中国组织工程研究 ›› 2022, Vol. 26 ›› Issue (30): 4912-4920.doi: 10.12307/2022.774
• 干细胞综述 stem cell review • 上一篇
牙周膜干细胞促进牙周组织再生的影响因素
吴梦鑫1,梁文红2,杨 琨3,韩盈盈4
- 1遵义医科大学附属口腔医院,贵州省遵义市 563000;遵义医科大学附属口腔医院,2牙体牙髓科,3牙周科,贵州省遵义市 563000;4遵义医科大学生命科学研究院口腔疾病研究特色重点实验室,贵州省遵义市 563000
-
收稿日期:2021-08-13接受日期:2021-09-18出版日期:2022-10-28发布日期:2022-03-29 -
通讯作者:韩盈盈,博士,副教授,遵义医科大学生命科学研究院口腔疾病研究特色重点实验室,贵州省遵义市 563000 -
作者简介:吴梦鑫,男,1996 年生,四川省绵阳市人,汉族,遵义医科大学在读硕士,主要从事牙周组织再生方面的研究。 -
基金资助:贵州省科技计划项目(黔科合基础[2020]1Y292),项目名称:IL-36亚家族在牙周组织炎症中的表达及作用机制,项目负责人:韩盈盈
Influencing factors of periodontal ligament stem cells promoting periodontal tissue regeneration
Wu Mengxin1, Liang Wenhong2, Yang Kun3, Han Yingying4
- 1Hospital of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China; 2Department of Dentistry and Endodontics, 3Department of Periodontology, Hospital of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China; 4Key Laboratory of Oral Diseases, Institute of Life Sciences, Zunyi Medical University, Zunyi 563000, Guizhou Province, China
-
Received:2021-08-13Accepted:2021-09-18Online:2022-10-28Published:2022-03-29 -
Contact:Han Yingying, MD, Associate professor, Key Laboratory of Oral Diseases, Institute of Life Sciences, Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Wu Mengxin, Master candidate, Hospital of Stomatology, Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:the Science and Technology Foundation of Guizhou Province, No. [2020]1Y292 (to HYY)
摘要:
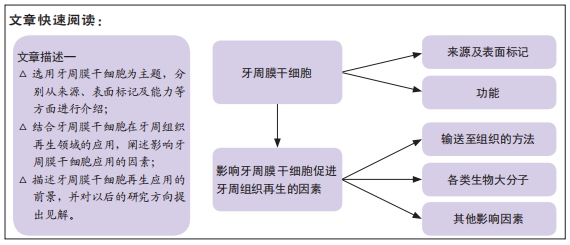
文题释义:
牙周膜干细胞:又称牙周膜间充质细胞,是由Seo等在2004年首次分离出的一种独特的细胞群,易于获得,并显示出间充质干细胞的重要特性,如自我更新、多潜能和免疫调节。不仅在在牙周复合体的再生方面显示出潜力,而且在其他牙齿和非牙齿组织的再生方面也显示出了潜力。
牙周组织再生:牙周病变过程中牙周组织因炎症而破坏,如何获得牙周组织结构和功能的重建——即牙周组织再生,是牙周病研究领域中的重要课题。
背景:牙周病是一种牙齿支持组织的慢性炎症,这些组织的破坏导致牙齿缺失。牙周组织再生是牙周治疗的最终目标。
目的:综述牙周膜干细胞的生物学特性及其在促进牙周组织再生方面的各种影响因素。
方法:应用计算机检索PubMed、CNKI、万方、ScienceDirect、Medline数据库,以“Periodontal ligament stem cells,Periodontal tissue regeneration”及“牙周膜干细胞,牙周组织再生”为关键词,检索2004-2021年发表的相关文献。根据纳入排除标准筛选文献,最终纳入72篇文献进行综述。
结果与结论:牙周炎的特征是炎症导致牙齿支撑结构的进行性损害,直到牙齿脱落。牙周再生治疗的一个宏伟目标是修复牙周组织中丢失或受损的支持组织,包括牙槽骨、牙周膜和牙骨质,并可能有效减少牙周炎引起的牙齿丢失。干细胞用于牙周再生是目前再生研究的热点,也是牙周炎的一种潜在治疗方法。牙周膜干细胞可能是最适合作为细胞来源的细胞,生物学的深入研究也强调了牙周膜干细胞是很有前途的免疫调节剂。然而,有各种各样的影响因素增强或抑制牙周膜干细胞在牙周组织再生中所发挥的作用,了解各种因素对应的影响机制将有利于在此领域拥有更深入的研究。
缩略语:牙周膜干细胞:periodontal ligament stem cells,PDLSCs;基质细胞衍生因子1:stromal cell-derived factor 1,SDF-1
https://orcid.org/0000-0002-6780-1385 (吴梦鑫)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
吴梦鑫, 梁文红, 杨 琨, 韩盈盈. 牙周膜干细胞促进牙周组织再生的影响因素[J]. 中国组织工程研究, 2022, 26(30): 4912-4920.
Wu Mengxin, Liang Wenhong, Yang Kun, Han Yingying. Influencing factors of periodontal ligament stem cells promoting periodontal tissue regeneration[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(30): 4912-4920.

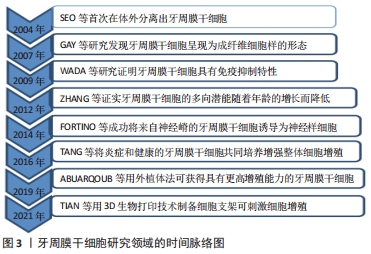
2.1 PDLSCs的来源 2004年,一项开创性的研究表明,牙周膜组织含有干细胞,通常被称为“牙周膜干细胞”或“PDLSCs”,在体外有可能分化为成牙骨质细胞样细胞、脂肪细胞和胶原形成细胞,并在体内产生新的牙骨质/牙周膜样间隔[7]。进一步的研究表明,它们呈现成纤维细胞样的形态,表达CD90、CD29、CD44、CD166、CD105和CD13抗原,这些抗原也被鉴定为骨髓基质前体[9]。随后的研究证明,PDLSCs具有免疫抑制特性,可以抑制外周血单个核细胞的增殖[10]。ZHANG 等[11]证实PDLSCs的增殖和迁移潜能以及分化能力随着年龄的增长而降低。随后,FORTINO等[12]利用表皮生长因子和碱性成纤维细胞生长因子的组合,成功将来自神经嵴的PDLSCs诱导为神经样细胞,使得PDLSCs成为神经再生医学中一个有吸引力的来源。另外,TANG等[13]将炎症PDLSCs和健康PDLSCs以不同比例共同培养,与健康PDLSCs单一培养相比,炎症PDLSCs在共培养物中的存在增强了整体细胞增殖。获得PDLSCs最常用的方法是通过酶消化法在胎牛血清中扩增。然而近期有研究表明,通过外植体法分离并在异种游离血小板裂解物中扩增的PDLSCs可作为成骨细胞再生的干细胞来源;这些细胞显示出更高的增殖能力,在传代过程中保持其干性特征,并显示成骨标记物的表达水平增加,细胞扩增后未显示任何核型异常[14]。最近,有研究者使用3D生物打印技术打印SA/Gel/na-HA生物折叠,并对其物理性能进行表征,包括表面形态、溶胀和机械性能。最后,将人PDLSCs(hPDLSCs)与SA/Gel/na-HA打印浆料混合,形成“生物墨水”,制备SA/Gel/na-HA/hPDLSCs细胞生物支架。研究结果证实,该支架可有效刺激细胞存活、增殖和成骨细胞分化[15]。牙周膜干细胞研究领域的时间脉络,见图3。
2.2 PDLSCs的表面标记 目前,尚无特异的细胞表面标志物能将PDLSCs与其他牙周构成细胞区分开来。相反,可以使用标记的组合来识别和分离它们,因为它们对应于牙周膜异质细胞群体中的一小部分细胞[7]。分离PDLSCs的最低标准包括间充质干细胞表面标志物阳性,如CD90/Thy1、CD73、CD105和CD166/VCAM-1[16]。已知的在间充质干细胞和PDLSCs上都表达的其他标志物包括CD44、CD29、Stro-3和Stro-4/HSP90等[10,17]。在最早的一项表征PDLSCs的研究中,观察到它们表达周细胞标记物Stro-1和CD146,以及典型的肌腱抗原Skeraxs[7]。进一步观察到PDLSCs还表达周细胞和干细胞共有的其他标记,包括NG2和CD140b/血小板衍生生长因子受体β[18]。PDLSCs还表达以下神经嵴来源的细胞标记:Nestin、CD271/p75NTR、Slug和Sox10等[19]。多能胚胎干细胞标志物NANOG,OCT-4,SOX2,SSEA-1,SSEA-3,SSEA-4,TRA-1-60和TRA-181也被报道在PDLSCs中表达,这可能表明这些细胞处于更未分化的状态[20]。
2.3 PDLSCs的能力 干细胞表现出不对称的细胞分裂,产生一个命运相同的子细胞(自我更新)和一个致力于分化为非干细胞的子细胞[21]。许多研究人员报道了PDLSCs以及其他类型干细胞的自我更新能力。令人惊讶的是,人PDLSCs显示出比人骨髓间充质干细胞和人牙髓干细胞更高的生长潜力[22]。此外,人骨髓间充质干细胞在大约50个群体倍增时停止增殖,而人PDLSCs的增殖能力保持在100个群体倍增以上[23]。
