中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (29): 7663-7672.doi: 10.12307/2026.262
• 组织构建综述 tissue construction review • 上一篇 下一篇
非编码RNA在阿尔茨海默病中的作用及中医药干预
王绍娜1,李瑞洋1,安兰花2,张金生1,2
- 1河南中医药大学,河南省郑州市 450046;2河南中医药大学第三附属医院,河南省郑州市 450000
-
收稿日期:2025-10-15修回日期:2025-12-06出版日期:2026-10-18发布日期:2026-03-06 -
通讯作者:张金生,博士,主任医师,教授,博士生导师,河南中医药大学,河南省郑州市 450046;河南中医药大学第三附属医院,河南省郑州市 450000 -
作者简介:王绍娜,女,1996年生,河南省郑州市人,汉族,河南中医药大学在读博士,主要从事中医药防治心脑血管疾病研究。 -
基金资助:中原科技创新领军人才项目(214200510022),项目负责人:张金生;河南省中医药科学研究专项(2022ZY1110),项目负责人:张金生;河南省科技攻关项目(232102311213),项目负责人:安兰花
Role of non-coding RNAs in Alzheimer’s disease and treatment with traditional Chinese medicine
Wang Shaona1, Li Ruiyang1, An Lanhua2, Zhang Jinsheng1, 2
- 1Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China; 2The Third Affiliated Hospital of Henan University of Chinese Medicine, Zhengzhou 450000, Henan Province, China
-
Received:2025-10-15Revised:2025-12-06Online:2026-10-18Published:2026-03-06 -
Contact:Zhang Jinsheng, PhD, Chief physician, Professor, Doctoral supervisor, Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China; The Third Affiliated Hospital of Henan University of Chinese Medicine, Zhengzhou 450000, Henan Province, China -
About author:Wang Shaona, MD candidate, Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China -
Supported by:Zhongyuan Science and Technology Innovation Leading Talent Program, No. 214200510022 (to ZJS); Special Project for Scientific Research on Traditional Chinese Medicine of Henan Province, No. 2022ZY1110 (to ZJS); Science and Technology Research and Development Project of Henan Province, No. 232102311213 (to ALH).
摘要:
文题释义:
阿尔茨海默病:是一种慢性、进行性神经退行性疾病,以大脑皮质和海马区神经元进行性丢失为病理基础,临床表现为认知功能障碍和行为损害,是痴呆的主要类型。目前其发病率持续上升,严重影响患者的生活质量,给家庭和社会带来沉重负担,成为重要的全球公共卫生挑战。
非编码RNA:是一种缺乏编码能力的功能性RNA分子,在基因组中广泛存在且转录丰度较高;并可通过调控转录过程、影响mRNA稳定性、修饰染色质状态以及参与信号转导等多种机制,在表观遗传调控、基因表达网络维持及细胞功能稳态中发挥关键作用。
背景:阿尔茨海默病病因多样且发病机制复杂,至今尚未完全阐明。近年来,非编码RNA被证实在调控β-淀粉样蛋白异常沉积、Tau蛋白过度磷酸化、神经炎症激活、线粒体功能障碍及突触损伤等过程中发挥关键作用,为阐释疾病机制和药物研发提供了新视角。同时,中医药通过调控非编码RNA网络展现了多通路干预优势。
目的:综述近年来调节性非编码RNA和转运RNA在阿尔茨海默病病理机制中的相关研究,总结中药单体、复方及针灸调控不同非编码RNA发挥抗阿尔茨海默病的现状,以期为今后阿尔茨海默病临床治疗策略优化及新型药物研发提供理论依据与方向参考。
方法:以“非编码RNA、微小RNA、长链非编码RNA、环状RNA、转运RNA、阿尔茨海默病、中医药”为中文检索词,以“ncRNA,miRNA,lncRNA,circRNA,tRNA,Alzheimer’s disease,traditional Chinese Medicine”为英文检索词,检索中国知网和PubMed数据库2015年1月至2025年7月发表的相关文献,根据纳入及排除标准最终纳入101篇文献进行综述分析。
结果与结论:①阿尔茨海默病的发生与发展源于一个多因素相互关联的病理网络,主要包括β-淀粉样蛋白沉积、Tau蛋白过度磷酸化、神经炎症激活、线粒体功能障碍、氧化应激、突触结构与功能异常以及钙稳态失衡等关键机制,这些因素之间亦可彼此交织、协同促进疾病进程;②多种调节性非编码RNA如微小RNA、长链非编码RNA、环状RNA以及管家非编码RNA中的转运RNA可通过不同层面调控上述病理过程,进而影响疾病进展;③诸多中药单体活性成分如小檗碱、梓醇、三七总皂苷、人参皂苷Rg1、β-细辛醚、雷公藤内酯醇、丹参酮IIA;中药复方如安神定志方、调心方、补肾填髓方以及针刺和艾灸等中医疗法均能通过上调或下调特定非编码RNA的表达,干预阿尔茨海默病的多个病理环节,发挥神经保护作用,从而延缓疾病的发生与发展。
https://orcid.org/0009-0006-4819-1610(王绍娜);https://orcid.org/0000-0002-8720-9055(张金生)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
王绍娜, 李瑞洋, 安兰花, 张金生, . 非编码RNA在阿尔茨海默病中的作用及中医药干预[J]. 中国组织工程研究, 2026, 30(29): 7663-7672.
Wang Shaona, Li Ruiyang, An Lanhua, Zhang Jinsheng. Role of non-coding RNAs in Alzheimer’s disease and treatment with traditional Chinese medicine[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(29): 7663-7672.
2.2 调节性非编码RNA在阿尔茨海默病中的相关研究进展 在阿尔茨海默病中,多种非编码RNA通过竞争性内源RNA机制构成一个高度互联的动态调控网络[22]。该网络以微小RNA为中心,长链非编码RNA、环状RNA及mRNA等分子通过共享的微小RNA反应元件相互竞争性结合微小RNA,起到“分子海绵”的作用,从而削弱微小RNA对其靶基因mRNA的转录后抑制,最终放大或缓冲对下游基因的调控效应[23]。竞争性内源RNA网络不仅涵盖RNA分子间的竞争性结合及微小RNA介导的基因沉默,还整合了表观遗传修饰和蛋白质相互作用等多层次机制,共同调控疾病发生与发展中的关键病理过程。此外,竞争性内源RNA交互具有一对多、可逆和上下依赖的特性:单个竞争性内源RNA可调控多个微小RNA活性,单个微小RNA也可靶向多个转录本,多个微小RNA还可靶向同一转录本;同时,微小RNA本身可通过反馈环路影响竞争性内源RNA的表达,增强了基因调控的可塑性与特异性[24]。这种多维、级联的互作模式不仅显著提高了基因表达网络的复杂性
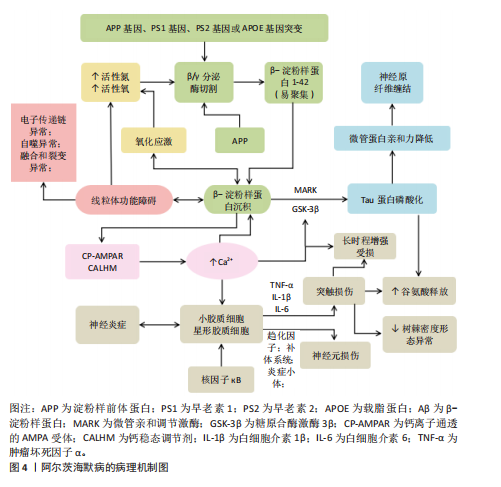
与稳健性,也有助于更全面了解其在阿尔茨海默病多因素、多通路互作的病因机制。
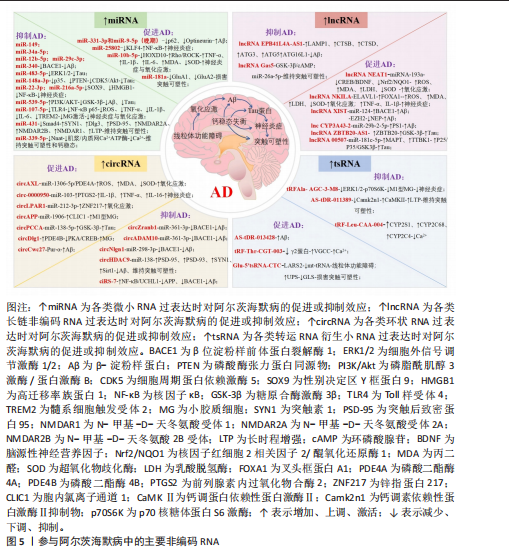
2.2.1 微小RNA与阿尔茨海默病 微小RNA是一类内源性短链非编码RNA,可通过靶向结合mRNA的3’-UTR介导mRNA降解或翻译抑制,从而在转录后水平调控基因表达。其表达水平受到严格调控,并可灵敏响应细胞内外环境变化[25]。在阿尔茨海默病患者中,血液和脑脊液内的微小RNA呈现特异性表达变化,因此具备作为疾病诊断生物标志物的潜力;同时因其固有的多靶点属性使微小RNA能直接或间接影响多个阿尔茨海默病关键基因表达,进而参与多种病理过程的调控。
(1)调控β-淀粉样蛋白代谢和Tau蛋白磷酸化:微小RNA失调与调节β-淀粉样蛋白生成、切割、清除及Tau蛋白磷酸化的关键基因有关。在多种阿尔茨海默病模型中,miR-149、miR-34a-5p和miR-12b-5p、miR-29c-3p、miR-340表达均下调[26-29],并与β位淀粉样前体蛋白裂解酶1水平呈负相关;其过表达可靶向抑制β位淀粉样前体蛋白裂解酶1表达,减少β-淀粉样蛋白沉积。而阿尔茨海默病模型小鼠中miR-331-3p和miR-9-5p的表达呈现明显的病程依赖性动态变化[30]:在疾病早期,两者下调可解除对自噬受体蛋白sequestosome 1 (p62/SQSTM1)和Optineurin(OPTN)的抑制,从而增强自噬活性促进β-淀粉样蛋白清除,发挥神经保护作用;而在晚期,两者表达上调则过度抑制自噬,导致自噬能力受损引发β-淀粉样蛋白堆积。因此抑制两者表达可增强自噬流、促进β-淀粉样蛋白清除能力并改善认知功能,提示其具有作为分期治疗靶点的潜力。此外,miR-539-5p在APP/PS1小鼠模型中下调并与淀粉样前体蛋白水平呈负相关,恢复其表达可通过激活磷脂酰肌醇3激酶/蛋白激酶通路抑制糖原合酶激酶3β活性,从而减少β-淀粉样蛋白生成、抑制Tau蛋白过度磷酸化并改善学习记忆功能障碍,表明miR-539-5p-磷脂酰肌醇3激酶/蛋白激酶B/糖原合酶激酶3β轴可能是阿尔茨海默病的潜在治疗靶点[31]。有研究发现,在早期阿尔茨海默病患者血浆中,miR-483-5p表达量增加最为显著,并在mRNA和蛋白质水平上抑制细胞外信号调节激酶1/2,降低其磷酸化活性,进而减少Tau磷酸化阻碍神经原纤维缠结的形成[32]。另有研究发现在APP/PS1小鼠模型中,miR-148a-3p表达下调解除了对p35和磷酸酶张力蛋白同源物的抑制,进而分别过度激活细胞周期蛋白依赖激酶5和蛋白激酶B信号通路,共同促使Tau蛋白过度磷酸化。尤为重要的是,环磷酸腺苷反应元件结合蛋白作为磷酸酶张力蛋白同源物/蛋白激酶B下游效应子被激活后,能直接结合miR-148a-3p启动子并促进其表达,形成一个自调节的前馈循环以挽救认知障碍和Tau病理学[33]。
