAI等[70]合成具有荧光抗CD11的二氧化硅钠也被用作阻塞性泌尿系统疾病动物模型中炎症和纤维化的成像工具。MAROM等[71]基于金纳米粒子的传感器被用来测量62例慢性肾脏病及早期受试者呼出的空气中的差异,被用于慢性肾脏病的早期诊断有很大的潜力。 2.4.2 急性肾损伤 HU等[72]制备了靶向肾小管上皮细胞的透明质酸受体修饰的透明质酸-姜黄素聚合物,显著提高了姜黄素在肾脏的积聚水平,透明质酸-姜黄素聚合物的在肾脏积聚的水平是游离姜黄素的13.9倍,静脉给药24 h后可有效改善大鼠的急性肾损伤模型。横纹肌溶解引起的肾损害的特征是早期炎症反应,高浓度的M1巨噬细胞(促炎性)和部分向M2表型(抗炎性)分化,后期CD163膜受体表达增加,因此Rubio-Navarro开发了用抗CD163抗体修饰的镀金氧化铁纳米颗粒,其特异性靶向受损肾脏中的CD163,减轻了急性肾损伤[73]。同时CD163被证明存在于人横纹肌溶解急性肾损伤,所以被应用在人急性肾损伤中具有深远的前景。在缺血再灌注肾损伤后,在肾损伤部位局部产生大量活化的凝血酶。因此,CHEN等[74]提出设计全氟化碳纳米颗粒与凝血酶抑制剂外部结合,这种结合使得纳米粒子能够定位到小鼠受损的肾区,减少病变。还有MANNE等[75]研究了在腹膜炎诱导的大鼠急性肾损伤模型中,氧化铈纳米粒子靶向肾脏降低了急性肾损伤的损伤程度。 2.4.3 肾纤维化 血小板衍生生长因子β受体在肾纤维化组织中高度上调。TEEKAMP等[76]研究表明通过皮下注射给药的聚合物微粒,该微粒能够释放由血小板衍生生长因子(配体)结合到人血清白蛋白上的缀合物,该缀合物可用作抗纤维化化合物的载体。从微粒中释放后,与非靶向缀合物相比,靶向缀合物能够在血浆中循环,并最终以高度特异性的方式到达小鼠纤维化肾脏。TAN等[77]合成了金纳米粒子用D,L-硫辛酸稳定,通过-COOH残基和谷胱甘肽上的NH2残基之间的交联反应,这些颗粒被谷胱甘肽功能化,腹腔注射金纳米粒子后,观察到这些纳米粒子的在近端肾小管高效靶向性。这一事实可归因于其表面的谷胱甘肽与肾近端小管细胞中表达的各种关键受体(如燕窝素和巨蛋白)之间的相互作用。QIAO等[78]合成的由金属离子、多柔比星和儿茶酚衍生的低分子量壳聚糖组成的用于治疗肾纤维化的纳米复合物能够定向纳米载体的作用并具有高的肾保留能力。这一发现表明肾脏靶向作用可能是由壳聚糖胺引起的,壳聚糖纳米颗粒进入肾脏细胞是由于巨蛋白介导的胞吞作用[79]。 2.4.4 多囊肾 TSAI等[80]研究表明,抗miRNA质粒和氧化铁/海藻酸钠纳米粒与抗肾抗体结合。这些纳米复合材料导致肾小管细胞的有效肾靶向,是治疗多囊肾疾病是新的潜在疗法。 2.4.5 肾小球肾炎 在炎症条件下,足细胞过度表达血管细胞黏附分子1受体,而抗血管细胞黏附分子1抗体的使用与纳米粒子的结合使得脂质纳米载体的肾脏靶向治疗成为可能[42]。 2.4.6 肾病综合征 肾病综合征表现为大量蛋白尿,与足细胞关系密切,足细胞大量表达白蛋白的特异性受体。因此通过酰胺化反应制备了负载甲强龙的白蛋白纳米粒。这些纳米颗粒对表达白蛋白的特异性受体的人足细胞的亲和力是对照细胞的36倍,明显增加了肾脏的给药效率。但是,这些纳米载体在静脉给药后24 h不仅在肾脏中积累,而且还保留在肝脏中[81],见表2。"

| [1] KELLUM JA, BELLOMO R, RONCO C. Definition and classification of acute kidney injury. Nephron Clin Pract. 2008;109(4):c182-c187. [2] XIAO Y-Q, CHENG W, WU X, et al. Novel risk models to predict acute kidney disease and its outcomes in a Chinese hospitalized population with acute kidney injury. Sci Rep. 2020;10(1):15636. [3] RILEY S, DIRO E, BATCHELOR P, et al. Renal impairment among acute hospital admissions in a rural Ethiopian hospital. Nephrology (Carlton). 2013;18(2):92-96. [4] JHA V, GARCIA-GARCIA G, ISEKI K, et al. Chronic kidney disease: global dimension and perspectives. Lancet. 2013;382(9888):260-272. [5] CORESH J, SELVIN E, STEVENS L A, et al. Prevalence of chronic kidney disease in the United States. JAMA. 2007;298(17):2038-2047. [6] HAAS M, MOOLENAAR F, MEIJER DKF, et al. Specific drug delivery to the kidney. Cardiovasc Drugs Ther. 2002;16(6):489-496. [7] MURO S, GARNACHO C, J A,. et al. Control of endothelial targeting and intracellular delivery of therapeutic enzymes by modulating the size and shape of ICAM-1-targeted carriers. Mol Ther. 2008;16(8):1450-1458. [8] CHEN Z, PENG H, ZHANG C. Advances in kidney-targeted drug delivery systems. Int J Pharm. 2020;587:119679. [9] 郭苗苗,张芸,徐丹,等.肾靶向纳米给药系统的研究进展[J].医药导报, 2012,31(6):768-770. [10] PETI-PETERDI J, KIDOKORO K, RIQUIER-BRISON A. Novel in vivo techniques to visualize kidney anatomy and function. Kidney Int. 2015;88(1):44-51. [11] ICHIMURA K, STAN RV, KURIHARA H, et al. Glomerular endothelial cells form diaphragms during development and pathologic conditions. J Am Soc Nephrol. 2008;19(8):1463-1471. [12] MINER JH. The glomerular basement membrane. Exp Cell Res. 2012; 318(9):973-978. [13] ARMELLONI S, CORBELLI A, GIARDINO L, et al. Podocytes:recent biomolecular developments. Biomol Concepts. 