中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (34): 9024-9031.doi: 10.12307/2026.814
• 组织构建综述 tissue construction review • 上一篇 下一篇
运动调控miRNA在急性心肌梗死后心脏重构中的作用与机制
栾传凯,朱 磊
- 曲阜师范大学体育科学学院,山东省曲阜市 273100
-
收稿日期:2025-09-10修回日期:2025-12-12出版日期:2026-12-08发布日期:2026-04-14 -
通讯作者:朱磊,博士,教授,博士生导师,曲阜师范大学体育科学学院,山东省曲阜市 273100 -
作者简介:栾传凯,男,1997年生,山东省潍坊市人,汉族,曲阜师范大学在读博士,主要从事运动调控及运动原理等方面研究。 -
基金资助:山东省专业学位研究生教学案例库项目(SDYAL20103),项目负责人:朱磊
Role and mechanism of exercise-regulated miRNAs in cardiac remodeling after acute myocardial infarction
Luan Chuankai, Zhu Lei
- College of Physical Education and Sport Science, Qufu Normal University, Qufu 273100, Shandong Province, China
-
Received:2025-09-10Revised:2025-12-12Online:2026-12-08Published:2026-04-14 -
Contact:Zhu Lei, PhD, Professor, Doctoral supervisor, College of Physical Education and Sport Science, Qufu Normal University, Qufu 273100, Shandong Province, China -
About author:Luan Chuankai, PhD candidate, College of Physical Education and Sport Science, Qufu Normal University, Qufu 273100, Shandong Province, China -
Supported by:Shandong Provincial Professional Postgraduate Teaching Case Database Project, No. SDYAL20103 (to ZL)
摘要:
文题释义:
急性心肌梗死:是由于冠状动脉血流突然中断,造成心肌细胞缺血和坏死,从而引起心脏功能障碍的疾病。相关研究显示,心脏重构是急性心肌梗死患者后期恢复的主要表现,涉及心肌细胞凋亡、纤维化、血管生成障碍等病理进程。
miRNA:是长度约22个核苷酸的非编码RNA,通过与靶基因的3’非翻译区结合,调控基因的表达。miRNA在多种生理和病理进程中发挥重要作用,尤其在急性心肌梗死后心脏重构中表现出重要作用,如miR-1、miR-133a、miR-29家族等。相关研究发现,运动能够通过调节miRNA的表达,改善急性心肌梗死后的心脏功能。
背景:急性心肌梗死的病理表现主要包括心肌细胞凋亡、纤维化、血管生成障碍、钙稳态失衡及心脏肥大等。miRNA是调控基因表达的关键分子,在急性心肌梗死进展中展现出重要作用。运动能够通过调控miRNA表达来改善急性心肌梗死后心功能,但作用机制仍不完全确定。
目的:综述miRNA在急性心肌梗死病理表现中的作用,并探讨运动通过调控miRNA治疗急性心肌梗死的分子机制,为急性心肌梗死的精准治疗提供理论参考。
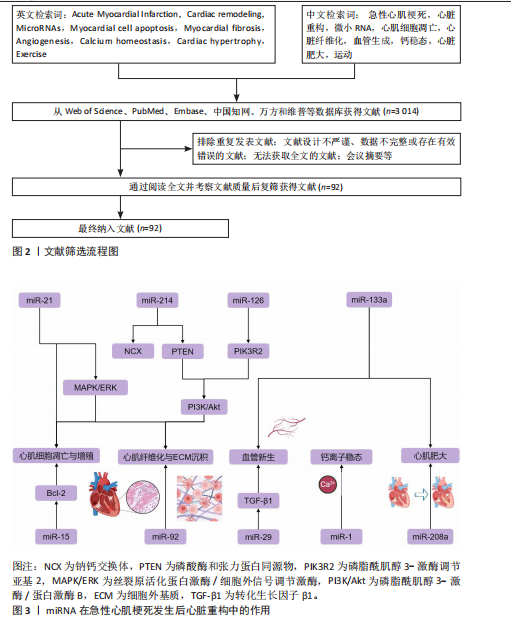