PDLSCs具有较高的自我更新潜能可能与机械负荷有关;牙周膜组织持续暴露于咀嚼或咬合引起的机械力中,静态机械应变可促进PDLSCs的增殖率[24]。研究发现,白细胞介素11、血管紧张素Ⅱ(Angiotensin Ⅱ,AngⅡ)和血管紧张素Ⅱ受体2型
(Angiotensin Ⅱreceptor 2,AGTR2)参与了机械负荷诱导的PDLSCs增殖,机械负荷下的人牙周膜细胞通过Ang II/AGTR2信号通路促进白细胞介素11的产生和分泌,牙周膜细胞来源的白细胞介素11对PDLSCs的增殖有刺激作用[25]。此外,年轻供者来源的PDLSCs表现出比老年供者来源的PDLSCs更高的集落形成单位和增殖活性[11]。
通过首次从牙周韧带中分离干细胞,SEO等[7]证明了一群多潜能的间充质干细胞可以进行成脂分化,并获得成骨细胞-成牙骨质细胞样表型。随后,事实证明,在适当的培养条件下,PDLSCs可以分化为成骨细胞、软骨细胞和脂肪细胞[9]。已证实PDLSCs具有成纤维细胞活性。在适当的生长因子刺激下,这些细胞表达成纤维样蛋白基因[如Ⅰ型胶原(Col1)、Ⅲ型胶原、成纤维细胞特异性蛋白1、牙周韧带相关蛋白1]和弹性蛋白,并对纤维连接蛋白有很强的免疫荧光标记。事实上,结缔组织生长因子的刺激增加了PDLSCs胶原的合成,这表明它们有可能分化为牙周韧带纤维形成细胞[26]。在神经源性诱导培养基中培养,牙周膜来源的细胞经历了与神经元相似的形态学变化。移植到成年小鼠脑内的PDLSCs能够存活、迁移并分化为具有神经样表型的细胞[27]。最近,在合适的培养基中培养的PDLSCs表现出与啮齿动物脑细胞相似的细胞和细胞核形态,它们能够形成类似轴突分支和树突的结构域,β-微管蛋白 Ⅲ和F-肌动蛋白的阳性染色进一步证实了这一点;此外,形态学分析表明,这些细胞可以通过突触样相互作用相互连接,并表达相关蛋白,如突触素和突触素1[28]。为了将来将这些细胞用于神经再生医学,有人通过用特定的生长因子(表皮生长因子和β成纤维细胞生长因子)处理PDLSCs,使其分化为神经胶质细胞和神经元样细胞[29]。PDLSCs的体外扩增还引起形态学变化,使其肌成纤维细胞表型破裂,从而上调其收缩活性,降低NANOG、SOX2和OCT-4的表达,这些因子与胚胎干细胞的多能性有关,但也在间充质干细胞中表达[30]。
多年来,免疫调节已被认为是骨髓干细胞样细胞的典型特征。虽然牙周膜细胞执行这种功能的能力仍在审查中,但最近的发现表明,来自该组织的骨髓间充质干细胞有可能具有相关的活性。在牙周炎病因中的关键病原体——牙龈卟啉单胞菌来源的脂多糖的刺激下,PDLSCs与人单核细胞来源的树突状细胞共培养,下调了树突状细胞上非经典的主要组织相容性复合体糖蛋白CD1b的表达,这支持了细胞与细胞之间的相互作用,反过来又导致T淋巴细胞增殖缺陷[31]。事实上,在PDLSCs的存在下,CD4+ T细胞的增殖指数可以显著降低,并且在丝裂原刺激的外周血单个核细胞中可以观察到CD4+CD25高/CD4+CD25低T细胞的比率降低。除了这些作用,PDLSCs在与脂多糖共刺激时,通过细胞外信号调节激酶1/2的下游激活上调环氧化酶2(COX-2)和白细胞介素6,并降低外周血单个核细胞内CD14+细胞的频率[32]。然而,牙龈卟啉单胞菌总蛋白提取物攻击CD105富集的PDLSCs的上清在体外增加了中性粒细胞的募集,这表明分泌组介导的信号通过旁分泌免疫调节是重要的[33]。类似地,在共培养实验中,PDLSCs通过分泌吲哚胺2,3-双加氧酶、肝细胞和转化生长因子(TGF-β)等代谢物,对单核细胞增殖率表现出非细胞接触性或分泌体依赖性的抑制活性[10]。
牙周膜干细胞的能力,见表1。

2.4 影响PDLSCs的因素

2.4.1 PDLSCs输送至组织的方法 干细胞的发现和治疗应用为牙周再生提供了新的概念。目前牙周病的干细胞治疗主要依赖于将培养扩增的细胞输送到牙周缺损以促进伤口愈合,许多研究记录了使用口腔内或口腔外干细胞的积极结果。体外制备的单细胞悬液或悬浮在培养基中的细胞可以很容易地直接注射到损伤部位,这种方法简单、微创。然而,注射细胞悬浮液也有相关的缺点,如注射后细胞供应不足、植入力差、注射的细胞扩散到周围的健康组织以及失去对细胞命运的控制[34]。因此,细胞支架、水凝胶以及纤维膜等生物材料技术应运而生。
(1)细胞支架:有人通过熔融电写入(MEW)技术构建高度有序的支架,并研究链间距以及纳米结构氟化磷酸钙(F/CaP)涂层对人PDLSCs黏附/增殖和成骨分化的影响。在初始细胞-支架相互作用筛选以确定最合适的设计后,选择500 μm链间距的新型聚(ε-己内酯)支架。碱处理后,将支架浸入预先确定的溶液中,以便形成涂层。纳米结构氟化磷酸钙涂层的存在导致成骨基因的显著上调和细菌生长的减弱;体内研究结果证实,氟化磷酸钙涂层支架在植入大鼠下颌牙周开窗缺损模型时具有生物相容性,并可引导牙周组织再生[35]。四面体框架核酸(tetrahedral frame nucleic acid,tFNAs)可以调节多种生物学行为,有人研究了炎症条件下四面体框架核酸对牙周组织的影响,结果表明,四面体框架核酸可降低PDLSCs中促炎细胞因子的释放和细胞活性氧水平,从而促进成骨分化;此外,动物实验表明四面体框架核酸可以改善牙周组织的炎症,保护牙周组织,尤其是通过减少炎症浸润和抑制破骨细胞的形成来减少牙槽骨的吸收[36]。
(2)水凝胶:另外,有研究合成了一种智能的牙龈菌蛋白酶敏感水凝胶(PEGPD@SDF-1),作为环境敏感的按需给药载体,该水凝胶以聚乙二醇二丙烯酸酯为骨架,二硫苏糖醇和新设计的功能肽模块通过Michael型加成反应合成,进一步负载基质细胞衍生因子1(stromal cell-derived factor 1,SDF-1);具有锚定肽-短抗菌肽-锚定肽结构的功能肽模块可被牙龈菌蛋白酶特异性切割,并且短抗菌肽被释放出水凝胶以响应牙龈菌蛋白酶的抗菌作用。通过扫描电子显微镜、傅里叶变换红外光谱、溶胀率分析、降解评估以及短抗菌肽和SDF-1的释放曲线来表征水凝胶的特性。结果表明PEGPD@SDF-1水凝胶具有较好的生物相容性,可以促进PDLSCs的增殖、迁移和成骨分化;抗菌试验表明,该抗菌剂PEGPD@SDF-1水凝胶在牙龈菌蛋白酶的刺激下释放短抗菌肽,从而强烈抑制细菌生长;此外,研究表明PEGPD@SDF-1水凝胶抑制生殖,创造低炎症环境,促进CD90+/CD34-基质细胞的募集,并诱导成骨[37]。
有研究报道了一种由聚乙二醇水凝胶微粒组成的新的MAP水凝胶平台(即TzMAP水凝胶),他们将血小板衍生生长因子BB和PDLSCs纳入TzMAP水凝胶中,以证明其提供再生疗法的潜力,特别是牙周组织再生。体外鉴定显示血小板衍生生长因子BB具有良好的保留率,PDLSCs具有良好的生长和铺展能力;此外,血小板衍生生长因子BB负载使PDLSCs在水凝胶中的增殖增加了90%,使每个细胞的平均体积增加了一倍多;总的来说,这些结果表明TzMAP水凝胶是输送干细胞和再生因子的多功能新平台[38]。
另有研究制备冻干血小板浓缩物并以 5,10 或 15 g/L 的质量浓度掺入热敏壳聚糖/β-甘油磷酸(β-GP)水凝胶中,将PDLSCs从人类第三磨牙中分离出来并封装在不同的水凝胶组中。结果表明,与未加载的对照组相比,在水凝胶中加载冻干血小板浓缩物降低了初始黏度,并且不影响任何组的溶胶-凝胶转变;所有装载冻干血小板浓缩物的水凝胶组均表现出转化生长因子β1和血小板衍生生长因子BB的持续释放2周,且不同浓度之间存在显著差异;与未加载的水凝胶相比,在水凝胶中加载 10 和 15 g/L的冻干血小板浓缩物显著增加了PDLSCs 的活力;因此,可以得出结论,在壳聚糖/β-GP水凝胶中加载冻干血小板浓缩物可以提供增强的可注射性、生长因子的持续释放和封装干细胞活力的增加[39]。6-Bromoindirubin-3’-oxime(BIO)是糖原合成酶激酶3的抑制剂,可通过增强β-连环蛋白(β-catenin)活性来激活Wnt信号通路;聚乳酸-羟基乙酸(Polylactic acid glycolic acid,PLGA)和透明质酸是很有前途的生物材料,因此,作者开发了一种嵌入 BIO 封装的聚乳酸-乙醇酸微球的新型透明质酸(PLGA-BIO-HA)水凝胶,并将该制剂注射到患有实验性牙周炎小鼠的牙龈沟中,该系统的释放速度在第1周内很快,并持续到第4周;体内实验表明,该水凝胶系统可抑制牙周炎症,促进骨再生,并能诱导骨形成标志物碱性磷酸酶(ALP)、矮小相关转录因子2(Runx2)和骨钙素在小鼠牙周炎模型中的表达[40]。
(3)纤维膜:以聚己内酯为壳,聚乙二醇为核,通过同轴静电纺丝技术再以Emdogain(EMD)包裹的复合电纺核壳纳米纤维膜也对PDLSCs成骨分化有影响。纳米纤维膜的表征显示出平均直径约为1 μm的核壳形态;检查荧光素异硫氰酸酯偶联的牛血清白蛋白在 22 d内从核壳纳米纤维中的释放表明,与固体纳米纤维相比,封装蛋白质的释放曲线有所改善;当在含有核壳纳米纤维的EMD上培养时,PDLSCs显示出显著的成骨分化改善,茜素红S染色增加,成骨基因表达增强,即骨钙素、RUNX2、碱性磷酸酶和顾桥蛋白;核壳纳米纤维膜可以通过同时的机械屏障和控释功能改善牙周再生治疗的效果[41]。另有研究也采用同轴静电纺丝技术制造了掺入氧化镁(MgO)纳米颗粒的聚己内酯/明胶核壳纳米纤维素牙周膜,称为同轴MgO,CCK-8细胞增殖和EdU染色显示,同轴MgO膜与使用单喷嘴静电纺丝技术的膜,即 Blending-MgO 和 Blending-Blank相比具有更好的PDLSCs增殖率,因为其明胶壳具有良好的生物相容性和亲水性[42]。有研究还通过静电纺丝将血管生成小分子二甲基草酰甘氨酸(dimethyl oxalyl glycine,DMOG)和骨诱导无机纳米材料纳米硅酸盐(nano silicate,nSi)结合到聚乳酸-羟基乙酸纤维中,开发牙周组织再生的双功能结构DMOG/nSi-PLGA膜,并通过检测PDLSCs的成骨和血管生成分化潜能,评估DMOG/nSi-PLGA膜对牙周组织再生的影响;此外,将纤维膜移植到大鼠牙周缺损中,通过组织学评估、显微计算机断层扫描和免疫组织化学分析评估组织再生,结果表明,DMOG/nSi-PLGA膜具有较好的力学性能和生物相容性;与单次施用相比,接种在DMOG/nSi PLGA膜上的PDLSCs表现出成骨和血管生成标记物的上调表达、更高的碱性磷酸酶活性和更多的血管形成;此外,研究表明DMOG/nSi-PLGA膜促进CD90+/CD34-基质细胞的募集,诱导血管生成和成骨,并在牙周缺损中再生牙骨质-韧带-骨复合体[43]。