2014;5(4):319-330. [14] AKILESH S, HUBER T B, WU H, et al. Podocytes use FcRn to clear IgG from the glomerular basement membrane. P Natl Acad Sci USA. 2008; 105(3):967-972. [15] LEVIDIOTIS V, POWER DA. New insights into the molecular biology of the glomerular filtration barrier and associated disease. Nephrology (Carlton). 2005; 10(2):157-166. [16] SATCHELL SC, BRAET F. Glomerular endothelial cell fenestrations: an integral component of the glomerular filtration barrier. Am J Physiol-Renal. 2009;296(5): F947-F956. [17] HIRONAKA K, MAKINO H, YAMASAKI Y, et al. Pores in the glomerular basement membrane revealed by ultrahigh-resolution scanning electron microscopy. Nephron. 1993;64(4):647-649. [18] LEWIS E J, XU X. Abnormal glomerular permeability characteristics in diabetic nephropathy: implications for the therapeutic use of low-molecular weight heparin. Diabetes care. 2008;31 Suppl 2:S202-S207. [19] TRYGGVASON K. Unraveling the mechanisms of glomerular ultrafiltration: nephrin, a key component of the slit diaphragm. J Am Soc Nephrol. 1999;10(11):2440-2445. [20] SAITO A, SATO H, IINO N, et al. Molecular mechanisms of receptor-mediated endocytosis in the renal proximal tubular epithelium. J Biomed Biotechnol. 2010;2010:403272. [21] SUBRAMANIAN VS, MARCHANT JS, SAID HM. Apical membrane targeting and trafficking of the human proton-coupled transporter in polarized epithelia. Am J Physiol Cell Physiol. 2008;294(1):C233-C240. [22] WOOD I S, TRAYHURN P. Glucose transporters (GLUT and SGLT): expanded families of sugar transport proteins. Br J Nutr. 2003;89(1):3-9. [23] HAGENBUCH B. Drug uptake systems in liver and kidney:a historic perspective. Clin Pharmacol Ther. 2010;87(1):39-47. [24] TERADA T, INUI KI. Physiological and pharmacokinetic roles of H+/organic cation antiporters (MATE/SLC47A). Biochem Pharmacol. 2008;75(9):1689-1696. [25] ABBOUD HE. Mesangial cell biology. Exp Cell Res. 2012;318(9):979-985. [26] ZHAO JH. Mesangial Cells and Renal Fibrosis. Adv Exp Med Biol. 2019; 1165:165-194. [27] BAGAVANT H, KALANTARINIA K, SCINDIA Y, et al. Novel therapeutic approaches to lupus glomerulonephritis: translating animal models to clinical practice. Am J Kidney Dis. 2011;57(3):498-507. [28] DUNCAN R, GASPAR R. Nanomedicine(s) under the microscope. Mol Pharm. 2011;8(6):2101-2141. [29] AZARMI S, ROA W H, LöBENBERG R. Targeted delivery of nanoparticles for the treatment of lung diseases. Adv Drug Del Rev. 2008;60(8):863-875. [30] CHOI HS, LIU W, MISRA P, et al. Renal clearance of quantum dots. Nat Biotechnol. 2007;25(10):1165-1170. [31] ZUCKERMAN JE, DAVIS ME. Targeting therapeutics to the glomerulus with nanoparticles. Adv Chronic Kidney Dis. 2013;20(6):500-507. [32] CHOI CHJ, ZUCKERMAN J E, WEBSTER P, et al. Targeting kidney mesangium by nanoparticles of defined size. Proc Natl Acad Sci USA. 2011;108(16):6656-6661. [33] GUO L, LUO S, DU Z, et al. Targeted delivery of celastrol to mesangial cells is effective against mesangioproliferative glomerulonephritis. Nat Commun. 2017;8(1):878. [34] LIANG X, WANG H, ZHU Y, et al. Short- and long-term tracking of anionic ultrasmall nanoparticles in kidney. ACS Nano. 2016;10(1):387-395. [35] BORGMAN MP, COLEMAN T, KOLHATKAR RB, et al. Tumor-targeted HPMA copolymer-(RGDfK)-(CHX-A’’-DTPA) conjugates show increased kidney accumulation. J Control Release. 2008;132(3):193-199. [36] KISSEL M, PESCHKE P, SUBR V, et al. Detection and cellular localisation of the synthetic soluble macromolecular drug carrier pHPMA. Eur J Nucl Med Mol Imag. 2002;29(8):1055-1062. [37] PIKE DB, GHANDEHARI H. HPMA copolymer-cyclic RGD conjugates for tumor targeting. Adv Drug Del Rev. 2010;62(2):167-183. [38] MANIL L, DAVIN JC, DUCHENNE C, et al. Uptake of nanoparticles by rat glomerular mesangial cells in vivo and in vitro. Pharm Res. 1994;11(8):1160-1165. [39] SANDOVAL RM, KENNEDY MD, LOW PS, et al. Uptake and trafficking of fluorescent conjugates of folic acid in intact kidney determined using intravital two-photon microscopy. Am J Physiol Cell Physiol. 2004;287(2):C517-C526. [40] OROOJALIAN F, REZAYAN AH, MEHRNEJAD F, et al. Efficient megalin targeted delivery to renal proximal tubular cells mediated by modified-polymyxin Bpolyethylenimine based nano-gene-carriers. Mater Sci Eng C Mater Biol Appl. 2017;79:770-782. [41] RICARD I, PAYET MD, DUPUIS G. VCAM-1 is internalized by a clathrin-related pathway in human endothelial cells but its alpha 4 beta 1 integrin counter-receptor remains associated with the plasma membrane in human T lymphocytes. Eur J Immunol. 1998;28(5):1708-1718. [42] VISWESWARAN GRR, GHOLIZADEH S, RUITERS MHJ, et al. Targeting Rapamycin to Podocytes Using a Vascular Cell Adhesion Molecule-1 (VCAM-1)-Harnessed SAINT-Based Lipid Carrier System. PLoS One. 2015;10(9):e0138870. [43] BAINES RJ, BRUNSKILL NJ. The molecular interactions between filtered proteins and proximal tubular cells in proteinuria. Nephron Exp Nephrol. 2008;110(2):e67-e71. [44] WANG G, LI Q, CHEN D, et al. Kidney-targeted rhein-loaded liponanoparticles for diabetic nephropathy therapy via size control and enhancement of renal cellular uptake. Theranostics. 2019;9(21):6191-6208. [45] WANG J, MASEHI-LANO JJ, CHUNG EJ. Peptide and antibody ligands for renal targeting: nanomedicine strategies for kidney disease. Biomater Sci. 2017;5(8):1450-1459. [46] BIDWELL G L, MAHDI F, SHAO Q, et al. A kidney-selective biopolymer for targeted drug delivery. Am J Physiol Renal Physiol. 2017;312(1):F54-F64. [47] WISCHNJOW A, SARKO D, JANZER M, et al. Renal targeting: peptide-based drug delivery to proximal tubule cells. Bioconjug Chem. 2016;27(4):1050-1057. [48] SHARMA P C, JAIN A, JAIN S, et al. Ciprofloxacin: review on developments in synthetic, analytical, and medicinal aspects. J Enzyme Inhib Med Chem. 2010;25(4):577-589. [49] ALIDORI S, AKHAVEIN N, THOREK DLJ, et al. Targeted fibrillar nanocarbon RNAi treatment of acute kidney injury. J Urol. 2016;196(4):1313-1314. [50] GENG Y, DALHAIMER P, CAI S, et al. Shape effects of filaments versus spherical particles in flow and drug delivery. Nat Nanotechnol. 