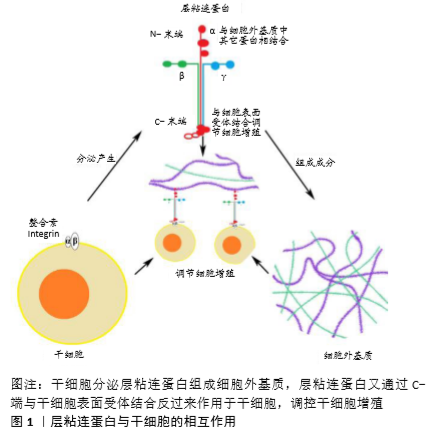
[1] MADEIRA C, SANTHAGUNAM A, SALGUEIRO JB, et al. Advanced cell therapies for articular cartilage regeneration. Trends Biotechnol. 2015;33(1):35-42.
[2] FELSON DT. Clinical practice. Osteoarthritis of the knee. N Engl J Med. 2006; 354(8):841-848.
[3] MAMIDI MK, DAS AK, ZAKARIA Z, et al. Mesenchymal stromal cells for cartilage repair in osteoarthritis. Osteoarthritis Cartilage. 2016;24(8): 1307-1316.
[4] FLANAGAN LA, REBAZA LM, DERZIC S, et al. Regulation of human neural precursor cells by laminin and integrins. J Neurosci Res. 2006; 83(5):845-856.
[5] SUH HN, KIM MO, HAN HJ. Laminin-111 stimulates proliferation of mouse embryonic stem cells through a reduction of gap junctional intercellular communication via RhoA-mediated Cx43 phosphorylation and dissociation of Cx43/ZO-1/drebrin complex. Stem Cells Dev. 2012; 21(11):2058-2070.
[6] LYNCH K, PEI M. Age associated communication between cells and matrix: a potential impact on stem cell-based tissue regeneration strategies. Organogenesis. 2014;10(3):289-298.
[7] ABDAL DAYEM A, LEE S, CHOI HY, et al. The Impact of Adhesion Molecules on the In Vitro Culture and Differentiation of Stem Cells. Biotechnol J. 2018;13(2).
[8] AUMAILLEY M. The laminin family. Cell Adh Migr. 2013;7(1):48-55.
[9] AUMAILLEY M, BRUCKNER-TUDERMAN L, CARTER WG, et al. A simplified laminin nomenclature. Matrix Biol. 2005;24(5):326-332.
[10] LEITON CV, ARANMOLATE A, EYERMANN C, et al. Laminin promotes metalloproteinase-mediated dystroglycan processing to regulate oligodendrocyte progenitor cell proliferation. J Neurochem. 2015;135(3):522-538.
[11] SEEGER T, HART M, PATARROYO M, et al. Mesenchymal Stromal Cells for Sphincter Regeneration: Role of Laminin Isoforms upon Myogenic Differentiation. PLoS One. 2015;10(9):e0137419.
[12] YAP L, TAY HG, NGUYEN MTX, et al. Laminins in Cellular Differentiation. Trends Cell Biol. 2019;29(12):987-1000.
[13] GANDHI JK, KNUDSEN T, HILL M, et al. Human Fibrinogen for Maintenance and Differentiation of Induced Pluripotent Stem Cells in Two Dimensions and Three Dimensions. Stem Cells Transl Med. 2019;8(6):512-521.
[14] KVIST AJ, NYSTRÖM A, HULTENBY K, et al. The major basement membrane components localize to the chondrocyte pericellular matrix--a cartilage basement membrane equivalent? Matrix Biol. 2008;27(1):22-33.
[15] BARRY F, MURPHY M. Mesenchymal stem cells in joint disease and repair. Nat Rev Rheumatol. 2013;9(10):584-594.
[16] FOLDAGER CB, TOH WS, CHRISTENSEN BB, et al. Collagen Type IV and Laminin Expressions during Cartilage Repair and in Late Clinically Failed Repair Tissues from Human Subjects. Cartilage. 2016;7(1):52-61.
[17] CHU WC, ZHANG S, SNG TJ, et al. Distribution of pericellular matrix molecules in the temporomandibular joint and their chondroprotective effects against inflammation. Int J Oral Sci. 2017;9(1):43-52.
[18] MOAZEDI-FUERST FC, GRUBER G, STRADNER MH, et al. Effect of Laminin-A4 inhibition on cluster formation of human osteoarthritic chondrocytes. J Orthop Res. 2016;34(3):419-426.