(4)条件培养基:与成纤维细胞的条件培养基相比,PDLSCs的条件培养基移植显示了牙周组织再生的增强;通过Ki67免疫染色、观察核碎裂和凝聚(凋亡)以及在Matrigel上的网络形成来检测内皮细胞的功能,使用ELISA试剂盒测定血管内皮细胞生长因子水平。与成纤维细胞的条件培养基相比,PDLSCs的条件培养基中的脐静脉内皮细胞显示出更高的细胞活力以及较高数量的Ki67阳性细胞和较低的凋亡细胞;此外,脐静脉内皮细胞在PDLSCs的条件培养基中形成的毛细血管样结构多于成纤维细胞的条件培养基;PDLSCs的条件培养基比成纤维细胞的条件培养基含有更高水平的血管生成生长因子,表明PDLSCs的条件培养基具有血管生成作用,并且该作用是PDLSCs的条件培养基对牙周组织的潜在再生机制[44]。另外,有研究采用超速离心法(100倍浓度)从PDLSCs、牙龈间充质干细胞和牙龈成纤维细胞中收集无细胞条件培养基;采用外科手术方法在90只大鼠左下颌骨第一磨牙颊侧造成牙周缺损,将含有浓缩无细胞条件培养基(α-MEM、牙龈成纤维细胞条件培养基、牙龈间充质干细胞条件培养基、PDLSCs条件培养基)的胶原膜移植到牙周缺损中。结果表明,牙龈间充质干细胞条件培养基移植可显著促进大鼠牙周组织再生,其效果与PDLSCs条件培养基相同[45]。人尿源性干细胞的细胞外基质由密集的纤维束组成,纤维束中含有丰富的纤维连接蛋白。人尿源性干细胞的细胞外基质和人PDLSCs的细胞外基质均可促进人PDLSCs的增殖、附着、扩散和分化,在两者之间,人尿源性干细胞的细胞外基质在更大程度上促进了增殖、成骨和血管生成。虽然纤维连接蛋白似乎是人尿源性干细胞的细胞外基质的丰富成分,但其性能不如人尿源性干细胞的细胞外基质。人尿源性干细胞的细胞外基质作为一种合适的生物材料,在其中培养人PDLSCs,可以增强它们的生物学功能[46]。
将牙周膜干细胞输送至组织的各类方法比较,见表2。

2.4.2 各类生物大分子对牙周膜干细胞的影响
(1)RNA和脂多糖:从第一次分离出PDLSCs至今,已有许多分子被研究显示在PDLSCs中起增强或抑制作用。YU 等[47]利用生物信息学工具预测CircMAP3K11(HSA_CIRC_002284)是miR-511的上游调控基因,并发现CircMAP3K11在正常人和牙周炎患者中的表达存在显著差异。以从牙周炎患者中分离出的PDLSCs作为炎症微环境的细胞模型,研究circMAP3K11/miR-511-3p/TLR4轴在炎症条件下对PDLSCs增殖、凋亡和迁移的影响,与正常人牙周组织相比,牙周炎患者牙周组织中circMAP3K11和Toll样受体4的表达水平较高,而miR-511-3p的表达水平较低;研究表明,circMAP3K11能够促进PDLSCs的增殖和迁移,减少PDLSCs的凋亡,并且circMAP3K11可以上调与牙周再生密切相关的转录因子(Runx2、OSX、ATF4和BSP)的表达;RT-PCR和Western blot显示,circMAP3K11可以逆转miR-511-3p对Toll样受体4表达的抑制作用,这与生物信息学工具和荧光素酶报告分析的结果一致;同时,两项研究都表明circMAP3K11可以逆转miR-511-3p在牙周炎中的作用,这进一步证实circMAP3K11作为miR-511-3p的“海绵”,正向调节Toll样受体4的表达。坏死性凋亡是一种程序性坏死,由受体相互作用蛋白激酶 1(Receptor interacting protein kinase,RIP1) 和受体相互作用蛋白激酶 3(RIP3)调节,并且可以被necrostatin-1(Nec-1)特异性抑制。有研究显示RIP1和RIP3在牙龈卟啉单胞菌脂多糖刺激的PDLSCs中完全表达,并且水平显著升高。Nec-1 抑制RIP1-RIP3相互作用,并进一步抑制炎症状态下PDLSCs的坏死性凋亡。此外,Nec-1预处理改善了脂多糖处理的PDLSCs的成骨分化,可以有效促进PDLSCs在裸鼠体内的牙骨质样结构异位再生能力[48]。同时,RIP3/caspase8可以调节PDLSCs的免疫反应,而阻断RIP3/caspase8可以保护PDLSCs的生物学特性,在炎症微环境中有效促进牙周组织再生[49]。然而,大肠杆菌衍生的脂多糖通过增强 TAZ(具有PDZ基序的转录激活剂)活性促进了PDLSCs的成骨分化;TAZ的升高和激活主要由Wnt/β-catenin 通路介导,所以,PDLSC控制的牙槽骨组织再生在细菌条件下不一定会减少,且可以通过Wnt和TAZ进行调节[50]。
(2)蛋白质:FBXO5是F-box蛋白家族的成员,参与间充质干细胞的成骨分化。LIU等[51]证实人PDLSCs成骨诱导后FBXO5的表达上调;FBXO5敲低减弱了细胞迁移,抑制了碱性磷酸酶活性和矿化,并降低了RUNX2、OSX和骨钙素的表达,而FBXO5两种转录本亚型的过表达显著加速了细胞迁移,增强了碱性磷酸酶活性和矿化,并增加了人PDLSC中RUNX2、OSX和骨钙素的表达。有研究通过在裸鼠皮下移植细胞和羟基磷灰石/β-磷酸三钙复合物研究体内效应,通过iTRAQ蛋白质组学技术探索所涉及的机制,并通过Western blot和免疫荧光进行验证,发现整合素α5强制表达可以增强PDLSCs的增殖、迁移和成骨能力,而抑制整合素α5表达则具有相反的作用,黏着斑激酶、磷脂酰肌醇3激酶/蛋白激酶B的磷酸化,丝裂原激活的蛋白激酶激酶/细胞外信号调节蛋白激酶1和2(MEK1/2/ERK1/2)在此过程中至关重要;PDLSCs中整合素α5的强制表达增加了体内类骨质和牙周膜样组织的形成,蛋白质组学和生物信息学分析表明,细胞骨架和细胞周期变化参与其中,角蛋白、Ⅱ型细胞骨架6B和结蛋白可以区分这一过程,并作为PDLSCs分化的新标记[52]。DUAN等[53]首先证实富血小板纤维蛋白在体外极大地增强了细胞增殖、骨唾液蛋白、骨钙素和RUNX2的mRNA和蛋白质表达水平以及碱性磷酸酶的活性,然后使用组织工程技术在体内修复了5组牙周缺损,其中富血小板纤维蛋白联合大鼠PDLSCs的移植导致手术后12,24 d骨桥蛋白、胶原蛋白Ⅰ和 RUNX2的更高表达;显微计算机断层扫描和组织学分析显示,在手术后24 d,富血小板纤维蛋白+细胞组的新骨形成明显增多。有研究显示用D-甘露糖预处理后,人PDLSCs可以抑制T细胞增殖并影响T细胞向Treg细胞分化;发现在D-甘露糖预处理的人PDLSCs中可以检测到较少的白细胞介素6;若在D-甘露糖预处理的人PDLSCs 中检测到白细胞介素6水平升高,则诱导的Treg细胞数量会减少[54]。大麻素受体1是内源性大麻素系统的重要组成部分,参与骨代谢和炎症组织愈合。大麻素受体1过表达或大麻素受体1激动剂(10 μmol/L R-1 Meth)促进PDLSCs的骨/牙本质分化,删除大麻素受体1或应用大麻素受体1拮抗剂(10 μmol/L AM251)则抑制PDLSCs的骨/牙本质分化;大麻素受体1的激活增强了PDLSCs中肿瘤坏死因子α和干扰素γ受损的成骨/牙本质分化潜能。此外,大麻素受体1激活p38丝裂原活化蛋白激酶(mitogen-activated protein kinases,MAPK)和c-Jun氨基末端激酶(c-Jun N-terminal kinase,JNK)信号,抑制过氧化物酶体增殖物激活受体γ和Erk1/2信号。抑制JNK信号可阻断大麻素受体1激活的JNK和p38 MAPK信号,而大麻素受体1可激活p38 MAPK和JNK信号,而肿瘤坏死因子α和干扰素γ刺激可抑制这两种信号。在炎症环境中,大麻素受体1能够通过p38 MAPK和JNK信号增强PDLSCs的成骨/牙本质分化能力[55]。
(3)有机化合物:有研究显示PDLSCs在质量浓度为6.25 g/L的PDLSCs衍生条件培养基和质量浓度为94 mg/L的浓缩生长因子中有较高的增殖率,10%条件培养基+90%浓缩生长因子复合物能显著促进PDLSCs的增殖,结果表明,浓缩生长因子、条件培养基或其组合在一定浓度下对细胞有增殖作用[56]。
山奈酚是一种黄酮类化合物,广泛存在于水果、蔬菜和草药中。有研究表明,10 mol/L山奈酚显著促进PDLSCs的增殖、碱性磷酸酶活性和矿物质沉积,碱性磷酸酶、RUNX2、SP7转录因子、骨γ-羧基谷氨酸蛋白和β-catenin的表达水平均上调;在用端锚聚合酶抑制剂 (XAV939)阻断Wnt/β-catenin通路后,山奈酚的作用明显逆转,说明山奈酚通过激活Wnt/β-catenin信号促进PDLSCs的增殖和成骨[57]。有研究从健康成人供者的第三磨牙中分离PDLSCs,并在添加棕榈酸酯(0、100或250 μmol/L)的正常或成骨培养基中培养21 d,通过ELISA和末端脱氧核苷酸转移酶dUTP缺口末端标记法检测细胞凋亡,并通过测量碱性磷酸酶活性、Ⅰ型前胶原C肽和骨钙素的产生、矿化和Runx2的mRNA表达来评估成骨分化,此外,在第3天检测白细胞介素6和白细胞介素8的mRNA表达。结果表明,棕榈酸酯抑制培养的PDLSC中Runx2的增殖、碱性磷酸酶活性、Ⅰ型前胶原C肽和骨钙素的产生、矿化和mRNA表达;棕榈酸酯还诱导PDLSCs凋亡和白细胞介素6和白细胞介素8的mRNA表达[58]。另外,有研究证实作为2型糖尿病首选用药的二甲双胍可以在体外促进PDLSCs的增殖、迁移和成骨分化[59]。
