[1] WEN S, DOONER M, PAPA E, et al. Biodistribution of mesenchymal stem cell-derived extracellular vesicles in a radiation injury bone marrow murine model. Int J Mol Sci. 2019;20(21):1-15.
[2] ZHOU R, LONG H, ZHANG B, et al. Salvianolic acid B, an antioxidant derived from Salvia militarize, protects mice against gammaradiationinduced damage through Nrf2/Bach1. Mol Med Rep. 2019;19(2):1309-1317.
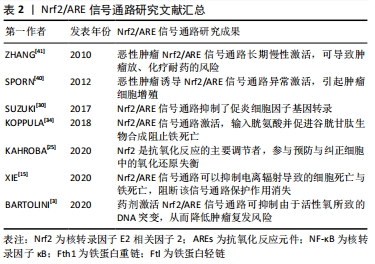
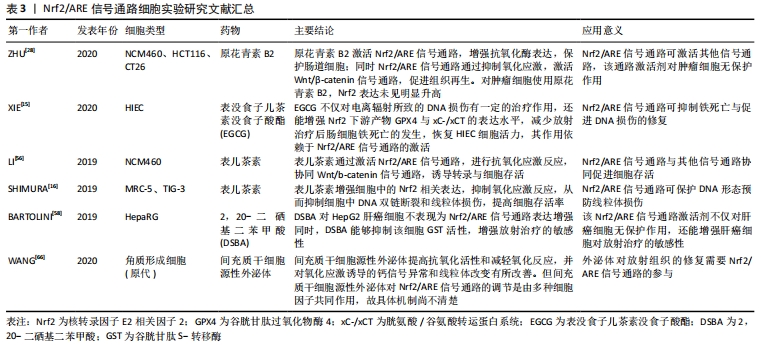
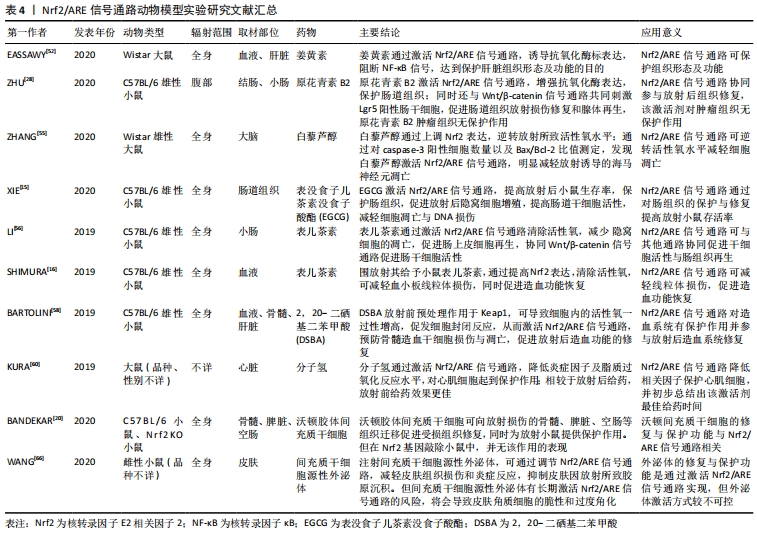
[3] BARTOLINI D, TEW KD, MARINELLI R, et al.Nrf2-modulation by seleno-hormetic agents and its potential for radiation protection. Biofactors. 2020;46(2):239-245.
[4] ANURANJANI, BALA M. Concerted action of Nrf2-ARE pathway, MRN complex, HMGB1 and inflammatory cytokines-implication in modification of radiation damage. Redox Biol. 2014;2:832-846.
[5] ZHANG T, SHI L, LI Y, et al. Polysaccharides extracted from Rheum tanguticum ameliorate radiation-induced enteritis via activation of Nrf2/HO-1. J Radiat Res. 2021;62(1):46-57.
[6] DODSON M, CASTRO-PORTUGUEZ R, ZHANG DD. Nrf2 plays a critical role in mitigating lipid peroxidation and ferroptosis. Redox Biol. 2019; 23:101107.
[7] HYBERTSON B M, GAO B, BOSE SK, et al. Oxidative stress in health and disease: the therapeutic potential of Nrf2activation. Mol Aspects Med. 2011;32(4-6):234-246.
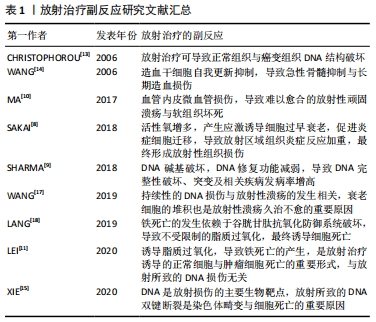
[8] SAKAI Y, YAMAMORI T, YOSHIKAWA Y, et al. NADPH oxidase 4 mediates ROS production in radiation-induced senescent cells and promotes migration of inflammatory cells. Free Radic Res. 2018;52(1):92-102.
[9] SHARMA N, CHAKRAVARTHY S, LONGLEY MJ, et al. The C-terminal tail of the NEIL1 DNA glycosylase interacts with the human mitochondrial single-strandedDNAbinding protein.DNARepair (Amst). 2018;65:11-19.
[10] MA X, JIN Z, LI G, et al. Classification of chronic radiation-induced ulcers in the chest wall after surgery in breast cancers. Radiat Oncol. 2017;12(1):135.
[11] LEI G, ZHANG Y, KOPPULA P, et al. The role of ferroptosis in ionizing radiation-induced cell death and tumor suppression. Cell Res. 2020; 30(2):146-162.
[12] LEBARON TW, KURA B, KALOCAYOVA B, et al. A New approach for the prevention and treatment of cardiovascular disorders. molecular hydrogen significantly reduces the effects of oxidative stress. Molecules. 2019;24(11):2076.
[13] CHRISTOPHOROU MA, RINGSHAUSEN I, FINCH AJ, et al. The pathological response toDNAdamage does not contribute to p53-mediated tumour suppression. Nature. 2006;443(7108):214-217.
[14] WANG Y, SCHULTE BA, LARUE AC, et al. Total body irradiation selectively induces murine hematopoietic stem cell senescence. Blood. 2006;107(1):358-366.
[15] XIE LW, CAI S, ZHAO TS, et al. Green tea derivative (-)-epigallocatechin-3-gallate (EGCG) confers protection against ionizing radiation-induced intestinal epithelial cell death both in vitro and in vivo. Free Radic Biol Med. 2020;161:175-186.
[16] SHIMURA T, KOYAMA M, AONO D, et al. Epicatechin as a promising agent to countermeasure radiation exposure by mitigating mitochondrial damage in human fibroblasts and mouse hematopoietic cells. FASEB J. 2019;33(6):6867-6876.
[17] WANG Z, CHEN Z, JIANG Z, et al. Cordycepin prevents radiation ulcer by inhibiting cell senescence viaNrf2and AMPK in rodents. Nat Commun. 2019;10(1):2538.
[18] LANG X, GREEN MD, WANG W, et al. Radiotherapy and immunotherapy promote tumoral lipid oxidation and ferroptosis via synergistic repression of SLC7A11. Cancer Discov. 2019;9(12):1673-1685.
[19] MISHRA K, ALSBEIH G. Appraisal of biochemical classes of radioprotectors: evidence, current status and guidelines for future development. 3 Biotech. 2017;7(5):292.
[20] BANDEKAR M, MAURYA DK, SHARMA D, et al. Xenogeneic transplantation of human WJ‐MSCs rescues mice from acute radiation syndrome via Nrf-2-dependent regeneration of damaged tissues. Am J Transplant. 2020;20(8):2044-2057.
[21] KOUKOURAKIS MI. Radiation damage and radioprotectants: new concepts in the era of molecular medicine. Br J Radiol. 2012;85(1012):313-330.
[22] OBRADOR E, SALVADOR R, VILLAESCUSA JI, et al. Radioprotection and radiomitigation: from the bench to clinical practice. Biomedicines. 2020;8(11):461.
[23] SINGH VK, SEED TM. Pharmacological management of ionizing radiation injuries: current and prospective agents and targeted organ systems. Expert Opin Pharmacother. 2020;21(3):317-337.
[24] SYKIOTIS GP, BOHMANN D. Stress-activated cap’n’collar transcription factors in aging and human disease. Sci Signal. 2010;3(112):re3.
[25] KAHROBA H, DAVATGARAN-TAGHIPOUR Y. Exosomal Nrf2: from anti-oxidant and anti-inflammation response to wound healing and tissue regeneration in aged-related diseases. Biochimie. 2020;171-172:103-109.
[26] LOBODA A, DAMULEWICZ M, PYZA E, et al. Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: an evolutionarily conserved mechanism. Cell Mol Life Sci. 2016;73(17):3221-3247.
[27] HAMADA N, SHIMURA T, NAKASHIRO C, et al. Induction of oxidative stress biomarkers following whole-body irradiation in mice. Plos One. 2020;15(10):e0240108.
