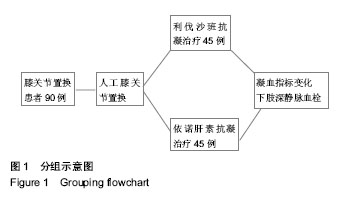
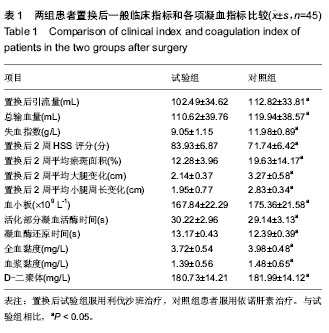
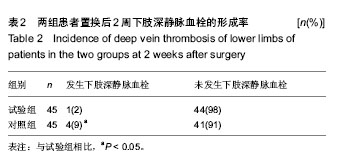
| [1] 赵邦术,刘新伟.股神经阻滞复合喉罩全麻在老年患者膝关节置换中的作用[J].检验医学与临床,2014,11(19):2665-2667.
[2] Lassen MR, Davidson BL, Gallus A,等.口服Ⅹa因子直接抑制剂阿哌沙班预防全膝关节置换后患者血栓形成的效果和安全性[J].中华关节外科杂志(电子版),2013,(4):570-576.
[3] Schneeweiss S, Gagne JJ, Patrick AR, et al. Comparative efficacy and safety of dabigatran, rivaroxaban and apixaban in patients with atrital fibrillation using network metaanalysis. Int Angiol. 2012;31(4):330-339.
[4] 陈俊晶.健康教育路径在膝关节置换患者护理中的应用[J].解放军护理杂志,2013,30(21):50-52.
[5] 代宏杰,靳宪辉,崔胜光,等. 人工膝关节置换中髌骨置换效果的Meta分析[J].中国组织工程研究,2012,39(16):7283-7287.
[6] 吴志远,贾杰,欧阳桂林,等.推拿手法对全膝关节置换后患者康复及D-二聚体水平的影响[J].成都医学院学报,2012,7(4): 536-539.
[7] 李毓灵,张健.晚期弥漫型色素沉着绒毛结节性滑膜炎行全膝关节置换的初步讨论[J].激光杂志,2014,35(1):63-65.
[8] 施华萍.人工全新膝关节置换患者的围手术期护理[J].中国实用护理杂志,2012,28(3):33-34.
[9] 王攀.利伐沙班与低分子肝素预防人工全膝关节置换后深静脉血栓形成的研究[J].中国依序前沿杂志(电子版),2014,6(4): 111-114.
[10] 高乐才,吴文元,魏金栋,等.置换后抗凝对膝关节置换后隐形失血影响的临床观察[J].河北医药,2013,35(6):880-882.
[11] 李佩佳,高玉镭,王东辰,等.利伐沙班与低分子肝素在预防人工全膝关节置换后深静脉血栓形成的分析[J].中国矫形外科杂志, 2012,20(11):998-1000.
[12] 王炜,翁习生,林进,等.单侧膝关节置换后依诺肝素和利伐沙班抗凝效果的随机对照研究[J].中国骨与关节外科,2012,5(3): 198-202.
[13] 李虎,王健,肖军,等.比较磺达肝葵钠与依诺肝素预防骨科大置换后静脉血栓栓塞症疗效与安全性的Meta分析[J].南方医科大学学报,2013,33(3):370-375.
[14] 范少地,王晓坤,白峰,等.膝关节置换后应用药物预防血栓形成的对比研究与国际分析[J].中国组织工程研究,2012,16(9): 1661-1670.
[15] 李军,王健,史占军,等.全髋关节置换后应用利伐沙班安全性的Meta分析[J].中国组织工程研究,2012,16(9):1618-1621.
[16] 杨礼庆,王程,深涛,等.口服拉伐沙班预防人工全髋关节置换后深静脉血栓的临床研究[J].中国临床药理学杂志,2013,29(4): 260-262.
[17] Kennedy B, GargoumFS, Kennedy L, et al. Emerging articoagulants. Curr Med Chem. 2012;19(20):3388-
3416.
[18] Brinkman HJ. Global assays and the management of oral anticoagulation. Thromb J. 2015;13:9.
[19] Jourdi G, Siguret V, Martin AC, et al. Association rate constants rationalise the pharmacodynamics of apixaban and rivaroxaban. Thromb Haemost. 2015;113(6).
[20] Farag M, Gorog DA. Rivaroxaban in atrial fibrillation cardioversion: insights from the X-VeRT trial. Future Cardiol. 2015;11(2):147-151.
[21] Washam JB, Stevens SR, Lokhnygina Y, et al. Digoxin use in patients with atrial fibrillation and adverse cardiovascular outcomes: a retrospective analysis of the Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET AF). Lancet. 2015. doi: 10.1016/S0140-6736(14)61836-5.
[22] Laliberté F, Cloutier M, Crivera C, et al. Effects of rivaroxaban versus warfarin on hospitalization days and other health care resource utilization in patients with nonvalvular atrial fibrillation: an observational study from a cohort of matched users. Clin Ther. 2015. doi: 10.1016/j.clinthera.2015.02.001.
[23] Demirtas S, Karahan O, Yaz?c? S, et al. Investigation of possible prophylactic, renoprotective, and cardioprotective effects of thromboprophylactic drugs against ischemia- reperfusion injury. Kaohsiung J Med Sci. 2015;31(3):115-122.