(4)无机化合物:一氧化氮(NO)调节发育胚胎和成体干细胞的许多生物过程。人类 PDLSCs表达诱导型一氧化氮合酶 (iNOS) 和内皮型一氧化氮合酶 (eNOS)并产生NO。用一氧化氮合酶抑制剂L-N-单甲基精氨酸阻断NO的产生对PDLSCs增殖和凋亡没有影响,但显著减弱了成骨分化能力并刺激了PDLSCs的成脂分化能力;用NO供体硝普钠增加NO的生理水平显著促进了成骨分化能力,但降低了PDLSCs的成脂分化能力。研究发现NO是通过 JNK/ MAPK信号通路来平衡PDLSCs中的成骨细胞和脂肪细胞谱系分化的[60]。有研究利用过氧化氢诱导的氧化应激来模拟大鼠PDLSCs中活性氧的增加,数据表明,当PDLSCs用过氧化氢处理时,一种长的非编码RNA lncRNA JHDM1D反义1(JHDM1D-AS1)迅速下调,这与PDLSCs凋亡呈负相关;此外,数据显示JHDM1D-AS1通过抑制热休克蛋白40家族成员DNAJC10调节PDLSCs凋亡;此外,过表达DNAJC10抑制Bcl-2蛋白水平和eIF2α磷酸化水平,进而促进PDLSCs凋亡;结果显示JHDM1D-AS1在活性氧诱导的细胞凋亡中具有保护作用,并证实JHDM1D-AS1/DNAJC10/phosphorylated-eIF2α/Bcl-2通路作为PDLSCs中的抗凋亡信号轴发挥作用[61]。另外,为响应过氧化氢刺激,Creb的表达以剂量依赖性方式降低。过表达Creb显著降低了PDLSCs中凋亡基因的活性氧水平和蛋白质表达水平。ERK通路的磷酸化对于激活Creb诱导的保护是必不可少的,结果揭示了Creb在活性氧诱导的细胞凋亡中的保护作用,并验证了ERK/Creb/细胞凋亡调节因子Bcl-2通路在PDLSC中作为抗凋亡信号传导起作用[62]。此外,含F-box/WD重复结构域7的过表达增加了过氧化氢给药后的细胞活力,并抑制了caspase-3和-9的激活;F-box/WD重复结构域7的过度表达抑制了Notch 信号;此外,含有F-box/WD重复结构域7的保护作用可以通过Notch特异性抑制剂 PF-03084014恢复,这些观察结果表明含有F-box/WD重复结构域7对过氧化氢诱导的大鼠PDLSCs氧化应激具有保护作用[63]。
2.4.3 其他影响因素 除去以上输送方法和一些分子的影响外,PDLSCs还受到其他很多因素的影响。低强度脉冲超声是一种加速骨折愈合的非侵入性治疗方法。WANG 等之前的一项研究表明,低强度脉冲超声具有促进牙周组织再生的潜力。2018年该小组成员对低强度脉冲超声促进牙周组织再生的潜在分子机制进行研究,用低强度脉冲超声处理PDLSCs,AMD3100阻断SDF-1/C-X-C基序趋化因子受体4(CXCR4)通路,反转录-定量聚合酶链反应检测SDF-1的表达水平;小干扰RNA转染使SDF-1的上游基因twist家族的bHLH转录因子1(Twist1)失活。结果表明,低强度脉冲超声处理在mRNA和蛋白质水平上促进了TWIST1和SDF-1的表达;此外,低强度脉冲超声处理增强了PDLSCs 的细胞迁移;TWIST1的敲低损害了SDF-1的表达和PDLSCs的细胞迁移能力;TWIST1可能是PDLSCs中SDF-1的上游调节器[64]。LI 等[65]将含有或不含有PDLSCs的羟基磷灰石移植到裸鼠背部的皮下囊中,并进行组织学分析,苏木精-伊红染色显示细胞外基质合成增加,实时PCR检测到低强度脉冲超声处理后细胞外基质相关基因显著增加;此外,低强度脉冲超声可促进成骨分化相关基因和蛋白的表达;碱性磷酸酶和茜素红染色也发现低强度脉冲超声增强了PDLSCs的成骨作用;体内移植后,再生出更致密的类似牙周膜的胶原纤维。发光二极管作为一种新型光疗光源正受到关注。有研究在PDLSCs细胞被诱导分化后,以红色发光二极管(峰值波长:650 nm)照射,测试了0-10 J/cm范围内的能量密度照射对成骨分化和矿化的影响,结果表明,8 J/cm发光二极管照射导致PDLSCs增殖显著增加,并增强 Runx2和Osterix mRNA表达、碱性磷酸酶活性、Ⅰ型原胶原C肽和骨钙素产生、钙沉积和茜素红染色;此外,发光二极管照射诱导ERK1/2的激活,并且发光二极管照射对PDLSCs增殖、分化和矿化的影响可以通过用PD98059处理来抑制[66]。
有研究用微阵列分析比较了人类PDLSCs细胞系(2-14和2-23)与低分化潜能细胞系(2-52)的基因图谱,确定了印迹基因中胚层特异性转录物(MEST),胚层特异性转录物在2-23细胞的细胞质中表达;用siRNA敲除2-23细胞中的胚层特异性转录物可抑制干细胞标记物的表达,如CD105、CD146、p75NTR、N-钙粘蛋白和NANOG,并抑制增殖潜能,以及成骨细胞、脂肪细胞和软骨细胞的多向分化能力;另一方面,胚层特异性转录物在2-52细胞中的过度表达增强了干细胞标记物和牙周膜相关标记物的表达以及多向分化能力;此外,胚层特异性转录物过表达的2-52细胞表现出从纺锤形到干细胞样圆形的形态变化,与2-14和2-23细胞形态相似;这些结果表明,胚层特异性转录物在维持PDLSCs的干细胞方面起着关键作用,并将牙周膜细胞转化为PDLSCs样细胞[67]。PRDM(包含PRDI-BF1和RIZ同源域)基因在细胞增殖和分化中起关键作用。有研究显示,敲低PRDM9通过促进细胞周期进程或通过下调p21和p27以及上调细胞周期蛋白E促进细胞周期进程和从G1到S期的快速转变来增强细胞增殖;此外,PRDM9的消耗增加了迁移和趋化性PDLSCs的潜力,并揭示了重要的下游基因[68]。
YANG等[69]将PDLSC与人尿源性干细胞以不同比例(1∶0.5、1∶1和1∶2)间接共培养后,3个共培养组中的PDLSC数量从第5天开始明显增加,程度相似,与无人尿源性干细胞对照组相比,随着人尿源性干细胞比例的增加,成骨培养基中成骨和成骨基因及蛋白的表达显著增加。此外,PDLSCs/人尿源性干细胞比为1∶2的成骨基质PDLSCs片材含有最致密的胶原层,并显示出增加的成骨和成骨蛋白表达;体内移植显示,以1∶2的PDLSCs/人尿源性干细胞比率非接触共培养的PDLSCs片形成更多新的致密结构,并表达更高水平的成骨和成骨蛋白[69]。有研究探讨不同牙周细菌上清液对PDLSCs免疫应答和细胞分化潜能的影响,结果表明,牙周细菌上清液不仅能抑制PDLSC的成骨,而且能增强PDLSCs的迁移和免疫应答;此外,这项研究还表明,生物过程中的这些变化很可能依赖于牙周细菌毒力的增加[70]。
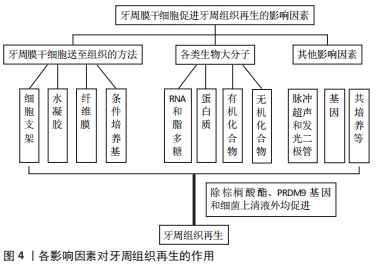
各影响因素对牙周组织再生的作用,见图4。
| [1] LAUDENBACH JM, KUMAR SS. Common Dental and Periodontal Diseases. Dermatol Clin. 2020;38(4):413-420. [2] GBD 2016 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2017;390(10100):1211-1259. [3] HERNÁNDEZ-MONJARAZ B, SANTIAGO-OSORIO E, MONROY-GARCÍA A, et al. Mesenchymal Stem Cells of Dental Origin for Inducing Tissue Regeneration in Periodontitis: A Mini-Review. Int J Mol Sci. 2018;19(4): 944. [4] AMANO A. Periodontal diseases and systemic diseases. Clin Calcium. 2017;27(10):1383-1391. [5] BARTOLD P M, GRONTHOS S, IVANOVSKI S, et al. Tissue engineered periodontal products. J Periodontal Res. 2016;51(1):1-15. [6] CHEN FM, JIN Y. Periodontal tissue engineering and regeneration: current approaches and expanding opportunities. Tissue Eng Part B Rev. 2010;16(2):219-255. [7] SEO BM, MIURA M, GRONTHOS S, et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet. 2004; 364(9429):149-155. [8] PARK CH, KIM KH, LEE YM, et al. Advanced Engineering Strategies for Periodontal Complex Regeneration. Materials(Basel). 2016;9(1):57. [9] GAY I C, CHEN S, MACDOUGALL M. Isolation and characterization of multipotent human periodontal ligament stem cells. Orthod Craniofac Res. 2007;10(3):149-160. [10] WADA N, MENICANIN D, SHI S, et al. Immunomodulatory properties of human periodontal ligament stem cells. J Cell Physiol. 2009;219(3): 667-676. [11] ZHANG J, AN Y, GAO L N, et al. The effect of aging on the pluripotential capacity and regenerative potential of human periodontal ligament stem cells. Biomaterials. 2012;33(29):6974-6986. [12] FORTINO VR, CHEN RS, PELAEZ D, et al. Neurogenesis of neural crest-derived periodontal ligament stem cells by EGF and bFGF. J Cell Physiol. 2014;229(4):479-488. [13] TANG HN, XIA Y, XU J, et al. Assessment of cellular materials generated by co-cultured ‘inflamed’ and healthy periodontal ligament stem cells from patient-matched groups. Exp Cell Res. 2016;346(1):119-129. [14] ABUARQOUB DA, ASLAM N, BARHAM RB, et al. The effect of platelet lysate in culture of PDLSCs: an in vitro comparative study. Peer J. 2019; 7:e7465. [15] TIAN Y, LIU M, LIU Y, et al. The performance of 3D bioscaffolding based on a human periodontal ligament stem cell printing technique. J Biomed Mater Res A. 2021;109(7):1209-1219. [16] SUAID FF, RIBEIRO FV, GOMES TR, et al. Autologous periodontal ligament cells in the treatment of Class III furcation defects: a study in dogs. J Clin Periodontol. 2012;39(4):377-384. [17] MENICANIN D, MROZIK KM, WADA N, et al. Periodontal-ligament-derived stem cells exhibit the capacity for long-term survival, self-renewal, and regeneration of multiple tissue types in vivo. Stem Cells Dev. 2014;23(9):1001-1011. [18] KOMAKI M. Pericytes in the Periodontal Ligament. Adv Exp Med Biol. 2019;1122:169-186. [19] HUANG CY, PELAEZ D, DOMINGUEZ-BENDALA J, et al. Plasticity of stem cells derived from adult periodontal ligament. Regen Med. 2009; 4(6):809-821. [20] KAWANABE N, MURATA S, MURAKAMI K, et al. Isolation of multipotent stem cells in human periodontal ligament using stage-specific embryonic antigen-4. Differentiation. 2010;79(2):74-83. [21] YAMASHITA YM, FULLER MT, JONES DL. Signaling in stem cell niches: lessons from the Drosophila germline. J Cell Sci. 2005;118(Pt 4): 665-672. [22] ELEUTERIO E, TRUBIANI O, SULPIZIO M, et al. Proteome of human stem cells from periodontal ligament and dental pulp. PLoS One. 2013;8(8): e71101. [23] SHI S, BARTOLD PM, MIURA M, et al. The efficacy of mesenchymal stem cells to regenerate and repair dental structures. Orthod Craniofac Res. 2005;8(3):191-199. [24] LIU J, LI Q, LIU S, et al. Periodontal Ligament Stem Cells in the Periodontitis Microenvironment Are Sensitive to Static Mechanical Strain. Stem Cells Int. 2017;2017:1380851. [25] MONNOUCHI S, MAEDA H, YUDA A, et al. Mechanical induction of interleukin-11 regulates osteoblastic/cementoblastic differentiation of human periodontal ligament stem/progenitor cells. J Periodontal Res. 2015;50(2):231-239. [26] LIU J, ZHAO Z, RUAN J, et al. Stem cells in the periodontal ligament differentiated into osteogenic, fibrogenic and cementogenic lineages for the regeneration of the periodontal complex. J Dent. 2020;92: 103259. [27] BUENO C, RAMIREZ C, RODRÍGUEZ-LOZANO FJ, et al. Human adult periodontal ligament-derived cells integrate and differentiate after implantation into the adult mammalian brain. Cell Transplant. 2013; 22(11):2017-2028. [28] BUENO C, MARTÍNEZ-MORGA M, MARTÍNEZ S. Non-proliferative neurogenesis in human periodontal ligament stem cells. Sci Rep. 2019; 9(1):18038. [29] NG TK, YANG Q, FORTINO VR, et al. MicroRNA-132 directs human periodontal ligament-derived neural crest stem cell neural differentiation. J Tissue Eng Regen Med. 2019;13(1):12-24. [30] IWASAKI K, KOMAKI M, AKAZAWA K, et al. Spontaneous differentiation of periodontal ligament stem cells into myofibroblast during ex vivo expansion. J Cell Physiol. 2019;234(11):20377-20391. [31] SHIN C, KIM M, HAN JA, et al. Human periodontal ligament stem cells suppress T-cell proliferation via down-regulation of non-classical major histocompatibility complex-like glycoprotein CD1b on dendritic cells. J Periodontal Res. 2017;52(1):135-146. [32] KUKOLJ T, TRIVANOVIĆ D, DJORDJEVIĆ IO, et al. Lipopolysaccharide can modify differentiation and immunomodulatory potential of periodontal ligament stem cells via ERK1,2 signaling. J Cell Physiol. 2018;233(1): 447-462. [33] MISAWA MYO, SILVÉRIO RUIZ KG, NOCITI FH JR, et al. Periodontal ligament-derived mesenchymal stem cells modulate neutrophil responses via paracrine mechanisms. J Periodontol. 