(2)调节炎症反应和氧化应激:核因子κB信号通路激活是驱动神经炎症的核心枢纽[34]。在阿尔茨海默病中下调的miR-22-3p和miR-216a-5p分别丧失对靶基因性别决定区Y框蛋白9和高迁移率族蛋白1的抑制作用[35-36],导致二者表达升高并直接激活核因子κB通路;同时,上调的miR-25802通过直接结合Krüppel样因子4 mRNA负向调节Krüppel样因子4,进一步促进核因子κB介导的炎症反应来推动小胶质细胞向促炎M1表型极化[37]。可见这3种微小RNA可通过不同机制共同增强核因子κB信号通路的活性,放大神经炎症反应,从而加剧阿尔茨海默病的病理损伤及认知功能障碍。此外有研究发现,抑制miR-10b-5p可解除其对靶基因同源盒基因D10的转录抑制,上调同源盒基因D10表达,从而进一步抑制Ras同源基因(Ras homolog gene,Rho)/Rho相关卷曲螺旋形成蛋白激酶信号通路,最终显著降低白细胞介素1β、白细胞介素6、肿瘤坏死因子α和丙二醛表达水平,并提高超氧化物歧化酶活性[38]。证实miR-10b-5p/同源盒基因D10/Ras同源基因/Rho相关卷曲螺旋形成蛋白激酶轴可通过同步抑制炎症与氧化应激,在阿尔茨海默病中发挥协同神经保护作用。HU等[39]研究表明,miR-107-5p过表达可直接靶向并抑制Toll样受体4表达,从而阻止核因子κB p65的核转位,进一步降低活性氧、肿瘤坏死因子α、白细胞介素1β、白细胞介素6和髓系细胞触发受体 2水平,并减弱小胶质细胞异常激活,最终有效减轻氧化应激和异常炎症反应,在阿尔茨海默病中发挥神经保护作用。
(3)靶向突触可塑性和钙稳态:维持和调节突触结构与功能的多种因素,包括神经递质、蛋白质及信号通路等,均受到多种微小RNA的调控。其中miR-181a作为α-氨基-3-羟基-5-甲基-4-异恶唑丙酸(alpha-amino-3-
hydroxy-5-methyl-4-isoxazolepropionic acid,AMPA)受体的关键负调控因子,通过直接结合离子型谷氨酸受体AMPA型亚基1(glutamate ionotropic receptor AMPA type subunit 1,GluA1)和离子型谷氨酸受体AMPA型亚基2(glutamate ionotropic receptor AMPA type subunit 2,GluA2)亚基的mRNA,抑制其翻译并促进降解,从而显著降低海马中受体的表达水平,损害突触可塑性导致记忆形成与巩固障碍;相反抑制miR-181a表达则可有效逆转上述效应,改善记忆缺陷[40]。有研究发现,miR-431过表达可通过特异性结合Smad4 mRNA的3’-UTR区域并抑制其翻译,显著降低Smad4蛋白水平[41]。该作用直接促进突触素1、突触后致密蛋白95、N-甲基-D-天冬氨酸受体1、N-甲基-D-天冬氨酸受体2A和N-甲基-D-天冬氨酸2B受体等分子表达,并增强长时程增强效应;同时亦可间接恢复Dlg3基因表达,从而在突触前与突触后层面双向协同提升突触稳定性,最终逆转突触可塑性损伤并改善记忆功能。ZOU等[42]研究发现在携带突变早老素1的阿尔茨海默病小鼠模型中,皮质内特异性降低的miR-339-5p通过直接上调其靶蛋白Neuronatin(Nnat)水平,进一步激活肌浆/内质网Ca2+-ATP酶(SERCA),引发Ca2+储存增加,破坏钙稳态,并导致突触功能障碍;同时亦可导致脾后皮质神经元反应的习惯化缺陷,加速阿尔茨海默病进展。
2.2.2 长链非编码RNA与阿尔茨海默病 与其他非编码RNA相比长链非编码RNA表现出更高的组织特异性,并能通过表观遗传修饰、微小RNA结合、蛋白质互作以及转录、转录后和翻译后调节等功能来影响阿尔茨海默病发展。最近的研究证实,多种长链非编码RNA在阿尔茨海默病中异常表达并通过不同的病理程序影响疾病进展[43],提示靶向功能性长链非编码RNA可能是探索阿尔茨海默病治疗的新策略。
(1)调控β-淀粉样蛋白代谢和Tau蛋白磷酸化:长链非编码RNA通过调控β-淀粉样蛋白产生、清除及Tau蛋白磷酸化等关键病理过程,在阿尔茨海默病中发挥独特作用。长链非编码RNA XIST通过双重分子机制促进阿尔茨海默病中β-淀粉样蛋白沉积[44-45]:一方面,其过表达可吸附miR-124,从而解除miR-124对β位淀粉样前体蛋白裂解酶1的抑制作用,导致β位淀粉样前体蛋白裂解酶1表达上调,增加β-淀粉样蛋白生成;另一方面,长链非编码RNA XIST直接与Zeste增强子同源物 2结合,促使组蛋白H3K27三甲基化修饰在脑啡肽酶基因启动子区域富集,从而抑制脑啡肽酶基因表达,削弱β-淀粉样蛋白清除能力。另有研究证实,靶向抑制lnc CYP3A43-2可释放游离的miR-29b-2-5p,进而有效抑制早老素1表达,显著减少γ-分泌酶介导的β-淀粉样蛋白生成,并改善认知功能[46]。此外EPB41L4A-AS1是维持基础自噬水平所必需的长链非编码RNA,其敲除会抑制溶酶体标志物(LAMP1、CTSB、CTSD)和自噬关键基因(ATG3、ATG5、ATG16L1)的表达,从而损害自噬-溶酶体途径功能,导致β-淀粉样蛋白清除障碍[47]。
长链非编码RNA 00507和长链非编码RNA ZBTB20-AS1过表达则可介导不同分子通路促进Tau蛋白过度磷酸化。其中长链非编码RNA 00507可作为竞争性内源RNA吸附miR-181c-5p,解除其对靶基因微管相关蛋白Tau和Tau微管蛋白激酶1的抑制,导致两者水平升高,进而激活P25/P35/糖原合酶激酶3β信号通路,驱动Tau蛋白过度磷酸化[48]。而长链非编码RNA ZBTB20-AS1可通过上调其邻近基因ZBTB20表达,形成自我增强的正反馈循环,并直接激活糖原合酶激酶3β,导致Tau多位点磷酸化[49]。可见两者最终均以糖原合酶激酶3β为共同枢纽,双重促进Tau蛋白病理进程。
(2)调节炎症反应和氧化应激:长链非编码RNA常在神经炎症、氧化应激和神经退行性疾病等密切相关的病理过程中扮演着调控者的角色。研究发现,长链非编码RNA NEAT1和长链非编码RNA NKILA过高表达均可促进氧化应激反应,具体表现为细胞内活性氧、丙二醛及乳酸脱氢酶水平升高,超氧化物歧化酶活性降低[50-51]。但两者实现此效应的机制存在差异:长链非编码RNA NEAT1主要通过上调微小RNA-193a表达并与之结合,从而抑制环磷酸腺苷反应元件结合蛋白/脑源性神经营养因子及核因子红细胞2相关因子2/醌氧化还原酶1[NAD(P)H:quinone oxidoreductase1,NQO1]信号通路活性;而长链非编码RNA NKILA则是通过结合胚胎致死异常视觉蛋白1,增强叉头框蛋白A1 mRNA的稳定性,继而促进其下游靶基因肿瘤坏死因子α诱导蛋白1的表达。此外,长链非编码RNA NEAT1还能诱导肿瘤坏死因子α、白细胞介素1β等炎性细胞因子表达,进一步加剧阿尔茨海默病病理进展。
(3)靶向突触可塑性:长链非编码RNA可通过调控突触可塑性维持突触效能与神经元活动。有研究发现,长链非编码RNA Gas5可作为环磷酸腺苷信号通路的一种活动响应性下游效应分子[52]。其在神经元激活后被环磷酸腺苷信号特异性诱导表达,并由驱动蛋白 KIF1A转运至树突。在树突局部,长链非编码RNA Gas5主要发挥两种功能:一是作为竞争性内源RNA直接结合miR-26a-5p,解除其对重要突触功能相关mRNA的翻译抑制;二是与糖原合酶激酶3β等多种蛋白相互作用形成动态的复合物,进而特异性地介导环磷酸腺苷信号所诱导的树突分支形成、突触结构维持及兴奋性突触传递增强。而长链非编码RNA Gas5缺陷会损害突触结构和功能,表明磷酸腺苷/长链非编码RNA Gas5/miR-26a-5p轴是维持兴奋性突触结构与可塑性的调控节点。
2.2.3 环状RNA与阿尔茨海默病 环状RNA是一类通过前体mRNA反向剪接形成的单链共价封闭环形RNA分子,其缺乏多聚腺苷酸尾和5’-3’末端结构,因此具有高度稳定性和高丰度性,并在突触区域呈现特异性表达[53]。功能上,环状RNA可通过充当微小RNA“海绵”、调控基因转录、参与蛋白翻译或与RNA结合蛋白相互作用等多种机制,多维度影响基因表达和细胞功能[54-56]。研究表明其可直接参与调控β-淀粉样蛋白代谢异常、Tau蛋白病理性聚集、神经炎症及氧化应激等阿尔茨海默病核心病理过程[57]。
(1)调控β-淀粉样蛋白代谢和Tau蛋白磷酸化:环状RNA可通过充当微小RNA“海绵”参与调控β-淀粉样蛋白生成与清除以及Tau蛋白磷酸化过程。多种环状RNA被证实通过此机制影响β-淀粉样蛋白代谢:其中circZranb1和circADAM10可靶向结合miR-361-3p,而circNlgn1则可结合miR-298-3p,均可抑制相应微小RNA的活性,从而下调β位淀粉样前体蛋白裂解酶1的表达与功能,减少β-淀粉样蛋白生成[58]。而circHDAC9可充当miR-138的分子海绵,吸附并抑制其活性,以解除对沉默信息调节因子1的抑制作用,减少β-淀粉样蛋白过量产生,形成由circHDAC9/miR-138/沉默信息调节因子1构成的调控通路,干预阿尔茨海默病中淀粉样前体蛋白的异常加工过程[59]。有研究发现,ciRS-7作为miR-7海绵对β-淀粉样蛋白稳态进行双向动态调控,其过表达时通过激活蛋白酶体和溶酶体降解途径,或抑制核因子κB翻译导致泛素羧基末端水解酶L1表达下降,进而降低淀粉样前体蛋白和β位淀粉样前体蛋白裂解酶1的蛋白水平,抑制β-淀粉样蛋白的生成;而其低表达时可导致游离miR-7升高,下调参与β-淀粉样蛋白降解的关键因子泛素结合酶E2A,从而损害β-淀粉样蛋白的清除能力[60-61]。
另有研究发现, 环状RNA-蛋白质互作机制也可能在阿尔茨海默病病理起关键作用,一方面,神经元中高富集的circCwc27可通过结合转录因子Pur-α并将其滞留于细胞质,阻碍其对淀粉样前体蛋白等基因启动子的招募,导致转录失调,进而驱动β-淀粉样蛋白异常沉积及认知功能障碍;反之circCwc27下调时,Pur-α被释放并恢复其转录调节功能,重建神经保护性转录程序,最终减少β-淀粉样蛋白生成并缓解认知损伤[62]。另一方面,circPCCA通过竞争性结合miR-138-5p,解除其对糖原合酶激酶3β的抑制,从而增强糖原合酶激酶3β活性并促进Tau蛋白过度磷酸化[63]。
(2)调节神经炎症:近期研究表明,特定环状RNA可通过不同分子途径调控神经炎症与神经元功能。一方面,circAPP可通过吸附miR-1906解除其对胞内氯离子通道1的转录后抑制,从而增强氯离子通道1表达及通道功能,进一步驱动小胶质细胞向促炎性M1表型极化,加剧神经炎症级联反应。功能实验证实,敲除circAPP可有效下调M1型标志物CD16及促炎因子(肿瘤坏死因子α、白细胞介素1β)的表达,同时上调M2型标志物Arg1和抗炎因子白细胞介素10的表达,充分表明circAPP通过miR-1906/氯离子通道1信号轴调控小胶质细胞极化平衡,参与阿尔茨海默病神经炎症失调和认知功能障碍恶化等病理进程[64]。另一方面,circDlg1上调则通过抑制Smad泛素化调节因子2介导的磷酸二酯酶4B泛素化降解,导致磷酸二酯酶4B异常积累及其酶活性增强,进而过度水解环磷酸腺苷,抑制了具有抗炎作用的蛋白激酶A/环磷酸腺苷反应元件结合蛋白信号通路,最终驱动小胶质细胞异常活化并加剧神经炎症;沉默circDlg1可增强小胶质细胞对β-淀粉样蛋白沉积的保护性反应并有效缓解神经炎症[65]。此外,circ-0000950可通过直接结合miR-103并抑制其表达,导致具有神经毒性的促炎基因前列腺素内过氧化物合酶2水平上调,进而促进肿瘤坏死因子α、白细胞介素1β及白细胞介素16表达,加剧神经元凋亡、抑制神经突生长,并放大神经炎症,最终损害神经元功能加速疾病进展[66]。相比之下,敲低circ-0004381不仅能减轻了海马神经元损伤,亦可通过miR-647/早老素1轴促进小胶质细胞向M2型极化,从而改善阿尔茨海默病模型小鼠的认知功能[67]。