[28] ZHU X, TIAN X, YANG M, et al. Procyanidin B2 promotes intestinal injury repair and attenuates colitis-associated tumorigenesis via suppression of oxidative stress in mice. Antioxid Redox Signal. 2020;35(2):75-92.
[29] YUAN L, DUAN X, ZHANG R, et al. Aloe polysaccharide protects skin cells from UVB irradiation through Keap1/Nrf2/ARE signal pathway. J Dermatol Treat. 2019;31(3):300-308.
[30] SUZUKI T, YAMAMOTO M. Stress-sensing mechanisms and the physiological roles of the Keap1-Nrf2 system during cellular stress. J Biol Chem. 2017;292(41):16817-16824.
[31] SLEZAK J, KURA B, LEBARON TW, et al. Oxidative stress and pathways of molecular hydrogen effects in medicine. Curr Pharm Des. 2020; 27(5):610-625.
[32] URSINI F, MAIORINO M. Lipid peroxidation and ferroptosis: the role of GSH and GPx4. Free Radic Biol Med. 2020;152:175-185.
[33] ARENSMAN MD, YANG XS, LEAHY DM, et al. Cystine-glutamate antiporter xCT deficiency suppresses tumor growth while preserving antitumor immunity. Proc Natl Acad Sci U S A. 2019;116(19):9533-9542.
[34] KOPPULA P, ZHANG Y, ZHUANG L, et al. Amino acid transporter SLC7A11/xCT at the crossroads of regulating redox homeostasis and nutrient dependency of cancer. Cancer Commun (Lond). 2018;38(1):12.
[35] KERINS MJ, OOI A. The roles of Nrf2 in modulating cellular iron homeostasis. Antioxid Redox Signal. 2018;29(17):1756-1773.
[36] SAHA S, BUTTARI B, PANIERI E, et al. An overview of Nrf2signaling pathway and its role in inflammation. Molecules. 2020;25(22):5474.
[37] PANIERI E, TELKOPARAN-AKILLILAR P, SUZEN S, et al. The Nrf2/KEAP1 axis in the regulation of tumor metabolism: mechanisms and therapeutic perspectives. Biomolecules. 2020;10(5):791.
[38] HAYES JD, DINKOVA-KOSTOVA AT. The Nrf2 regulatory network provides an interface between redox and intermediary metabolism. Trends Biochem Sci. 2014;39(4):199-218.
[39] JIANG GP, LIAO YJ, HUANG LL, et al. Effects and molecular mechanism of pachymic acid on ferroptosis in renal ischemia reperfusion injury. Mol Med Rep. 2021;23(1):63.
[40] SPORN MB, LIBY KT. Nrf2and cancer: the good, the bad and the importance of context. Nat Rev Cancer. 2012;12(8):564-571.
[41] ZHANG DD. TheNrf2-Keap1-ARE signaling pathway: the regulation and dual function of Nrf2in cancer. Antioxid Redox Signal. 2010;13(11): 1623-1626.
[42] MIURA S, YAMAGUCHI M, YOSHINO H, et al. Dose-dependent increase of Nrf2target gene expression in mice exposed to ionizing radiation. Radiat Res. 2019;191(2):176-188.
[43] KIRSCH DG, SANTIAGO PM, DI TOMASO E, et al. p53 controls radiation-induced gastrointestinal syndrome in mice independent of apoptosis. Science. 2010;327(5965):593-596.
[44] ABDEL-MAGIED N, SHEDID SM. The effect of naringenin on the role of nuclear factor (erythroid-derived 2)-like2 (Nrf2) and haem oxygenase 1 (HO-1) in reducing the risk of oxidative stress-related radiotoxicity in the spleen of rats. Environ Toxicol. 2019;34(7):788-795.
[45] BAILLY C. Potential use of edaravone to reduce specific side effects of chemo-, radio- and immuno-therapy of cancers. Int Immunopharmacol. 2019;77:105967.
[46] O’BRIEN J, WENDELL SG. Electrophile modulation of inflammation: a two-hit approach. Metabolites. 2020;10(11):453.
[47] ZHANG DD. Mechanistic studies of theNrf2-Keap1 signaling pathway. Drug Metab Rev. 2006;38(4):769-789.
[48] LU H, ZHOU Q, HE J, et al. Recent advances in the development of protein-protein interactions modulators: mechanisms and clinical trials. Signal Transduct Target Ther. 2020;5(1):213.
[49] JIANG ZY, LU MC, YOU QD. Discovery and Development of Kelch-like ECH-Associated Protein 1. Nuclear Factor Erythroid 2-Related Factor 2 (KEAP1:NRF2) Protein-Protein Interaction Inhibitors: achievements, Challenges, and Future Directions. J Med Chem. 2016;59(24):10837-10858.
[50] MENG X, WADDINGTON JC, TAILOR A, et al. CDDO-imidazolide targets multiple amino acid residues on the Nrf2 adaptor, Keap1. J Med Chem. 2020;63(17):9965-9976.
[51] NOUREDDIN SA, EL-SHISHTAWY RM, AL-FOOTY KO. Curcumin analogues and their hybrid molecules as multifunctional drugs. Eur J Med Chem. 2019;182:111631.
[52] EASSAWY MMT, SALEM AA, ISMAIL AFM. Biochemical study on the protective effect of curcumin on acetaminophen and gamma-irradiation induced hepatic toxicity in rats. Environ Toxicol. 2020. doi: 10.1002/tox.23077.
[53] DINKOVA-KOSTOVA AT, WANG XJ. Induction of the Keap1/Nrf2/ARE pathway by oxidizable diphenols. Chem Biol Interact. 2011;192(1-2): 101-106.
[54] ZHENG Y, MORRIS A, SUNKARA M, et al. Epigallocatechin-gallate stimulates NF-E2-related factor and heme oxygenase-1 via caveolin-1 displacement. J Nutr Biochem. 2012;23(2):163-168.
[55] ZHANG Y, ZHU XB, ZHAO JC, et al. Neuroprotective effect of resveratrol against radiation after surgically induced brain injury by reducing oxidative stress, inflammation, and apoptosis throughNrf2/HO-1/NF-kappaB signaling pathway. J Biochem Mol Toxicol. 2020;34(12):e22600.
[56] LI Y, MA S, ZHANG Y, et al. (-)-Epicatechin mitigates radiation-induced intestinal injury and promotes intestinal regeneration via suppressing oxidative stress. Free Radic Res. 2019;53(8):851-864.
[57] TINDELL R, WALL SB, LI Q, et al. Selenium supplementation of lung epithelial cells enhances nuclear factor E2-related factor 2 (Nrf2) activation following thioredoxin reductase inhibition. Redox Biol. 2018;19:331-338.
[58] BARTOLINI D, WANG Y, ZHANG J, et al. A seleno-hormetine protects bone marrow hematopoietic cells against ionizing radiation-induced toxicities. PLoS One. 2019;14(4):e0205626.
[59] SCHILLING D, HEROLD B, COMBS SE, et al. Selenium does not affect radiosensitivity of breast cancer cell lines. Radiat Environ Biophys. 2019;58(3):433-438.
[60] KURA B, BAGCHI AK, SINGAL PK, et al. Molecular hydrogen: potential in mitigating oxidative-stress-induced radiation injury. Can J Physiol Pharmacol. 2019;97(4):287-292.
[61] ABDEL-MAGEED AS, SENAGORE AJ, PIETRYGA DW, et al. Intravenous administration of mesenchymal stem cells genetically modified with extracellular superoxide dismutase improves survival in irradiated mice. Blood. 2009;113(5):1201-1203.
[62] SAHA S, BHANJA P, KABARRITI R, et al. Bone marrow stromal cell transplantation mitigates radiation-induced gastrointestinal syndrome in mice. PLoS One. 2011;6(9):e24072.
[63] 刘涛,张霓霓,黄桂林.细胞外囊泡与放射性组织损伤的关系[J].中国组织工程研究,2021,25(13):2121-2126.
[64] FAIS S, O’DRISCOLL L, BORRAS FE, et al. Evidence-based clinical use of nanoscale extracellular vesicles in nanomedicine. ACS Nano. 2016; 10(4):3886-3899.
[65] CHEN B, SUN Y, ZHANG J, et al. Human embryonic stem cell-derived exosomes promote pressure ulcer healing in aged mice by rejuvenating senescent endothelial cells. Stem Cell Res Ther. 2019;10(1):142.
[66] WANG T, JIAN Z, BASKYS A, et al. MSC-derived exosomes protect against oxidative stress-induced skin injury via adaptive regulation of theNrf2defense system. Biomaterials. 2020;257:120264.
[67] BODEGA G, ALIQUE M, PUEBLA L, et al. Microvesicles: ROS scavengers and ROS producers. J Extracell Vesicles. 2019;8(1):1626654.
|