2019;90(7): 747-755. [34] DU J, SHAN Z, MA P, et al. Allogeneic bone marrow mesenchymal stem cell transplantation for periodontal regeneration. J Dent Res. 2014; 93(2):183-188. [35] DAGHRERY A, FERREIRA JA, DE SOUZA ARAÚJO IJ, et al. A Highly Ordered, Nanostructured Fluorinated CaP-Coated Melt Electrowritten Scaffold for Periodontal Tissue Regeneration. Adv Healthc Mater. 2021; e2101152. [36] ZHOU M, GAO S, ZHANG X, et al. The protective effect of tetrahedral framework nucleic acids on periodontium under inflammatory conditions. Bioact Mater. 2021;6(6):1676-1688. [37] LIU S, WANG YN, MA B, et al. Gingipain-Responsive Thermosensitive Hydrogel Loaded with SDF-1 Facilitates In Situ Periodontal Tissue Regeneration. ACS Appl Mater Interfaces. 2021;13(31):36880-36893. [38] ISAAC A, JIVAN F, XIN S, et al. Microporous Bio-orthogonally Annealed Particle Hydrogels for Tissue Engineering and Regenerative Medicine. ACS Biomater Sci Eng. 2019;5(12):6395-6404. [39] AMMAR MM, WALY GH, SANIOUR SH, et al. Growth factor release and enhanced encapsulated periodontal stem cells viability by freeze-dried platelet concentrate loaded thermo-sensitive hydrogel for periodontal regeneration. Saudi Dent J. 2018;30(4):355-364. [40] SHEN S, ZHANG Y, ZHANG S, et al. 6-Bromoindirubin-3’-oxime Promotes Osteogenic Differentiation of Periodontal Ligament Stem Cells and Facilitates Bone Regeneration in a Mouse Periodontitis Model. ACS Biomater Sci Eng. 2021;7(1):232-241. [41] LAM L RW, SCHILLING K, ROMAS S, et al. Electrospun core-shell nanofibers with encapsulated enamel matrix derivative for guided periodontal tissue regeneration. Dent Mater J. 2021. doi: 10.4012/dmj.2020-412. [42] PENG W, REN S, ZHANG Y, et al. MgO Nanoparticles-Incorporated PCL/Gelatin-Derived Coaxial Electrospinning Nanocellulose Membranes for Periodontal Tissue Regeneration. Front Bioeng Biotechnol. 2021;9: 668428. [43] SHANG L, LIU Z, MA B, et al. Dimethyloxallyl glycine/nanosilicates-loaded osteogenic/angiogenic difunctional fibrous structure for functional periodontal tissue regeneration. Bioact Mater. 2021;6(4): 1175-1188. [44] IWASAKI K, AKAZAWA K, NAGATA M, et al. Angiogenic Effects of Secreted Factors from Periodontal Ligament Stem Cells. Dent J (Basel). 2021;9(1):9. [45] QIU J, WANG X, ZHOU H, et al. Enhancement of periodontal tissue regeneration by conditioned media from gingiva-derived or periodontal ligament-derived mesenchymal stem cells: a comparative study in rats. Stem Cell Res Ther. 2020;11(1):42. [46] XIONG X, YANG X, DAI H, et al. Extracellular matrix derived from human urine-derived stem cells enhances the expansion, adhesion, spreading, and differentiation of human periodontal ligament stem cells. Stem Cell Res Ther. 2019;10(1):396. [47] YU B, HU J, LI Q, et al. CircMAP3K11 Contributes to Proliferation, Apoptosis and Migration of Human Periodontal Ligament Stem Cells in Inflammatory Microenvironment by Regulating TLR4 via miR-511 Sponging. Front Pharmacol. 2021;12:633353. [48] YAN B, ZHANG H, DAI T, et al. Necrostatin-1 promotes ectopic periodontal tissue like structure regeneration in LPS-treated PDLSCs. PLoS One. 2018;13(11):e0207760. [49] YAN B, WEI K, HOU L, et al. Receptor-Interacting Protein 3/Caspase-8 May Regulate Inflammatory Response and Promote Tissue Regeneration in the Periodontal Microenvironment. Med Sci Monit. 2018;24:5247-5257. [50] XING Y, ZHANG Y, JIA L, et al. Lipopolysaccharide from Escherichia coli stimulates osteogenic differentiation of human periodontal ligament stem cells through Wnt/β-catenin-induced TAZ elevation. Mol Oral Microbiol. 2019;34(1): doi: 10.1111/omi.12249. [51] LIU L, LIU K, YAN Y, et al. Two Transcripts of FBXO5 Promote Migration and Osteogenic Differentiation of Human Periodontal Ligament Mesenchymal Stem Cells. Biomed Res Int. 2018;2018:7849294. [52] WANG H, LI J, ZHANG X, et al. Priming integrin alpha 5 promotes the osteogenic differentiation of human periodontal ligament stem cells due to cytoskeleton and cell cycle changes. J Proteomics. 2018; 179:122-130. [53] DUAN X, LIN Z, LIN X, et al. Study of platelet-rich fibrin combined with rat periodontal ligament stem cells in periodontal tissue regeneration. J Cell Mol Med. 2018;22(2):1047-1055. [54] GUO L, HOU Y, SONG L, et al. D-Mannose Enhanced Immunomodulation of Periodontal Ligament Stem Cells via Inhibiting IL-6 Secretion. Stem Cells Int. 2018;2018:7168231. [55] YAN W, CAO Y, YANG H, et al. CB1 enhanced the osteo/dentinogenic differentiation ability of periodontal ligament stem cells via p38 MAPK and JNK in an inflammatory environment. Cell Prolif. 