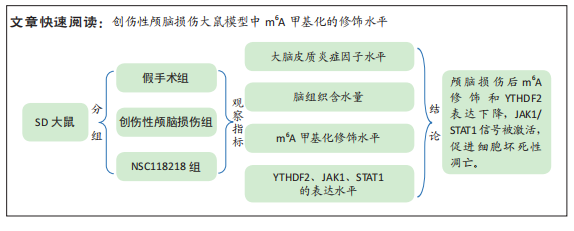
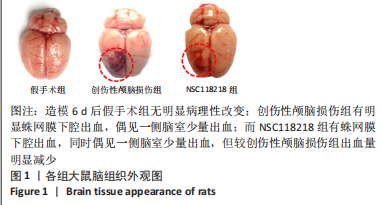
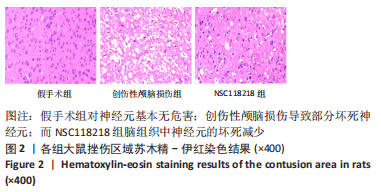
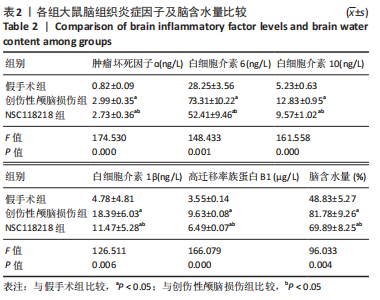
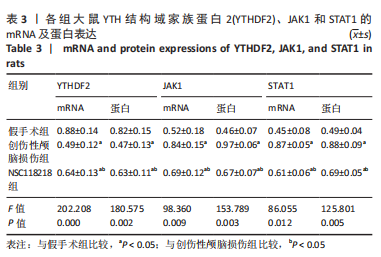
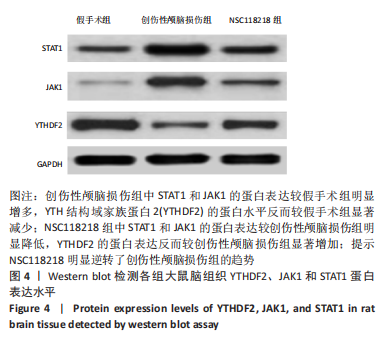
[1] CORPS KN, ROTH TL, MCGAVERN DB. Inflammation and neuroprotection in traumatic brain injury. JAMA Neurol. 2015;2(3):355-362.
[2] YU J, CHEN M, HUANG H, et al. Dynamic m6A modification regulates local translation of mRNA in axons. Nucleic Acids Rese. 2018;46(3): 1412-1423.
[3] 邓宇, 杜先超. RNA甲基化在中枢神经系统作用中的研究进展[J]. 沈阳医学院学报,2019,21(6):489-499.
[4] LI MM, ZHAO X, WANG W, et al. Ythdf2-mediated m6A mRNA clearance modulates neural development in mice. Genome Biol. 2018;19(1):69-78.
[5] 徐新, 高伟伟, 吕莉, 等. 小鼠颅脑创伤模型的构建及运动功能评估[J]. 中华实验外科杂志,2016,33(1):252-255.
[6] WANG WT, SUN L, SUN CH. PDIA3-regulted inflammation and oxidative stress contribute to the traumatic brain injury (TBI) in mice. Biochem Biophys Res Commun. 2019;518(4):657-663.
[7] BEDIRLI N, BAGRIACIK EL, YILMAZ G, et al. Sevoflurane exerts brain-protective effects against sepsis-associated encephalopathy and memory impairment through caspase3/9 and Bax/Bcl signaling pathway in a rat model of sepsis. J Int Med Res. 2018;46(7):2828-2842.
[8] LI XL, WANG B, YANG FB, et al. HOXA11-AS aggravates microglia-induced neuroinflammation after traumatic brain injury. Neural Regen Res. 2022;17(5):1096-1105.
[9]李恒希. 单纯性脑震荡大鼠海马IL-1β的表达及AH6809干预的研究[D]. 昆明:昆明医科大学,2017.
[10] JENSTER M, BONIFACIO SL, RUEL T, et al. Maternal or neonatal infection: association with neonatal encephalopathy outcomes. Pediatr Res. 2018;83(3):747-762.
[11] CHEN X, CHEN C, FAN S, et al. Omega-3 polyunsaturated fatty acid attenuates the inflammatory response by modulating microglia polarization through SIRT1-mediated deacetylation of the HMGB1/NF-κB pathway following experimental traumatic brain injury. J Neuroinflammation. 2018;15(1):116-125.
[12] HELM M, MOTORIN Y. Detecting RNA modifications in the epitranscriptome:predict and validate. Nat Rev Genet. 2017;18(5): 275-291.