方法:检索Web of Science、PubMed、中国知网、万方等数据库,选取2000-2025年间关于miRNA与急性心肌梗死相关病理特征(如纤维化、血管生成、钙稳态、心脏肥大)以及运动调控机制的文献。中文检索词为“急性心肌梗死,心脏重构,微小RNA,心肌细胞凋亡,心脏纤维化,血管生成,钙稳态,心脏肥大,运动”;英文检索词为“Acute Myocardial Infarction,Cardiac remodeling,MicroRNAs,Myocardial cell apoptosis,Myocardial fibrosis,Angiogenesis,Calcium homeostasis,Cardiac hypertrophy,Exercise”,最终纳入92篇文献进行分析。
结果与结论:miRNA(miR-1、miR-133a、miR-21、miR-29家族)通过调控细胞凋亡、纤维化、血管生成和钙稳态等相关信号通路参与急性心肌梗死发病后的心脏重构。运动通过上调miR-126促进血管生成,上调miR-29抑制心肌纤维化,上调miR-214-3p恢复钙稳态以及上调miR-222促进心肌生理性肥大,从而改善急性心肌梗死发病后的心脏功能。运动与药物[如三氟甲氧基苯基-3-(1-丙酰哌啶-4-基)脲]或者营养物质(如维生素D3)联合应用能够调控miRNA的治疗效果。后期建议进一步研究miRNA的时空性差异及家族间差异,制定精准靶向干预方案;基于miRNA表达谱制定精准运动干预方案,结合人工智能进行疗效预测评估;探索将运动干预与基因编辑技术或者外泌体递送技术结合,以精确调控miRNA的表达,继而促进心脏修复和功能恢复。
https://orcid.org/0009-0002-6471-0418(栾传凯)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
栾传凯, 朱 磊. 运动调控miRNA在急性心肌梗死后心脏重构中的作用与机制[J]. 中国组织工程研究, 2026, 30(34): 9024-9031.
Luan Chuankai, Zhu Lei. Role and mechanism of exercise-regulated miRNAs in cardiac remodeling after acute myocardial infarction[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(34): 9024-9031.
急性心肌梗死的病理进程主要包括心肌细胞缺血坏死、炎症反应以及后期阶段的心脏重构,最终结果可能出现心力衰竭、心律失常等并发症[15]。急性心肌梗死前期的治疗通常包括溶栓、抗血小板治疗以及冠脉介入手术等,目的在于恢复血流、缓解心肌损伤同时预防心脏功能衰竭[16-17]。
2.2 miRNA在急性心肌梗死病理表现中的作用
2.2.1 miRNA的生物学功能以及作用机制 miRNA是一类长度为18-24个核苷酸的RNA小分子,通过与靶基因的3′非翻译区结合,抑制靶基因的翻译或促进靶基因的降解,表现出基因表达调控机制[5]。miRNA参与多种生理、病理进程如细胞增殖、分化、凋亡、免疫、代谢以及肿瘤的发生等[18]。研究发现,miRNA在急性心肌梗死中参与了心脏细胞功能调控、心脏重构进程[19-20]。
miRNA通过调控心肌细胞、成纤维细胞、内皮细胞以及免疫细胞,能够在急性心肌梗死后的修复进程中展现出重要作用[21]。此外,miRNA通过调控关键的信号通路(如转化生长因子β、磷脂酰肌醇3-激酶/蛋白激酶B、丝裂原活化蛋白激酶、Notch等)调控心脏重构进程[7,22-23]。因此,miRNA可作为潜在的治疗靶标,有望改善心脏功能、抑制不良重构。
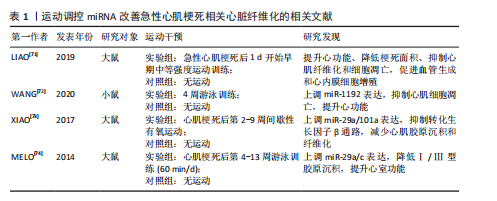
2.2.2 miRNA在急性心肌梗死后心脏重构中的作用 急性心肌梗死发生后,心脏发生许多组织病理学改变,主要包括心肌细胞死亡、心脏组织纤维化、血管生成失衡及心脏肥大。miRNA在急性心肌梗死发生后心脏重构中的作用见图3。
