| [1]Younger EM, Chapman MW Morbidity at bone graft donor sites. J Orthop Trauma. 1989;3(3):192-195.[2]Pryor LS, Gage E, Langevin CJ, et al. Review of bone substitutes. Craniomaxillofac Trauma Reconstr. 2009;2(3): 151-160.[3]Perry A, Mahar A, Massie J, et al. Biomechanical evaluation of kyphoplasty with calcium sulfate cement in a cadaveric osteoporotic vertebral compression fracture model. Spine J. 2005;5(5):489-493.[4]Stubbs D, Deakin M, Chapman-Sheath P, et al. In vivo evaluation of resorbable bone graft substitutes in a rabbit tibial defect model. Biomaterials. 2004;25(20):5037-5044.[5]Goodrich JT, Sandler AL, Tepper O. A review of reconstructive materials for use in craniofacial surgery bone fixation materials, bone substitutes, and distractors. Childs Nerv Syst. 2012;28(9):1577-1588.[6]Magnan B, Bondi M, Maluta T, et al. Acrylic bone cement: current concept review. Musculoskelet Surg. 2013;97(2):93-100.[7]Wu XT, Jiang XJ, Zhang SD, et al. Biomechanical evaluation of vertebroplasty using calcium sulfate cement for thoracolumbar burst fractures. Chin J Traumatol. 2007;10(6): 327-333.[8]Taylor RS, Fritzell P, Taylor RJ. Balloon kyphoplasty in the management of vertebral compression fractures: an updated systematic review and meta-analysis. Eur Spine J. 2007;16(8): 1085-1100.[9]Heini PF, Berlemann U. Bone substitutes in vertebroplasty. Eur Spine J. 2001;10 Suppl 2:S205-213.[10]Xie P, Zhao Y, Li G. Efficacy of percutaneous vertebroplasty in patients with painful vertebral metastases: A retrospective study in 47 cases. Clin Neurol Neurosurg. 2015;138:157-161.[11]Dressmann H. Ueber Knochenplombierung bei Hohlenformigen Defekten des Knochens. Beitr Klin Chir. 1892; 9: 804-810.[12]Turner TM, Urban RM, Gitelis S, et al. Resorption evaluation of a large bolus of calcium sulfate in a canine medullary defect. Orthopedics. 2003;26(5 Suppl):s577-579.[13]Zhang J, He F, Zhang W, et al. Mechanical force enhanced bony formation in defect implanted with calcium sulphate cement. Bone Res. 2015;3:14048.[14]Zampelis V, Tägil M, Lidgren L, et al. The effect of a biphasic injectable bone substitute on the interface strength in a rabbit knee prosthesis model. J Orthop Surg Res. 2013;8:25.[15]Liu W, Wu C, Liu W, et al. The effect of plaster (CaSO4 •1/2H2O) on the compressive strength, self-setting property, and in vitro bioactivity of silicate-based bone cement.J Biomed Mater Res B Appl Biomater. 2013;101(2):279-286.[16]崔文岗,石岩,肖德明.硫酸钙人工骨修复骨缺损的应用研究进展[J]. 生物骨科材料与临床研究, 2013, 10(5): 21-23.[17]Ryu KS, Shim JH, Heo HY, et al. Therapeutic efficacy of injectable calcium phosphate cement in osteoporotic vertebral compression fractures: prospective nonrandomized controlled study at 6-month follow-up. World Neurosurg. 2010;73(4): 408-411.[18]Cardoso DA, van den Beucken JJ, Both LL, et al. Gelation and biocompatibility of injectable alginate-calcium phosphate gels for bone regeneration. J Biomed Mater Res A. 2014; 102(3):808-817.