[24] Douxfils J, Chatelain B, Hjemdahl P, et al. Does the Russell Viper Venom time test provide a rapid estimation of the intensity of oral anticoagulation? A cohort study. Thromb Res. 2015. doi: 10.1016/j.thromres.2015.02.020.
[25] Singer DE, Hellkamp AS, Yuan Z, et al. Alternative calculations of individual patient time in therapeutic range while taking warfarin: results from the rocket af trial. J Am Heart Assoc. 2015;4(3): e001349.
[26] Loffredo L, Perri L, Violi F. Impact of new oral anticoagulants on gastrointestinal bleeding in atrial fibrillation: A meta-analysis of interventional trials. Dig Liver Dis. 2015. doi: 10.1016/j.dld.2015.01.159.
[27] Ma G, Zhang R, Wu X, et al. Direct factor Xa inhibitors (rivaroxaban and apixaban) versus enoxaparin for the prevention of venous thromboembolism after total knee replacement: a meta-analysis of 6 randomized clinical trials. Thromb Res. 2015. doi: 10.1016/j.thromres.2015.02.008.
[28] Ferreira J, Mirco A. Systematic review of cost-effectiveness analyses of novel oral anticoagulants for stroke prevention in atrial fibrillation. Rev Port Cardiol. 2015. doi: 10.1016/j.repc.2014.08.008.
[29] Patel AD, Tan MK, Angaran P, et al. Risk Stratification and stroke prevention therapy care gaps in canadian atrial fibrillation patients (from the co-ordinated national network to engage physicians in the care and treatment of patients with atrial fibrillation chart audit). Am J Cardiol. 2015;115(5): 641-646.
[30] Lauffenburger JC, Farley JF, Gehi AK, et al. Factors driving anticoagulant selection in patients with atrial fibrillation in the united states. Am J Cardiol. 2015. doi: 10.1016/j.amjcard.2015.01.539.
[31] Charters MA, Frisch NB, Wessell NM, et al. Rivaroxaban versus enoxaparin for venous thromboembolism Prophylaxis after hip and knee arthroplasty. J Arthroplasty. 2015. doi: 10.1016/j.arth.2015.02.009.
[32] Coleman CM, Khalaf S, Mould S, et al. Novel oral anticoagulants for DC cardioversion procedures: utilization and clinical outcomes compared with warfarin. Pacing Clin Electrophysiol. 2015. doi: 10.1111/pace.12618.
[33] Yamada N, Hirayama A, Maeda H, et al. Oral rivaroxaban for Japanese patients with symptomatic venous thromboembolism - the J-EINSTEIN DVT and PE program. Thromb J. 2015;13:2.
[34] Bacchus F, Schulman S. Clinical experience with the new oral anticoagulants for treatment of venous thromboembolism. Arterioscler Thromb Vasc Biol. 2015;35(3):513-519.
[35] Fermann GJ, Erkens PM, Prins MH, et al. Treatment of pulmonary embolism with rivaroxaban: outcomes by simplified pulmonary embolism severity index score from a post hoc analysis of the EINSTEIN PE study. Acad Emerg Med. 2015;22(3):299-307.
[36] Lobo N, Ejiofor K, Thurairaja R, et al. Life-threatening haematuria caused by vancomycin-induced thrombocytopenia. BMJ Case Rep. 2015. doi: 10.1136/bcr-2014-208192.
[37] Favaloro EJ, Lippi G. Laboratory testing in the era of direct or non-vitamin k antagonist oral anticoagulants: a practical guide to measuring their activity and avoiding diagnostic errors. Semin Thromb Hemost. 2015;41(2):208-227.
[38] Chin PK. Which patients may benefit from dose adjustment of non-vitamin k antagonist oral anticoagulants? Semin Thromb Hemost. 2015;41(2):195-207.
[39] Hidalgo F, Gómez-Luque A, Ferrandis R, et al. Perioperative management of direct oral anticoagulant in emergency surgery and bleeding. Haemostasis monitoring and treatment. Rev Esp Anestesiol Reanim. 2015. doi: 10.1016/j.redar.2015.01.002.
[40] Wells PS, Gebel M, Prins MH, et al. Influence of statin use on the incidence of recurrent venous thromboembolism and major bleeding in patients receiving rivaroxaban or standard anticoagulant therapy. Thromb J. 2014;12:26.
[41] Harder S. Pharmacokinetic and pharmacodynamic evaluation of rivaroxaban: considerations for the treatment of venous thromboembolism. Thromb J. 2014;12:22.
[42] Hoffman M, Monroe DM. Reversing targeted oral anticoagulants. Hematology Am Soc Hematol Educ Program. 2014;2014(1):518-523.
[43] Siegal DM, Konkle BA. What is the effect of rivaroxaban on routine coagulation tests? Hematology Am Soc Hematol Educ Program. 2014;2014(1):334-336.
[44] Beyer-Westendorf J, Förster K, et al. Drug persistence with rivaroxaban therapy in atrial fibrillation patients-results from the Dresden non-interventional oral anticoagulation registry. Europace. 2015.
[45] Bultas J. Anticoagulant therapy in secondary prevention of coronary events. Vnitr Lek. 2014;60(12):1023-1032.
[46] Imberti D, Ambrosoli A, Cimminiello C, et al. Periprocedural management of rivaroxaban-treated patients. Expert Opin Pharmacother. 2015;16(5):685-6891.
[47] Raschi E, Poluzzi E, Koci A, et al. Liver injury with novel oral anticoagulants: assessing post-marketing reports in the US Food and Drug Administration Adverse Event Reporting System. Br J Clin Pharmacol. 2015. doi: 10.1111/bcp.12611 |