| [1]Abdullah KG,Steinmetz MP,Benzel EC,et al.The state of lumbar fusion extenders. Spine (Phila Pa 1976).2011;36(20):E1328-1334.[2]Buser Z,Brodke DS,Youssef JA,et al.Synthetic bone graft versus autograft or allograft for spinal fusion: a systematic review.J Neurosurg Spine. 2016; 25(4):509-516.[3]Mao K,Cui F,Li J,et al.Preparation of combined β-TCP/α-CSH artificial bone graft and its performance in a spinal fusion model.J Biomater Appl. 2012; 27(1):37-45.[4]毛克亚,李江涛,杨云,等.β-TCP/α-CSH复合植骨材料的制备与理化性能检测[J].生物医学工程与临床, 2011,15(3):205-209.[5]Parfitt AM.Bone histomorphometry: proposed system for standardization of nomenclature, symbols, and units.Calcif Tissue Int.1988;42(5):284-286.[6]Pierannunzii L,Zagra L.Bone grafts, bone graft extenders, substitutes and enhancers for acetabular reconstruction in revision total hip arthroplasty. EFORT Open Rev.2016;1(12):431-439.[7]Fillingham Y,Jacobs J.Bone grafts and their substitutes.Bone Joint J. 2016;98-B(1 Suppl A):6-9.[8]Rh OG,Dard M,Larjava H.Hydoxyapatite/beta-tricalcium phosphate biphasic ceramics as regenerative material for the repair of complex bone defects.J Biomed Mater Res B Appl Biomater.2018;106(6): 2493-2512.[9]Barbanti BG,Griffoni C,Nataloni A,et al.Biomaterials as bone graft substitutes for spine surgery: from preclinical results to clinical study.J Biol Regul Homeost Agents.2017;31(4 suppl 1):167-181.[10]Pulyala P,Singh A,Dias-Netipanyj MF,et al.In-vitro cell adhesion and proliferation of adipose derived stem cell on hydroxyapatite composite surfaces.Mater Sci Eng C Mater Biol Appl.2017;75: 1305-1316.[11]Oliveira HL,WLO DR,Cuevas-Suárez CE,et al.Histological Evaluation of Bone Repair with Hydroxyapatite: A Systematic Review.Calcif Tissue Int. 2017;101(4):341-354.[12]Li B,Liu Z,Yang J,et al.Preparation of bioactive β-tricalcium phosphate microspheres as bone graft substitute materials.Mater Sci Eng C Mater Biol Appl.2017;70(Pt 2): 1200-1205.[13]Fathima H, Harish. Osteostimulatory effect of bone grafts on fibroblast cultures. J Nat Sci Biol Med.2015;6(2):291-294.[14]Dahabreh Z,Panteli M,Pountos I,et al.Ability of bone graft substitutes to support the osteoprogenitor cells: An in-vitro study.World J Stem Cells. 2014;6(4):497-504.[15]Barradas AM,Monticone V,Hulsman M,et al. Molecular mechanisms of biomaterial-driven osteogenic differentiation in human mesenchymal stromal cells.Integr Biol (Camb).2013;5(7):920-931.[16]毛文文,茹江英.羟基磷灰石类陶瓷在骨组织工程中的研究与更广泛应用[J].中国组织工程研究,2018,22(30):4855-4863.[17]Rakhmatia YD,Ayukawa Y,Jinno Y,et al.Micro-computed tomography analysis of early stage bone healing using micro-porous titanium mesh for guided bone regeneration: preliminary experiment in a canine model. Odontology. 2017;105(4):408-417.[18]Demirel M,Aksakal B.Effect of porosity on the structure, mechanical properties and cell viability of new bioceramics as potential bone graft substitutes.Acta Bioeng Biomech. 2018;20(2):11-22.[19]Pihlman H,Keränen P,Paakinaho K,et al.Novel osteoconductive β-tricalcium phosphate/poly(L-lactide-co-e-caprolactone) scaffold for bone regeneration: a study in a rabbit calvarial defect.J Mater Sci Mater Med. 2018;29(10):156.