[19]Evaniew N, Tan V, Parasu N, et al. Use of a calcium sulfate-calcium phosphate synthetic bone graft composite in the surgical management of primary bone tumors. Orthopedics. 2013;36(2):e216-222.[20]黄强,李程,周宗科,等.掺锶硫酸钙复合骨修复材料的制备及体外特性研究[J].生物医学工程学杂志,2009,26(3):575-579.[21]张民,王建生,卫小春,等.注射性硫酸钙/羟基磷灰石骨替代材料的性能研究[J].中国康复,2006,21(1): 16-17.[22]陈华,田学忠,张伯勋,等.硫酸钙骨内植入修复兔包容性骨缺损的力学分析[J].中国矫形外科杂志, 2008, 16(5): 373-375.[23]赵增辉,蒋电明,权正学,等. 灭菌对聚氨基酸/硫酸钙复合材料理化性能的影响[J].第三军医大学学报, 2011, 33(2): 119-123.[24]严永刚,李鸿,吕国玉,等.氨基酸共聚物-硫酸钙复合材料及制备方法:中国, CN101560326B [P].2011-01-19.[25]周春光,宋跃明,屠重棋,等. 多元氨基酸共聚物/磷酸钙复合材料椎间融合器的设计制备及压缩强度测试[J]. 生物医学工程学杂志, 2011, 28(6): 1136-1140.[26]Bell WH. Resorption Characteristics of Bone and Bone Substitutes. Oral Surg Oral Med Oral Pathol. 1964;17: 650-657.[27]姜星杰,吴小涛,张绍东,等. 硫酸钙骨水泥降解成骨性能的实验研究[J].中华创伤骨科杂志,2007, 9(8): 717-720.[28]普有登,汤逊. 硫酸钙骨移植替代材料的研究及应用进展[J].中国矫形外科杂志,2011,19(11): 931-933.[29]Carinci F, Piattelli A, Stabellini G, et al. Calcium sulfate: analysis of MG63 osteoblast-like cell response by means of a microarray technology. J Biomed Mater Res B Appl Biomater. 2004;71(2):260-267.[30]Sidqui M, Collin P, Vitte C, et al. Osteoblast adherence and resorption activity of isolated osteoclasts on calcium sulphate hemihydrate. Biomaterials. 1995;16(17):1327-1332.[31]Ricci JL,Weiner MJ,Iorio DD,et al. Evaluation of Timed Release Calcium Sulfate (CS-TR) Bone Graft Substitutes. Microscopy & Microanalysis. 2005; 11(11):1256-1257.[32]Palmieri A, Pezzetti F, Brunelli G, et al. Calcium sulfate acts on the miRNA of MG63E osteoblast-like cells. J Biomed Mater Res B Appl Biomater. 2008;84(2):369-374.[33]Walsh WR, Morberg P, Yu Y, et al. Response of a calcium sulfate bone graft substitute in a confined cancellous defect. Clin Orthop Relat Res. 2003;(406):228-236.[34]Lazáry A, Speer G, Varga PP, et al. Effect of vertebroplasty filler materials on viability and gene expression of human nucleus pulposus cells. J Orthop Res. 2008;26(5):601-607.[35]Dasmah A, Sennerby L, Rasmusson L, et al. Intramembraneous bone tissue responses to calcium sulfate: an experimental study in the rabbit maxilla. Clin Oral Implants Res. 2011;22(12):1404-1408.[36]Rauschmann MA, Wichelhaus TA, Stirnal V, et al. Nanocrystalline hydroxyapatite and calcium sulphate as biodegradable composite carrier material for local delivery of antibiotics in bone infections. Biomaterials. 2005;26(15): 2677-2684.[37]Mazurkiewicz T, Matuszewski L, Matuszewska A, et al. Implanted bisphosphonates in bone cement affect bone markers in rat serum. Int Orthop. 2013;37(5):969-974. |
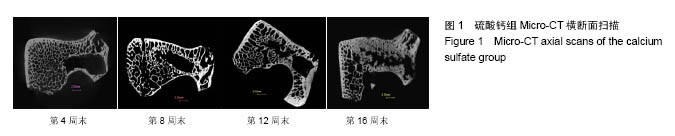
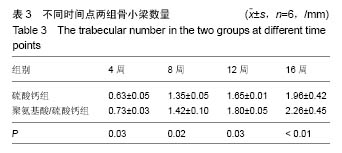
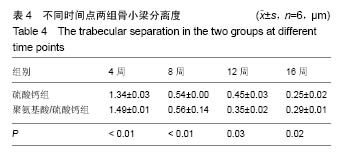
.jpg)
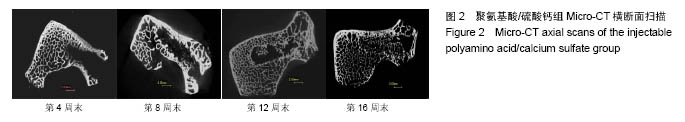
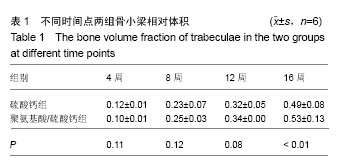
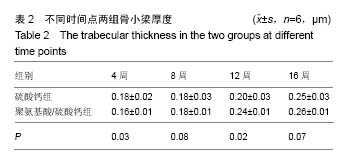
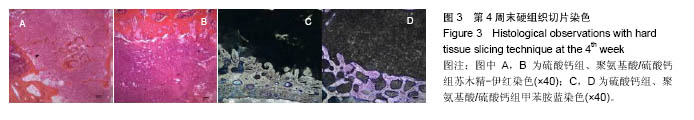
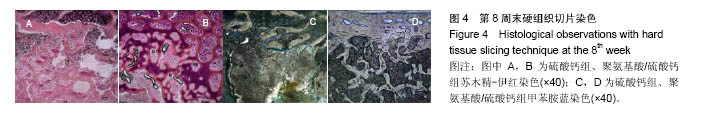

.jpg)