中国组织工程研究 ›› 2020, Vol. 24 ›› Issue (30): 4795-4801.doi: 10.3969/j.issn.2095-4344.2836
• 人工假体 artificial prosthesis • 上一篇 下一篇
血小板计数可作为预测关节假体周围感染的新指标
曹厚然1,邓 鹏2,叶鹏程2,揭 珂1,曾建春2,冯文俊2,陈锦伦2,齐新宇1,李 杰2,谭雪秋1,张海涛1,曾意荣2
- 1广州中医药大学第一临床医学院,广东省广州市 510006;2广州中医药大学第一附属医院,广东省广州市 510405
-
收稿日期:2020-01-07修回日期:2020-01-16接受日期:2020-03-04出版日期:2020-10-28发布日期:2020-09-19 -
通讯作者:曾意荣,博士,主任医师,广州中医药大学第一附属医院三骨科,广东省广州市 510405 -
作者简介:曹厚然,男,1993年生,广东省广州市人,汉族,广州中医药大学在读硕士,主要从事髋膝关节疾病的研究。 -
基金资助:广州中医药大学第一附属医院创新强院工程培育项目(2019IIT35)
Platelet count as a novel potential predictor of periprosthetic joint infection
Cao Houran1, Deng Peng2, Ye Pengcheng2, Jie Ke1, Zeng Jianchun2, Feng Wenjun2, Chen Jinlun2, Qi Xinyu1, Li Jie2, Tan Xueqiu1, Zhang Haitao1, Zeng Yirong2
- 1First Clinical Medical College of Guangzhou University of Chinese Medicine, Guangzhou 510006, Guangdong Province, China; 2First Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China
-
Received:2020-01-07Revised:2020-01-16Accepted:2020-03-04Online:2020-10-28Published:2020-09-19 -
Contact:Zeng Yirong, MD, Chief physician, First Affiliated Hospital of Guangzhou University of Chinese Medicine, Guangzhou 510405, Guangdong Province, China -
About author:Cao Houran, Master candidate, First Clinical Medical College of Guangzhou University of Chinese Medicine, Guangzhou 510006, Guangdong Province, China -
Supported by:the Moral Education Program of Innovation and Strong Academy Project of First Affiliated Hospital of Guangzhou University of Chinese Medicine, No. 2019IIT35
摘要:
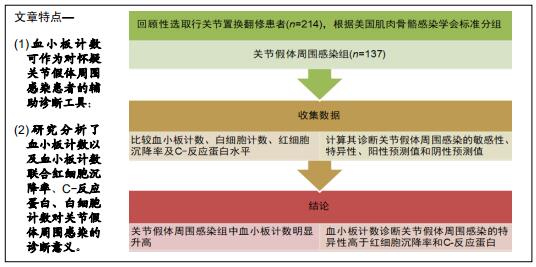
文题释义:
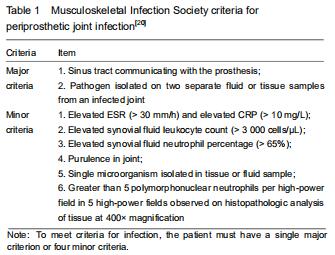
关节假体周围感染:是关节置换后灾难性并发症之一,由于缺乏特异性指标,细菌培养阳性率低,其诊断是临床研究的难点。
血小板计数:是指计数单位容积血液中血小板的数量。目前研究发现在一些感染性疾病中,血清中血小板计数会升高,提示血小板计数可作为预测感染性疾病的诊断指标。
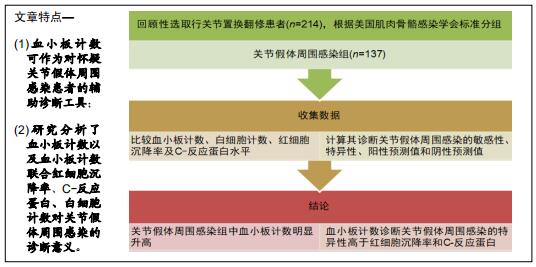
背景:对于患者而言,以关节腔液检测关节假体周围感染是侵入性的并且是痛苦的。由于血小板计数是一种常规的血液检测措施,已被用作感染性疾病的预测指标,故推测其可能作为关节假体周围感染的指标之一。
目的:评价血小板计数联合白细胞计数、红细胞沉降率、C-反应蛋白对诊断关节假体周围感染的准确性。
方法:回顾性收集了2013年3月至2018年12月在广州中医药大学第一附属医院行关节置换翻修的患者。根据美国肌肉骨骼感染学会的标准,其中77例确诊为关节假体周围感染,其余137例为无菌性松动。收集并比较两组的血小板计数、白细胞计数、红细胞沉降率及C-反应蛋白水平,计算其诊断关节假体周围感染的敏感性、特异性、阳性预测值和阴性预测值。
结果与结论:关节假体周围感染组中血小板计数明显升高,其对诊断关节假体周围感染的敏感性和特异性分别为64.94%和86.13%。血小板计数诊断关节假体周围感染的特异性高于红细胞沉降率和C-反应蛋白。因此认为血小板计数对诊断关节假体周围感染具有一定的参考价值。对于怀疑关节假体周围感染的患者,可通过检测血小板计数辅助诊断。
orcid: 0000-0002-6287-0322(Cao Houran)中国组织工程研究杂志出版内容重点:人工关节;骨植入物;脊柱;骨折;内固定;数字化骨科;组织工程
中图分类号:
引用本文
曹厚然, 邓 鹏, 叶鹏程, 揭 珂, 曾建春, 冯文俊, 陈锦伦, 齐新宇, 李 杰, 谭雪秋, 张海涛, 曾意荣. 血小板计数可作为预测关节假体周围感染的新指标[J]. 中国组织工程研究, 2020, 24(30): 4795-4801.