2019;52(6): e12691. [56] AGHAMOHAMADI Z, KADKHODAZADEH M, TORSHABI M, et al. A compound of concentrated growth factor and periodontal ligament stem cell-derived conditioned medium. Tissue Cell. 2020;65:101373. [57] NIE F, ZHANG W, CUI Q, et al. Kaempferol promotes proliferation and osteogenic differentiation of periodontal ligament stem cells via Wnt/β-catenin signaling pathway. Life Sci. 2020;258:118143. [58] TAKEUCHI T, MASUNO K, UMEDA M, et al. Palmitate induces apoptosis and inhibits osteogenic differentiation of human periodontal ligament stem cells. Arch Oral Biol. 2020;112:104681. [59] ZHANG R, LIANG Q, KANG W, et al. Metformin facilitates the proliferation, migration, and osteogenic differentiation of periodontal ligament stem cells in vitro. Cell Biol Int. 2019; doi: 10.1002/cbin. 11202. [60] YANG S, GUO L, SU Y, et al. Nitric oxide balances osteoblast and adipocyte lineage differentiation via the JNK/MAPK signaling pathway in periodontal ligament stem cells. Stem Cell Res Ther. 2018;9(1):118. [61] SHI B, SHAO B, YANG C, et al. Upregulation of JHDM1D-AS1 protects PDLSCs from H(2)O(2)-induced apoptosis by decreasing DNAJC10 via phosphorylation of eIF2α. Biochimie. 2019;165:48-56. [62] FU X, FENG Y, SHAO B, et al. Activation of the ERK/Creb/Bcl‑2 pathway protects periodontal ligament stem cells against hydrogen peroxide‑induced oxidative stress. Mol Med Rep. 2019;19(5): 3649-3657. [63] FENG Y, FU X, LOU X. Notch pathway deactivation mediated by F-box/WD repeat domain-containing 7 ameliorates hydrogen peroxide-induced apoptosis in rat periodontal ligament stem cells. Arch Oral Biol. 2019;100:93-99. [64] WANG Y, LI J, QIU Y, et al. Low‑intensity pulsed ultrasound promotes periodontal ligament stem cell migration through TWIST1‑mediated SDF‑1 expression. Int J Mol Med. 2018;42(1):322-330. [65] LI H, ZHOU J, ZHU M, et al. Low-intensity pulsed ultrasound promotes the formation of periodontal ligament stem cell sheets and ectopic periodontal tissue regeneration. J Biomed Mater Res A. 2021;109(7): 1101-1112. [66] YAMAUCHI N, TAGUCHI Y, KATO H, et al. High-power, red-light-emitting diode irradiation enhances proliferation, osteogenic differentiation, and mineralization of human periodontal ligament stem cells via ERK signaling pathway. J Periodontol. 2018;89(3):351-360. [67] HASEGAWA D, HASEGAWA K, KANEKO H, et al. MEST Regulates the Stemness of Human Periodontal Ligament Stem Cells. Stem Cells Int. 2020;2020:9672673. [68] ZHANG J, ZHANG C, YANG H, et al. Depletion of PRDM9 enhances proliferation, migration and chemotaxis potentials in human periodontal ligament stem cells. Connect Tissue Res. 2020;61(5):498-508. [69] YANG X, XIONG X, ZHOU W, et al. Effects of human urine-derived stem cells on the cementogenic differentiation of indirectly-cocultured periodontal ligament stem cells. Am J Transl Res. 2020;12(2):361-378. [70] RAMENZONI LL, RUSSO G, MOCCIA MD, et al. Periodontal bacterial supernatants modify differentiation, migration and inflammatory cytokine expression in human periodontal ligament stem cells. PLoS One. 2019;14(7):e0219181. [71] CHEN FM, GAO LN, TIAN BM, et al. Treatment of periodontal intrabony defects using autologous periodontal ligament stem cells: a randomized clinical trial. Stem Cell Res Ther. 2016;7:33. [72] SáNCHEZ N, FIERRAVANTI L, NúñEZ J, et al. Periodontal regeneration using a xenogeneic bone substitute seeded with autologous periodontal ligament-derived mesenchymal stem cells: A 12-month quasi-randomized controlled pilot clinical trial. J Clin Periodontol. 2020; 47(11):1391-1402. |
| [1] | 范亚茹, 李瑞欣, 李凤集, 罗睿, 刘浩, 严颖彬. 载吲哚菁绿聚乳酸-羟基乙酸共聚物微球的表征及其光热效应 [J]. 中国组织工程研究, 2022, 26(在线): 1-6. |
| [2] | 姚晓玲, 彭建城, 许岳荣, 杨志东, 张顺聪. 可变角度零切迹前路椎间融合内固定系统治疗脊髓型颈椎病:30个月随访[J]. 中国组织工程研究, 2022, 26(9): 1377-1382. |
| [3] | 朱 婵, 韩栩珂, 姚承佼, 周 倩, 张 强, 陈 秋. 人体唾液成分与骨质疏松/骨量低下[J]. 中国组织工程研究, 2022, 26(9): 1439-1444. |
| [4] | 金 涛, 刘 林, 朱晓燕, 史宇悰, 牛建雄, 张同同, 吴树金, 杨青山. 骨关节炎与线粒体异常[J]. 中国组织工程研究, 2022, 26(9): 1452-1458. |
