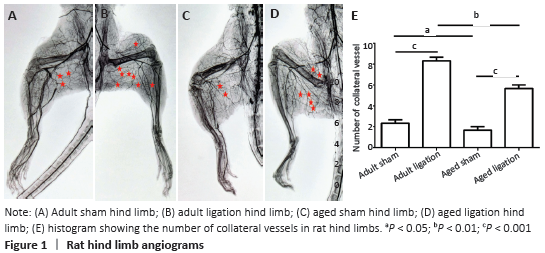
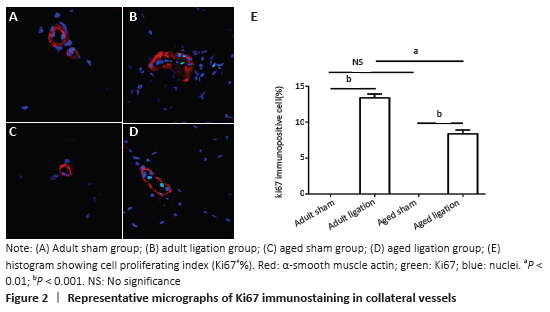
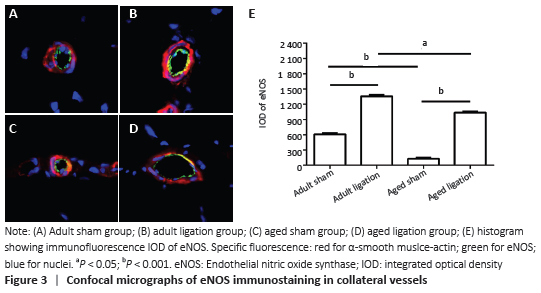
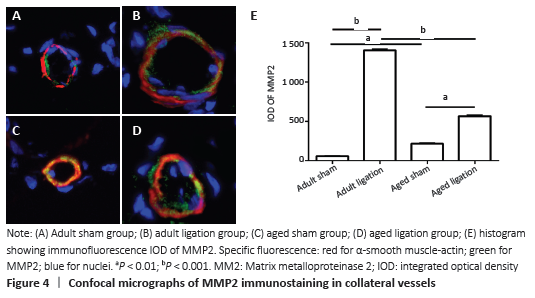
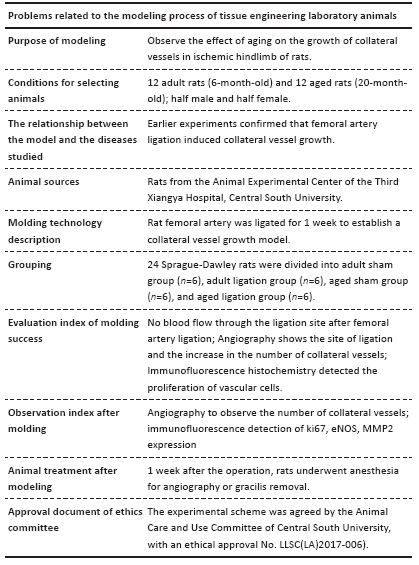
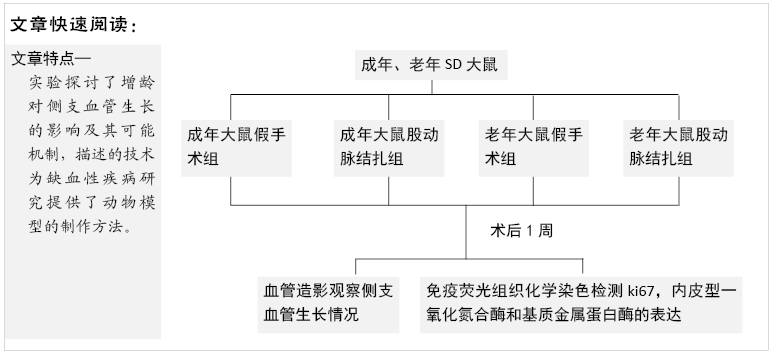
The results of this study demonstrate that: (1) in aged rats, collateral vessel growth in hindlimb ischemic model was model was impaired, showing the number of collateral vessel less than that in adult rats; (2) compared with the adult ligation group, the aged ligation group showed that the number of collateral vessels was reduced, Ki-67 positive cells in vascular walls were reduced, and the expression levels of eNOS and MMP2 were also decreased.