Cao Houran, Deng Peng, Ye Pengcheng, Jie Ke, Zeng Jianchun, Feng Wenjun, Chen Jinlun, Qi Xinyu, Li Jie, Tan Xueqiu, Zhang Haitao, Zeng Yirong. Platelet count as a novel potential predictor of periprosthetic joint infection[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(30): 4795-4801.
The study was approved by the Institutional Review Board of the First Affiliated Hospital of Guangzhou University of Chinese Medicine (approval No. 2019205). This single-center, retrospective, cohort study enrolled patients who underwent revision arthroplasty because of delayed or chronic infection of joint arthroplasties and aseptic loosening of an implant from March 2013 to December 2018 in the First Affiliated Hospital of Guangzhou University of Chinese Medicine. PJI was diagnosed based on the MSIS criteria noted in Table 1[20]. Patients were excluded if they had hematological disease or underwent a blood transfusion within the last year, used ongoing antiplatelet drugs, had a history of recent trauma or dislocation (within 2 weeks), used lipid emulsion, associated with hepatic disease or other site infections.
A total of 214 patients were recruited for the final analysis. Based on the MSIS criteria, 77 patients were classified into the PJI group (129 hips and 8 knees), while 137 patients were classified into the aseptic loosening group. The mean age in the PJI group was 60.6±13.1 years (range, 21-80 years); of them, 30 were men and 47 were women. The mean age in the aseptic loosening group was 64.0±11.7 years (range, 30-86 years); of them, 64 were men and 73 were women. No significant intergroup differences were detected in gender or age (P > 0.05). The characteristics of the enrolled patients are shown in Table 2.
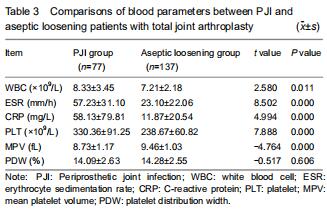
Serum PLT was significantly higher in the PJI group (330.36 ± 91.25 × 109/L) than in the aseptic loosening group (238.67 ± 60.82 × 109/L) (P < 0.001). WBC, ESR, and CRP values were also significantly higher among patients with PJI (P < 0.05). The mean MPV was also significantly higher in the PJI group than in the aseptic loosening group (P < 0.001). No significant intergroup differences were detected in the PDW values (P > 0.05; Table 3).
The ROC curves showed that ESR had the highest AUC (0.829), followed by CRP (0.820), and PLT (0.804). The AUCs of these three markers were all between 0.8 and 0.9, indicating good diagnostic value for PJI. In contrast, MPV had an AUC of 0.695, which was the second lowest value but slightly higher than the AUC of WBC (0.619). The AUCs of these two markers were both lower than 0.7, indicating poor diagnostic value (Figure 1).
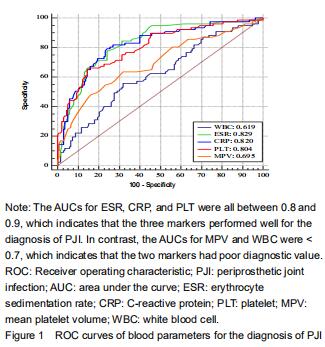
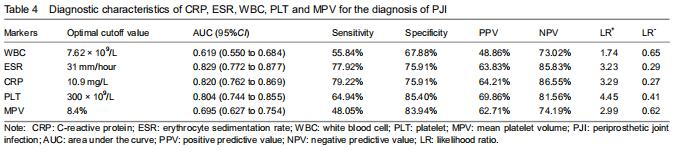
The serum PLT demonstrated a sensitivity of 64.94% and a specificity of 85.40% for diagnosing PJI. Using Youden’s index, the optimal cutoff value for PLT was 300 × 109/L, which resulted in a good PPV and NPV of 69.56% and 81.56%, respectively. Based on the ROC analysis, when WBC was above 7.62 × 109/L, the sensitivity, specificity, PPV, and NPV were 55.84%, 67.88%, 48.86%, and 73.02%, respectively. Using a cutoff value for ESR of 31 mm/h, the sensitivity, specificity, PPV, and NPV were 77.92%, 75.91%, 63.83%, and 85.83%; using a cutoff value for CRP at 10.9 mg/L, the sensitivity, specificity, PPV, and NPV were 79.22%, 75.91%, 64.21%, and 86.55%, respectively. For MPV, the optimal cutoff value was 8.4%, resulting in sensitivity, speci?city, PPV, and NPV of 48.05%, 83.94%, 62.71%, and 74.19%, respectively (Table 4).
