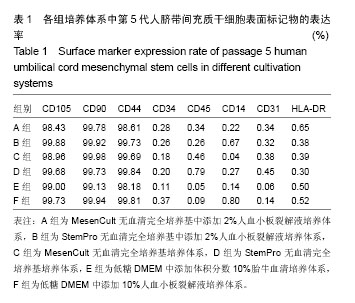
| [1] Parekkadan B,Milwid JM.Mesenchymal stem cells as therapeutics.Annu Rev Biomed Eng.2010;12:87-117.[2] Servais S,Beguin Y,Delens L,et al.Novel approaches for preventing acute graft-versus-host disease after allogeneic hematopoieticstem cell transplantation.Expert Opin Investig Drugs.2016;25(8):957-972. [3] Wystrychowski W,Patlolla B,Zhuge Y,et al.Multipotency and cardiomyogenic potential of human adipose-derived stem cells from epicardium, pericardium, and omentum.Stem Cell Res Ther.2016;7(1):84-95. [4] Jiang R,Han Z,Zhuo G,et al.Transplantation of placenta-derived mesenchymal stem cells in type 2 diabetes: a pilot study.Front Med.2011;5(1):94-100.[5] Deng P,Torrest A,Pollock K,et al.Clinical trial perspective for adult and juvenile Huntington's disease using genetically-engineered mesenchymal stem cells.Neural Regen Res.2016;11(5):702-705. [6] Labrador S,Alonso ML,Alvarez S,et al.Mesenchymal stem cell therapy in retinal and optic nerve diseases: An update of clinical trials.World J Stem Cells.2016;8(11):376-383.[7] Moussa L,Pattappa G,Doix B, et al.A biomaterial-assisted mesenchymal stromal cell therapy alleviates colonic radiation-induced damage.Biomaterials.2017;115:40-52.[8] Mao X,Liu Y,Chen C,et al.Mesenchymal Stem Cells and Their Role in Dental Medicine. Dent Clin North Am.2017;61(1): 161-172. [9] Cui GH,Wang YY,Li CJ,et al.Efficacy of mesenchymal stem cells in treating patients with osteoarthritis of the knee: A meta-analysis.Exp Ther Med.2016;12(5):3390-3400. [10] Oner A,Gonen ZB,Sinim N,et al.Subretinal adipose tissue-derived mesenchymal stem cell implantation in advanced stage retinitis pigmentosa: a phase I clinical safety study.Stem Cell Res Ther.2016;7(1):178.[11] Dothel G,Raschi E,Rimondini R,et al.Mesenchymal stromal cell-based therapy: Regulatory and translational aspects in gastroenterology.World J Gastroenterol.2016; 22(41):9057- 9068.[12] Can A,Balci D.Isolation,culture,and characterization of human umbilical cord stroma-derived mesenchymal stem cells. Methods Mol Biol.2011;698:51-62.[13] Gong W, Han Z, Zhao H,et al.Banking human umbilical cord-derived mesenchymal stromal cells for clinical use.Cell Transplant.2012;21(1):207-216.[14] Zhou HX,Liu ZG,Liu XJ,et al.Umbilical cord-derived mesenchymal stem cell transplantation combined with hyperbaric oxygen treatment for repair of traumatic brain injury.Neural Regen Res.2016;11(1): 107-113.[15] Guo ZY,Sun X,Xu XL,et al.Human umbilical cord mesenchymal stem cells promote peripheral nerve repair via paracrine mechanisms. Neural Regen Res. 2015;10(4): 651-658. [16] Zhao Q,Wang XY,Yu XX,et al.Expression of human telomerase reverse transcriptase mediates the senescence of mesenchymal stem cells through the PI3K/AKT signaling pathway.Int J Mol Med.2015;36(3):857-864.[17] Mizuno M,Katano H,Otabe K,et al.Platelet-derived growth factor (PDGF)-AA/AB in human serum are potential indicators of the proliferative capacity of human synovial mesenchymal stem cells.Stem Cell Res Ther.2015;6:243-253.[18] Esmaeli A,Moshrefi M,Shamsara A,et al.Xeno-free culture condition for human bone marrow and umbilical cord matrix-derived mesenchymalstem/stromal cells using human umbilical cord blood serum.Int J Reprod Biomed (Yazd). 2016; 14(9):567-576.[19] Fazzina R,Iudicone P,Fioravanti D,et al.Potency testing of mesenchymal stromal cell growth expanded in human platelet lysate from different human tissues. Stem Cell Res Ther.2016; 7(1):122.[20] Smith JR,Pfeifer K,Petry F,et al.Standardizing Umbilical Cord Mesenchymal Stromal Cells for Translation to Clinical Use: Selection of GMP-Compliant Medium and a Simplified Isolation Method.Stem Cells Int.2016;2016:6810980.[21] Kocaoemer A,Kern S,Klüter H,et al.Human AB serum and thrombin-activated platelet-rich plasma are suitable alternatives to fetal calf serum for the expansion of mesenchymal stem cells from adipose tissue.Stem Cells. 