| [5] | 张立创, 徐 浩, 马迎辉, 熊梦婷, 韩海慧, 鲍嘉敏, 翟伟韬, 梁倩倩. 免疫调控淋巴回流功能治疗类风湿关节炎的机制及前景[J]. 中国组织工程研究, 2022, 26(9): 1459-1466. |
| [6] | 王 景, 熊 山, 曹 金, 冯林伟, 王 信. 白细胞介素3在骨代谢中的作用及机制[J]. 中国组织工程研究, 2022, 26(8): 1260-1265. |
| [7] | 朱 婵, 韩栩珂, 姚承佼, 张 强, 刘 静, 邵 明. 针刺治疗帕金森病:动物实验显示的作用机制[J]. 中国组织工程研究, 2022, 26(8): 1272-1277. |
| [8] | 安维政, 何 萧, 任 帅, 刘建宇. 肌源干细胞在周围神经再生中的潜力[J]. 中国组织工程研究, 2022, 26(7): 1130-1136. |
| [9] | 范一鸣, 刘方煜, 张洪宇, 李 帅, 王岩松. 脊髓损伤后室管膜区内源性神经干细胞反应的系列问题[J]. 中国组织工程研究, 2022, 26(7): 1137-1142. |
| [10] | 黄晨玮, 费彦亢, 朱梦梅, 李鹏昊, 于 兵. 谷胱甘肽在干细胞“干性”及调控中的重要作用[J]. 中国组织工程研究, 2022, 26(7): 1119-1124. |
| [11] | 惠小珊, 白 京, 周思远, 王 阶, 张金生, 何庆勇, 孟培培. 中医药调控干细胞诱导分化的理论机制[J]. 中国组织工程研究, 2022, 26(7): 1125-1129. |
| [12] | 田 川, 朱向情, 杨再玲, 鄢东海, 李 晔, 王严影, 杨育坤, 何 洁, 吕冠柯, 蔡学敏, 舒丽萍, 何志旭, 潘兴华. 骨髓间充质干细胞调控猕猴卵巢的衰老[J]. 中国组织工程研究, 2022, 26(7): 985-991. |
| [13] | 郭 嘉, 丁琼桦, 刘 泽, 吕思懿, 周泉程, 高玉花, 白春雨. 间充质干细胞来源外泌体的生物学特性及免疫调控作用[J]. 中国组织工程研究, 2022, 26(7): 1093-1101. |
| [14] | 吴玮玥, 郭晓东, 包崇云. 工程化外泌体在骨修复再生中的应用[J]. 中国组织工程研究, 2022, 26(7): 1102-1106. |
| [15] | 周洪琴, 吴丹丹, 杨 琨, 刘 琪. 传递特定miRNA的外泌体可调控成骨并促进成血管[J]. 中国组织工程研究, 2022, 26(7): 1107-1112. |
牙周炎是最常见的口腔疾病之一,如果不治疗可能会对个体产生不可逆转的后遗症和整体心理和生理影响,从而降低生活质量。晚期牙周病导致牙齿脱落的负担很重[1]。根据全球疾病负担研究(2016),严重的牙周病是世界上第11位最流行的疾病[2]。牙周炎会持续破坏牙周组织,如果不治疗会导致牙齿脱落[3]。牙周炎还与各种系统性疾病的发生和预后密切相关,包括心血管疾病、癌症、肥胖、糖尿病和慢性肾炎等[4]。因此,探索有效、安全、可转化为临床的牙周治疗方法是世界各国迫切的健康需求。牙周治疗的宏伟目标是再生多种牙周组织,包括受损牙周组织中的牙槽骨、牙骨质和牙周膜(PDL)[5]。虽然非手术牙周治疗可以通过物理方法去除病原体和坏死组织来防止疾病进展,但在治疗部位只有少量牙周组织可以再生[3]。引导组织再生(GTR)等技术在牙周手术中的应用可以不规则地修复牙槽骨和软组织,但总体结果并不一定令人满意,而且缺乏临床预见性[6]。尽管新的生物材料和生长因子丰富了牙周缺损的治疗方法,但临床试验表明它们的疗效仍然存在争议,丢失牙周组织的结构和功能再生仍然具有挑战性[5]。
干细胞可以自我更新和分化为多种细胞类型,因此具有巨大的治疗潜力。2004年,从人牙周组织中鉴定出干细胞,称为牙周膜干细胞(periodontal ligament stem cells,PDLSCs),开创了牙周再生研究的新纪元[7]。从那时起,人们发现其他干细胞在适当的诱导条件下具有形成多个牙周组织的能力。除了干细胞的再生能力外,干细胞接受免疫调节的能力在获得成功的结果中也起着同样重要的作用。目前,干细胞的使用被认为是牙周治疗的主流策略,特别是对于牙周复合体的完全再生,这不仅意味着重建合适的牙槽骨,而且还意味着通过定向插入新形成的牙周膜组织来诱导牙根表面的牙骨质形成[8]。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1.1 检索人及检索时间 第一作者在2021年8月进行检索
1.1.2 检索文献时限 为2004-01-01/2021-08-27。
1.1.3 检索数据库 ①PubMed数据库(pubmed.ncbi.nlm.nih.gov),是一个提供生物医学方面的论文搜寻以及摘要,并且免费搜寻的数据库。②CNKI数据库(www.cnki.net),中国知识基础设施工程,以全面打通知识生产、传播、扩散与利用各环节信息通道,打造支持全国各行业知识创新、学习和应用的交流合作平台为总目标。③万方数据库(www.wanfangdata.com.cn),涵盖期刊、会议纪要、论文、学术成果、学术会议论文的大型网络数据库;也是和中国知网齐名的中国专业的学术数据库。④ScienceDirect数据库(www.ScienceDirect.com),是荷兰一家全球著名的学术期刊出版商,每年出版大量的学术图书和期刊,大部分期刊被SCI、SSCI、EI收录,是世界上公认的高品位学术期刊。⑤Medline数据库(ovidsp.dc2.ovid.com),是美国国立医学图书馆(The National Library of Medicine,简称NLM)生产的国际性综合生物医学信息书目数据库,是当前国际上最权威的生物医学文献数据库。
1.1.4 检索词 英文检索词:“Periodontal ligament stem cells”“Periodontal tissue regeneration”“(Periodontal ligament stem cells) OR (Periodontal tissue regeneration)”,“(Periodontal ligament stem cells) AND (Periodontal tissue regeneration)”。中文检索词:“牙周膜干细胞”“牙周组织再生”“牙周膜干细胞或牙周组织再生”“牙周膜干细胞和牙周组织再生”。
1.1.5 检索文献类型 文献类型包括:研究原著,综述,荟萃分析。
1.1.6 手工检索情况 无。
1.1.7 检索策略 以PubMed数据库为例,文献检索策略见图1。

1.1.8 检索文献量 共检索到文献808篇,其中英文文献425篇,中文文献383篇。
1.2 文献筛选标准
1.2.1 纳入标准 有关牙周膜干细胞介绍的文献;与牙周组织再生有关的文献;有关牙周膜干细胞在牙周组织再生方面的文献
1.2.2 排除标准 ①与该文主题不相干的文章;②重复性研究;③较为陈旧的文章。
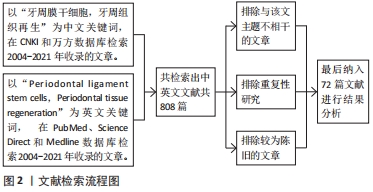
1.3 质量评估及数据的提取 共检索到 808篇文献,通过阅读文章标题及摘要进行初步筛选,资料收集者共同评估相关文献的重复性、非相关性、陈旧性;通读全文内容后,将72篇文献纳入并进行综述。文献检索流程,见图2。
牙周组织再生疗法是通过应用一些生物材料、生长因子和细胞而发展起来的。目前,关于这一疗法的研究主要集中在探讨牙周膜干细胞的再生能力上,研究已有十余年,但至今仍局限在动物模型和细胞研究上,缺乏相关的临床研究。作者通过查阅文献,仅发现2篇文献对PDLSCs应用于临床的安全性和可行性进行了临床研究,并证实其是安全的且不会产生不良反应,但与对照组(不含PDLSCs的治疗组)相比,实验组(含PDLSCs的治疗组)未检测到具有统计学意义的差异[71-72]。而且以此篇综述所引用的文章看,研究主要偏向于PDLSCs促进牙周组织再生的影响因素,甚少研究PDLSCs抑制牙周组织再生的影响因素,前人的综述主要集中在PDLSCs的再生潜力、动物模型研究等,而此次综述从一个不同的角度——影响因素入手,探讨促进或抑制PDLSCs影响牙周组织再生的影响因素,总结了各影响因素对PDLSCs在牙周组织再生方面的促进或抑制作用,为更多的临床研究铺路。然而各种各样的影响因素太多,故此次综述以近两年最新的研究文章为主,揭示了该领域的研究趋势,为后来的研究人员提供研究思路。目前来看,这一领域的基础研究还有很长的路要走。为了确保临床应用的安全性、重复性和成本效益,制定更好的培养、储存、扩增和分化PDLSCs的方案是一项挑战。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程

文题释义:
牙周膜干细胞:又称牙周膜间充质细胞,是由Seo等在2004年首次分离出的一种独特的细胞群,易于获得,并显示出间充质干细胞的重要特性,如自我更新、多潜能和免疫调节。不仅在在牙周复合体的再生方面显示出潜力,而且在其他牙齿和非牙齿组织的再生方面也显示出了潜力。
牙周组织再生:牙周病变过程中牙周组织因炎症而破坏,如何获得牙周组织结构和功能的重建——即牙周组织再生,是牙周病研究领域中的重要课题。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
ScienceDirect数据库(www.ScienceDirect.com),是荷兰一家全球著名的学术期刊出版商,每年出版大量的学术图书和期刊,大部分期刊被SCI、SSCI、EI收录,是世界上公认的高品位学术期刊。 #br# Medline数据库(ovidsp.dc2.ovid.com),是美国国立医学图书馆(The National Library of Medicine,简称NLM)生产的国际性综合生物医学信息书目数据库,是当前国际上最权威的生物医学文献数据库。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||