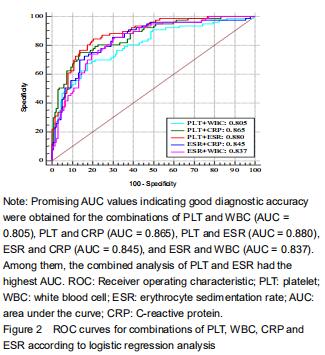
Further analyses of the combined diagnostic value of PLT, WBC, ESR, and CRP were performed to improve their diagnostic accuracies. The combination of serum PLT and WBC/ESR improved the specificity and specificity (Table 5). PLT combined with the CRP improved the specificity but decreased the sensitivity. Promising AUC values indicating good diagnostic accuracy were obtained for the combinations of PLT and WBC (AUC = 0.805), PLT and CRP (AUC = 0.865), PLT and ESR (AUC = 0.880), ESR and CRP (AUC = 0.845), and ESR and WBC (AUC = 0.837) (Figure 2). Among them, the combined analysis of PLT and ESR had the highest AUC. The sensitivity, specificity, and AUC were 84.42%, 79.96%, and 0.880 (95%CI, 0.829-0.920).
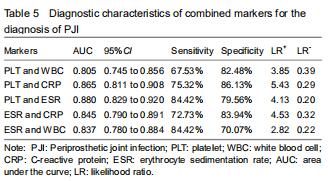
Further analyses of the combined diagnostic value of PLT, WBC, ESR, and CRP were performed to improve their diagnostic accuracies. The combination of serum PLT and WBC/ESR improved the specificity and specificity (Table 5). PLT combined with the CRP improved the specificity but decreased the sensitivity. Promising AUC values indicating good diagnostic accuracy were obtained for the combinations of PLT and WBC (AUC = 0.805), PLT and CRP (AUC = 0.865), PLT and ESR (AUC = 0.880), ESR and CRP (AUC = 0.845), and ESR and WBC (AUC = 0.837) (Figure 2). Among them, the combined analysis of PLT and ESR had the highest AUC. The sensitivity, specificity, and AUC were 84.42%, 79.96%, and 0.880 (95%CI, 0.829-0.920).
|
[1] KIM K, ZHU M, CAVADINO A, et al. Failed debridement and implant retention does not compromise the success of subsequent staged revision in infected total knee arthroplasty. J Arthroplasty. 2019;34(6):1214-1220.e1.
[2] PETIS SM, PERRY KI, MABRY TM, et al. Two-stage exchange protocol for periprosthetic joint infection following total knee arthroplasty in 245 knees without prior treatment for infection. J Bone Joint Surg Am. 2019; 101(3):239-249.
[3] PREOBRAZHENSKY PM, BOZHKOVA SA, KAZEMIRSKY AV, et al. Functional outcome of two-stage reimplantation in patients with periprosthetic joint infection after primary total knee arthroplasty. Int Orthop. 2019;43(11):2503-2509.
[4] SRIVASTAVA K, BOZIC KJ, SILVERTON C, et al. Reconsidering strategies for managing chronic periprosthetic joint infection in total knee arthroplasty: using decision analytics to find the optimal strategy between one-stage and two-stage total knee revision. J Bone Joint Surg Am. 2019;101(1):14-24.
[5] RENARD G, LAFFOSSE JM, TIBBO M, et al. Periprosthetic joint infection in aseptic total hip arthroplasty revision. Int Orthop. 2020;44(4):735-741.
[6] NIKOLAUS OB, MCLENDON PB, HANSSEN AD, et al. Factors associated with 20-year cumulative risk of infection after aseptic index revision total knee arthroplasty. J Arthroplasty. 2016;31(4):872-877.
[7] PARVIZI J, GEHRKE T; International Consensus Group on Periprosthetic Joint Infection. Definition of periprosthetic joint infection. J Arthroplasty. 2014;29(7):1331.
[8] DI BENEDETTO P, BUTTIRONI MM, CAUSERO A. Biomarkers and infections in orthopedics: our experience and literature review. J Biol Regul Homeost Agents. 2018; 32(6 Suppl. 1):51-56.
[9] SALEH A, GEORGE J, FAOUR M, et al. Serum biomarkers in periprosthetic joint infections. Bone Joint Res. 2018;7(1): 85-93.
[10] KHEIR MM, TAN TL, SHOHAT N, et al. Routine diagnostic tests for periprosthetic joint infection demonstrate a high false-negative rate and are influenced by the infecting organism. J Bone Joint Surg Am. 2018;100(23):2057-2065.