[20]Okada T,Kanai T,Tachikawa N,et al.Histological and Histomorphometrical Determination of the Biogradation of β-Tricalcium Phosphate Granules in Maxillary Sinus Floor Augmentation: A Prospective Observational Study. Implant Dent.2017;26(2):275-283.[21]Tanaka T,Komaki H,Chazono M,et al.Basic research and clinical application of beta-tricalcium phosphate (β-TCP).Morphologie.2017;101(334): 164-172.[22]Matsunaga A,Takami M,Irié T,et al.Microscopic study on resorption of β-tricalcium phosphate materials by osteoclasts.Cytotechnology. 2015 ; 67(4):727-732.[23]Jelusic D,Zirk ML,Fienitz T,et al.Monophasic ß-TCP vs. biphasic HA/ß-TCP in two-stage sinus floor augmentation procedures - a prospective randomized clinical trial. Clin Oral Implants Res.2017;28(10):e175-e183.[24]Fouad H,AlFotawi R,Alothman OY,et al.Porous Polyethylene Coated with Functionalized Hydroxyapatite Particles as a Bone Reconstruction Material.Materials (Basel).2018;11(4). pii: E521. doi: 10.3390/ma11040521.[25]Leventis M,Fairbairn P,Mangham C,et al.Bone Healing in Rabbit Calvaria Defects Using a Synthetic Bone Substitute: A Histological and Micro-CT Comparative Study.Materials (Basel).2018;11(10). pii: E2004. doi: 10.3390/ma11102004.[26]Shih TC,Teng NC,Wang PD,et al.In vivo evaluation of resorbable bone graft substitutes in beagles: histological properties.J Biomed Mater Res A. 2013; 101(8):2405-2411.[27]Pförringer D,Harrasser N,Mühlhofer H,et al.Osteoinduction and -conduction through absorbable bone substitute materials based on calcium sulfate: in vivo biological behavior in a rabbit model. J Mater Sci Mater Med. 2018; 29(2):17.[28]Tan V,Evaniew N,Finlay K,et al.Chronology of the Radiographic Appearances of the Calcium Sulfate-Calcium Phosphate Synthetic Bone Graft Composite Following Resection of Bone Tumors: A Follow-up Study of Postoperative Appearances.Can Assoc Radiol J.2016;67(1):21-27.[29]谭海涛,孟志斌,李俊,等.β磷酸三钙和α半水硫酸钙复合人工骨生物相容性及在脊柱融合模型中的应用[J].中国组织工程研究, 2017,21(26):4119-4124.[30]Orellana BR,Hilt JZ,Puleo DA.Drug release from calcium sulfate-based composites. J Biomed Mater Res B Appl Biomater.2015;103(1):135-142.[31]Wu CC,Huang YK,Chang WJ,et al.Limitation of the antibiotic-eluting bone graft substitute: An example of gentamycin-impregnated calcium sulfate.J Biomed Mater Res B Appl Biomater.2018;106(1): 80-87.[32]王骞,耿广起,丛晓明,等.载三联抗痨药硫酸钙/聚氨基酸缓释材料在兔脊柱结核模型体内的缓释性能[J].中国组织工程研究, 2017,21(10):1520-1526.[33]Civinini R,Capone A,Carulli C,et al.The kinetics of remodeling of a calcium sulfate/calcium phosphate bioceramic.J Mater Sci Mater Med.2017;28(9):137.[34]Sakata M,Tonomura H,Itsuji T,et al.Bone Regeneration of Osteoporotic Vertebral Body Defects Using Platelet-Rich Plasma and Gelatin β-Tricalcium Phosphate Sponges. Tissue Eng Part A.2018; 24(11-12): 1001-1010. |
.jpg)
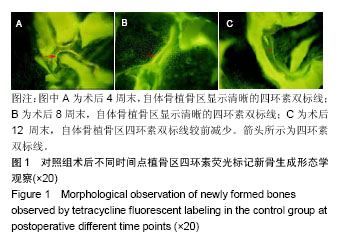
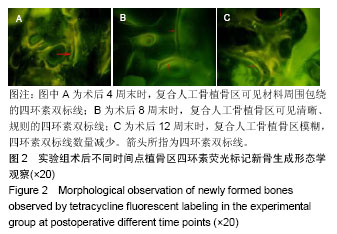
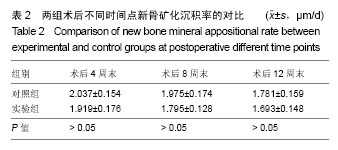
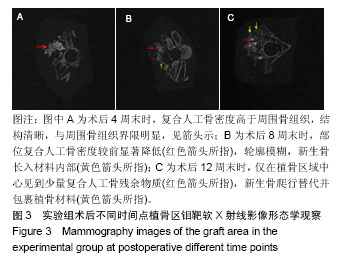

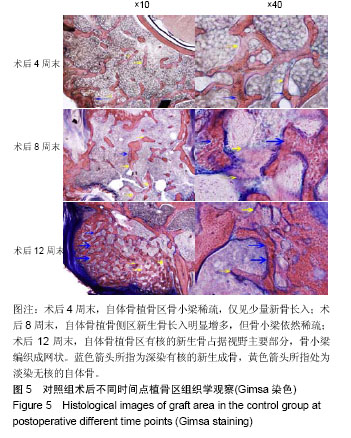
.jpg)
.jpg)