[13] CUI Q, SHI H, YE P, et al. m6A RNA methylation regulates the self-renewal and tumorigenesis of glioblastoma stem cells. Cell Rep. 2017; 18(11):2622-2634.
[14] 孟祥雯, 张雪洁, 张波, 等. 多柔比星体内外对心肌甲基化转移酶样3依赖的m6A RNA甲基化修饰的影响 [J]. 中国药理学与毒理学杂志,2018,32(12):930-937.
[15] JIN D, GUO J, WU Y, et al. m6A mRNA methylation initiated by METTL3 directly promotes YAP translation and increases YAP activity by regulating the MALAT1-miR-1914-3p-YAP axis to induce NSCLC drug resistance and metastasis. J Hematol Oncol. 2019;12(1):1-22.
[16] LEE H, BAO S, QIAN Y, et al. Stage-specific requirement for Mettl3-dependent m6A mRNA methylation during haematopoietic stem cell differentiation. Nature Cell Boil. 2019;21(6):700-709.
[17] WANG S, ZHANG J, WU X, et al. Differential roles of YTHDF1 and YTHDF3 in embryonic stem cell-derived cardiomyocyte differentiation. RNA Boil. 2021;18(9):1354-1363.
[18] ZHONG L, LIAO D, ZHANG M, et al. YTHDF2 suppresses cell proliferation and growth via destabilizing the EGFR mRNA in hepatocellular carcinoma. Cancer Lett. 2019;442:252-261.
[19] WORPENBERG L, PAOLANTONI C, LONGHI S, et al. Ythdf is a N6-methyladenosine reader that modulates Fmr1 target mRNA selection and restricts axonal growth in Drosophila. EMBO J. 2021;40(4): e104975.
[20] PARIS J, MORGAN M, CAMPOS J, et al. Targeting the RNA m6A reader YTHDF2 selectively compromises cancer stem cells in acute myeloid leukemia. Cell Stem Cell. 2019;25(1):137-148. e6.
[21] 史佳宾, 王大勇, 夏晴, 等. m6A修饰对中枢神经系统功能及疾病的影响 [J]. 遗传,2020,42(12):1156-1167.
[22] WANG X, LU ZK, GOMEZ A, et al. N6-methyladenosine-dependent regulation of messenger RNA stability. Nature. 2014;505(7481):117-120.
[23] DIXIT D, PRAGER BC, GIMPLE RC, et al. The RNA m6A Reader YTHDF2 Maintains Oncogene Expression and Is a Targetable Dependency in Glioblastoma Stem Cells. Cancer Discov. 2021;11(2):480-499.
[24] LI M, ZHAO X, WANG W, et al. Ythdf2-mediated m6A mRNA clearance modulates neural development in mice. Genome Boil. 2018;19(1): 1-16.
[25] JERE SW, ABRAHAMSE H, HOURELD NN. The JAK/STAT signaling pathway and photobiomodulation in chronic wound healing. Cytokine Growth Factor Rev. 2017;38:73-79.
[26] WANG Y, KONG XQ, WU F, et al. SOCS1/JAK2/STAT3 axis regulates early brain injury induced by subarachnoid hemorrhage via inflammatory responses. Neural Regen Res. 2021;16(12):2453-2464.
[27] DELEN E, DOĞANLAR O. The dose dependent effects of ruxolitinib on the invasion and tumorigenesis in gliomas cells via inhibition of interferon gamma-depended JAK/STAT signaling pathway. J Korean Neurosurg Soc. 2020;63(4):444-458.
[28] YAO DW, O’CONNOR LJ, PRICE AL, et al. Quantifying genetic effects on disease mediated by assayed gene expression levels. Nat Genet. 2020;52(6):626-633.
[29] WANG K, WANG C, XIAO F, et al. JAK2/STAT2/STAT3 are required for myogenic differentiation. J Biol Chem. 2008;283(49):34029-34036.
[30] AARONSON DS, HORVATH CM. A road map for those who don’t know JAK-STAT. Science. 2002;296(5573):1653-1655.
[31] GONG L, MANAENKO A, FAN R, et al. Osteopontin attenuates inflammation via JAK2/STAT1 pathway in hyperglycemic rats after intracerebral hemorrhage. Neuropharmacology. 2018;138:160-169.
[32] AL-MASSRI KF, AHMED LA, EL-ABHAR HS. Pregabalin and lacosamide ameliorate paclitaxel-induced peripheral neuropathy via inhibition of JAK/STAT signaling pathway and Notch-1 receptor. Neurochem Int. 2018;120:164-171.
[33] CUI DM, ZENG T, REN J, et al. KLF 4 Knockdown Attenuates TBI‐Induced Neuronal Damage through p53 and JAK‐STAT 3 Signaling. CNS Neurosci Ther. 2017;23(2):106-118. |