[11] AHMAD SS, HIRSCHMANN MT, BECKER R, et al. A meta-analysis of synovial biomarkers in periprosthetic joint infection: Synovasure™ is less effective than the ELISA-based alpha-defensin test. Knee Surg Sports Traumatol Arthrosc. 2018;26(10):3039-3047.
[12] ZHANG Z, JI Y, WANG Z, et al. The association between platelet indices and deep surgical site infection after open induction internal fixation for traumatic limb fractures. Infect Drug Resist. 2018;11:2533-2538.
[13] DEMIRDAL T, SEN P. The significance of neutrophil-lymphocyte ratio, platelet-lymphocyte ratio and lymphocyte-monocyte ratio in predicting peripheral arterial disease, peripheral neuropathy, osteomyelitis and amputation in diabetic foot infection. Diabetes Res Clin Pract. 2018;144:118-125.
[14] GUCLU M, FARUQ AGAN A. Association of severity of helicobacter pylori infection with peripheral blood neutrophil to lymphocyte ratio and mean platelet volume. Euroasian J Hepatogastroenterol. 2017;7(1):11-16.
[15] CELIK U, CELIK T, TOLUNAY O, et al. Platelet indices in the diagnosis of ventriculoperitoneal shunt infection in children. Turk Neurosurg. 2017;27(4):590-593.
[16] LEE MY, KIM YJ, LEE HJ, et al. Mean platelet volume in mycobacterium tuberculosis infection. Biomed Res Int. 2016;2016:7508763.
[17] KIM JK, JEON JS, KIM JW, et al. Correlation between abnormal platelet count and respiratory viral infection in patients from Cheonan, Korea. J Clin Lab Anal. 2016;30(3): 185-189.
[18] LEE IR, SHIN JI, PARK SJ, et al. Mean platelet volume in young children with urinary tract infection. Sci Rep. 2015;5: 18072.
[19] ZHANG W, GUO Y, KUSS M, et al. Platelet-rich plasma for the treatment of tissue infection: preparation and clinical evaluation. Tissue Eng Part B Rev. 2019;25(3):225-236.
[20] OSMON DR, BERBARI EF, BERENDT AR, et al. Diagnosis and management of prosthetic joint infection: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2013;56(1):e1-e25.
[21] PARVIZI J, TAN TL, GOSWAMI K, et al. The 2018 Definition of periprosthetic hip and knee infection: an evidence-based and validated criteria. J Arthroplasty. 2018;33(5):1309-1314.e2.
[22] YOUDEN WJ. Index for rating diagnostic tests. Cancer. 1950;3(1):32-35.
[23] ADAMS JE, CAPO JT, RIZZO M. International consensus on periprosthetic joint infection. Acta Orthop Belg. 2014; 80(3):293-300.
[24] YOON JR, YANG SH, SHIN YS. Diagnostic accuracy of interleukin-6 and procalcitonin in patients with periprosthetic joint infection: a systematic review and meta-analysis. Int Orthop. 2018;42(6):1213-1226.
[25] XIE K, DAI K, QU X, et al. Serum and synovial fluid interleukin-6 for the diagnosis of periprosthetic joint infection. Sci Rep. 2017;7(1):1496.
[26] RANDAU TM, FRIEDRICH MJ, WIMMER MD, et al. Interleukin-6 in serum and in synovial fluid enhances the differentiation between periprosthetic joint infection and aseptic loosening. PLoS One. 2014;9(2):e89045.
[27] PANNU TS, VILLA JM, RIESGO AM, et al. Serum D-dimer in the diagnosis of periprosthetic knee infection: where are we today? J Knee Surg. 2020;33(2):106-110.
[28] XIONG L, LI S, DAI M. Comparison of D-dimer with CRP and ESR for diagnosis of periprosthetic joint infection. J Orthop Surg Res. 2019;14(1):240.
[29] QIN L, LI F, GONG X, et al. Combined measurement of D-dimer and C-reactive protein levels: highly accurate for diagnosing chronic periprosthetic joint infection. J Arthroplasty. 2020;35(1):229-234.
[30] SHAHI A, KHEIR MM, TARABICHI M, et al. Serum D-dimer test is promising for the diagnosis of periprosthetic joint infection and timing of reimplantation. J Bone Joint Surg Am. 2017;99(17):1419-1427.
[31] LEE YS, LEE YK, HAN SB, et al. Natural progress of D-dimer following total joint arthroplasty: a baseline for the diagnosis of the early postoperative infection. J Orthop Surg Res. 2018;13(1):36.
[32] DEIRMENGIAN C, KARDOS K, KILMARTIN P, et al. Diagnosing periprosthetic joint infection: has the era of the biomarker arrived?. Clin Orthop Relat Res. 2014;472(11): 3254-3262. [33] WYATT MC, BESWICK AD, KUNUTSOR SK, et al. The alpha-defensin immunoassay and leukocyte esterase colorimetric strip test for the diagnosis of periprosthetic infection: a systematic review and meta-analysis. J Bone Joint Surg Am. 2016;98(12):992-1000.