[19] SANTIAGO LY, NOWAK RW, PETER RUBIN J, et al. Peptide-surface modification of poly(caprolactone) with laminin-derived sequences for adipose-derived stem cell applications. Biomaterials. 2006;27(15): 2962-2969.
[20] KÖLLMER M, KESKAR V, HAUK TG, et al. Stem cell-derived extracellular matrix enables survival and multilineage differentiation within superporous hydrogels. Biomacromolecules. 2012;13(4): 963-973.
[21] JENG L, OLSEN BR, SPECTOR M. Engineering endostatin-expressing cartilaginous constructs using injectable biopolymer hydrogels. Acta Biomater. 2012;8(6): 2203-2212.
[22] TOH WS, FOLDAGER CB, PEI M, et al. Advances in mesenchymal stem cell-based strategies for cartilage repair and regeneration. Stem Cell Rev Rep. 2014; 10(5):686-696.
[23] LI J, HANSEN KC, ZHANG Y, et al. Rejuvenation of chondrogenic potential in a young stem cell microenvironment. Biomaterials. 2014;35(2):642-653.
[24] PIZZUTE T, LI J, ZHANG Y, et al. Fibroblast Growth Factor Ligand Dependent Proliferation and Chondrogenic Differentiation of Synovium-Derived Stem Cells and Concomitant Adaptation of Wnt/Mitogen-Activated Protein Kinase Signals. Tissue Eng Part A. 2016;22(15-16):1036-1046.
[25] KO JY, KIM KI, PARK S, et al. In vitro chondrogenesis and in vivo repair of osteochondral defect with human induced pluripotent stem cells. Biomaterials. 2014;35(11):3571-3581.
[26] 李双星,戚媛,朴丰源,等.利用层粘连蛋白扩增罗曼鹤鸡骨髓间充质干细胞[J].中国组织工程研究,2014,18(45):7222-7226.
[27] CHOI BH, CHOI YS, KANG DG, et al. Cell behavior on extracellular matrix mimic materials based on mussel adhesive protein fused with functional peptides. Biomaterials. 2010;31(34):8980-8988.
[28] HASHIMOTO J, KARIYA Y, MIYAZAKI K. Regulation of proliferation and chondrogenic differentiation of human mesenchymal stem cells by laminin-5 (laminin-332). Stem Cells. 2006;24(11):2346-2354.
[29] DOMOGATSKAYA A, RODIN S, BOUTAUD A, et al. Laminin-511 but not -332, -111, or -411 enables mouse embryonic stem cell self-renewal in vitro. Stem Cells. 2008;26(11):2800-2809.
[30] PEI M. Environmental preconditioning rejuvenates adult stem cells’ proliferation and chondrogenic potential. Biomaterials. 2017;117: 10-23.
[31] PACCOLA MESQUITA FC, HOCHMAN-MENDEZ C, MORRISSEY J, et al. Laminin as a Potent Substrate for Large-Scale Expansion of Human Induced Pluripotent Stem Cells in a Closed Cell Expansion System. Stem Cells Int. 2019;2019:9704945.
[32] LAPERLE A, HSIAO C, LAMPE M, et al. α-5 Laminin Synthesized by Human Pluripotent Stem Cells Promotes Self-Renewal. Stem Cell Reports. 2015;5(2): 195-206.
[33] LAM MT, LONGAKER MT. Comparison of several attachment methods for human iPS, embryonic and adipose-derived stem cells for tissue engineering. J Tissue Eng Regen Med. 2012;6 Suppl 3:s80-86.
[34] RODIN S, ANTONSSON L, NIAUDET C, et al. Clonal culturing of human embryonic stem cells on laminin-521/E-cadherin matrix in defined and xeno-free environment. Nat Commun. 2014;5:3195.
[35] MIYAZAKI T, FUTAKI S, SUEMORI H, et al. Laminin E8 fragments support efficient adhesion and expansion of dissociated human pluripotent stem cells. Nat Commun. 2012;3:1236.
[36] PEI M, LI JT, SHOUKRY M, et al. A review of decellularized stem cell matrix: a novel cell expansion system for cartilage tissue engineering. Eur Cell Mater. 2011; 22:333-343.
[37] KANG BJ, RYU HH, PARK SS, et al. Effect of matrigel on the osteogenic potential of canine adipose tissue-derived mesenchymal stem cells. J Vet Med Sci. 2012; 74(7):827-836.
[38] HEYDARKHAN-HAGVALL S, GLUCK JM, DELMAN C, et al. The effect of vitronectin on the differentiation of embryonic stem cells in a 3D culture system. Biomaterials. 2012;33(7):2032-2040.