(3)介导氧化应激:circLPAR1和circAXL表达水平与β-淀粉样蛋白诱导产生的氧化应激严重程度相关。在阿尔茨海默病有关的人体样本和体外实验模型中均一致发现,circLPAR1表达显著升高,并通过竞争性结合miR-212-3p,阻断了其对下游靶标锌 指 蛋 白 217的抑制作用,从而促进神经元凋亡和氧化应激等病理过程[68]。而在阿尔茨海默病细胞模型中敲除circAXL可显著降低活性氧和丙二醛水平,并恢复超氧化物歧化酶活性,其主要保护机制在于circAXL通过调控miR-1306-5p/磷酸二酯酶4A信号轴,有效减轻了由氧化应激引起的神经毒性[69]。
(4)靶向突触可塑性:阿尔茨海默病中记忆障碍的病理基础是突触丢失,而circHDAC9可通过其分子海绵作用缓解这一过程。主要机制为circHDAC9通过吸附miR-138并抑制其活性,进而促进突触素1、突触后致密蛋白93和突触后致密蛋白95表达,有助于修复突触损伤,减轻树突棘退化,最终改善阿尔茨海默病中的突触可塑性[59]。
基于上述研究进展,不同类型的非编码RNA可通过靶向mRNA沉默、竞争性内源RNA海绵效应及蛋白质互作构成动态网络调控阿尔茨海默病的病理级联:通过调节淀粉样前体蛋白、β位淀粉样前体蛋白裂解酶1和早老素1等影响β-淀粉样蛋白生成;调节自噬及泛素蛋白酶体系等途径影响β-淀粉样蛋白清除;调节糖原合酶激酶3β等相关通路影响Tau蛋白磷酸化状态;调节胶质细胞活化及核因子κB通路影响促炎细胞因子的水平;调节活性氧和脂质过氧化、抗氧化酶活性,影响氧化和抗氧化平衡;调节相关突触蛋白、钙调蛋白和长时程增强等表达影响突触可塑性。但鉴于阿尔茨海默病多因异质性和非线性病理特征,且单一类型非编码RNA的应用受限于靶点覆盖不足,未来应整合微小RNA、长链非编码RNA与环状RNA的多维生物标志物网络,通过协同解码病理级联的交互作用,可实现更全面的疾病评估与精准干预。
2.3 转运RNA衍生小RNA在阿尔茨海默病中的相关研究进展 转运RNA衍生小RNA是一类源于前体或成熟转运RNA的小RNA分子,具有典型的三环四茎结构,由Dicer、血管生成素或核糖核酸酶5等酶在特定位点裂解而成。根据生成机制和长度差异,转运RNA衍生小RNA主要分为应激诱导的RNA(tRNA-derivedstress-induced RNA,tiRNA)和转运RNA衍生的片段(tRNA-derivedfragment,tRF)两大类[70]。该类分子在神经系统中特异性分布,并参与调控线粒体功能等生物学过程;其异常剪切或表达失调不仅会损害神经元功能,还可诱发神经炎症反应[71]。
2.3.1 驱动线粒体损伤 阿尔茨海默病患者内侧前额叶皮质的谷氨酰胺能神经元中,Glu-5′tsRNA-CTC水平显著高于非阿尔茨海默病个体,且呈现出年龄依赖性累积的特征。该分子由血管生成素在细胞质内切割产生后易位至线粒体,通过竞争性结合线粒体亮氨酰-转运RNA合成酶2,干扰其与线粒体转运RNA的正常互作,导致mt-tRNA氨酰化障碍;此过程特异性破坏了线粒体编码的蛋白质翻译,引发线粒体嵴结构塌陷和能量代谢障碍。同时,Glu-5′tsRNA-CTC累积所诱发的嵴组织缺陷通过激活泛素-蛋白酶体系统触发K48连接的多聚泛素化,导致关键代谢酶谷氨酰胺酶降解。而谷氨酰胺酶的耗竭显著削弱了谷氨酰胺向谷氨酸的转化效率,致使突触谷氨酸匮乏,最终损害兴奋性突触传递并导致记忆功能障碍[72]。
2.3.2 参与胶质细胞介导的多重病理级联 关键转运RNA衍生的RNA片段介导的胶质细胞稳态失衡可能是阿尔茨海默病神经病理的重要驱动因素。其中ZHANG等[73]和DENG等[74]不同研究团队发现AS-tDR-011389(i-tRF)和tRFAla-AGC-3-M8均可调控胶质细胞稳态,其中前者可上调P2ry1基因直接损害星形胶质细胞网络功能并扰乱其稳态;后者与Eph受体家族中EphA7的表达呈负相关,在APP/PS1转基因小鼠模型和阿尔茨海默病患者中tRFAla-AGC-3-M8均显著下调,解除了对EphA7的抑制并促进其表达,进而过度激活下游的细胞外信号调节激酶1/2-/p70核糖体蛋白S6激酶信号通路并引发异常磷酸化,最终驱动小胶质细胞向促炎的M1型极化,破坏小胶质细胞稳态加剧神经炎症。不同的是AS-tDR-011389 (i-tRF)还可上调钙调素依赖性蛋白激酶Ⅱ抑制物,以抑制钙调蛋白依赖性蛋白激酶Ⅱ活性,进而抑制长时程增强损害突触可塑性[75]。此外,其他转运RNA衍生的RNA片段(如靶向Rpsa基因促β-淀粉样蛋白毒性的AS-tDR-013428)及tRFAla-AGC-3-M8(促Tau磷酸化)可协同加重神经元损伤。因此,胶质细胞稳态的协同破坏不仅是转运RNA衍生的RNA片段诱发神经炎症和关联性突触损伤的直接动因,也可能与β-淀粉样蛋白/Tau介导的神经元毒性损伤构成驱动阿尔茨海默病进展的多维度病理网络。
2.3.3 靶向钙稳态 LU等[76]使用高通量测序技术在阿尔茨海默病的APP/PS1转基因小鼠模型中进行了转运RNA衍生的RNA片段差异分析,发现tRF-Leu-CAA-004表达下调而tRF-Thr-CGT-003表达上调。下调的tRF-Leu-CAA-004可抑制视黄酸代谢酶CYP2S1、CYP2C68和CYP2C40的表达,同时亦能靶向兰尼碱受体1 mRNA,导致Ryr1蛋白表达升高,进而增强了内质网钙释放,引起血浆Ca2+水平升高;作为补偿机制,上调的tRF-Thr-CGT-003通过增加 γ2蛋白的表达,抑制电压门控Ca2+通道的活性,从而减少Ca2+内流,这种tRF-Thr-CGT-003介导的钙内流抑制,平衡了由tRF-Leu-CAA-004下调引起的钙升高,共同维持了细胞内钙稳态,可见转运RNA衍生的RNA片段在阿尔茨海默病钙稳态失调中扮演重要角色,并显示出作为疾病诊断生物标志物及潜在治疗靶点的价值。
基于上述研究发现,转运RNA衍生小RNA是阿尔茨海默病级联性病理网络的驱动力。在分子层面(干扰蛋白翻译、泛素化)、细胞层面(引发线粒体能量危机、胶质细胞炎症、神经元死亡)和系统层面(导致突触传递衰竭、钙稳态失调)触发一系列连锁反应,最终表现为神经炎症、β-淀粉样蛋白/Tau毒性、突触可塑性丧失、代谢紊乱以及钙稳态崩溃。可见靶向关键转运RNA衍生小RNA有望成为调控阿尔茨海默病病理进程的新策略。参与阿尔茨海默病中的主要非编码RNA见图5。
2.4 中医药以非编码RNA为靶点调控阿尔茨海默病的相关研究进展 在阿尔茨海默病的病理进程中,β-淀粉样蛋白沉积属有形“痰浊”壅滞脑络;Tau蛋白异常磷酸化致神经原纤维缠结则属“瘀毒”缠聚,痰瘀毒浊搏结、深伏于窍、损伤神机。神经炎症的激活可属“痰火”或“火毒”上攻,灼损脑络;线粒体功能障碍乃元气衰惫、生化乏源;氧化应激属虚火内炽、津液受灼之征。突触乃神机传导之枢,其结构功能异常所致认知衰退,其本在于髓海不足、灵机失养;钙稳态失衡引发神经元兴奋性毒性,则属阴阳失调、内风妄动之象,扰乱神明。基于此,中医治疗强调清热化痰、活血解毒以祛其邪,通络开窍、益气滋阴、填精补髓以扶其正,诸法相合,共奏调和阴阳之效。
2.4.1 中药单体活性成分
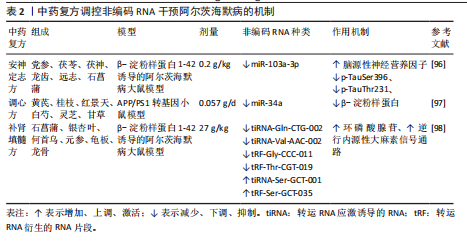
(1)小檗碱:小檗碱(BBR)是存在于中药黄连中的一种天然的异喹啉生物碱,凭借其抗炎、抗氧化、抗胆碱酯酶活性和神经保护等多种药理作用,以及能够通过血脑屏障进入海马和丘脑等关键脑区的特性,成为治疗阿尔茨海默病的潜在药物[77-78]。研究表明,在β-淀粉样蛋
白诱导的阿尔茨海默病模型中,小檗碱可通过调节多种非编码RNA发挥神经保护作用。具体而言,在β-淀粉样蛋白25-35诱导的模型中,小檗碱干预后显著上调细胞内miR-137表达,从而降低淀粉样前体蛋白水平[79];亦可下调长链非编码RNA β位淀粉样前体蛋白裂解酶1反义转录物表达,解除对miR-132-3p的抑制,进而降低乳酸脱氢酶活性、Ca2+浓度和活性氧水平,且高浓度小檗碱作用尤为显著[80]。然而在β-淀粉样蛋白42诱导的HN细胞模型中,小檗碱则通过增加circ HDAC9表达并抑制miR-142-5p,减少促炎因子白细胞介素1β、白细胞介素6和肿瘤坏死因子α产生[81]。综合以上效应可见,黄连清热化痰、解毒通络、泻火护神的传统药效,为从“解毒祛痰”调控不同非编码RNA论治阿尔茨海默病提供了分子依据。
(2)梓醇:梓醇(CAT)是从中药地黄中提取的主要小分子环烯醚萜类化合物,现代药理学发现其具有抗衰老、抗炎、增强免疫及神经系统保护作用[82]。MENG等[83]研究中发现,梓醇在体内外均能延缓阿尔茨海默病进展,其主要机制是通过上调miR-138-5p表达,一方面抑制Tau蛋白异常沉积,另一方面促进小窝蛋白1表达,从而增强突触可塑性、维持神经元完整性。可见梓醇从分子层面揭示了其通过调控微小RNA以“补肾填髓”法论治阿尔茨海默病的作用机制(3)三七总皂苷:三七总皂苷(PNS)是中药三七的主要活性成分,具有抗炎、抗氧化、抗血小板聚集和神经保护等多种药理作用[84]。HUANG等[85]研究表明,在SAMP8小鼠模型中,三七总皂苷可显著调控海马组织中环状RNA表达谱,尤其下调环状RNA-013636并上调环状RNA-012180的表达,且这两种环状RNA均参与阿尔茨海默病相关信号通路。该结果表明,三七总皂苷可能通过调节环状RNA表达以抑制阿尔茨海默病病理进展,而结合其“散瘀止血、消肿定痛”之功效,未来研究可侧重于深入解析其通过环状RNA调控Tau蛋白磷酸化及神经纤维缠结降解的具体分子机制。
(4)人参皂苷Rg1:人参皂苷Rg1是人参总皂苷中活性最强的单体成分,具有天然抗氧化、抗衰老及抗凋亡等药理活性[86]。近期体内外研究表明,其可通过靶向下调miR-9-5p,解除对沉默信息调节因子1的抑制,进而提升ATP水平、线粒体膜电位及呼吸控制率;并增强PTEN诱导的假定激酶1、E3泛素连接酶、微管相关蛋白轻链3-Ⅱ/Ⅰ、和Beclin-1等自噬相关蛋白表达,最终显著改善阿尔茨海默病中线粒体功能障碍,延缓疾病进展[87]。此次研究阐明了“益气充髓”法通过调控miR-9-5p/沉默信息调节因子1/线粒体自噬轴治疗阿尔茨海默病,为该治法的现代科学内涵提供了实验依据。
(5) β-细辛醚:β-细辛醚是石菖蒲挥发油中发挥药效作用的关键活性成分,具有调节自噬、突触功能和胆碱能系统,以及抑制炎症反应和氧化应激等多重治疗作用[88]。WANG等[89]在SH-SY5Y细胞模型中发现,β-细辛醚表现出一种独特的双向调控作用:其虽可升高长链非编码RNA β位淀粉样前体蛋白裂解酶1反义转录物基因表达,却能有效阻断与β位淀粉样前体蛋白裂解酶1 mRNA互作的稳定性,从而拮抗该长链非编码RNA的致病功能;这种“表达上调”与“功能抑制”并存的效应,最终显著降低了早老素1、β-淀粉样蛋白、β位淀粉样前体蛋白裂解酶1、淀粉样前体蛋白及自噬阻滞蛋白p62表达;同时上调自噬相关蛋白(LC3-Ⅰ/Ⅱ、Beclin-1)及突触蛋白(syn)水平。可见“化痰开窍”法可通过调控长链非编码RNA β位淀粉样前体蛋白裂解酶1反义转录物/自噬通路双向促进β-淀粉样蛋白代谢与突触功能修复。
(6)雷公藤内酯醇和丹参酮ⅡA:雷公藤内酯醇和丹参酮ⅡA分别源自雷公藤和丹参的二萜类抗炎化合物,在不同阿尔茨海默病模型中,两者均可通过抑制长链非编码RNA NEAT1表达,解除其对下游特定微小RNA的抑制作用,从而减轻神经炎症并改善病理损伤[90-91]。具体机制为:雷公藤内酯通过下调长链非编码RNA NEAT1,解除对miR-361-3p的抑制,进而抑制其靶点肿瘤坏死因子受体相关因子2的表达[92];丹参酮ⅡA则通过抑制长链非编码RNA NEAT1,减弱其对miR-291a-3p的“分子海绵”吸附效应,增强miR-291a-3p活性,进而抑制Rab22a表达[93]。尽管两者抑制的微小RNA靶点不同,但最终均汇聚于核因子κB炎症信号通路并抑制其活化,降低促炎因子(白细胞介素1β、白细胞介素6、肿瘤坏死因子α)释放,减轻神经炎症反应最终改善认知功能。该作用揭示了以“解毒祛瘀”之法抑制长链非编码RNA NEAT1介导的神经炎症通路,干预阿尔茨海默病现代机制。中药单体调控非编码RNA干预阿尔茨海默病的机制见表1。
2.4.2 中药复方
(1)安神定志方:安神定志方出自《医学心悟》,具有安神定志、化痰镇惊之功效,由党参、茯苓、茯神、龙齿、远志、石菖蒲组成,常用于治疗精神情志类和阿尔茨海默病等疾病[94-95]。王欣波等[96]研究表明,安神定志方可显著改善β-淀粉样蛋白1-42诱导的阿尔茨海默病大鼠海马CA1区组织形态损伤,其机制与下调miR-103a-3p表达有关,进而促进脑源性神经营养因子表达,并显著降低p-TauSer396和p-TauThr231水平,从而减轻神经元损伤并改善记忆功能障碍。
(2)调心方:调心方是在孔圣枕中丹与益气聪明汤基础上化裁而来,由黄芪、桂枝、红景天、白芍、灵芝、甘草组成,具有益气通阳、化痰开窍之功效,临床长期用于治疗阿尔茨海默病。HU等[97]通过APP/PS1转基因小鼠模型研究调心方对阿尔茨海默病的治疗作用,结果发现调心方可有效改善认知障碍并明显减少皮质和海马区β-淀粉样蛋白沉积,延缓了早期阿尔茨海默病发展。其机制可能与下调miR-34a表达,进而抑制其介导的氧化应激有关。
(3)补肾填髓方:补肾填髓方承袭《千金方》孔圣枕中丹之精义经化裁重组而来,由石菖蒲、银杏叶、何首乌、元参、龟板、龙骨组成,以补肾填髓为主,兼具化痰开窍之效。ZHANG等[98]通过基因组学分析发现,补肾填髓方在β-淀粉样蛋白1-42诱导的阿尔茨海默病大鼠模型中可通过调控海马区tsRNA,发挥改善学习记忆障碍的作用。研究共筛选出387个海马区tsRNA,其中57个受BSTSF显著调控,重点聚焦并鉴定了BSTSF直接干预的6个核心tsRNA(tiRNA-Gln-CTG-002、tiRNA-Val-AAC-002、tRF-Gly-CCC-011、tRF-Thr-CGT-019被下调;tiRNA-Ser-GCT-001、tRF-Ser-GCT-035被上调)。经靶基因预测及功能验证,发现这些tsRNA通过靶向CHRNA5、GRIA4等44个关键基因,激活环磷酸腺苷信号通路及逆行内源性大麻素信号通路,进而调节神经递质传递、突触可塑性和免疫反应,最终介导神经保护作用。该研究首次从tsRNA角度揭示了补肾填髓方抗阿尔茨海默病的分子机制,为中医药治疗阿尔茨海默病提供了新的作用靶点和机制依据。中药复方调控非编码RNA干预阿尔茨海默病的机制见表2。
2.4.3 针灸
(1)电针:电针疗法是将脉冲电流通过毫针导入腧穴,结合针刺与电刺激以增强疗效,在阿尔茨海默病的防治中取得了一定的临床效果。刘兴媛[99]的研究揭示,电针干预可下调长链非编码RNA SNHG1表达,进而通过双重机制发挥神经保护作用:一方面阻断其与RNA结合蛋白IGF2BP2形成复合物,解除对Beclin1 mRNA的抑制,促进自噬激活并改善长时程增强;另一方面,抑制其对核因子E2相关因子2 mRNA的降解,提升核因子E2相关因子2蛋白表达,从而抑制细胞糖酵解和铁死亡发生,减少β-淀粉样蛋白和活性氧的积累,最终延缓阿尔茨海默病的进展。
(2)艾灸:艾灸通过调整气机、祛除痰瘀、充养脑髓等机制防治阿尔茨海默病的研究已获得广泛关注且成果日益丰富[100]。贾玉梅等[101]在APP/PS1双转基因小鼠模型中发现,艾灸“百会”“大椎”“风府”等督脉穴位(15-20 min/d,持续14 d)可显著下调海马区长链非编码RNA H19表达,抑制哺乳动物雷帕霉素靶蛋白信号通路过度激活,同时促进转录因子EB)核转位,进而增强自噬关键蛋白微管相关蛋白轻链3-Ⅱ/Ⅰ及溶酶体相关蛋白表达,减少自噬底物p62堆积,最终恢复自噬流加速β-淀粉样

蛋白清除,改善认知功能。
以上研究从中医病机理论与现代分子机制结合的角度,阐述了清热解毒、活血化瘀、益气补虚以及开窍豁痰类中药活性成分和复方通过调节不同非编码RNA干预阿尔茨海默病的多个病理环节,揭示了中医不同治法在现代分子生物学层面的内涵。该领域仍存在明显局限:①非编码RNA调控网络与中药多靶点特性的交互机制尚未明晰;②针灸调控非编码RNA的研究仍处于起步阶段,现有证据有限,但初步结果展现了巨大潜力,亟待更多设计严谨的研究予以验证,并确立标准化的穴位选择与刺激参数方案。

| [1] DZIEWA M, ZŁOTEK M, HERBET M, et al. Molecular and Cellular Foundations of Aging of the Brain: Anti-aging Strategies in Alzheimer’s Disease. Cell Mol Neurobiol. 2024;44(1):80. [2] ZHENG Q, WANG X. Alzheimer’s disease: insights into pathology, molecular mechanisms, and therapy. Protein Cell. 2025;16(2):83-120. [3] JORFI M, MAASER-HECKER A, TANZI RE. The neuroimmune axis of Alzheimer’s disease. Genome Med. 2023;15(1):6. [4] JU Y, TAM KY. Pathological mechanisms and therapeutic strategies for Alzheimer’s disease. Neural Regen Res. 2022;17(3):543-549. [5] ZHU CC, FU SY, CHEN YX, et al. Advances in Drug Therapy for Alzheimer’s Disease. Curr Med Sci. 2020;40(6):999-1008. [6] SUGANDHI VV, GADHAVE DG, UGALE AR, et al. Advances in Alzheimer’s therapy: Exploring neuropathological mechanisms to revolutionize the future therapeutic landscape. Ageing Res Rev. 2025;109:102775. [7] LIU Y, CHEN X, CHE Y, et al. LncRNAs as the Regulators of Brain Function and Therapeutic Targets for Alzheimer’s Disease. Aging Dis. 2022;13(3):837-851. [8] WANG E, LEMOS DUARTE M, ROTHMAN LE, et al. Non-coding RNAs in Alzheimer’s disease: perspectives from omics studies. Hum Mol Genet. 2022;31(R1):R54-R61. [9] LAURETTI E, DABROWSKI K, PRATICÒ D. The neurobiology of non-coding RNAs and Alzheimer’s disease pathogenesis: Pathways, mechanisms and translational opportunities. Ageing Res Rev. 2021;71: 101425. [10] 吴锦润,孟语晨,寇作阳,等.化瘀和祛痰中药调控自噬治疗阿尔茨海默病的研究进展[J].中华中医药杂志,2023,38(6):2751-2756. [11] YUKSEL M, TACAL O. Trafficking and proteolytic processing of amyloid precursor protein and secretases in Alzheimer’s disease development: An up-to-date review. Eur J Pharmacol. 2019;856:172415. [12] 张雷,范占芳,张作鹏,等.阿尔兹海默症发病机制及相关治疗药物的研究进展[J].中国药物化学杂志,2021,31(6):438-446+469. [13] 原野,余姊阳,邵思迈,等.星形胶质细胞对阿尔兹海默病的双向作用[J].中国比较医学杂志,2021,31(6):133-138. [14] LENG F, EDISON P. Neuroinflammation and microglial activation in Alzheimer disease: where do we go from here? Nat Rev Neurol. 2021;17(3):157-172. [15] CHEN Z, BALACHANDRAN YL, CHONG WP, et al. Roles of Cytokines in Alzheimer’s Disease. Int J Mol Sci. 2024;25(11):5803. [16] WANG X, WANG L, YU X, et al. Glutaminyl cyclase inhibitor exhibits anti-inflammatory effects in both AD and LPS-induced inflammatory model mice. Int Immunopharmacol. 2019;75:105770. [17] HUANG C, YAN S, ZHANG Z. Maintaining the balance of TDP-43, mitochondria, and autophagy: a promising therapeutic strategy for neurodegenerative diseases. Transl Neurodegener. 2020;9(1):40. [18] OLUFUNMILAYO EO, GERKE-DUNCAN MB, HOLSINGER RMD. Oxidative Stress and Antioxidants in Neurodegenerative Disorders. Antioxidants (Basel). 2023;12(2):517. [19] WANG M, ZHANG H, LIANG J, et al. Calcium signaling hypothesis: A non-negligible pathogenesis in Alzheimer’s disease. J Adv Res. 2025:S2090-1232(25)00026-8. [20] CASCELLA R, CECCHI C. Calcium Dyshomeostasis in Alzheimer’s Disease Pathogenesis. Int J Mol Sci. 2021;22(9):4914. [21] QIU Q, YANG M, GONG D, et al. Potassium and calcium channels in different nerve cells act as therapeutic targets in neurological disorders. Neural Regen Res. 2025;20(5):1258-1276. [22] CAI HY, CHEN SR, WANG Y, et al. Integrated analysis of the lncRNA-associated ceRNA network in Alzheimer’s disease. Gene. 2023; 876:147484. [23] REN S, LIN P, WANG J, et al. Circular RNAs: Promising Molecular Biomarkers of Human Aging-Related Diseases via Functioning as an miRNA Sponge. Mol Ther Methods Clin Dev. 2020;18:215-229. [24] LIN W, LIU H, TANG Y, et al. The development and controversy of competitive endogenous RNA hypothesis in non-coding genes. Mol Cell Biochem. 2021;476(1):109-123. [25] CUI Y, QI Y, DING L, et al. miRNA dosage control in development and human disease. Trends Cell Biol. 2024;34(1):31-47. [26] DU W, LEI C, DONG Y. MicroRNA-149 is downregulated in Alzheimer’s disease and inhibits β-amyloid accumulation and ameliorates neuronal viability through targeting BACE1. Genet Mol Biol. 2021;44(1): e20200064. [27] LI P, XU Y, WANG B, et al. miR-34a-5p and miR-125b-5p attenuate Aβ-induced neurotoxicity through targeting BACE1. J Neurol Sci. 2020; 413:116793. [28] CAO Y, TAN X, LU Q, et al. MiR-29c-3p May Promote the Progression of Alzheimer’s Disease through BACE1. J Healthc Eng. 2021; 2021:2031407. [29] TAN X, LUO Y, PI D, et al. MiR-340 Reduces the Accumulation of Amyloid-β Through Targeting BACE1 (β-site Amyloid Precursor Protein Cleaving Enzyme 1) in Alzheimer’s Disease. Curr Neurovasc Res. 2020;17(1):86-92. [30] CHEN ML, HONG CG, YUE T, et al. Inhibition of miR-331-3p and miR-9-5p ameliorates Alzheimer’s disease by enhancing autophagy [published correction appears in Theranostics. 2021 Oct 2;11(20):9774. Theranostics. 2021; 11(5):2395-2409. [31] JIANG Y, ZHANG Y, SU L. MiR-539-5p Decreases amyloid β-protein production, hyperphosphorylation of Tau and Memory Impairment by Regulating PI3K/Akt/GSK-3β Pathways in APP/PS1 Double Transgenic Mice. Neurotox Res. 2020;38(2):524-535. [32] NAGARAJ S, WANT A, LASKOWSKA-KASZUB K, et al. Candidate Alzheimer’s Disease Biomarker miR-483-5p Lowers TAU Phosphorylation by Direct ERK1/2 Repression. Int J Mol Sci. 2021;22(7):3653. [33] ZENG L, JIANG H, ASHRAF GM, et al. Implications of miR-148a-3p/p35/PTEN signaling in tau hyperphosphorylation and autoregulatory feedforward of Akt/CREB in Alzheimer’s disease. Mol Ther Nucleic Acids. 2021;27:256-275. [34] SUN E, MOTOLANI A, CAMPOS L, et al. The Pivotal Role of NF-kB in the Pathogenesis and Therapeutics of Alzheimer’s Disease. Int J Mol Sci. 2022;23(16):8972. [35] XIA P, CHEN J, LIU Y, et al. MicroRNA-22-3p ameliorates Alzheimer’s disease by targeting SOX9 through the NF-κB signaling pathway in the hippocampus. J Neuroinflammation. 2022;19(1):180. [36] SHAO P. MiR-216a-5p ameliorates learning-memory deficits and neuroinflammatory response of Alzheimer’s disease mice via regulation of HMGB1/NF-κB signaling. Brain Res. 2021;1766:147511. [37] ZHAO K, LIU J, SUN T, et al. The miR-25802/KLF4/NF-κB signaling axis regulates microglia-mediated neuroinflammation in Alzheimer’s disease. Brain Behav Immun. 2024;118:31-48. [38] RUAN Z, LI Y, HE R, et al. Inhibition of microRNA-10b-5p up-regulates HOXD10 to attenuate Alzheimer’s disease in rats via the Rho/ROCK signalling pathway. J Drug Target. 2021;29(5):531-540. [39] HU GJ, JIANG XY, DU SY, et al. miR-107-5p ameliorates neurological damage, oxidative stress, and immune responses in mice with Alzheimer’s disease by suppressing the Toll-like receptor 4 (TLR4)/nuclear factor-kappaB(NF-κB) pathway. Kaohsiung J Med Sci. 2024;40(2): 119-130. [40] RODRIGUEZ-ORTIZ CJ, PRIETO GA, MARTINI AC, et al. miR-181a negatively modulates synaptic plasticity in hippocampal cultures and its inhibition rescues memory deficits in a mouse model of Alzheimer’s disease. Aging Cell. 2020; 19(3):e13118. [41] GE J, XUE Z, SHU S, et al. MiR-431 attenuates synaptic plasticity and memory deficits in APPswe/PS1dE9 mice. JCI Insight. 2023;8(12): e166270. [42] ZOU HY, GUO L, ZHANG B, et al. Aberrant miR-339-5p/neuronatin signaling causes prodromal neuronal calcium dyshomeostasis in mutant presenilin mice. J Clin Invest. 