The femoral artery ligation model in experimental animals is a common model to observe the growth of collateral vessels induced by acute hindlimb ischemia. Scientists used this model to find that hind limb blood flow in aged animals is less than that in adult animals after acute ischemia of hindlimbs, and the recovery of hind limb function is weakened, which is believed to be due to the influence of aging on angiogenesis[24]. Similarly, another acute hindlimb ischemia model induced by ligation of iliac arteries and veins found that the ability of aged rats could still maintain potent angiogenesis to some extent, but the recovery of hindlimb blood flow and function in aged rats is far less than that of adult rats, and aged rats mainly induce angiogenesis, while collateral vessel growth is impaired, indicating that aging leads to defects in arteriogenesis[25]. In this study, we found that the number of collateral vessels was smaller in aged rats than in adult rats at 7 days after femoral artery ligation. This further proves that aging leads to decreased collateral vessel growth capacity.
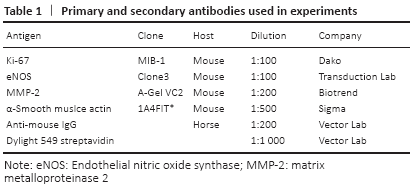
Nitric oxide and NOS can regulate the function of vascular endothelial cells. Li et al. [26] found that fluid shear stress increased nitric oxide production by increasing the activity of eNOS. Dai et al. [27] found that compared with wild-type mice, eNOS knockout mice showed blood flow and more severe shrinkage injury after 3 weeks of femoral artery ligation, which was caused by reduced collateral vessel remodeling. This indicates that the growth of collateral vessels due to shear stress depends on the expression of eNOS. It was found in the experiment that the expression of eNOS in the collateral vessels of aged rats was significantly lower than that of adult rats in the sham groups and the ligation groups. Therefore, the low expression of eNOS in aged rats may affect the growth of collateral vessels.
MMP2 is a group of zinc-containing enzymes that are involved in the degradation of the extracellular matrix[28]. In normal arterioles, smooth muscle cells are surrounded by a basement membrane, which restricts the migration and proliferation of smooth muscle cells; the inner elastic membrane between the intimal and medial membranes also limits smooth muscle cell migration. MMP2 can degrade the basement membrane and elastic membrane.
CHENG et al. [29] applied hind limb ischemia models with MMP2 +/+ and MMP2-/-mice and found that there were less developed collateral vessels in MMP2-/-mice. This shows that the loss of MMP2 impairs the growth of collateral vessels. This is consistent with the findings of Haas et al.[30] that MMP2 promotes the growth of collateral vessels. In the present study, we found that after ligation of the femoral artery, the expression of MMP2 in the collateral vessels of adult rats was significantly enhanced, causing the degradation of elastic components and extracellular matrix, which was beneficial to the outward growth of collateral vessels. However, the expression of MMP2 was also increased in aged rats after the same operation, but not as obvious as in adult rats.
In this present study, we found that the expression of eNOS and MMP2 in the collateral vessel of the aged ligation group was lower than that in the adult ligation group. Therefore, we speculate that the low expression of eNOS and MMP2 during the growth of collateral vessels may be an important mechanism for the damage of the growth of the collateral vessels in the aged rats.
In conclusion, our data demonstrate that aging impairs the growth of collateral vessels in the rat model of hind limb ischemia. The low expression of eNOS and MMP2 in the collateral vessels may be an important mechanism of impaired collateral vessel growth. However, we should continue to study the mechanism by which aging leads to impaired growth of collateral vessels, thereby providing a theoretical basis for the treatment of senile ischemic diseases.
中国组织工程研究杂志出版内容重点:组织构建;骨细胞;软骨细胞;细胞培养;成纤维细胞;血管内皮细胞;骨质疏松;组织工程
 文题释义:
文题释义: