中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (34): 9032-9040.doi: 10.12307/2026.832
• 组织构建综述 tissue construction review • 上一篇 下一篇
滑膜液外泌体介导滑膜细胞与软骨细胞信号互作在膝骨关节炎发生发展中的作用
王燕飞1,2,靳连海1,2,李清雅1,2,付远飞1,2,谭黄圣1,2,邓鹏伟1,2,高 坤1,2
- 1深圳市中医院,广东省深圳市 518000;2广州中医药大学第四临床医学院,广东省深圳市 518000
-
收稿日期:2025-09-29修回日期:2026-01-05出版日期:2026-12-08发布日期:2026-04-14 -
通讯作者:高坤,博士,主治医师,深圳市中医院,广东省深圳市 518000;广州中医药大学第四临床医学院,广东省深圳市 518000 -
作者简介:王燕飞,女,1998年生,2024年河南中医药大学毕业,主要从事中医骨伤相关疾病研究。 -
基金资助:深圳市科技计划资助(JCYJ20240813152355072),项目负责人:高坤;国家自然科学基金青年项目(82104884),项目
负责人:高坤
Synovial fluid exosome-mediated crosstalk between synoviocytes and chondrocytes in development and progression of knee osteoarthritis
Wang Yanfei1, 2, Jin Lianhai1, 2, Li Qingya1, 2, Fu Yuanfei1, 2, Tan Huangsheng1, 2, Deng Pengwei1, 2, Gao Kun1, 2
- 1Shenzhen Traditional Chinese Medicine Hospital, Shenzhen 518000, Guangdong Province, China; 2Fourth Clinical Medical College of Guangzhou University of Chinese Medicine, Shenzhen 518000, Guangdong Province, China
-
Received:2025-09-29Revised:2026-01-05Online:2026-12-08Published:2026-04-14 -
Contact:Gao Kun, MD, Attending physician, Shenzhen Traditional Chinese Medicine Hospital, Shenzhen 518000, Guangdong Province, China; Fourth Clinical Medical College of Guangzhou University of Chinese Medicine, Shenzhen 518000, Guangdong Province, China -
About author:Wang Yanfei, Shenzhen Traditional Chinese Medicine Hospital, Shenzhen 518000, Guangdong Province, China; Fourth Clinical Medical College of Guangzhou University of Chinese Medicine, Shenzhen 518000, Guangdong Province, China -
Supported by:Shenzhen Science and Technology Program, No. JCYJ20240813152355072 (to GK); National Natural Science Foundation of China (Youth Project), No. 82104884 (to GK)
摘要:
文题释义:
滑膜液外泌体:由滑膜腔内的多种细胞(如滑膜细胞、软骨细胞、免疫细胞等)分泌,富含蛋白质、脂质、核酸等信息分子,具有高度稳定性、组织靶向性和生物活性。
膝骨关节炎:是一种常见的慢性退行性、非特异性炎症的关节疾病,在慢性炎症环境的影响下,膝关节可出现滑膜炎症、软骨退变以及关节功能的逐渐丧失。
背景:越来越多的证据表明滑膜液外泌体介导滑膜细胞-软骨细胞信号互作在膝骨关节炎中的重要作用。滑膜细胞与软骨细胞的信号互作,从分子层面揭示了滑膜液外泌体在膝骨关节炎进展中的双向调控作用。
目的:从多角度、多层次、全方位阐释滑膜液外泌体介导滑膜细胞与软骨细胞的信号交互作用,以期为相关领域的研究与临床应用提供新思路与新视角。
方法:检索PubMed数据库2001年1月至2025年6月的文献,以“Osteoarthritis*,Exosomes,Synovial cells,Chondrocytes”为检索词,检索2001年1月至2025年6月发表的所有相关文献,纳入原始研究、综述及临床试验等文献,排除无关机制及与主题相关性低的文献,最终筛选93篇文献进行综合分析。
结果与结论:在膝骨关节炎发生与发展过程中,滑膜细胞与软骨细胞之间借助滑膜液外泌体建立了密切的信号交流,这种信号互作显著影响膝骨关节炎的进展,既能加剧软骨退变和滑膜炎症,也可能在特定条件下发挥保护性调控作用。因此,该机制的双面性凸显了对它进行精准干预的必要性,而针对外泌体介导的细胞间信号通路的关键分子,有望为膝骨关节炎提供新的治疗策略与靶点。
https://orcid.org/0009-0003-3812-7015(王燕飞)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
王燕飞, 靳连海, 李清雅, 付远飞, 谭黄圣, 邓鹏伟, 高 坤, . 滑膜液外泌体介导滑膜细胞与软骨细胞信号互作在膝骨关节炎发生发展中的作用[J]. 中国组织工程研究, 2026, 30(34): 9032-9040.
Wang Yanfei, Jin Lianhai, Li Qingya, Fu Yuanfei, Tan Huangsheng, Deng Pengwei, Gao Kun. Synovial fluid exosome-mediated crosstalk between synoviocytes and chondrocytes in development and progression of knee osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 9032-9040.
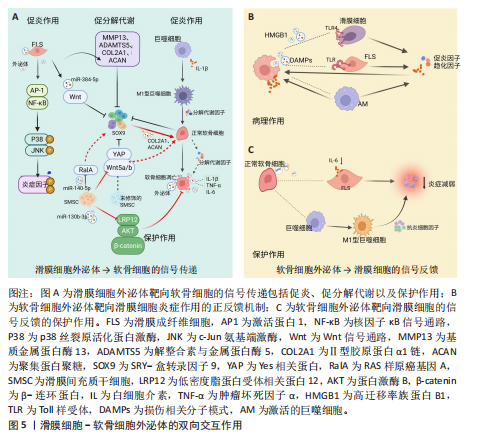
2.2 滑膜细胞外泌体在滑膜细胞-软骨细胞信号互作中的作用机制 在膝骨关节炎复杂的病理环境中,滑膜组织不仅是炎症的源头,也是调控软骨稳态的关键参与者,尤其是在滑膜细胞(包括滑膜成纤维细胞、巨噬细胞及滑膜间充质干细胞)释放的外泌体中,携带多种蛋白、miRNA等生物活性成分,可通过旁分泌或外泌体方式与软骨细胞发生双向交流[6-8]。不同来源和状态的滑膜细胞外泌体可在促炎、促降解和修复等方面对软骨产生显著影响,是膝骨关节炎局部微环境失衡或修复过程中的重要“信号递送者”。
2.2.1 滑膜细胞来源外泌体介导的促炎和促分解代谢信号 滑膜细胞,特别是滑膜成纤维细胞在膝骨关节炎病理进程中发挥着核心作用。在膝骨关节炎的炎症环境刺激(如白细胞介素1β或肿瘤坏死因子α)下,滑膜成纤维细胞分泌的外泌体富含促炎因子,如白细胞介素6、白细胞介素8及膜结合型肿瘤坏死因子α[9]。外泌体与软骨细胞表面的受体如Toll 样受体4和Toll 样受体7以及白细胞介素1结合,激活下游激活蛋白1与核因子κB信号通路,进而诱导基质金属蛋白酶13和血小板反应蛋白解整合素金属肽酶5等基质降解酶的表达,以及抑制软骨细胞中Ⅱ型胶原蛋白α1链和聚集蛋白聚糖的合成[10],这一过程增强了炎症反应并且能够直接靶向软骨细胞外基质并促进其降解[11]。而外泌体的靶向作用使基质金属蛋白酶3和基质金属蛋白酶13在局部聚集并达到较高浓度,再次增强降解作用[12-13]。此外,这些促
炎外泌体还能够激活其他致病细胞,如少突胶质细胞瘤和类风湿关节炎滑膜成纤维细胞,促使它们侵入细胞外基质,进一步加剧关节损伤的病理进程[14],同时滑膜M1型巨噬细胞来源外泌体也可诱导软骨细胞高表达基质金属蛋白酶13、解整合素与金属蛋白酶5,并下调SRY-盒转录因子9表达,从转录层面破坏软骨稳态调控网络[15]。滑膜间充质干细胞虽具有一定修复潜力,但未经修饰的滑膜间充质干细胞来源外泌体可通过激活Wnt5a/b信号通路,进一步激活Yes相关蛋白通路,同样抑制SRY-盒转录因子9表达,导致软骨修复效率下降,加重软骨降解趋势[16-18]。滑膜细胞外泌体通过干扰SRY-盒转录因子9等关键转录因子,打破合成与降解之间的动态平衡,是膝骨关节炎中软骨破坏的核心驱动机制之一,这种“炎症-降解”双向驱动机制不仅是膝骨关节炎早期软骨破坏的启动因素,也在疾病持续进展中形成持续性的正反馈环,成为膝骨关节炎治疗中急需干预的核心靶点(图5A)。
2.2.2 滑膜细胞外泌体介导的修复与保护作用 滑膜间充质干细胞来源外泌体在膝骨关节炎干预中表现出积极的保护作用。研究表明,滑膜间充质干细胞具有较高的软骨分化能力[19],能够通过多种机制促进软骨细胞增殖、迁移和基质合成,从而延缓膝骨关节炎的进展。外泌体中miR-140-5p可通过靶向抑制RAS样原癌基因A的表达,激活SRY-盒转录因子9信号通路,进而促进软骨细胞中Ⅱ型胶原蛋白α1链和聚集蛋白聚糖的合成,促进基质重建,降低膝骨关节炎的病理评分[17]。此外,滑膜间充质干细胞来源外泌体中的miR-130b-3p通过抑制低密度脂蛋白受体相关蛋白12/蛋白激酶B/β-连环蛋白轴发挥抗炎和抗凋亡作用,虽然在一定程度上可能会减少细胞外基质的分泌,但综合作用倾向于减轻和维持软骨细胞活性,表现出整体有利的组织保护趋势[20]。近期亦有研究发现来源于滑膜成纤维细胞的外泌体miR-146a通过抑制肿瘤坏死因子受体相关因子6表达,进而抑制核因子κB信号通路,减少软骨细胞凋亡,促进巨噬细胞M2极化,延缓软骨退化,为膝骨关节炎的治疗提供了一种新型治疗策略[21] (图5A)。
2.2.3 滑膜细胞外泌体介导的基因调控——mRNA与非编码RNA网络的联合调控机制 在膝骨关节炎的病理过程中,外泌体作为细胞间的天然通讯工具,发挥着至关重要的作用。外泌体能够通过携带并传递mRNA和非编码RNA等生物分子,调节靶细胞的代谢和炎症反应[22]。在膝骨关节炎的进程中,外泌体的这种作用不仅揭示了细胞间的复杂信号传递机制,还为理解膝骨关节炎的病理过程和寻找新的治疗靶点提供了新视角。
(1)mRNA的传递机制:外泌体携带的mRNA,尤其是环氧化酶2 mRNA,是骨关节炎中细胞功能调节的重要媒介[9]。滑膜细胞分泌的外泌体通过传递环氧化酶2 mRNA到软骨细胞,在细胞内部翻译为环氧化酶2蛋白,进而促进前列腺素E2的合成[23-24],引发炎症反应,加剧软骨基质降解以及增强疼痛感知,最终加重膝骨关节炎的病理进程。
(2) 非编码RNA网络的作用:除了mRNA,外泌体还携带大量非编码RNA[25-26],包括miRNA、长链非编码RNA和环状RNA,这些非编码RNA在膝骨关节炎的病理过程中同样至关重要[27-28]。研究发现,miRNA能够通过与靶向mRNA结合,抑制翻译和切割,进而调控细胞的各种生理过程[29-30]。例如,外泌体中富集的miR-1246可以通过抑制Wnt/β-连环蛋白信号通路中的糖原合酶激酶3β和轴抑制蛋白2,促进软骨细胞分泌促炎递质和基质金属蛋白酶[31];而miR-155-5p通过外泌体传递,能够抑制软骨细胞凋亡并通过靶向核心结合因子亚基α1来预防膝骨关节炎[32];在大鼠软骨细胞模型中,富含miR-126-3p的外泌体同样促进了软骨细胞的迁移和增殖,抑制细胞凋亡和促炎因子的表达[33];miR-150-3p可通过降低三重基序蛋白14的表达,抑制β干扰素的分泌,使炎症递质和合成代谢因子Ⅱ型胶原蛋白及聚集蛋白聚糖表达降低[34]。
长链非编码RNA在膝骨关节炎的病理过程中也发挥着重要的作用。例如,在膝骨关节炎患者血液单核细胞和血清外泌体中HOTAIR的表达显著增加,并通过与软骨细胞组蛋白甲基化酶结合,抑制聚集蛋白聚糖,促进软骨基质降解[35],加剧膝骨关节炎进程。研究进一步发现,HOTAIR能够通过吸附miR-1277-5p,间接增加小谷氨酸富集tetratricopeptide重复序列包含蛋白β(Small glutamine-rich tetratricopeptide repeat-containing protein beta,SGTB)的表达,进一步促进软骨细胞凋亡[35]。此外,HOTAIR还可以通过抑制miR-124的表达或通过miR-17-5p/岩藻糖基转移酶2/β-连环蛋白轴促进炎症反应和细胞凋亡[26];另一种长链非编码RNA PVT1,则通过调节miR-149和miR-488的表达,促进软骨细胞凋亡、基质降解及炎症反应[26]。
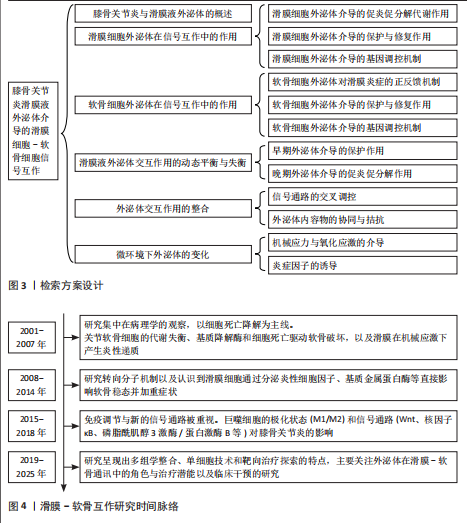
滑膜细胞来源外泌体通过mRNA与多类非编码RNA(miRNA、长链非编码RNA等)的协同传递,实质上构建了一个跨细胞的表观遗传调控网络,可以精准调控软骨细胞的转录活动、凋亡程序及炎症应答。该网络不仅具备多通路交叉性,还反映出膝骨关节炎进程中滑膜-软骨轴信号失衡的关键分子机制,提示外泌体RNA组分在膝骨关节炎中的作用远不止是信号载体,更可能是病理进展的“驱动节点”与潜在“主调控器”。因此,针对外泌体RNA谱系的解析与干预,有望打破传统炎症控制与软骨保护的局限,开启膝骨关节炎“信号调控型治疗”新策略(表1,2)[15,21,23,31-39]。
2.3 软骨细胞外泌体在滑膜细胞-软骨细胞信号互作中的机制 在膝骨关节炎的发病过程中,软骨细胞不仅作为关节的主要结构单元参与损伤,还通过软骨细胞分泌的外泌体与滑膜细胞进行复杂的信号传递。软骨细胞外泌体既可通过促炎和促分解代谢途径加剧软骨破坏,也可通过保护机制减轻炎症和促进软骨修复,体现出在膝骨关节炎中的双重性作用。
2.3.1 软骨细胞来源外泌体对滑膜细胞炎症的正反馈机制 在炎症或损伤状态下,软骨细胞会释放含有多种生物活性分子的外泌体,这些外泌体通过多条信号通路与滑膜细胞互动,加剧关节的炎症反应。软骨细胞外泌体中携带的损伤相关分子模式(如纤连蛋白和Ⅱ型胶原蛋白片段)以及高迁移率族蛋白B1能够警示滑膜细胞感知软骨损伤[40],同时可以激活滑膜成纤维细胞上的整合素和Toll样受体信号通路,进一步推动炎症级联反应,加剧膝骨关节炎的病理进展[41-42]。此外,软骨细胞碎片可激活巨噬细胞,刺激巨噬细胞分泌更多的促炎递质(如肿瘤坏死因子α、白细胞介素6、一氧化氮、β-干扰素),这些递质反过来又进一步诱导软骨细胞分泌大量的促炎代谢因子和基质降解酶,从而形成恶性循环[43-45],这种机制打破了传统观点中软骨细胞仅作为损伤靶点的认知,强调了软骨细胞在膝骨关节炎进程中通过外泌体信号主动参与滑膜病变,这种炎症因子诱导的基质降解酶释放与滑膜细胞活化之间形成的正反馈环路,为理解膝骨关节炎的慢性进展提供了新视角(图5B)。
2.3.2 软骨细胞外泌体介导的保护与软骨修复作用 尽管软骨细胞外泌体在膝骨关节炎中有促炎作用,但正常软骨细胞来源外泌体则表现出显著的保护效应。正常软骨细胞外泌体富含转化生长因子β3,这一细胞因子在软骨发育、修复及维持中发挥核心作用。转化生长因子β3通过抑制滑膜成纤维细胞中白细胞介素6的分泌,减轻滑膜炎症[46]。此外,正常软骨细胞外泌体通过促进巨噬细胞向M2(抗炎型)表型转化,分泌抗炎细胞因子(如白细胞介素10和转化生长因子β)进一步减少炎症反应,促进组织修复[47]。同时,软骨细胞外泌体中的肿瘤坏死因子刺激基因6蛋白通过与滑膜细胞表面受体结合,抑制核因子κB信号通路激活,下调促炎因子的表达,在一定程度上对抗滑膜细胞的持续炎症反应,负向调控周围组织的促炎性反馈,从而形成一种保护机制,延缓膝骨关节炎的进程[48](图5C)。
2.3.3 软骨细胞外泌体介导的基因调控——mRNA与非编码RNA网络的联合调控机制 软骨细胞通过外泌体将功能性核酸传递至滑膜细胞,调节滑膜细胞行为、细胞外基质代谢、免疫应答与血管生成等过程。除传统的mRNA翻译调控外,非编码RNA所组成的调控网络,包括miRNA、长链非编码RNA和环状RNA,在膝骨关节炎发病机制中的作用日益显现,并被认为是潜在的治疗干预靶点。
(1)mRNA的传递机制:在膝骨关节炎早期进展中,滑膜细胞的结构稳定性与细胞-基质黏附状态对炎症反应和组织重构具有关键影响。研究发现,软骨细胞释放的外泌体中富含软骨寡聚基质mRNA,它被滑膜细胞摄取后,可在局部翻译为软骨寡聚基质蛋白,调控滑膜细胞与细胞外基质的黏附和结构稳定性[49]。软骨寡聚基质蛋白不仅是软骨与滑膜功能偶联的
重要结构蛋白,还可以通过抑制促炎细胞因子的释放,缓解滑膜组织的炎性损伤[50-51]。因此,软骨寡聚基质蛋白对滑膜细胞信号转导与增殖活性调节为改善局部病理状态提供了新的思路。
(2)非编码RNA网络的作用:非编码RNA在膝骨关节炎的发病机制中扮演着重要角色,通过精细调控基因表达,影响软骨细胞的代谢、凋亡、炎症反应以及滑膜细胞的病理行为[25-26]。近年来,研究发现外泌体中的非编码RNA,包括miRNA、长链非编码RNA和环状RNA,能够通过细胞间通讯网络调节膝骨关节炎的进展[27-28]。
miRNA在膝骨关节炎的炎症反应、细胞凋亡、血管翳形成和基质代谢失衡中发挥多重作用[29]。软骨细胞外泌体中的miR-449a-5p可抑制自噬相关基因4B的表达,阻断巨噬细胞的自噬清除过程,并通过活化炎性小体增强白细胞介素1β的成熟与释放[52-53],加剧滑膜炎与软骨破坏,推动膝骨关节炎的进展,因此膝骨关节炎软骨细胞来源外泌体可通过miR-449a-5p/自噬相关基因4B/自噬途径增强软骨细胞凋亡,抑制细胞增殖,并促进巨噬细胞中成熟白细胞介素1β的产生[12-13,18]。miR-140-5p和miR-214-3p可靶向血管内皮生长因子A和酪氨酸激酶受体B,抑制磷脂酰肌醇3激酶/蛋白激酶B等信号通路,抑制滑膜血管新生、调节成纤维细胞迁移,减缓“血管翳”形成,从而发挥抗炎和抗血管生成的保护作用[54-57]。miR-140-5p还可通过靶向调控滑膜成纤维细胞中的岩藻糖基转移酶1基因,抑制细胞增殖、迁移、凋亡,促进原代软骨细胞增殖。岩藻糖基转移酶1是一种糖基化合成酶,在介导炎症细胞黏附和软骨细胞增殖中发挥重要作用[58]。软骨细胞中miR-27b通过靶向抑制基质金属蛋白酶13的表达,调节软骨细胞的代谢平衡[58-60]。miR-92a-3p通过靶向磷酸酶和张力蛋白同源物激活磷脂酰肌醇3激酶/蛋白激酶B信号通路,抑制滑膜细胞的炎症反应和过度增殖,减轻滑膜炎症和软骨破坏[61]。
长链非编码RNA通过竞争性结合miRNA,形成内源竞争RNA调控轴,在膝骨关节炎炎症与代谢调节中发挥枢纽作用。研究显示,长链非编码RNA MALAT1通过miR-150-5p/蛋白激酶B3轴促进软骨细胞增殖、抑制凋亡和基质降解[62]。有研究发现miR-124-3p可以抑制MALAT1与转录因子KLF5的结合并抑制CXCL11的转录,从而抑制膝骨关节炎软骨细胞焦亡和软骨损伤[63]。长链非编码RNA PVT1通过吸附miR-93-5p解除对高迁移率族蛋白B1的抑制,激活Toll 样受体4/核因子κB信号通路,增强促炎细胞因子表达,加重滑膜炎和软骨细胞凋亡[14],PVT1还可能通过影响miR-149、miR-488等miRNA进一步促进细胞外基质降解,提示PVT1在膝骨关节炎进展中的多通路放大效应[26]。
环状RNA因结构稳定性和miRNA的“海绵”功能,近年来被认为是膝骨关节炎中的新型调控因子。hsa_circ_0005567可作为miR-492的“分子海绵”,解除对细胞因子信号抑制因子2的抑制,促进巨噬细胞向抗炎型M2极化,减少炎症并减缓软骨凋亡[64];另一路径通过miR-495/自噬相关蛋白14轴激活软骨细胞自噬,保护关节结构。而环状RNA BRWD1在白细胞介素1β刺激下显著上调,通过调控miR-1277/肿瘤坏死因子受体相关因子6轴,激活核因子κB炎症通路,抑制自噬,诱导滑膜细胞过度增殖并加剧炎症反应,构成膝骨关节炎病程中重要的炎症正反馈环[14,65]。
这些研究揭示了软骨细胞可以通过外泌体精准释放非编码RNA,参与滑膜细胞-软骨细胞之间的炎症调控过程,体现了膝骨关节炎微环境中细胞-分子-信号通路三位一体的网络调控格局。特别值得强调的是,这些非编码RNA在多个病理环节中呈现出“一分子多靶点、一通路多环节”的特点,为膝骨关节炎的干预提供了组合式、多通道的靶点选择(表3,4)[14,16,22,38,52,54-55,57-58,60-62,66-67]。
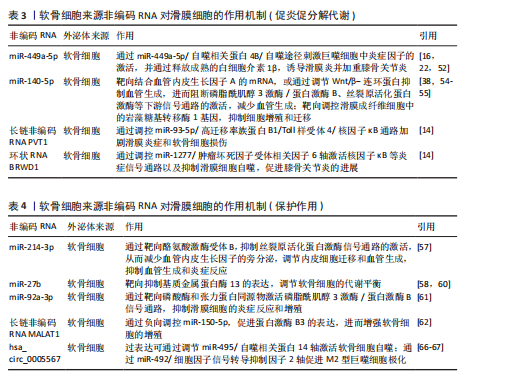
2.4 滑膜液外泌体交互作用的动态平衡与失衡 在膝骨关节炎的病理进程中,滑膜液外泌体扮演着至关重要的角色,滑膜液外泌体动态平衡与失衡直接影响疾病的进展。外泌体的生物学特性及其携带的生物活性分子在不同供体细胞类型和疾病分期下有着显著差异,因而在膝骨关节炎早期和晚期发挥不同的作用。
2.4.1 早期膝骨关节炎进程中以保护性外泌体为主导 在早期膝骨关节炎中,滑膜存在较为显著的炎症,而滑膜细胞和间充质干细胞来源外泌体通过携带抗炎因子、抗氧化酶以及促进软骨修复的生物活性分子,在减轻软骨损伤、缓解炎症方面起着重要的作用。滑膜间充质干细胞来源外泌体携带高水平的C-X-C模式趋化因子配体8、白细胞介素6、C-C模式趋化因子配体2以及抗炎因子(肿瘤坏死因子刺激基因6蛋白)和抗氧化酶(超氧化物歧化酶2)等[61],这些分子能够有效减轻滑膜细胞和软骨细胞的炎症反应及氧化应激,保护软骨结构[68-70]。另一项研究显示,在滑膜间充质干细胞的软骨形成过程中,WNT5A的表达在软骨分化早期阶段(第3-14天)保持稳定,这可能有助于软骨形成;然而在晚期阶段(第14-21天),WNT5A的表达迅速增加,可能会促进基质金属蛋白酶13的表达,导致聚集素、Ⅱ型胶原蛋白α1链、Ⅸ型胶原蛋白α1链和软骨寡聚基质蛋白表达减少[61]。因此,早期膝骨关节炎的滑膜液外泌体通过调节特定分子能够改善软骨修复并减缓病情进展。然而,随着疾病进展,保护性作用逐渐减弱,软骨细胞的代谢能力降低,最终导致表型丧失(图6)。
2.4.2 晚期膝骨关节炎进程中以促炎促分解代谢外泌体占优势 在膝骨关节炎的晚期,外泌体的表达模式发生显著改变,外泌体中趋化因子水平更高,且促炎促分解代谢外泌体逐渐占据主导地位,加剧软骨损伤。GAO等[71]证明来自终末期膝骨关节炎患者的外泌体具有更高水平的趋化因子,这些外泌体能够募集炎症细胞并通过外泌体膜表面传递,直接结合软骨细胞受体,激活核因子κB等炎症信号通路,促进基质降解酶的表达,最终加速软骨基质降解。此外,在晚期膝骨关节炎中,滑膜成纤维细胞过度增殖并分化,导致细胞外基质合成代谢受损,促进滑膜纤维化[72]。滑膜成纤维细胞来源外泌体中长链非编码RNA PCGEM1水平随着膝骨关节炎的进展而逐渐增加[73]。PCGEM1通过miR-142-5p/ Runt相关转录因2轴加速白细胞介素1β诱导的软骨细胞凋亡和软骨基质降
解[74]。因此,晚期膝骨关节炎中的外泌体呈现出促炎促分解代谢的特征,导致关节内环境的进一步恶化(图6)。
综上,早期膝骨关节炎的外泌体主要起到抗炎作用,并为软骨修复和关节保护提供了有力支持。然而,随着疾病的进展,外泌体的保护作用逐渐减弱,促炎和促分解代谢外泌体逐渐占据主导地位,导致软骨基质降解加速。因此,研究外泌体在膝骨关节炎中的动态平衡与失衡机制,对于探索早期干预和治疗策略具有重要意义。
2.5 外泌体交互作用的整合机制 在膝骨关节炎的病理进展中,滑膜细胞与软骨细胞之间的外泌体介导交流构成了一个高度动态且复杂的信号调控网络。外泌体的交互作用不仅涉及细胞间信号通路的交叉调控、内容物之间的协同与拮抗作用,还受到微环境因素等多个维度的影响,表现出显著的异质性。
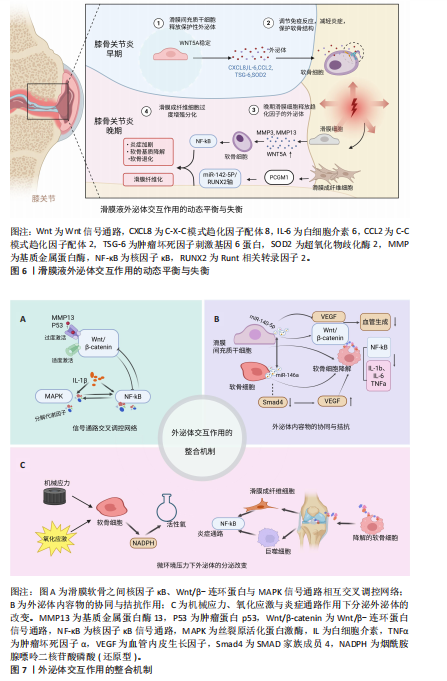
2.5.1 滑膜软骨细胞之间的信号通路交叉调控网络 在膝骨关节炎的病理过程中,多个炎症和代谢信号通路共同构成了一个复杂而精细的调控网络,其中尤以核因子κB、Wnt/β-连环蛋白与丝裂原活化蛋白激酶三大通路为核心,这3条信号轴之间存在的交互作用,不仅决定了局部炎症反应的强度,也决定了软骨细胞的命运选择,包括细胞增殖、分化与凋亡等[75]。滑膜细胞外泌体可以显著激活软骨细胞中的核因子κB信号通路[76],进而抑制Wnt/β-连环蛋白通路,阻碍软骨细胞的修复和再生;而Wnt/β-连环蛋白通路的适度激活可通过抑制核因子κB通路,减轻炎症反应,促进软骨细胞的存活和功能维持[77-78],但Wnt/β-连环蛋白信号通路的过度激活反而会诱导软骨细胞退变,增强基质金属蛋白酶13和P53表达,促进膝骨关节炎的进展[75,79-80]。丝裂原活化蛋白激酶通路(包括细胞外信号调节激酶、丝裂原活化蛋白激酶、c-Jun氨基端激酶)则能进一步激活核因子κB通路,加剧炎症反应,而核因子κB通路激活也可通过上调炎症因子的表达,进一步激活丝裂原活化蛋白激酶通路,构成了一个相互放大的炎症回路[75,81]。
这一系统揭示了以上3条通路并非单向递进,而是通过外泌体之间的反馈性调节实现交错共振(图7A)。
更重要的是,滑膜细胞外泌体和软骨细胞外泌体正是这些信号通路交叉调控的中介桥梁。它们通过携带特定的功能性分子,影响上述信号通路的活性水平。
2.5.2 外泌体内容物的协同与拮抗机制 在膝骨关节炎这一多因素驱动、慢性进展的疾病模型中,外泌体内容物之间的功能耦合显得尤为关键。研究者不仅需要识别关键miRNA的靶点作用,更应深入解析它在细胞交叉调控中的协同与对抗模式,以揭示调控网络的动态本质。滑膜间充质干细胞来源外泌体miR-140-5p与软骨细胞来源外泌体miR-146a作为当前研究中的代表性分子,展现出在炎症控制与软骨代谢调节中的双向功能调控特征,成为外泌体协同与拮抗机制研究的理想范式[58,82]。
(1)外泌体内容物的协同作用:在膝骨关节炎的慢性炎症背景下,滑膜间充质干细胞外泌体miR-140-5p与软骨细胞外泌体miR-146a均对调控炎症通路具有显著效果。研究发现,miR-140-5p可通过靶向下调Toll样受体4 mRNA的表达水平,抑制核因子κB通路激活,显著减少促炎因子的表达[38,83]。与此同时,miR-146a通过作用于多个软骨降解相关基因(如解整合素与金属蛋白酶、岩藻糖基转移酶1和Runt相关转录因子2),降低基质降解酶的表达,缓解软骨细胞的炎症损伤[84-88]。令人关注的是,当miR-140-5p与miR-146a联合表达时,膝骨关节炎软骨细胞中的核因子κB信号通路磷酸化水平与炎症因子表达均达到最显著的抑制程度,抗炎与保护效应优于单独miRNA处理[38]。
从机制角度看,两种miRNA协同靶向Toll 样受体4/核因子κB通路,构建了一个多点调控、互为补充的炎症抑制网络,这种协同机制不仅提示联合干预策略的潜力,也为外泌体多
miRNA疗法提供了理论支持。
(2)外泌体内容物的拮抗作用: miR-140-5p与miR-146a之间的关系并非始终协同。在某些病理背景下,miR-146a可能产生拮抗性影响。具体而言,miR-146a可靶向Smad4的3’-非翻译区域,抑制Smad4表达,进而引发血管内皮生长因子水平升高,促进血管生成与软骨降解,加剧膝骨关节炎进展与疼痛[20]。此外,miR-146a还可能抑制某些细胞外基质相关蛋白表达,加剧软骨基质破坏[58]。相对地,miR-140-5p在多项研究中被证实可通过直接靶向血管内皮生长因子A mRNA,或调节Wnt/β-连环蛋白信号,抑制血管新生、维持软骨无血管特性[54-55];除此之外,miR-140-5p通过激活Yes相关蛋白信号,促进软骨细胞增殖和迁移的同时不影响细胞外基质合成,还可以通过miR-140-5p/RAS样原癌基因A/SRY-盒转录因子9轴调节基质生成,降低OARSI评分,有效延缓膝骨关节炎进展并改善组织结构[14]。从功能角度看,miR-146a在某些条件下具有双面性:一方面抗炎,另一方面促进血管生成和基质退化;而miR-140-5p则表现出更为稳定和保护性的生物学特征。因此,二者在特定微环境下可能由协同转向功能性拮抗。
(3)软骨代谢方向的动态平衡:一个值得强调的现象是,miR-140-5p与miR-146a在软骨细胞中的相对表达水平可能决定代谢倾向。当miR-140-5p占优势时,软骨细胞维持较强的合成代谢状态,SRY-盒转录因子9活性高,细胞外基质合成活跃,Wnt信号抑制适度,炎症反应减弱,修复能力增强,整体代谢倾向于软骨修复。当miR-146a上调失控时,Smad4/血管内皮生长因子轴被激活,血管生成增加,软骨细胞趋向凋亡,软骨降解加剧[25,88]。因此,这种miRNA调控轴之间的相对优势构成了“代谢开关”,一定程度上决定了软骨细胞的命运,这为膝骨关节炎治疗提供了一个重要的分子干预切入点——通过调节特定miRNA的表达比值,重新塑造软骨细胞的命运。
miR-140-5p与miR-146a在膝骨关节炎病理状态中的协同与拮抗作用,揭示了外泌体内容物之间并非孤立运作,而是参与到一个高度复杂、具备反馈调控与环境依赖特征的分子网络中。这类miRNA之间的比例关系与表达模式,构成了膝骨关节炎软骨代谢状态的决定因素,提示应将关注点从“单因子靶向”转向“多因子网络协调”,并充分考虑生物学行为的动态性、可塑性与可干预性(图7B)。
2.5.3 微环境压力下外泌体的分泌改变 在外泌体介导的膝骨关节炎病理进程中,组织微环境作为一个动态调节因子,决定着外泌体的产生、内容物组成以及功能效应的方向性。外泌体并非静态实体,而是在多重环境应激下产生表型转变,外泌体功能也随微环境变化而改变,这种动态变化虽参与到膝骨关节炎炎症与组织破坏环节,但也提供了潜在的干预窗口和诊断靶点。
(1)机械应力与氧化应激介导的功能转变:在关节活动过程中,机械应力本是维持软骨稳态的重要生理刺激,然而在过度载荷或异常冲击条件下,软骨细胞面临强烈的应力刺激,逐步走向程序性死亡(包括凋亡与焦亡),损伤程度与应力强度呈正相关[89]。研究表明,为了应对这一机械负荷,软骨细胞会通过NOX系统过度生成活性氧,继而导致线粒体损伤与代谢紊乱,影响外泌体的生物合成途径及其miRNA/蛋白的选择性包装。活性氧不仅调控细胞的代谢命运,也直接改变外泌体的功能属性,使它从支持修复转向诱导退化[90]。这种机械应力诱导下的氧化应激,改变了外泌体的质量与功能,提示“力学-代谢-分泌”三联调控模式是膝骨关节炎中不可忽视的机制模块。
(2)炎症因子诱导的促炎外泌体的释放:传统观点将膝骨关节炎视为一种“机械磨损性疾病”,但现代研究越来越多地支持膝骨关节炎为慢性低度炎症驱动的退行性疾病[14]。在软骨降解过程中,大量细胞外基质降解片作为损伤相关分子模式被滑膜巨噬细胞与成纤维样滑膜细胞识别,激活下游炎症通路[91],这一过程中释放出的炎症因子与趋化因子可诱导滑膜组织分泌促炎型外泌体,这种炎症驱动的外泌体,加剧了软骨细胞表型失稳与基质降解,并通过正反馈增强滑膜炎症,为膝骨关节炎的持续进展提供“炎性信息桥梁”。
综上,一方面,膝骨关节炎微环境中的机械刺激、氧化应激与慢性炎症等因素深刻塑造了外泌体的分泌行为和内容物的构成,反映了外泌体来源细胞的应激状态,也决定了细胞的功能方向;另一方面,这种“环境-外泌体-靶细胞”轴构成了一个动态化的通路,使外泌体成为炎症持续与软骨恶化的稳定载体,说明外泌体本身也是微环境的反映者与塑造者(图7C)。

| [1] SANCHEZ-LOPEZ E, CORAS R, TORRES A, et al. Synovial inflammation in osteoarthritis progression. Nat Rev Rheumatol. 2022;18(5):258-275. [2] GUILAK F, NIMS RJ, DICKS A, et al. Osteoarthritis as a disease of the cartilage pericellular matrix. Matrix Biol. 2018;71-72:40-50. [3] NI Z, ZHOU S, LI S, et al. Exosomes: roles and therapeutic potential in osteoarthritis. Bone Res. 2020;8:25. [4] HEADLAND SE, JONES HR, NORLING LV, et al. Neutrophil-derived microvesicles enter cartilage and protect the joint in inflammatory arthritis. Sci Transl Med. 2015;7(315):315ra190. [5] OHTSUKI T, SATO I, TAKASHITA R, et al. Distribution and Incorporation of Extracellular Vesicles into Chondrocytes and Synoviocytes. Int J Mol Sci. 2024;25(22):11942. [6] RAIMONDO F, MOROSI L, CHINELLO C, et al. Advances in membranous vesicle and exosome proteomics improving biological understanding and biomarker discovery. Proteomics. 2011; 11(4):709-720. [7] CHEN Y, XUE K, ZHANG X, et al. Exosomes derived from mature chondrocytes facilitate subcutaneous stable ectopic chondrogenesis of cartilage progenitor cells. Stem Cell Res Ther. 2018;9(1):318. [8] ASGHAR S, LITHERLAND GJ, COLE JJ, et al. Small extracellular vesicles derived from synovial fibroblasts contain distinct miRNA profiles and contribute to chondrocyte damage in osteoarthritis. Arthritis Res Ther. 2024;26(1):167. [9] FU H, HU D, ZHANG L, et al. Role of extracellular vesicles in rheumatoid arthritis. Mol Immunol. 2018;93:125-132. [10] KATO T, MIYAKI S, ISHITOBI H, et al. Exosomes from IL-1β stimulated synovial fibroblasts induce osteoarthritic changes in articular chondrocytes. Arthritis Res Ther. 2014;16(4):R163. [11] HAN D, FANG Y, TAN X, et al. The emerging role of fibroblast-like synoviocytes-mediated synovitis in osteoarthritis: An update. J Cell Mol Med. 2020;24(17):9518-9532. [12] ZENG N, YAN ZP, CHEN XY, et al. Infrapatellar Fat Pad and Knee Osteoarthritis. Aging Dis. 2020; 11(5):1317-1328. [13] ASGHAR S, LITHERLAND GJ, LOCKHART JC, et al. Exosomes in intercellular communication and implications for osteoarthritis. Rheumatology (Oxford). 2020;59(1):57-68. [14] WU Y, LI J, ZENG Y, et al. Exosomes rewire the cartilage microenvironment in osteoarthritis: from intercellular communication to therapeutic strategies. Int J Oral Sci. 2022;14(1):40. [15] SI W, WEI H, CHEN W, et al. Exosomal microRNA-363 mediates the destructive effect of M1 macrophages on chondrocytes by repressing G3BP2. Exp Cell Res. 2024;442(2):114276. [16] NG CY, CHAI JY, FOO JB, et al. Potential of Exosomes as Cell-Free Therapy in Articular Cartilage Regeneration: A Review. Int J Nanomedicine. 2021;16:6749-6781. [17] TAO SC, YUAN T, ZHANG YL, et al. Exosomes derived from miR-140-5p-overexpressing human synovial mesenchymal stem cells enhance cartilage tissue regeneration and prevent osteoarthritis of the knee in a rat model. Theranostics. 2017;7(1):180-195. [18] ZHU Y, WANG Y, ZHAO B, et al. Comparison of exosomes secreted by induced pluripotent stem cell-derived mesenchymal stem cells and synovial membrane-derived mesenchymal stem cells for the treatment of osteoarthritis. Stem Cell Res Ther. 2017;8(1):64. [19] KURTH T, HEDBOM E, SHINTANI N, et al. Chondrogenic potential of human synovial mesenchymal stem cells in alginate. Osteoarthritis Cartilage. 2007;15(10):1178-1189. [20] ZENG Z, DAI Y, DENG S, et al. Synovial mesenchymal stem cell-derived extracellular vesicles alleviate chondrocyte damage during osteoarthritis through microRNA-130b-3p-mediated inhibition of the LRP12/AKT/β-catenin axis. Immunopharmacol Immunotoxicol. 2022; 44(2):247-260. [21] WANG H, ZHANG Y, ZHANG C, et al. Exosomes derived from miR-146a-overexpressing fibroblast-like synoviocytes in cartilage degradation and macrophage M1 polarization: a novel protective agent for osteoarthritis?. Front Immunol. 2024; 15:1361606. [22] FAN Y, LI Z, HE Y. Exosomes in the Pathogenesis, Progression, and Treatment of Osteoarthritis. Bioengineering (Basel). 2022;9(3):99. [23] SCHIOPPO T, UBIALI T, INGEGNOLI F, et al. The role of extracellular vesicles in rheumatoid arthritis: a systematic review. Clin Rheumatol. 2021;40(9):3481-3497. [24] FU Y, LEI J, ZHUANG Y, et al. Overexpression of HMGB1 A-box reduced IL-1β-induced MMP expression and the production of inflammatory mediators in human chondrocytes. Exp Cell Res. 2016;349(1):184-190. [25] KOLHE R, HUNTER M, LIU S, et al. Gender-specific differential expression of exosomal miRNA in synovial fluid of patients with osteoarthritis. Sci Rep. 2017;7(1):2029. [26] XIE F, LIU YL, CHEN XY, et al. Role of MicroRNA, LncRNA, and Exosomes in the Progression of Osteoarthritis: A Review of Recent Literature. Orthop Surg. 2020;12(3):708-716. [27] LIN Z, RODRIGUEZ NE, ZHAO J, et al. Selective enrichment of microRNAs in extracellular matrix vesicles produced by growth plate chondrocytes. Bone. 2016;88:47-55. [28] MITTON E, GOHR CM, MCNALLY MT, et al. Articular cartilage vesicles contain RNA. Biochem Biophys Res Commun. 2009;388(3):533-538. [29] BARTEL DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136(2):215-233. [30] PENG Y, CROCE CM. The role of MicroRNAs in human cancer. Signal Transduct Target Ther. 2016;1:15004. [31] PENG S, YAN Y, LI R, et al. Extracellular vesicles from M1-polarized macrophages promote inflammation in the temporomandibular joint via miR-1246 activation of the Wnt/β-catenin pathway. Ann N Y Acad Sci. 2021;1503(1):48-59. [32] WANG Z, YAN K, GE G, et al. Exosomes derived from miR-155-5p-overexpressing synovial mesenchymal stem cells prevent osteoarthritis via enhancing proliferation and migration, attenuating apoptosis, and modulating extracellular matrix secretion in chondrocytes. Cell Biol Toxicol. 2021; 37(1):85-96. [33] ZHOU Y, MING J, LI Y, et al. Exosomes derived from miR-126-3p-overexpressing synovial fibroblasts suppress chondrocyte inflammation and cartilage degradation in a rat model of osteoarthritis. Cell Death Discov. 2021;7(1):37. [34] WANG H, SHU J, ZHANG C, et al. Extracellular Vesicle-Mediated miR-150-3p Delivery in Joint Homeostasis: A Potential Treatment for Osteoarthritis. Cells. 2022;11(17):2766. [35] SONG J, KIM D, HAN J, et al. PBMC and exosome-derived Hotair is a critical regulator and potent marker for rheumatoid arthritis. Clin Exp Med. 2015;15(1):121-126. [36] KIM IK, KIM SH, CHOI SM, et al. Extracellular Vesicles as Drug Delivery Vehicles for Rheumatoid Arthritis. Curr Stem Cell Res Ther. 2016;11(4): 329-342. [37] KONTELES V, PAPATHANASIOU I, TZETIS M, et al. Synovial Fibroblast Extracellular Vesicles Induce Inflammation via Delivering miR-21-5p in Osteoarthritis. Cells. 2025;14(7):519. [38] PAPATHANASIOU I, BALIS C, TRACHANA V, et al. The synergistic function of miR-140-5p and miR-146a on TLR4-mediated cytokine secretion in osteoarthritic chondrocytes. Biochem Biophys Res Commun. 2020;522(3):783-791. [39] LAI C, LIAO B, PENG S, et al. Synovial fibroblast-miR-214-3p-derived exosomes inhibit inflammation and degeneration of cartilage tissues of osteoarthritis rats. Mol Cell Biochem. 2023;478(3):637-649. [40] MOUW JK, OU G, WEAVER VM. Extracellular matrix assembly: a multiscale deconstruction. Nat Rev Mol Cell Biol. 2014;15(12):771-785. [41] AULIN C, LASSACHER T, PALMBLAD K, et al. Early stage blockade of the alarmin HMGB1 reduces cartilage destruction in experimental OA. Osteoarthritis Cartilage. 2020;28(5):698-707. [42] PALUMBO A, ATZENI F, MURDACA G, et al. The Role of Alarmins in Osteoarthritis Pathogenesis: HMGB1, S100B and IL-33. Int J Mol Sci. 2023; 24(15):12143. [43] PENG Z, SUN H, BUNPETCH V, et al. The regulation of cartilage extracellular matrix homeostasis in joint cartilage degeneration and regeneration. Biomaterials. 2021;268:120555. [44] KAPOOR M, MARTEL-PELLETIER J, LAJEUNESSE D, et al. Role of proinflammatory cytokines in the pathophysiology of osteoarthritis. Nat Rev Rheumatol. 2011;7(1):33-42. [45] HAMASAKI M, TERKAWI MA, ONODERA T, et al. A Novel Cartilage Fragments Stimulation Model Revealed that Macrophage Inflammatory Response Causes an Upregulation of Catabolic Factors of Chondrocytes In Vitro. Cartilage. 2021;12(3):354-361. [46] DU X, CAI L, XIE J, et al. The role of TGF-beta3 in cartilage development and osteoarthritis. Bone Res. 2023;11(1):2. [47] MUSTONEN AM, NIEMINEN P. Extracellular Vesicles and Their Potential Significance in the Pathogenesis and Treatment of Osteoarthritis. Pharmaceuticals (Basel). 2021;14(4):315. [48] JIANG L, ZHANG Y, LIU T, et al. Exosomes derived from TSG-6 modified mesenchymal stromal cells attenuate scar formation during wound healing. Biochimie. 2020;177:40-49. [49] BURKE J, KOLHE R, HUNTER M, et al. Stem Cell-Derived Exosomes: A Potential Alternative Therapeutic Agent in Orthopaedics. Stem Cells Int. 2016;2016:5802529. [50] LIU X, WANG L, MA C, et al. Exosomes derived from platelet-rich plasma present a novel potential in alleviating knee osteoarthritis by promoting proliferation and inhibiting apoptosis of chondrocyte via Wnt/β-catenin signaling pathway. J Orthop Surg Res. 2019; 14(1):470. [51] KANG X, ZHANG K, WANG Y, et al. Single-cell RNA sequencing analysis of human chondrocytes reveals cell-cell communication alterations mediated by interactive signaling pathways in osteoarthritis. Front Cell Dev Biol. 2023;11:1099287. [52] NI Z, KUANG L, CHEN H, et al. The exosome-like vesicles from osteoarthritic chondrocyte enhanced mature IL-1β production of macrophages and aggravated synovitis in osteoarthritis. Cell Death Dis. 2019;10(7):522. [53] SKOTLAND T, HESSVIK NP, SANDVIG K, et al. Exosomal lipid composition and the role of ether lipids and phosphoinositides in exosome biology. J Lipid Res. 2019;60(1):9-18. [54] LU Y, QIN T, LI J, et al. MicroRNA-140-5p inhibits invasion and angiogenesis through targeting VEGF-A in breast cancer. Cancer Gene Ther. 2017; 24(9):386-392. [55] SUN Z, LIU B, LIU ZH, et al. Notochordal-Cell-Derived Exosomes Induced by Compressive Load Inhibit Angiogenesis via the miR-140-5p/Wnt/β-Catenin Axis. Mol Ther Nucleic Acids. 2020; 22:1092-1106. [56] MAPP PI, WALSH DA. Mechanisms and targets of angiogenesis and nerve growth in osteoarthritis. Nat Rev Rheumatol. 2012;8(7):390-398. [57] XIAO P, ZHU X, SUN J, et al. Cartilage tissue miR-214-3p regulates the TrkB/ShcB pathway paracrine VEGF to promote endothelial cell migration and angiogenesis. Bone. 2021;151:116034. [58] LI SH, WU QF. MicroRNAs target on cartilage extracellular matrix degradation of knee osteoarthritis. Eur Rev Med Pharmacol Sci. 2021; 25(3):1185-1197. [59] LI YH, TAVALLAEE G, TOKAR T, et al. Identification of synovial fluid microRNA signature in knee osteoarthritis: differentiating early- and late-stage knee osteoarthritis. Osteoarthritis Cartilage. 2016;24(9):1577-1586. [60] AKHTAR N, RASHEED Z, RAMAMURTHY S, et al. MicroRNA-27b regulates the expression of matrix metalloproteinase 13 in human osteoarthritis chondrocytes. Arthritis Rheum. 2010;62(5):1361-1371. [61] MAO G, ZHANG Z, HU S, et al. Exosomes derived from miR-92a-3p-overexpressing human mesenchymal stem cells enhance chondrogenesis and suppress cartilage degradation via targeting WNT5A. Stem Cell Res Ther. 2018;9(1):247. [62] ZHANG Y, WANG F, CHEN G, et al. LncRNA MALAT1 promotes osteoarthritis by modulating miR-150-5p/AKT3 axis. Cell Biosci. 2019;9:54. [63] ROZI R, ZHOU Y, RONG K, et al. miR-124-3p sabotages lncRNA MALAT1 stability to repress chondrocyte pyroptosis and relieve cartilage injury in osteoarthritis. J Orthop Surg Res. 2022; 17(1):453. [64] ZHANG J, CHENG F, RONG G, et al. Circular RNA hsa_circ_0005567 overexpression promotes M2 type macrophage polarization through miR-492/SOCS2 axis to inhibit osteoarthritis progression. Bioengineered. 2021;12(1):8920-8930. [65] GUO Z, WANG H, ZHAO F, et al. Exosomal circ-BRWD1 contributes to osteoarthritis development through the modulation of miR-1277/TRAF6 axis. Arthritis Res Ther. 2021;23(1):159. [66] ZHANG J, CHENG F, RONG G, et al. Hsa_circ_0005567 Activates Autophagy and Suppresses IL-1β-Induced Chondrocyte Apoptosis by Regulating miR-495. Front Mol Biosci. 2020;7:216. [67] DE ANDRÉS MC, IMAGAWA K, HASHIMOTO K, et al. Suppressors of cytokine signalling (SOCS) are reduced in osteoarthritis. Biochem Biophys Res Commun. 2011;407(1):54-59. [68] GÓMEZ-ARISTIZÁBAL A, SHARMA A, BAKOOSHLI MA, et al. Stage-specific differences in secretory profile of mesenchymal stromal cells (MSCs) subjected to early- vs late-stage OA synovial fluid. Osteoarthritis Cartilage. 2017;25(5):737-741. [69] LIAN C, WANG X, QIU X, et al. Collagen type II suppresses articular chondrocyte hypertrophy and osteoarthritis progression by promoting integrin β1-SMAD1 interaction. Bone Res. 2019;7:8. [70] PIÑEIRO-RAMIL M, GÓMEZ-SEOANE I, RODRÍGUEZ-CENDAL AI, et al. Mesenchymal stromal cells-derived extracellular vesicles in cartilage regeneration: potential and limitations. Stem Cell Res Ther. 2025;16(1):11. [71] GAO K, ZHU W, LI H, et al. Association between cytokines and exosomes in synovial fluid of individuals with knee osteoarthritis. Mod Rheumatol. 2020;30(4):758-764. [72] CHEN B, SUN Y, XU G, et al. Role of crosstalk between synovial cells and chondrocytes in osteoarthritis (Review). Exp Ther Med. 2024; 27(5):201. [73] ZHAO Y, XU J. Synovial fluid-derived exosomal lncRNA PCGEM1 as biomarker for the different stages of osteoarthritis. Int Orthop. 2018;42(12): 2865-2872. [74] ZENG G, DENG G, XIAO S, et al. Fibroblast-like Synoviocytes-derived Exosomal PCGEM1 Accelerates IL-1β-induced Apoptosis and Cartilage Matrix Degradation by miR-142-5p/RUNX2 in Chondrocytes. Immunol Invest. 2022;51(5): 1284-1301. [75] XI Y, HUANG X, TAN G, et al. Protective effects of Erdosteine on interleukin-1β-stimulated inflammation via inhibiting the activation of MAPK, NF-κB, and Wnt/β-catenin signaling pathways in rat osteoarthritis. Eur J Pharmacol. 2020;873:172925. [76] CHEN P, ZENG L, WANG T, et al. The communication role of extracellular vesicles in the osteoarthritis microenvironment. Front Immunol. 2025;16:1549833. [77] MAO Z, WANG P, PAN Q, et al. Pravastatin alleviates interleukin 1β-induced cartilage degradation by restoring impaired autophagy associated with MAPK pathway inhibition. Int Immunopharmacol. 2018;64:308-318. [78] ZHONG G, LIANG R, YAO J, et al. Artemisinin Ameliorates Osteoarthritis by Inhibiting the Wnt/β-Catenin Signaling Pathway. Cell Physiol Biochem. 2018;51(6):2575-2590. [79] USAMI Y, GUNAWARDENA AT, IWAMOTO M, et al. Wnt signaling in cartilage development and diseases: lessons from animal studies. Lab Invest. 2016;96(2):186-196. [80] STAMPELLA A, MONTEAGUDO S, LORIES R. Wnt signaling as target for the treatment of osteoarthritis. Best Pract Res Clin Rheumatol. 2017;31(5):721-729. [81] HAO L, MA C, LI Z, et al. Effects of type II collagen hydrolysates on osteoarthritis through the NF-κB, Wnt/β-catenin and MAPK pathways. Food Funct. 2022;13(3):1192-1205. [82] WU C, HE Y, YAO Y, et al. Exosomes treating osteoarthritis: hope with challenge. Heliyon. 2023;9(1):e13152. [83] CHEN Y, ZENG Z, SHEN X, et al. MicroRNA-146a-5p Negatively Regulates Pro-Inflammatory Cytokine Secretion and Cell Activation in Lipopolysaccharide Stimulated Human Hepatic Stellate Cells through Inhibition of Toll-Like Receptor 4 Signaling Pathways. Int J Mol Sci. 2016;17(7):1076. [84] BURSAVICH MG, GILBERT AM, LOMBARDI S, et al. Synthesis and evaluation of aryl thioxothiazolidinone inhibitors of ADAMTS-5 (Aggrecanase-2). Bioorg Med Chem Lett. 2007; 17(5):1185-1188. [85] WANG Z, HU J, PAN Y, et al. miR-140-5p/miR-149 Affects Chondrocyte Proliferation, Apoptosis, and Autophagy by Targeting FUT1 in Osteoarthritis. Inflammation. 2018;41(3):959-971. [86] HESS J, PORTE D, MUNZ C, et al. AP-1 and Cbfa/runt physically interact and regulate parathyroid hormone-dependent MMP13 expression in osteoblasts through a new osteoblast-specific element 2/AP-1 composite element. J Biol Chem. 2001;276(23):20029-20038. [87] TAKAHASHI A, DE ANDRÉS MC, HASHIMOTO K, et al. DNA methylation of the RUNX2 P1 promoter mediates MMP13 transcription in chondrocytes. Sci Rep. 2017;7(1):7771. [88] SKRINER K, ADOLPH K, JUNGBLUT PR, et al. Association of citrullinated proteins with synovial exosomes. Arthritis Rheum. 2006;54(12):3809-3814. [89] EWERS BJ, DVORACEK-DRIKSNA D, ORTH MW, et al. The extent of matrix damage and chondrocyte death in mechanically traumatized articular cartilage explants depends on rate of loading. J Orthop Res. 2001;19(5):779-784. [90] KOIKE M, NOJIRI H, OZAWA Y, et al. Mechanical overloading causes mitochondrial superoxide and SOD2 imbalance in chondrocytes resulting in cartilage degeneration. Sci Rep. 2015;5:11722. [91] JIA S, YANG Y, BAI Y, et al. Mechanical Stimulation Protects Against Chondrocyte Pyroptosis Through Irisin-Induced Suppression of PI3K/Akt/NF-κB Signal Pathway in Osteoarthritis. Front Cell Dev Biol. 2022;10:797855. [92] JIA S, YANG Y, LIU J, et al. PPARγ controls ESCRT-dependent fibroblast-like synoviocyte exosome biogenesis and alleviates chondrocyte osteoarthritis mediated by exosomal ANXA1. J Orthop Translat. 2025;53:187-205. [93] LONGFEI H, WENYUAN H, WEIHUA F, et al. Exosomes in cartilage microenvironment regulation and cartilage repair. Front Cell Dev Biol. 2025;13:1460416. |
| [1] | 宋浦蓁, 马贺宾, 陈宏广, 章亚东. 骨髓间充质干细胞外泌体联合转化生长因子β1对巨噬细胞的作用[J]. 中国组织工程研究, 2026, 30(7): 1616-1623. |
| [2] | 何家乐, 黄 茜, 董鸿斐, 陈 朗, 钟方宇, 李先慧. 脱细胞真皮基质联合脂肪干细胞外泌体促进烧伤创面愈合[J]. 中国组织工程研究, 2026, 30(7): 1699-1710. |
| [3] | 夏林枫, 王 露, 龙乾发, 唐荣武, 罗浩东, 汤 轶, 钟 俊, 刘 阳. 人脐带间充质干细胞来源外泌体减轻脓毒症脑病小鼠血脑屏障损伤[J]. 中国组织工程研究, 2026, 30(7): 1711-1719. |
| [4] | 陈钰璘, 何莹莹, 胡 凯, 陈枝凡, 聂 莎, 蒙衍慧, 李闰珍, 张小朵, 李宇稀, 唐耀平. 瓜蒌类外泌体囊泡防治动脉粥样硬化的作用及机制[J]. 中国组织工程研究, 2026, 30(7): 1768-1781. |
| [5] | 韩 腾, 马 洪, 杨若仪, 罗 祎, 李 超. 口腔鳞状细胞癌细胞来源外泌体递送血管生成素2参与肿瘤血管生成[J]. 中国组织工程研究, 2026, 30(7): 1755-1767. |
| [6] | 黄嘉雯, 潘之怡, 薛文君, 廉源沛, 徐建达. 植物源性囊泡与恶性肿瘤治疗:跨物种交流并调节宿主细胞反应[J]. 中国组织工程研究, 2026, 30(7): 1828-1838. |
| [7] | 王白燕, 杨 树, 王弋鸣, 吴梦晴, 肖 瑀, 郭梓璇, 张博艺, 冯书营. 外泌体递送CRISPR/Cas系统在靶细胞内可实现基因编辑[J]. 中国组织工程研究, 2026, 30(7): 1839-1849. |
| [8] | 王振泽, 刘奋德, 张 瑞, 李武军. 间充质干细胞治疗下肢动脉硬化闭塞症:系统评价和Meta分析[J]. 中国组织工程研究, 2026, 30(7): 1869-1876. |
| [9] | 李 豪, 陶红成, 曾 平, 刘金富, 丁 强, 牛驰程, 黄 凯, 康宏誉. 丝裂原活化蛋白激酶信号通路调控骨关节炎的发生发展:指导中药靶点治疗[J]. 中国组织工程研究, 2026, 30(6): 1476-1485. |
| [10] | 包卓玛, 侯孜明, 江 露, 李玮怡, 张宗星, 刘道忠, 袁 林. 枫杨总黄酮调控类风湿关节炎成纤维样滑膜细胞增殖、迁移及凋亡的作用及机制[J]. 中国组织工程研究, 2026, 30(4): 816-823. |
| [11] | 杨 肖, 白月辉, 赵甜甜, 王东昊, 赵 琛, 袁 硕. 颞下颌关节骨关节炎软骨退变:机制及再生的挑战[J]. 中国组织工程研究, 2026, 30(4): 926-935. |
| [12] | 张益瑞, 顾 叶, 钱正韬, 吴泽睿, 谢 恒, 唐奕涵, 顾赢楚, 方 涛, 王秋霏, 彭育沁, 耿德春, 徐耀增. 机械应力调控骨关节炎的分子机制与治疗靶点[J]. 中国组织工程研究, 2026, 30(34): 9048-9055. |
| [13] | 武瑞骐, 董盼锋, 张洪瑞, 吕永斌, 彭清林, 卓映宏, 陈跃平. 不同物理因子治疗膝骨关节炎:有效性和安全性的网状Meta分析[J]. 中国组织工程研究, 2026, 30(34): 9056-9066. |
| [14] | 田 宇, 郭 英, 伊尔夏提江·艾尼瓦尔, 艾合买提江·热甫海提, 麦尔达娜·买买提热依木. 具核梭杆菌调控小鼠结肠息肉形成的分子机制[J]. 中国组织工程研究, 2026, 30(29): 7603-7611. |
| [15] | 雍 侨, 孙 鑫, 汪国友, 张 磊, 沈骅睿, 刘 欢, 关钛元. 少阳生骨方抑制氧化应激延缓膝骨关节炎大鼠软骨衰老[J]. 中国组织工程研究, 2026, 30(28): 7251-7259. |
滑膜液外泌体作为细胞间通讯的关键载体,在膝骨关节炎病理环境中扮演着重要角色。滑膜液外泌体由滑膜腔内的多种细胞(如滑膜细胞、软骨细胞、免疫细胞等)分泌,富含蛋白质、脂质、核酸(如microRNA)等信息分子,具有高度稳定性、组织靶向性和生物活性[3-4]。越来越多的证据表明,滑膜液外泌体通过介导炎症调控、microRNA转运、细胞外基质代谢等途径,深度参与滑膜细胞与软骨细胞之间的双向信号互作,对关节微环境稳态产生决定性影响[5]。鉴于滑膜液外泌体在滑膜-软骨信号互作中的核心枢纽地位,是探究膝骨关节炎早期病理机制和发掘新型诊疗靶点的关键研究对象。
基于上述背景,此综述旨在系统总结滑膜液外泌体介导的滑膜细胞-软骨细胞信号互作在膝骨关节炎发生发展中的作用及分子机制。深入解析这一过程,不仅有助于阐明膝骨关节炎的病理本质,更将为开发基于外泌体的早期诊断标志物和靶向治疗策略提供重要的理论依据。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1 资料来源
1.1.1 检索人及检索时间 第一作者在2025年6月进行检索。
1.1.2 检索文献时限 2001年1月至2025年6月。
1.1.3 检索数据库 PubMed数据库。
1.1.4 检索词 英文检索词为“Exosomes,Synovial cells,Chondrocytes,Osteoarthritis”。
1.1.5 检索文献类型 原始研究论文、综述文章、临床试验、方法学论文。
1.1.6 检索策略 PubMed数据库检索策略见图1。
1.2 入组标准
1.2.1 纳入标准 ①研究内容:明确在骨关节炎环境下,滑膜液来源外泌体在滑膜细胞与软骨细胞之间的信号传递;②研究类型:基础研究、原始实验研究、综述、荟萃分析;③文献语言:英文文献;④时间范围:2001年1月至2025年6月。
1.2.2 排除标准 ①其他来源外泌体或细胞外囊泡(例如来源于血浆、胚胎干细胞等);②同一研究在不同期刊重复发表,仅保留最早或最完整的版本;③研究数据不完整、无法获取全文。
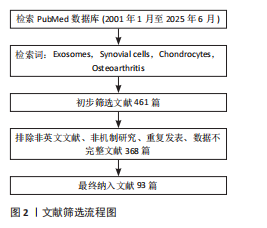
1.3 资料整合 共检索到461篇相关文献,排除368篇,实际纳入93篇,均为英文文献。文献检索流程见图2,检索方案设计见图3。
Conclusions and prospects
3.1 外泌体是滑膜-软骨信号互作的关键递质与双向调控者 外泌体作为细胞间通讯的核心载体,在膝骨关节炎滑膜细胞与软骨细胞的双向信号传递中扮演着不可或缺的角色。外泌体携带的丰富生物活性分子,深刻影响着膝骨关节炎的病理进程。滑膜细胞(尤其是炎症状态下)来源的外泌体富含促炎因子和基质降解酶,它们激活软骨细胞内的炎症信号通路,抑制合成代谢,促进分解代谢,直接加速软骨基质降解,并进一步放大滑膜炎症;软骨细胞在损伤应激下释放的外泌体携带损伤相关分子模式如高迁移率族蛋白B1、纤连蛋白/Ⅱ型胶原片段等,这些损伤相关分子模式通过结合滑膜细胞表面的模式识别受体,激活炎症信号通路,加剧滑膜炎;而特定来源(如滑膜间充质干细胞)的外泌体则发挥保护效应,通过靶向抑制分解代谢基因促进软骨细胞增殖与基质合成;同时正常软骨细胞来源外泌体也包含保护性分子(如肿瘤坏死因子刺激基因6蛋白、超氧化物歧化酶2)以抑制滑膜炎症和氧化应激,因此外泌体介导的信号网络呈现显著的双向调控潜力,“保护性”与“促损伤性”外泌体的动态平衡是决定膝骨关节炎进展的关键。深入解析外泌体介导的滑膜-软骨信号互作机制,是阐明膝骨关节炎病理本质和开发新型干预策略的核心环节。
3.2 未来将靶向外泌体信号互作,打破恶性循环 基于上述核心机制,未来研究应聚焦于干预促损伤性外泌体信号并增强保护性效应,为膝骨关节炎治疗提供新策略。如最新研究通过运动疗法促使滑膜成纤维细胞中过氧化物酶体增殖物激活受体γ上调,并通过转运必需内体分选复合物途径促使滑膜细胞外泌体中聚集膜联蛋白A1,膜联蛋白A1被软骨细胞摄取后,可以抑制细胞外调节蛋白激酶磷酸化,激活自噬、抑制凋亡的同时增强保护性信号[92]。除此之外,也可以通过靶向外泌体生成与递送,开发特异性靶向促炎/分解代谢外泌体或表面标志物的药物(如抗体、小分子抑制剂、适配体),阻断外泌体产生、释放或与靶细胞的结合;设计工程化纳米载体,模拟或改造外泌体,用于靶向递送保护性分子(如特定miRNA、肿瘤坏死因子刺激基因6蛋白、超氧化物歧化酶2)至滑膜或软骨细胞,精准调控细胞行为;深入研究并优化间充质干细胞来源外泌体的治疗潜力[93];深入解析信号网络与关键节点,系统研究核因子κB、Wnt/β-连环蛋白、丝裂原活化蛋白激酶等关键信号通路在外泌体介导的信号互作中的作用及动态变化等,或利用多组学技术(转录组、蛋白组、代谢组等)全面绘制膝骨关节炎不同阶段的外泌体信号图谱,为开发高选择性干预靶点提供理论依据。
总之,靶向滑膜液外泌体及其介导的滑膜-软骨信号互作,代表了膝骨关节炎诊疗领域极具前景的新方向。通过整合靶向递送、基因调控、细胞治疗优化和深度机制挖掘等多维度策略,未来研究有望开发出能够有效阻断恶性循环、促进关节稳态恢复的新型诊断工具和治疗手段,为膝骨关节炎患者带来福音。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
近年来,外泌体在膝骨关节炎研究中受到广泛关注,尤其是滑膜液来源外泌体因直接参与关节腔微环境调控而成为研究热点。已有证据表明,滑膜液外泌体能够携带 miRNA、长链非编码RNA 及蛋白质,在滑膜细胞与软骨细胞之间介导信号互作,调节炎症反应、细胞凋亡以及基质代谢,深刻影响膝骨关节炎的发生发展过程。目前,研究主要集中在外泌体所含非编码RNA的鉴定与功能验证。然而,不同病程阶段外泌体成分与功能是否存在动态差异,仍缺乏系统性证据。未来的发展趋势可能集中在三方面:外泌体的工程化修饰以增强治疗效应;外泌体组学与单细胞组学结合,解析关节腔微环境的精细调控;以及将滑膜液外泌体作为早期诊断标志物和干预手段的临床转化探索。本研究正位于该领域的前沿,有助于揭示膝骨关节炎进展的新机制,并为个体化治疗策略提供理论基础。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||