[34] DEIRMENGIAN C, KARDOS K, KILMARTIN P, et al. The alpha-defensin test for periprosthetic joint infection responds to a wide spectrum of organisms. Clin Orthop Relat Res. 2015;473(7):2229-2235.
[35] ZAGRA L, VILLA F, CAPPELLETTI L, et al. Can leucocyte esterase replace frozen sections in the intraoperative diagnosis of prosthetic hip infection? Bone Joint J. 2019; 101-B(4):372-377.
[36] GAUTAM VK, SAINI R, SHARMA S. Effectiveness of leucocyte esterase as a diagnostic test for acute septic arthritis. J Orthop Surg (Hong Kong). 2017;25(1): 2309499016685019.
[37] LI R, SHAO HY, HAO LB, et al. Plasma fibrinogen exhibits better performance than plasma d-dimer in the diagnosis of periprosthetic joint infection: a multicenter retrospective study. J Bone Joint Surg Am. 2019;101(7):613-619.
[38] CARLI AV, ABDELBARY H, AHMADZAI N, et al. Diagnostic accuracy of serum, synovial, and tissue testing for chronic periprosthetic joint infection after hip and knee replacements: a systematic review. J Bone Joint Surg Am. 2019;101(7):635-649.
[39] CHEN CY, ESSIEN MD, JOHNSON AJ, et al. Use of mean platelet volume in the assessment of intrauterine infection in newborns with combined thrombocytopenia and leukopenia at birth. J Matern Fetal Neonatal Med. 2019. doi:10.1080/14767058.2019.1608174.
[40] MURATOGLU M, KAYIPMAZ AE, KAVALCI C, et al. Platelet-to-lymphocyte ratio as a potential indicator of infection-associated emergency visits of renal transplant recipients. Exp Clin Transplant. 2019. doi:10.6002/ect.2018.0249.
[41] GUPTA P, GUDDATTU V, SARAVU K. Characterization of platelet count and platelet indices and their potential role to predict severity in malaria. Pathog Glob Health. 2019; 113(2):86-93.
[42] YUYUN D, ZHIHUA T, HAIJUN W, et al. Predictive value of the red blood cell distribution width-to-platelet ratio for hepatic fibrosis. Scand J Gastroenterol. 2019;54(1):81-86.
[43] RUBIO E, ALEJO-CANCHO I, AYLAGAS C, et al. Diagnostic value of platelet and leukocyte counts in the differential diagnosis of fever in the returning traveler. Am J Trop Med Hyg. 2019;100(2):470-475.
[44] ABDEL-RAZIK A, ELDARS W, RIZK E. Platelet indices and inflammatory markers as diagnostic predictors for ascitic fluid infection. Eur J Gastroenterol Hepatol. 2014;26(12): 1342-1347.
[45] KITAZAWA T, YOSHINO Y, TATSUNO K, et al. Changes in the mean platelet volume levels after bloodstream infection have prognostic value. Intern Med. 2013;52(13):1487-1493.
[46] COULDWELL G, MACHLUS KR. Modulation of megakaryopoiesis and platelet production during inflammation. Thromb Res. 2019;179:114-120. [47] SENINI V, AMARA U, PAUL M, et al. Porphyromonas gingivalis lipopolysaccharide activates platelet Cdc42 and promotes platelet spreading and thrombosis. J Periodontol. 2019;90(11):1336-1345. |
| [1] | 宋 珊, 胡方媛, 乔 军, 王 佳, 张升校, 李小峰. 基于生物信息学途径认识骨关节炎滑膜的生物学标志物[J]. 中国组织工程研究, 2021, 25(5): 785-790. |
| [2] | 马 瑞, 王家麟, 吴梦军, 葛 莹, 王 伟, 王坤正. 关节置换后假体周围感染病原菌分布及耐药情况与治疗周期的关系[J]. 中国组织工程研究, 2021, 25(3): 380-385. |
| [3] | 王秋霏, 顾 叶, 彭育沁, 薛 峰, 巨 荣, 朱 锋, 王熠军, 耿德春, 徐耀增. 人工假体磨损颗粒作用下Wnt/β-catenin信号通路对成骨细胞的影响[J]. 中国组织工程研究, 2021, 25(24): 3894-3901. |
| [4] | 程崇杰, 闫 延, 张启栋, 郭万首. D-二聚体在诊断假体周围感染中的应用价值及准确性:系统评价与荟萃分析[J]. 中国组织工程研究, 2021, 25(24): 3921-3928. |
| [5] | 刘志伟, 谢 瑞, 孙 凯, 李凯明, 汪雄伟, 展嘉文, 朱立国. 颈源性头痛诊断标准解读:诊断与鉴别诊断中的问题与认识[J]. 中国组织工程研究, 2021, 25(23): 3746-3751. |
| [6] | 蒋昇源, 李 丹, 姜建浩, 杨上游, 杨淑野. 假体无菌性松动过程中Co2+对成骨前体细胞的生物学反应[J]. 中国组织工程研究, 2021, 25(21): 3292-3299. |
| [7] | 陈 登, 张亚鑫, 戴纪杭, 陈铎允, 孙 钰. 人工髋关节置换后发热的相关因素分析[J]. 中国组织工程研究, 2021, 25(18): 2846-2850. |