2007;25(5):1270-1278. [22] Tateishi K,Ando W,Higuchi C,et al.Comparison of human serum with fetal bovine serum for expansion and differentiation of human synovial MSC: Potential feasibility for clinical applications.Cell Transplant.2008;17(5):549-557.[23] Yilmaz M,Ovali E,Akdogan E,et al.Autologous serum is more effective than fetal bovine serum on proliferation of bone marrow derived human mesenchymal stem cells.Saudi Med J.2008;29(2):306-309.[24] Juhl M,Tratwal J,Follin B,et al.Comparison of clinical grade human platelet lysates for cultivation of mesenchymal stromal cells from bone marrow and adipose tissue.Scand J Clin Lab Invest.2016;76(2):93-104. [25] Stute N,Holtz K,Bubenheim M,et al.Autologous serum for isolation and expansion of human mesenchymal stem cells for clinical use.Exp Hematol.2004;32(12):1212-1225.[26] Lange C,Cakiroglu F,Spiess AN,et al.Accelerated and safe expansion of human mesenchymal stromal cells in animal serum-free medium for transplantation and regenerative medicine.J Cell Physiol.2007;213(1):18-26.[27] Chase LG,Lakshmipathy U,Solchaga LA,et al.A novel serum-free medium for the expansion of human mesenchymal stem cells.Stem Cell Res Ther.2010;1(1):8.[28] Haack-Sorensen M,Friis T,Bindslev L,et al.Comparison of different culture conditions for human mesenchymal stromal cells for clinical stem cell therapy.Scand J Clin Lab Invest. 2008;68(3):192-203.[29] Merlo B,Pirondi S,Iacono E,et al. Viability, in vitro Differentiation and Molecular characterization of equine adipose tissue-derived mesenchymal stem cells cryopreserved in serum and serum-free medium. Cryo Letters. 02016;37(4):243-252.[30] Zanicotti DG,Coates DE.Growing Adipose-Derived Stem Cells Under Serum-Free Conditions.Methods Mol Biol. 2017; 1537: 439-446.[31] Escobar CH,Chaparro O.Xeno-Free Extraction, Culture, and Cryopreservation of Human Adipose-Derived Mesenchymal Stem Cells.Stem Cells Transl Med.2016; 5(3): 358-365.[32] Wu X,Kang H,Liu X,et al.Serum and xeno-free, chemically defined, no-plate-coating-based culture system for mesenchymal stromal cells from the umbilical cord.Cell Prolif. 2016;49(5):579-88.[33] Wu JY,Lu Y,Chen JS,et al.Pooled Umbilical Cord Blood Plasma for Culturing UCMSC and Ex Vivo Expanding Umbilical Cord Blood CD34? Cells.Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2015;23(4):1112-1119.[34] Chase LG,Yang S,Zachar V,et al.Development and characterization of a clinically compliant xeno-free culture medium in good manufacturing practice for human multipotent mesenchymal stem cells.Stem Cells Transl Med. 2012;1(10):750-758. [35] Simões IN,Boura JS,dos Santos F,et al.Human mesenchymal stem cells from the umbilical cord matrix: successful isolation and ex vivo expansion using serum-/xeno-free culture media. Biotechnol J.2013;8(4):448-458. [36] Swamynathan P,Venugopal P,Kannan S,et al.Are serum-free and xeno-free culture conditions ideal for large scale clinical grade expansion of Wharton's jelly derived mesenchymal stem cells? A comparative study. Stem Cell Res Ther.2014; 5(4):88. [37] Rakian R,Block TJ,Johnson SM,et al.Native extracellular matrix preserves mesenchymal stem cell "stemness" and differentiation potential under serum-free culture conditions. Stem Cell Res Ther.2015;6:235. [38] Gottipamula S,Ashwin KM,Muttigi MS,et al.Isolation, expansion and characterization of bone marrow-derived mesenchymal stromal cells in serum-free conditions.Cell Tissue Res. 2014;356(1):123-135. [39] Mizukami A,Fernandes-Platzgummer A,Carmelo JG,et al.Stirred tank bioreactor culture combined with serum-/xenogeneic-free culture medium enables an efficient expansion of umbilical cord-derived mesenchymal stem/stromal cells.Biotechnol J. 2016;11(8):1048-1059. [40] Gerby S,Attebi E,Vlaski M,et al.A new clinical-scale serum-free xeno-free medium efficient in ex vivo amplification of mesenchymal stromal cells does not support mesenchymal stem cells.Transfusion.2016.doi: 10.1111/trf.13902. [Epub ahead of print] |
.jpg)

.jpg)