2023; 133(3):e168441. [43] KHODAYI-SHAHRAK M, KHALAJ-KONDORI M, HOSSEINPOUR FEIZI MA, et al. Insights into the mechanisms of non-coding RNAs’ implication in the pathogenesis of Alzheimer’s disease. EXCLI J. 2022;21:921-940. [44] YUE D, GUANQUN G, JINGXIN L, et al. Silencing of long noncoding RNA XIST attenuated Alzheimer’s disease-related BACE1 alteration through miR-124. Cell Biol Int. 2020;44(2): 630-636. [45] YAN XW, LIU HJ, HONG YX, et al. lncRNA XIST induces Aβ accumulation and neuroinflammation by the epigenetic repression of NEP in Alzheimer’s disease. J Neurogenet. 2022;36(1):11-20. [46] WULI W, LIN SZ, CHEN SP, et al. Targeting PSEN1 by lnc-CYP3A43-2/miR-29b-2-5p to Reduce β Amyloid Plaque Formation and Improve Cognition Function. Int J Mol Sci. 2022;23(18):10554. [47] WANG Z, WANG R, NIU L, et al. EPB41L4A-AS1 is required to maintain basal autophagy to modulates Aβ clearance. NPJ Aging. 2024; 10(1):24. [48] YAN Y, YAN H, TENG Y, et al. Long non-coding RNA 00507/miRNA-181c-5p/TTBK1/MAPT axis regulates tau hyperphosphorylation in Alzheimer’s disease. J Gene Med. 2020;22(12): e3268. [49] WANG Y, CAI M, LOU Y, et al. ZBTB20-AS1 promoted Alzheimer’s disease progression through ZBTB20/GSK-3β/Tau pathway. Biochem Biophys Res Commun. 2023;640:88-96. [50] LI Y, FAN H, NI M, et al. Targeting lncRNA NEAT1 Hampers Alzheimer’s Disease Progression. Neuroscience. 2023;529:88-98. [51] ZHOU Y, WANG Y, WANG Y, et al. LncRNA NKILA Exacerbates Alzheimer’s Disease Progression by Regulating the FOXA1-Mediated Transcription of TNFAIP1. Neurochem Res. 2023;48(9):2895-2910. [52] CHANDA K, GRINMAN E, CLARK K, et al. The lncRNA Gas5 is an activity-responsive scaffold that mediates cAMP-dependent synaptic plasticity. Sci Signal. 2025;18(892):eadn2044. [53] HUANG JL, SU M, WU DP. Functional roles of circular RNAs in Alzheimer’s disease. Ageing Res Rev. 2020;60:101058. [54] XU X, ZHANG J, TIAN Y, et al. CircRNA inhibits DNA damage repair by interacting with host gene. Mol Cancer. 2020;19(1):128. [55] SHI Y, JIA X, XU J. The new function of circRNA: translation. Clin Transl Oncol. 2020;22(12): 2162-2169. [56] PRATS AC, DAVID F, DIALLO LH, et al. Circular RNA, the Key for Translation. Int J Mol Sci. 2020;21(22):8591. [57] WEN X, HUANG C, XIE H, et al. The Applications of CircRNA in the Diagnosis and Treatment of Alzheimer’s Disease. Mol Neurobiol. 2024; 61(9):6501-6510. [58] ROMERO SC, ETELEEB AM, NYKANEN NP, et al. circRNAs mediate the effect of chronic lysosomal dysfunction on Alzheimer’s disease pathology. Alzheimer’s Dementia. 2023; 19(Sup13):2. [59] LU Y, TAN L, WANG X. Circular HDAC9/microRNA-138/Sirtuin-1 Pathway Mediates Synaptic and Amyloid Precursor Protein Processing Deficits in Alzheimer’s Disease. Neurosci Bull. 2019;35(5):877-888. [60] SHI Z, CHEN T, YAO Q, et al. The circular RNA ciRS-7 promotes APP and BACE1 degradation in an NF-κB-dependent manner. FEBS J. 2017; 284(7):1096-1109. [61] ZHAO Y, ALEXANDROV PN, JABER V, et al. Deficiency in the Ubiquitin Conjugating Enzyme UBE2A in Alzheimer’s Disease (AD) is Linked to Deficits in a Natural Circular miRNA-7 Sponge (circRNA; ciRS-7). Genes (Basel). 2016; 7(12):116. [62] SONG C, ZHANG Y, HUANG W, et al. Circular RNA Cwc27 contributes to Alzheimer’s disease pathogenesis by repressing Pur-α activity. Cell Death Differ. 2022;29(2):393-406. [63] LI Y, FAN H, SUN J, et al. Circular RNA expression profile of Alzheimer’s disease and its clinical significance as biomarkers for the disease risk and progression. Int J Biochem Cell Biol. 2020;123:105747. [64] WU DP, WEI YS, HOU LX, et al. Circular RNA APP contributes to Alzheimer’s disease pathogenesis by modulating microglial polarization via miR-1906/CLIC1 axis. Alzheimers Res Ther. 2025;17(1):44. [65] SHI J, SONG C, ZHANG P, et al. Microglial circDlg1 modulates neuroinflammation by blocking PDE4B ubiquitination-dependent degradation associated with Alzheimer’s disease. Theranostics. 2025;15(8):3401-3423. [66] YANG H, WANG H, SHANG H, et al. Circular RNA circ_0000950 promotes neuron apoptosis, suppresses neurite outgrowth and elevates inflammatory cytokines levels via directly sponging miR-103 in Alzheimer’s disease. Cell Cycle. 2019;18(18):2197-2214. [67] LI N, ZHANG D, GUO H, et al. Inhibition of circ_0004381 improves cognitive function via miR-647/PSEN1 axis in an Alzheimer disease mouse model. J Neuropathol Exp Neurol. 2022;82(1):84-92. [68] WU L, DU Q, WU C. CircLPAR1/miR-212-3p/ZNF217 feedback loop promotes amyloid β-induced neuronal injury in Alzheimer’s Disease. Brain Res. 2021;1770:147622. [69] MENG S, WANG B, LI W. CircAXL Knockdown Alleviates Aβ-Induced Neurotoxicity in Alzheimer’s Disease via Repressing PDE4A by Releasing miR-1306-5p. Neurochem Res. 2022;47(6):1707-1720. [70] YUAN J, SONG Z, LIU J, et al. Transfer RNAs and transfer RNA-derived small RNAs in cerebrovascular diseases. Exp Neurol. 2024; 382:114971. [71] LI T, ZHEN H, WU W, et al. tsRNAs: A Prospective, Effective Therapeutic Intervention for Neurodegenerative Diseases. CNS Neurosci Ther. 2024;30(12):e70177. [72] LI D, GAO X, MA X, et al. Aging-induced tRNA-derived fragment impairs glutamate biosynthesis by targeting mitochondrial translation-dependent cristae organization.Cell Metab. 2024;36(5):1059-1075.e9. [73] ZHANG S, LI H, ZHENG L, et al. Identification of functional tRNA-derived fragments in senescence-accelerated mouse prone 8 brain. Aging (Albany NY). 2019;11(22):10485-10498. [74] DENG Z, LI Y, CHI W, et al. tRFAla-AGC-3-M8 attenuates neuroinflammation and neuronal damage in Alzheimer’s disease via the EphA7-ERK1/2-p70S6K signaling pathway. Alzheimers Res Ther. 2025;17(1):104. [75] ASTUDILLO D, KARMELIC D, CASAS BS, et al. CaMKII inhibitor 1 (CaMK2N1) mRNA is upregulated following LTP induction in hippocampal slices. Synapse. 2020;74(10): e22158. [76] LU H, LIU L, HAN S, et al. Expression of tiRNA and tRF in APP/PS1 transgenic mice and the change of related proteins expression. Ann Transl Med. 2021;9(18):1457. [77] SHOU JW, SHAW PC. Therapeutic Efficacies of Berberine against Neurological Disorders: An Update of Pharmacological Effects and Mechanisms. Cells. 2022;11(5):796. [78] BEHL T, SINGH S, SHARMA N, et al. Expatiating the Pharmacological and Nanotechnological Aspects of the Alkaloidal Drug Berberine: Current and Future Trends. Molecules. 2022; 27(12):3705. [79] 周景芬,张开,张林英,等.小檗碱通过上调miR-137抑制APP表达调控阿尔茨海默病的发生发展[J].中国老年学杂志,2021, 41(17):3766-3770. [80] GE Y, SONG X, LIU J, et al. The Combined Therapy of Berberine Treatment with lncRNA BACE1-AS Depletion Attenuates Aβ Induced Neuronal Injury Through Regulating the Expression of miR-132-3p in Neuronal Cells. Neurochem Res. 2020;45(4):741-751. [81] ZHANG N, GAO Y, YU S, et al. Berberine attenuates Aβ42-induced neuronal damage through regulating circHDAC9/miR-142-5p axis in human neuronal cells. Life Sci. 2020; 252:117637. [82] 赵婧含,李雪,吴文轩,等.生地黄提取物及其有效成分的药理作用研究进展[J].药物评价研究,2024,47(10):2443-2448. [83] MENG S, CHEN H, DENG C, et al. Catalpol Mitigates Alzheimer’s Disease Progression by Promoting the Expression of Neural Stem Cell Exosomes Released miR-138-5p. Neurotox Res. 2023;41(1):41-56. [84] 曹国琼,过利敏,张海静,等.三七皂苷类成分治疗阿尔茨海默病研究进展[J].中成药,2016,38(12):2647-2651. [85] HUANG JL, XU ZH, YANG SM, et al. Identification of Differentially Expressed Profiles of Alzheimer’s Disease Associated Circular RNAs in a Panax Notoginseng Saponins-Treated Alzheimer’s Disease Mouse Model. Comput Struct Biotechnol J. 2018;16:523-531. [86] 王超楠,赵大庆,王健,等.人参皂苷Rg1治疗阿尔茨海默病作用及机制的研究进展[J].中成药,2021,43(4):984-987. [87] WANG Y, SUN X, HE B, et al. Ginsenoside Rg1 Downregulates miR-9-5p Expression to Modulate SIRT1-Mediated Mitochondrial Dysfunction and Ameliorate Alzheimer’s Disease. Mol Neurobiol. 2025;62(10):13044-13059. [88] 王浩,高磊,张金莲,等.石菖蒲中α-细辛醚和β-细辛醚药理作用及机制研究进展[J].中国中药杂志,2025,50(9):2305-2316. [89] WANG Z, ZHOU J, ZHANG B, et al. Inhibitory effects of β-asarone on lncRNA BACE1-mediated induction of autophagy in a model of Alzheimer’s disease. Behav Brain Res. 2024; 463:114896. [90] 田文国,王春芳,陈金鹏,等.中药抗阿尔茨海默病的作用及其机制研究进展[J].中草药,2022,53(10):3195-3208. [91] 孟泽宇,孔莹,孟胜喜,等.中医防治阿尔茨海默病的新思路:表观遗传学[J].中西医结合心脑血管病杂志,2022,20(13):2385-2389. [92] ZHOU L, HUANG X, LI H, et al. Triptolide improves Alzheimer’s disease by regulating the NFκB signaling pathway through the lncRNA NEAT1/microRNA 3613p/TRAF2 axis. Exp Ther Med. 2023;26(3):440. [93] YANG LX, LUO M, LI SY. Tanshinone IIA improves Alzheimer’s disease via RNA nuclear-enriched abundant transcript 1/microRNA-291a-3p/member RAS oncogene family Rab22a axis. World J Psychiatry. 2024;14(4):563-581. [94] 张峥嵘,高峰,杨绍杰,等.基于网络药理学和分子对接技术研究安神定志方干预创伤后应激障碍焦虑样行为的机制[J].安徽中医药大学学报,2023,42(4):74-80. [95] 王欣波,齐明明,邵音,等.安神定志方对阿尔茨海默病认知障碍及神经炎症的影响[J].海南医科大学学报,2025,31(10):750-757. [96] 王欣波,赵宇,袁星星.安神定志方对阿尔茨海默病大鼠海马组织miR-103a-3p及其介导的Tau蛋白磷酸化的影响[J].中国中医药信息杂志,2021,28(2):62-68. [97] HU YR, XING SL, CHEN C, et al. Tiaoxin Recipe, a Chinese herbal formula, inhibits microRNA-34a expression in the APPswe/PS1ΔE9 mouse model of Alzheimer’s disease. J Integr Med. 2019;17(6):404-409. [98] ZHANG ZY, ZHANG CH, YANG JJ, et al. Genome-wide analysis of hippocampal transfer RNA-derived small RNAs identifies new potential therapeutic targets of Bushen Tiansui formula against Alzheimer’s disease. J Integr Med. 2021;19(2):135-143. [99] 刘兴媛.电针通过长非编码RNA SNHG1调控细胞自噬、糖代谢和铁死亡延缓阿尔茨海默病进展及机制研究[D].南昌:南昌大学,2021. [100] 王志杰,周杰,沈燕.艾灸对阿尔茨海默病的作用机制研究进展[J].中国中医药信息杂志,2023,30(3):176-181. [101] 贾玉梅,朱才丰,杨坤,等.艾灸督脉对APP/PS1双转基因小鼠mTOR/TFEB通路介导的自噬溶酶体功能及lncRNA H19表达的影响[J].针刺研究,2022,47(8):665-672. |
| [1] | 陶代菊, 苏海玉, 王宇琪, 沈志强, 何 波. 高/低表达miR-122-5p稳转PC12细胞株的构建和鉴定[J]. 中国组织工程研究, 2026, 30(7): 1790-1799. |
| [2] | 侯明凯, 耿斯羽, 秦靖涵, 申莉亚, 黄浩轩, 赵千奥, 张道培, 张怀亮. 前庭周围性眩晕随机对照试验结局指标:中医药治疗的现状分析[J]. 中国组织工程研究, 2026, 30(29): 7696-7705. |
| [3] | 徐东方, 赵 堃, 卢长柱, 王玉阁, 白连杰, 孟凡谋, 王 洋, 姚宏波. 阿尔茨海默病中m6A相关铁死亡基因表达与免疫浸润:机器学习和分子生物学验证[J]. 中国组织工程研究, 2026, 30(24): 6421-6432. |
| [4] | 杨 骏, 李 彬, 邢国刚, 蔡 捷, 刘 璐, 陈 鹏, 张 涛, 付渊博, 刘慧林, . 膜片钳技术在中医药领域的应用:相关文献可视化分析[J]. 中国组织工程研究, 2026, 30(23): 6010-6020. |
| [5] | 孙祖延, 黄文良, 徐 林, 李豪杰, 谢同亮, 杨治航, 邓 江. 胫骨横向骨搬移加速2型糖尿病模型兔足溃疡愈合:环状RNA的参与和调控[J]. 中国组织工程研究, 2026, 30(22): 5639-5649. |
| [6] | 胡亚琳, 黄凤琴, 杨伯银, 罗兴梅. 转录因子EB介导自噬溶酶体通路改善阿尔茨海默病[J]. 中国组织工程研究, 2026, 30(22): 5844-5858. |
| [7] | 徐亚伟, 孟世龙, 张 徐, 汪成杰, 袁一峰, 史晓林, 王 娇, 刘 康. 中药有效成分结合水凝胶修复骨缺损:成功与挑战[J]. 中国组织工程研究, 2026, 30(20): 5295-5303. |
| [8] | 郇康辉, 姜昱建, 卞伟华. DNA损伤激活的非编码RNA促进诱导多能干细胞衍生心肌细胞增殖并抑制凋亡[J]. 中国组织工程研究, 2026, 30(19): 4926-4933. |
| [9] | 廖兴传, 李光第, 吴亚滨, 刘星余, 万佳佳. 非编码RNA调控骨关节炎铁死亡的分子机制[J]. 中国组织工程研究, 2026, 30(18): 4713-4725. |
| [10] | 刘安南, 李建辉, 高 伟, 李 雪, 宋 婧, 邢丽萍, 李虹霖. 铁死亡与阿尔茨海默病的文献计量学分析[J]. 中国组织工程研究, 2026, 30(16): 4278-4288. |
| [11] | 李怡文, 刘飞祥, 张运克. 干细胞调控溶酶体功能治疗溶酶体贮积症[J]. 中国组织工程研究, 2026, 30(1): 145-152. |
| [12] | 于漫亚, 崔 兴. 骨髓微环境中不同细胞对多发性骨髓瘤骨病外泌体环状RNA的贡献及相互作用[J]. 中国组织工程研究, 2026, 30(1): 101-110. |
| [13] | 周盼盼, 崔应麟, 张文涛, 王姝瑞, 陈佳慧, 杨 潼. 细胞自噬在脑缺血损伤中的作用及中药调控机制[J]. 中国组织工程研究, 2025, 29(8): 1650-1658. |
| [14] | 于经邦, 吴亚云. 非编码RNA在肺纤维化过程中的调控作用[J]. 中国组织工程研究, 2025, 29(8): 1659-1666. |
| [15] | 刘凌云, 何贵新, 秦伟彬, 宋 惠, 张利文, 唐伟智, 杨斐斐, 朱子一, 欧阳彬. 中药改善心肌损伤:线粒体钙稳态介导巨噬细胞自噬与焦亡的作用途径[J]. 中国组织工程研究, 2025, 29(6): 1276-1284. |
非编码RNA(non-coding RNA,ncRNA)是指不具有编码蛋白质或多肽能力的RNA分子。常见的非编码RNA主要包括两大类:一类是调节性非编码RNA,如微小RNA(microRNA,miRNA)、
长链非编码RNA(long non-coding RNA,lncRNA)和环状RNA(circular RNA,circRNA),它们主要参与基因表达的调控;另一类是管家非编码RNA,如转运RNA(transfer RNA,tRNA)在细胞中组成型表达,执行基础的生物学功能[8]。近年来,随着生物信息学和高通量测序技术的飞速发展揭示,大量非编码RNA在中枢神经系统中呈现组织特异性富集,其表达谱异常与大脑衰老及神经退行性疾病的发生发展显著相关;这些分子在生理病理状态及发育进程中的动态表达模式与功能也已逐步被阐明[9]。最近研究发现,此类非编码RNA作为关键的基因表达调控因子,与阿尔茨海默病的致病级联反应有关。
中医学将阿尔茨海默病归属于“痴呆、呆病、善忘”等范畴,其病机本质为肾精亏虚,髓海失养,兼夹痰浊、瘀血、毒邪为标,属本虚标实之证[10]。中医治疗以补肾填精、化痰逐瘀、解毒通络为核心治则,并凭借多组分、多途径、多靶点的独特优势,临床疗效显著;现代网络药理学与分子对接技术系统阐明了其通过调控神经炎症、氧化应激及蛋白质异常沉积等多层次机制发挥治疗作用。基于此,此文聚焦阿尔茨海默病关键的病理机制,综述近几年来微小RNA、长链非编码RNA、环状RNA和转运RNA在阿尔茨海默病中的研究进展,以及中医药调控非编码RNA治疗阿尔茨海默病的报道,旨在为中医药现代化研究构建表观遗传调控桥梁,并为临床精准诊疗提供新策略。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1 资料来源
1.1.1 检索人及检索时间 第一作者于2025年7月进行检索。
1.1.2 文献检索时限 2015年1月至2025年7月。
1.1.3 检索数据库 中国知网和PubMed数据库。
1.1.4 检索词 中文检索词为“非编码RNA、微小RNA、长链非编码RNA、环状RNA、转运RNA、阿尔茨海默病、中医药”;英文检索词为“ncRNA,miRNA,lncRNA,circRNA,tRNA,Alzheimer’s disease,traditional Chinese Medicine”。
1.1.5 检索文献类型 综述研究、荟萃分析、基础研究及临床研究。
1.1.6 手工检索情况 无。
1.1.7 检索策略 中国知网和PubMed数据库检索策略,见图1。
1.1.8 检索文献量 共检索得到2 531篇文献,其中中文文献545篇、英文文献1 986篇。
1.2 入选标准
1.2.1 纳入标准 ①关于阿尔茨海默病发病机制的研究;②关于不同类型的非编码RNA参与阿尔茨海默病病理的研究;③中医药调控各类非编码RNA干预阿尔茨海默病进展的研究;④重点纳入阐述清晰、内容丰富、具有创新性的研究。
1.2.2 排除标准 ①与研究主题内容差异大的文献;②重复性研究及陈旧性文献;③文献资料无法获取的部分文献。
1.3 文献质量评估和数据的提取 检索中英文数据库得到与研究内容相关的2 531篇文献,严格按照纳入和排除标准进行筛选,最后纳入101篇进行综述,其中中文文献16篇、英文文献85篇。文献检索流程图见图2。检索方案设计见图3。
3.1 既往他人在该领域研究的贡献和存在的问题 非编码RNA在阿尔茨海默病发病机制中的调控作用取得了一定进展。机制方面,揭示了多种非编码RNA调控病理因素发挥神经保护作用,并系统绘制了竞争性内源RNA网络参与机制的分子图谱;应用层面,外周血非编码RNA标志物的发现,为早期诊断提供了新途径;治疗层面,部分中药及针灸疗法可通过多靶点调控非编码RNA网络延缓疾病进展。但仍存在一些问题:①研究存在明显类型失衡,多聚焦于微小RNA而缺乏对环状RNA、长链非编码RNA及转运RNA衍生小RNA等其他类型的系统探索;②病理机制研究覆盖不足,对调控神经递质、肠脑轴互作、铁死亡等新兴环节关注有限;③阿尔茨海默病的高度异质性及人鼠间物种差异,制约了动物模型中良好实验结果向临床的转化;④技术层面缺乏标准化检测体系与高质量数据库,且多数研究为静态分析,难以揭示非编码RNA在阿尔茨海默病动态演进及脑区特异调控中的时序规律。与此同时,中医药调控非编码RNA防治阿尔茨海默病的研究相对滞后,尚未与“病机-证候”理论体系深度融合,复方整体研究稀缺,“君臣佐使”配伍与非编码RNA网络互作机制亟待阐明;现有动物模型难以模拟中医证候特质,制约“同病异治”原则的实证转化。
3.2 作者综述区别于他人他篇的特点 该综述的特点在于对该领域内多种类型的非编码RNA(包括微小RNA、长链非编码RNA、环状RNA等)在阿尔茨海默病中的作用进行了系统梳理,并纳入了新兴的转运RNA衍生小RNA及其分子机制以充实论述。同时,该文填补了目前中医药通过调控非编码RNA干预该疾病领域缺乏系统性综述的空白。文章以阿尔茨海默病的病理机制为主线,重点阐释了不同非编码RNA及其构成的竞争性内源RNA网络在疾病发生发展中的功能,总结了中医药基于非编码RNA调控的潜在价值与既有证据;并强调未来应深入开展机制研究与临床转化,为中医药调控非编码RNA发展精准治疗策略和新药研发提供重要参考与方向指引。
3.3 综述的局限性 此综述尽可能系统梳理了非编码RNA调控阿尔茨海默病的作用及中医药干预的研究进展,但仍存在一定的局限性:首先,由于该领域的前沿性,多数机制研究仍停留于临床前模型,缺乏临床转化验证;其次,主要基于现有文献进行整合与分析,可能存在文献检索范围不全或选择偏倚,未能涵盖全部相关研究;同时,中医药具有多成分、多靶点的作用特点,其干预非编码RNA的具体活性成分尚未完全明确,机制阐释仍较初步。
3.4 综述的重要意义 阿尔茨海默病发病机制复杂且缺乏根治性疗法,当前研究面临病因不明、早期诊断生物标志物缺乏及单一靶点药物疗效不佳的困境。而非编码RNA调控网络的研究为阐明阿尔茨海默病的多因素致病机制提供了全新视角,尤其在揭示基因表达调控失衡与疾病进展关联方面展现出巨大潜力,为开发早期诊断工具和创新治疗靶点带来了新希望。在此背景下,综述中医药通过多成分、多靶点、多通路协同调控非编码RNA网络的整体干预机制,不仅有助于构建兼具现代靶向治疗精确性和传统医学整体调节优势的新策略,也为克服疾病的难治性挑战及推动中医药的现代化与国际化提供了重要的学术依据和转化前景。
3.5 课题专家组对未来的建议 作者课题组也在逐步开展阿尔茨海默病的相关研究,基于现有问题,课题组专家提出:未来研究应立足于中医病机理论,构建病证结合的动物模型,并整合多组学与生物信息学方法,系统阐释关键非编码RNA的细胞特异性功能及其与中医证候的关联机制;同时加强中药复方整体干预研究,开发高效递送系统提升活性成分的中枢神经靶向性,并通过大样本临床验证建立基于非编码RNA分子分型的中医药精准治疗体系。此外,需推动人源干细胞类器官模型及人源化动物模型的构建与应用,以克服物种差异,最终实现从病机理论向临床应用的转化。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
阿尔茨海默病目前临床治疗主要依赖乙酰胆碱酯酶抑制剂和N-甲基-D-天冬氨酸拮抗剂,虽可一定程度改善认知症状,却难以延缓疾病进展,且常伴有副作用。近年来,非编码RNA因其高度稳定性及易于检测的特性,成为阿尔茨海默病早期诊断的新型生物标志物,并在调控β-淀粉样蛋白沉积、Tau蛋白磷酸化、神经炎症及线粒体功能障碍等核心病理机制中发挥关键作用,为开发靶向治疗策略提供了新方向。尽管反义寡核苷酸和基因编辑技术在靶向致病性非编码RNA方面已取得初步进展,但仍面临递送效率低等技术瓶颈。我国在该领域的研究凸显了中西医结合的特色,尤其是中医药通过多靶点、整体性调控非编码RNA网络干预阿尔茨海默病,显示出独特优势。此文系统总结了当前研究所挖掘出的关键非编码RNA,并深入探讨了中医药通过靶向调控非编码RNA网络进行干预疾病的独特价值与应用前景,为未来研究指出多组学整合、精准干预策略创新以及中医药现代化与国际化等重要方向,以推动阿尔茨海默病防治实现重要突破。此文的核心在于跨学科整合,将非编码RNA分子机制的前沿研究与中医药整体观和辨证论治理念有机融合,也为阿尔茨海默病的系统性诊疗提供新范式。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||