| [8] | 刘子歌, 刘心蕊, 李 燕, 宋国瑞, 张 晨, 陈德胜. 汉防己甲素干预假体周围磨损颗粒诱导下骨溶解模型的体外实验[J]. 中国组织工程研究, 2021, 25(15): 2358-2363. |
| [9] | 周 祺, 高 益, 魏 康, 黎 俊, 徐建达, 蒋 阳, 瞿玉兴. 全膝关节置换治疗类风湿关节炎:关节功能及相关生化指标的变化[J]. 中国组织工程研究, 2020, 24(9): 1337-1341. |
| [10] | 房 燚, 赵文志, 潘德悦, 韩 鑫, 张 路, 何洪涛, 石 峰, 田廷啸. 肩锁关节脱位研究:如何达到解剖复位和持续性稳定及关节微动[J]. 中国组织工程研究, 2020, 24(5): 796-802. |
| [11] | 唐金龙, 杨冠杰, 刘 磊, 徐石庄, 赵凤朝. 全膝关节置换后双下肢长度及相关功能变化[J]. 中国组织工程研究, 2020, 24(15): 2310-2316. |
| [12] | 赵剑峰, 耿 宇, 陈前波, 杨静晖, 李 燕. 白内障伴青光眼患者采用超声乳化与小梁切除治疗后胰岛素抵抗及炎性因子的变化:自身对照研究[J]. 中国组织工程研究, 2020, 24(11): 1750-1755. |
| [13] | 师 慧,赵璐露,华宝桐,杜云蕙,郭 涛. 脊髓电刺激治疗房颤模型犬炎性因子水平的变化[J]. 中国组织工程研究, 2019, 23(7): 1023-1029. |
| [14] | 李 朋,杨淑野,张 锴,贾 龙,杜刚强,刘 栋,张新军,张德刚,王志刚. 不同磨损颗粒对体外培养人外周血单核细胞的影响[J]. 中国组织工程研究, 2019, 23(6): 894-900. |
| [15] | 简长春,陈 宇,陈 诚,胡 宁,黄 伟. 血栓弹力图评价关节置换后低分子肝素和利伐沙班的抗凝效果[J]. 中国组织工程研究, 2019, 23(4): 499-504. |
Periprosthetic joint infection (PJI), the main cause of modern total joint arthroplasty failure, affects 25%-38% of revision procedures[1-4]. In revision arthroplasty, it is important to distinguish periprosthetic infection from aseptic failure as their respective treatment protocols and impact on patient health differ greatly[5-6].
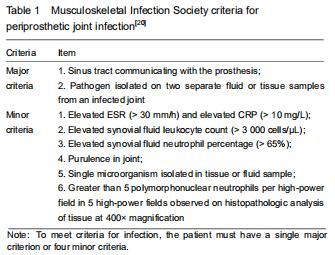
Although the diagnostic criteria of PJI are well defined by the Musculoskeletal Infection Society (MSIS)[7], the sensitivity and specificity of present serological markers for PJI require improvement. Inflammatory markers such as white blood cell (WBC) count, C-reactive protein (CRP), and erythrocyte sedimentation rate (ESR) have been generally used to screen for infection because of their simplicity and cost-effectiveness[8-9]. The recently identified synovial fluid biomarkers such as alpha-defensin demonstrate high sensitivity and specificity, but they also exhibit high false-negative and sensitivity rates that appear to be related to organism type[10-11]. Moreover, obtaining synovial fluid is invasive and painful for patients. Hence, a reliable serological test that can help to diagnose PJI is required.
 Previous studies have paid great attention to inflammatory markers and rarely focused on other serum parameters. Recent studies maintain that platelet (PLT) count and platelet indices such as mean platelet volume (MPV) and platelet distribution width (PDW) play an important role in infectious and inflammatory diseases[12-18]. Zhang et al.[19] first demonstrated that serum PLT is a promising marker for the diagnosis of deep surgical site infection after open induction internal fixation for traumatic limb fractures. However, few studies have focused on the relationship between PLT and PJI. This retroprospective study aimed to identify the sensitivity, specificity, and positive and negative predictive values of PLT for diagnosing PJI after total joint arthroplasty.
Previous studies have paid great attention to inflammatory markers and rarely focused on other serum parameters. Recent studies maintain that platelet (PLT) count and platelet indices such as mean platelet volume (MPV) and platelet distribution width (PDW) play an important role in infectious and inflammatory diseases[12-18]. Zhang et al.[19] first demonstrated that serum PLT is a promising marker for the diagnosis of deep surgical site infection after open induction internal fixation for traumatic limb fractures. However, few studies have focused on the relationship between PLT and PJI. This retroprospective study aimed to identify the sensitivity, specificity, and positive and negative predictive values of PLT for diagnosing PJI after total joint arthroplasty.
Design
A single-center, retrospective, cohort study.
Time and setting
The patients who underwent revision arthroplasty from March 2013 to December 2018 in the First Affiliated Hospital of Guangzhou University of Chinese Medicine were enrolled.
Subjects
A total of 214 patients were enrolled in this retroprospective study. A diagnosis of PJI was confirmed in 77 patients according to the MSIS criteria[20], while the remaining 137 patients were aseptic cases. Furthermore, data on WBC counts, ESR, CRP levels, and PLT of all patients were investigated. All blood samples were obtained from vein on the day of admission and stored in tubes containing ethylenediaminetetraacetic acid, and then were analyzed automatically using internationally certified devices.
The study was approved by the Institutional Review Board of the First Affiliated Hospital of Guangzhou University of Chinese Medicine (approval No. 2019205). This single-center, retrospective, cohort study enrolled patients who underwent revision arthroplasty because of delayed or chronic infection of joint arthroplasties and aseptic loosening of an implant from March 2013 to December 2018 in the First Affiliated Hospital of Guangzhou University of Chinese Medicine. PJI was diagnosed based on the MSIS criteria noted in Table 1[20]. Patients were excluded if they had hematological disease or underwent a blood transfusion within the last year, used ongoing antiplatelet drugs, had a history of recent trauma or dislocation (within 2 weeks), used lipid emulsion, associated with hepatic disease or other site infections.
Methods
Patient sex, age, and involved joint were recorded. Venous blood samples were obtained from peripheral vein on the day of admission and then analyzed for serum PLT, PDW, WBC, CRP, and ESR. Threshold values for the laboratory markers were based on the levels outlined by the International Consensus Meeting on PJI[21]. Results of intraoperative testing and findings were reviewed and included in the analysis. The following intraoperative findings were recorded: presence of purulent fluid in the joint as documented in the operative report, presence of acute inflammation on the frozen section or the pathology report, intraoperative synovial fluid, and/or tissue cultures. At our institution, at least three intraoperative tissue culture specimens were taken and cultured for 3 and 7 days.
Statistical analysis
Patients’ baseline characteristics and the results of the diagnostic tests were compared between the groups using independent-samples t-test. P values of < 0.05 were considered statistically significant. Receiver operating characteristic (ROC) curves were generated to determine the diagnostic value of each test for the assessment of PJI. The area under the curve (AUC) and 95% confidence interval (CI) were calculated. The discriminatory value of each curve was interpreted as excellent (0.9-1), good (0.8-0.89), fair (0.7-0.79), poor (0.6-0.69), or as failing or having no discriminatory capacity (0.5-0.59). Using the method described by Youden[22], the optimal cutoff point for each marker as a diagnostic marker of PJI was calculated. Additionally, the sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), and likelihood ratios (LR) were calculated. We also determined the diagnostic accuracy of the combined tests of PLT with other biomarkers. Data were analyzed using IBM SPSS Statistics 21 and MedCalc for Windows version 15.0 (MedCalc Software).
WBC, ESR, and CRP are initial tests recommended by the current guidelines of the American Association of Orthopaedic Surgeons and International Consensus Meeting on PJI because of the low false-negative and high sensitivity rates[20-21, 23]. However, other conditions such as recent surgery and inflammatory disease could also cause the same changes, making them low-specificity tests for the PJI diagnosis. Other serological markers for PJI including interleukin 6 were found to have high diagnostic value for distinguishing PJI from other causes of failure, with a specificity of 72%-85.7%[24-26]. However, it showed that interleukin 6 levels also increased in patients with aseptic loosening of total hip arthroplasty; thus, it had limited accuracy.
D-dimer, a fibrin degradation product, has been shown to be a promising predictor of PJI. Recent studies have suggested that D-dimer outperformed ESR and CRP, with a sensitivity of 89% and specificity of 93% for the diagnosis of PJI in knees and hips[27-28]. Furthermore, D-dimer was effective in the early detection of PJI if combined with ESR and CRP[29]. It was reported that D-dimer was also a promising biomarker to determine reimplantation timing[30]. However, the rapid change of D-dimer makes it invaluable for the diagnosis of PJI based on the natural curve[31].
Synovial fluid biomarkers such as alpha-defensin and leucocyte esterase are reliable markers of PJI. Deirmengian et al.[32] examined 43 biomarkers and reported 100% sensitivity and specificity for the alpha-defensin enzyme-linked immunosorbent assay for detecting PJI. The high accuracy of the alpha-defensin enzyme-linked immunosorbent assay was supported by a recent meta-analysis from Wyatt et al., who calculated a sensitivity of 100% and specificity of 96%[33]. Another advantage of this method is the fact that it delivers consistent results regardless of the organism type or virulence or prior antibiotic therapy[34]. However, the main drawbacks of this test are its high cost and the lack of widespread availability.