2007; 2(4):249-255. [51] JIANG D, GE Z, IM HJ, et al. DNA origami nanostructures can exhibit preferential renal uptake and alleviate acute kidney injury. Nat Biomed Eng. 2018;2(11):865-877. [52] PONNUSWAMY N, BASTINGS MMC, NATHWANI B, et al. Oligolysine-based coating protects DNA nanostructures from low-salt denaturation and nuclease degradation. Nat Commun. 2017;8:15654. [53] HE J, CHEN H, ZHOU W, et al. Kidney targeted delivery of asiatic acid using a FITC labeled renal tubular-targeting peptide modified PLGA-PEG system. Int J Pharm. 2020;584:119455. [54] HU JB, KANG XQ, LIANG J, et al. E-selectin-targeted Sialic Acid-PEG-dexamethasone Micelles for enhanced anti-inflammatory efficacy for acute kidney injury. Theranostics. 2017;7(8):2204-2219. [55] DAEMS N, PENNINCKX S, NELISSEN I, et al. Gold nanoparticles affect the antioxidant status in selected normal human cells. Int J Nanomedicine. 2019;14:4991-5015. [56] NIIDOME T, YAMAGATA M, OKAMOTO Y, et al. PEG-modified gold nanorods with a stealth character for in vivo applications. J Control Release. 2006;114(3):343-347. [57] GREF, LüCK, QUELLEC, et al. ‘Stealth’ corona-core nanoparticles surface modified by polyethylene glycol (PEG): influences of the corona (PEG chain length and surface density) and of the core composition on phagocytic uptake and plasma protein adsorption. Colloids Surf B Biointerfaces. 2000;18(3-4):301-313. [58] LI Y, PEI Y, ZHANG X, et al. PEGylated PLGA nanoparticles as protein carriers: synthesis, preparation and biodistribution in rats. J Control Release. 2001;71(2):203-211. [59] CHERUKURI P, GANNON CJ, LEEUW TK, et al. Mammalian pharmacokinetics of carbon nanotubes using intrinsic near-infrared fluorescence. Proc Natl Acad Sci U S A. 2006;103(50):18882-18886. [60] OLTRA NS, NAIR P, DISCHER DE. From stealthy polymersomes and filomicelles to “self” Peptide-nanoparticles for cancer therapy. Annu Rev Chem Biomol Eng. 2014;5:281-299. [61] PIAO JG, WANG L, GAO F, et al. Erythrocyte membrane is an alternative coating to polyethylene glycol for prolonging the circulation lifetime of gold nanocages for photothermal therapy. ACS Nano. 2014;8(10):10414-10425. [62] HU CMJ, ZHANG L, ARYAL S, et al. Erythrocyte membrane-camouflaged polymeric nanoparticles as a biomimetic delivery platform. Proc Natl Acad Sci U S A. 2011; 108(27):10980-10985. [63] LUK BT, HU CM J, FANG RH, et al. Interfacial interactions between natural RBC membranes and synthetic polymeric nanoparticles. Nanoscale. 2014;6(5):2730-2737. [64] SLACK JD, KANKE M, SIMMONS GH, et al. Acute hemodynamic effects and blood pool kinetics of polystyrene microspheres following intravenous administration. J Pharm Sci. 1981;70(6):660-664. [65] TANG S, PENG C, XU J, et al. Tailoring renal clearance and tumor targeting of ultrasmall metal nanoparticles with particle density. Angew Chem Int Ed Engl. 2016;55(52):16039-16043. [66] WILLIAMS RM, SHAH J, TIAN HS, et al. Selective nanoparticle targeting of the renal tubules. Hypertension. 2018;71(1):87-94. [67] BARUA S, MITRAGOTRI S. Challenges associated with penetration of nanoparticles across cell and tissue barriers: a review of current status and future prospects. Nano today. 