[39] BARROS D, CONDE-SOUSA E, GONÇALVES AM, et al. Engineering hydrogels with affinity-bound laminin as 3D neural stem cell culture systems. Biomater Sci. 2019; 7(12):5338-5349.
[40] HE L, TANG S, PRABHAKARAN MP, et al. Surface modification of PLLA nano-scaffolds with laminin multilayer by LbL assembly for enhancing neurite outgrowth. Macromol Biosci. 2013;13(11):1601-1609.
[41] NIE Y, WANG W, XU X, et al. Enhancement of human induced pluripotent stem cells adhesion through multilayer laminin coating. Clin Hemorheol Microcirc. 2018;70(4):531-542.
[42] WANG D, WANG Y, LIU H, et al. Laminin promotes differentiation of rat embryonic stem cells into cardiomyocytes by activating the integrin/FAK/PI3K p85 pathway. J Cell Mol Med. 2019;23(5):3629-3640.
[43] CELEBI B, MANTOVANI D, PINEAULT N. Effects of extracellular matrix proteins on the growth of haematopoietic progenitor cells. Biomed Mater. 2011;6(5):055011.
[44] MATHEWS S, BHONDE R, GUPTA PK, et al. Extracellular matrix protein mediated regulation of the osteoblast differentiation of bone marrow derived human mesenchymal stem cells. Differentiation. 2012;84(2):185-192.
[45] LUO D, RUAN S, LIU A, et al. Laminin functionalized biomimetic apatite to regulate the adhesion and proliferation behaviors of neural stem cells. Int J Nanomedicine. 2018;13:6223-6233.
[46] ABAY N, GUREL PEKOZER G, RAMAZANOGLU M, et al. Bone Formation from Porcine Dental Germ Stem Cells on Surface Modified Polybutylene Succinate Scaffolds. Stem Cells Int. 2016;2016:8792191.
[47] ORTINAU S, SCHMICH J, BLOCK S, et al. Effect of 3D-scaffold formation on differentiation and survival in human neural progenitor cells. Biomed Eng Online. 2010;9:70.
[48] KOMURA T, KATO K, KONAGAYA S, et al. Optimization of surface-immobilized extracellular matrices for the proliferation of neural progenitor cells derived from induced pluripotent stem cells. Biotechnol Bioeng. 2015;112(11):2388-2396.
[49] ARULMOLI J, WRIGHT HJ, PHAN DTT, et al. Combination scaffolds of salmon fibrin, hyaluronic acid, and laminin for human neural stem cell and vascular tissue engineering. Acta Biomater. 2016;43:122-138.
[50] ADDINGTON CP, PAUKEN CM, CAPLAN MR, et al. The role of SDF-1α-ECM crosstalk in determining neural stem cell fate. Biomaterials. 2014;35(10):3263-3272.
[51] TOH WS, FOLDAGER CB, HUI JH, et al. Exploiting Stem Cell-Extracellular Matrix Interactions for Cartilage Regeneration: A Focus on Basement Membrane Molecules. Curr Stem Cell Res Ther. 2016;11(8):618-625.
[52] RAZAVI M, QIAO Y, THAKOR AS. Three-dimensional cryogels for biomedical applications. J Biomed Mater Res A. 2019;107(12): 2736-2755.
[53] CHUN SY, LIM JO, LEE EH, et al. Preparation and Characterization of Human Adipose Tissue-Derived Extracellular Matrix, Growth Factors, and Stem Cells: A Concise Review. Tissue Eng Regen Med. 2019; 16(4):385-393.
[54] SUN Y, CHEN S, PEI M. Comparative advantages of infrapatellar fat pad: an emerging stem cell source for regenerative medicine. Rheumatology (Oxford). 2018;57(12):2072-2086.
[55] SCHREIBER RE, ILTEN-KIRBY BM, DUNKELMAN NS, et al. Repair of osteochondral defects with allogeneic tissue engineered cartilage implants. Clin Orthop Relat Res. 1999;(367 Suppl):S382-395.
[56] KIYOTAKE EA, BECK EC, DETAMORE MS. Cartilage extracellular matrix as a biomaterial for cartilage regeneration. Ann N Y Acad Sci. 2016;1383(1):139-159.
[57] SUN Y, YAN L, CHEN S, et al. Functionality of decellularized matrix in cartilage regeneration: A comparison of tissue versus cell sources. Acta Biomater. 2018; 74:56-73. |