Leucocyte esterase reportedly has predictive value for diagnosing PJI, with a sensitivity and specificity of 79.2% and 80.8% to 100% and 93.8%, retrospectively[35-36]. However, it could not be determined when either an inadequate amount of fluid was available or no fluid was retrieved from the joint.
In the present study, ROC curves demonstrated that ESR was the best blood parameter for diagnosing PJI, similar to that reported by Li et al.[37]. ESR and CRP alone present a sensitivity of 77.92% and 79.22% and specificity of 75.91% and 75.91%, respectively, close to those of the present study[38].
Blood biomarkers are simple and practical methods for diagnosing PJI. Previous studies have focused on inflammatory markers, but they have rarely focused on platelet count and platelet indices. Platelet and platelet indices are proposed biomarkers for predicting or monitoring some infections and inflammatory diseases[12-16, 18, 39-43]. Zhang et al.[12] suggested that PDW combined with PLT can be an important additional test for the diagnosis of deep surgical site infection after open induction internal fixation for traumatic limb fractures. Rubio et al.[43] found that initial platelet and leukocyte counts might be useful for making the clinical differential diagnosis of fever in the returning traveler. Gupta et al.[41] implied that PLT and plateletcrit could be used as markers of disease severity in malaria. Demirdal et al.[13] concluded that elevated platelet-to-lymphocyte ratio can predict osteomyelitis in diabetic foot infection. Celik et al.[15] revealed that MPV decreased in ventriculoperitoneal shunt infection and suggested that MPV and PDW can be used for the diagnosis of ventriculoperitoneal shunt infection in children with an accuracy of at least 75%. Lee et al.[16] found that MPV was positively correlated with CRP and could be an inflammatory marker to determine the disease activity in tuberculosis patients. MPV could also predict urinary tract infection[18]. Kim et al.[17] reported that the incidence of abnormal PLT count increased with age in respiratory virus infection patients and those with a prolonged hospital stay. Platelet and platelet indices increased in cirrhotic patients with ascitic fluid infection[44]. In addition, MPV level and average PLT decreased after bloodstream infection[45].
In the current study, PLT and MPV differed significantly between the PJI and aseptic loosening groups. PLT had an AUC of 0.804, which indicated that it had good performance in the diagnosis of PJI. Based on the ROC analysis, using a cutoff value of 300 × 109/L, PLT had better specificity and PPV than ESR and CRP alone. Moreover, the combined analysis of PLT and ESR had the highest AUC with a sensitivity and specificity of 84.42% and 79.56%, respectively, which proved that PLT could be a potential predictor for diagnosing PJI. To our knowledge, few studies have evaluated the role of platelet and platelet indices as a diagnostic test for PJI. In this retrospective study, PLT and PDW have potential value for diagnosing PJI and had good performance when combined with ESR.
Platelets, anucleate cell fragments circulating in the blood, play a basic role in hemostasis and thrombosis. The reason for the elevated platelet level in PJI patients remains unknown. Previous studies have demonstrated that megakaryocytes, the progenitor cells of platelets, are induced by the release of inflammatory cytokines and rupture and rapidly release platelets[46]. Megakaryocytes exposed to varying bone marrow environments produce different platelets that enter the circulation primed to respond to and combat inflammation, infection, or injury. We believe that platelets play an antibacterial role in PJI development as an immune response triggered by megakaryocytes. Some inflammatory states in PJI induce populations of committed MK progenitors, giving rise to platelets in response. In addition, pathogens could change and spread platelet shape as a result of great responsiveness of platelets to bacteria[47]. Thus, PDW would decrease in PJI patients. In contrast, infection could enhance bone marrow activity and cause thrombocytosis, which may explain the increased level of PLT in PJI. However, this hypothesis requires further confirmation by studies with larger sample sizes.
It is concluded that PLT is a valuable biomarker for the diagnosis of PJI. However, the use of PLT alone has some disadvantages. PLT combined with ESR could make all the statistical indices adequate and within range. It is acknowledged that the platelet indices cannot be the sole diagnostic tool for PJI.
Several limitations should be considered. First, this was a retrospective study, so selection bias may influence its results. Second, limited numbers of PJI cases were enrolled in this study due to the low infection rate in our department. Further analyses with larger sample sizes are needed.
关节假体周围感染:是关节置换后灾难性并发症之一,由于缺乏特异性指标,细菌培养阳性率低,其诊断是临床研究的难点。#br# 血小板计数:是指计数单位容积血液中血小板的数量。目前研究发现在一些感染性疾病中,血清中血小板计数会升高,提示血小板计数可作为预测感染性疾病的诊断指标。#br# 中国组织工程研究杂志出版内容重点:人工关节;骨植入物;脊柱;骨折;内固定;数字化骨科;组织工程
血小板计数:是指计数单位容积血液中血小板的数量。目前研究发现在一些感染性疾病中,血清中血小板计数会升高,提示血小板计数可作为预测感染性疾病的诊断指标。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||