2014;9(2):223-243. [68] MATSUURA S, KATSUMI H, SUZUKI H, et al. l-Serine-modified polyamidoamine dendrimer as a highly potent renal targeting drug carrier. Proc Natl Acad Sci U S A. 2018;115(41):10511-10516. [69] HAUGER O, DELALANDE C, DEMINIèRE C, et al. Nephrotoxic nephritis and obstructive nephropathy:evaluation with MR imaging enhanced with ultrasmall superparamagnetic iron oxide-preliminary findings in a rat model. Radiology. 2000;217(3):819-826. [70] SHIRAI T, KOHARA H, TABATA Y. Inflammation imaging by silica nanoparticles with antibodies orientedly immobilized. J Drug Targeting. 2012;20(6):535-543. [71] MAROM O, NAKHOUL F, TISCH U, et al. Gold nanoparticle sensors for detecting chronic kidney disease and disease progression. Nanomedicine (Lond). 2012;7(5): 639-650. [72] HU JB, LI SJ, KANG XQ, et al. CD44-targeted hyaluronic acid-curcumin prodrug protects renal tubular epithelial cell survival from oxidative stress damage. Carbohydr Polym. 2018;193:268-280. [73] RUBIO-NAVARRO A, CARRIL M, PADRO D, et al. CD163-macrophages are involved in rhabdomyolysis-induced kidney injury and may be detected by MRI with targeted gold-coated iron oxide nanoparticles. Theranostics. 2016;6(6):896-914. [74] CHEN J, VEMURI C, PALEKAR RU, et al. Antithrombin nanoparticles improve kidney reperfusion and protect kidney function after ischemia-reperfusion injury. Am J Physiol Renal Physiol. 2015;308(7):F765-F773. [75] MANNE NDPK, ARVAPALLI R, NEPAL N, et al. Cerium oxide nanoparticles attenuate acute kidney injury induced by intra-abdominal infection in Sprague-Dawley rats. J Nanobiotechnol. 2015;13:75. [76] TEEKAMP N, VAN DIJK F, BROESDER A, et al. Polymeric microspheres for the sustained release of a protein-based drug carrier targeting the PDGFβ-receptor in the fibrotic kidney. Int J Pharm. 2017;534(1-2):229-236. [77] TAN L, LAI X, ZHANG M, et al. A stimuli-responsive drug release nanoplatform for kidney-specific anti-fibrosis treatment. Biomater Sci. 2019;7(4):1554-1564. [78] QIAO H, SUN M, SU Z, et al. Kidney-specific drug delivery system for renal fibrosis based on coordination-driven assembly of catechol-derived chitosan. Biomaterials. 2014;35(25):7157-7171. [79] GAO S, HEIN S, DAGNAES-HANSEN F, et al. Megalin-mediated specific uptake of chitosan/siRNA nanoparticles in mouse kidney proximal tubule epithelial cells enables AQP1 gene silencing. Theranostics. 2014;4(10):1039-1051. [80] TSAI YC, TENG IL, JIANG ST, et al. Safe nanocomposite-mediated efficient delivery of microrna plasmids for autosomal dominant polycystic kidney disease (ADPKD) therapy. Adv Healthc Mater. 2019; 8(5):e1801358. [81] WU L, CHEN M, MAO H, et al. Albumin-based nanoparticles as methylprednisolone carriers for targeted delivery towards the neonatal Fc receptor in glomerular podocytes. Int J Mol Med. 2017;39(4):851-860. |
| [1] | Liu Hanfeng, Wang Jingjing, Yu Yunsheng. Artificial exosomes in treatment of myocardial infarction: current status and prospects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1118-1123. |
| [2] | Yin Tong, Yang Jilei, Li Yourui, Liu Zhuoran, Jiang Ming. Application of core-shell structured nanofibers in oral tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 766-770. |
| [3] | Tian Xin, Liu Tao, Yang Huilin, He Fan. In vitro evaluation of sustained release Kartogenin by gelatin methacryloyl microspheres for repairing nucleus pulposus degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 724-730. |
| [4] | Zhu Liwei, Wang Jiangyue, Bai Ding. Application value of nanocomposite gelatin methacryloyl hydrogels in different bone defect environments [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 753-758. |
| [5] | Chen Guanting, Zhang Linqi, Wang Xixi, Chen Xu. Autophagy, ferroptosis-related targets and renal function progression in patients with chronic kidney disease: bioinformatics analysis and experimental verification [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5122-5129. |
| [6] | Chen Pinrui, Pei Xibo, Xue Yiyuan. Function and advantages of magnetically responsive hydrogel in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 452-457. |
| [7] | Ilham • Maimaitiabudula, Huang Xiaoxia, Li Luyao, Teng Yong. Polymer-based antibiotic sustained-release carrier in treatment of chronic osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3597-3602. |
| [8] | Liu Chang, Li Linhua, Fu Ping . Research and development of nanoparticles with active targeting ability in nonneoplastic kidney disease [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3603-3608. |
| [9] | Wang Zhaofei, He Guoyun, Tian Fangcan, Li Guangfeng, Cao Zhonghua, Liu Xiangfei. Antibacterial properties of silver nanoparticle-coated stainless steel prepared via active screen plasma surface modification in vitro [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(22): 3464-3471. |
| [10] | Wang Jinlei, Li Ke, Zhao Liang. Platelet-camouflaged silver nanoparticle hydrogel accelerates wound healing in type 1 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2659-2666. |
| [11] | Wang Xindong, Liang Chengzhi, Zhang Yongxian. Functional characteristics and clinical applications of MXene nanoparticles in wound healing [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2739-2746. |
| [12] | Zhang Shuzhi, Qu Pengfei, Han Junquan, Wang Hong. Research and application of electrospinning drug delivery systems containing traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2759-2765. |
| [13] | Guo Yuxin, Wang Hao, Li Mingqi, Chen Yueying, Pan Juhong, Huang Xin, Wang Zhiwen, Zhou Qing. Ultrasound-optimized hydrogel scaffold used to promote transdermal delivery of gold nanoparticles [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(15): 2330-2337. |
| [14] | Liu Chang, Zhang Wen, Zhu Can, Sun Jie, Ding Yicheng, Shi Qin. Inhibitory effect of bovine serum albumin-chitosan nanoparticles loaded with EPZ6438 on osteosarcoma [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(10): 1512-1518. |
| [15] | Bei Ying, Li Wenjing, Li Meiyun, Su Meng, Zhang Jin, Huang Yu, Zhu Yanzhao, Li Jiali, Wu Yan. Prussian blue nanoparticles promote wound healing of diabetic skin [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(10): 1526-1532. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||