(1)心肌细胞凋亡以及增殖:心肌细胞凋亡是急性心肌梗死后心脏重构的核心进程之一。miRNA通过调控凋亡相关基因的表达来影响心肌细胞的存活以及死亡[24]。例如,miR-1以及miR-133a通过靶向Bcl-2家族蛋白,调控心肌细胞的凋亡进程[25]。miR-21通过靶向磷酸酶和张力蛋白同源物/蛋白激酶 B信号通路促进心肌细胞的存活以及增殖[26]。此外,部分miRNA (如miR-210)通过促进血管生成以及减少缺血区域细胞凋亡,促进心脏修复[27]。
(2)心脏纤维化以及细胞外基质沉积:心脏纤维化是急性心肌梗死发展为心脏重构的重要进程,是心肌成纤维细胞异常激活以及细胞外基质异常沉积造成的。miRNA通过调节转化生长因子β1信号通路在此过程中发挥了重要作用[28-31]。例如miR-1192通过抑制转化生长因子β1水平以及抑制心肌细胞凋亡从而减轻心肌纤维化[8,32]。
(3)血管生成:血管生成是急性心肌梗死后心脏修复的重要进程,通过输送氧气以及营养供应,加强心肌再生[33-34]。miR-126[35]、miR-15a[11]、miR-146a等miRNA通过靶向血管生成相关因子,调控血管新生[12]。miR-126通过磷脂酰肌醇3-激酶/蛋白激酶B通路促进内皮细胞增殖、构建血管网络[10,36]。
此外,运动通过调控miRNA表达,促进血管生成,改善心脏功能[36]。
(4)钙离子稳态:钙离子在心肌细胞的兴奋-收缩偶联中展现出核心作用,有助于保持细胞
的正常功能[37]。急性心肌梗死发生后,钙离子稳态破坏,导致钙超载及心肌细胞电生理异常[38]。miRNA在调控钙稳态中展现出重要作用,特别是在调控钠钙交换体以及肌浆网钙ATP酶等关键蛋白的表达方面[39]。例如,miR-214-3p通过调控钠钙交换体以及内质网钙ATP酶2a的表达,恢复钙离子稳态,缓解了钙超载带来的心脏损伤[40]。
(5)心脏肥大:心脏肥大是急性心肌梗死后心脏适应性重构的一部分,主要表现为心肌细胞增大以及心脏负荷的适应性提升[40-41]。miR-222通过靶向同源域相互作用蛋白激酶2等基因来抑制细胞凋亡并加强心肌修复,继而加强适应性肥大变化[42]。此外,miR-1以及miR-133等miRNA通过调控多种信号通路(如磷脂酰肌醇3-激酶/蛋白激酶B、丝裂原活化蛋白激酶等)影响心脏的肥大反应[6,43]。
miR-142-3p通过白细胞介素6激活丝裂原活化蛋白激酶/细胞外信号调节激酶信号通路,在心脏肥大调控中展现出重要作用[44]。
2.2.3 细胞线粒体自噬在心脏重构中的作用 急性心肌梗死后,心脏重构进程通常伴随心肌细胞死亡、细胞外基质的异常聚集以及细胞功能的改变[45]。研究显示,急性心肌梗死后心肌细胞氧化应激水平显著升高,线粒体受损会加剧活性氧生成,导致蛋白质变性、脂质过氧化和DNA损伤[46]。
线粒体自噬作为细胞自噬的关键形式之一,主要作用是去除受损线粒体、保持线粒体质量和细胞能量代谢的稳定[47-49]。研究显示,提高线粒体自噬能够减缓氧化应激水平,继而改善心脏功能[50]。miR-27a和miR-27b能够通过直接结合PTEN诱导的假定激酶1信使RNA的3′非翻译区,抑制其翻译能力,继而降低PTEN诱导的假定激酶1的表达[51]。
miR-34a-5p能够直接结合PTEN诱导的假定激酶1信使RNA的3′非翻译区和编码区,进而抑制PTEN诱导的假定激酶1的表达,影响PTEN诱导的假定激酶1/Parkin驱动的线粒体自噬进程[52]。miR-29家族主要通过抑制Parkin的转位来间接调控PTEN诱导的假定激酶1/Parkin驱动的线粒体自噬途径[53],在心脏重构中发挥重要作用。
2.2.4 炎症反应与心脏重构的关系 心肌损伤后,局部和全身免疫反应被激活,释放促炎因子如肿瘤坏死因子α和白细胞介素6等[54],这些促炎因子通过激活核因子κB和丝裂原活化蛋白激酶信号通路,促进心脏成纤维细胞增生及纤维化[55]。慢性炎症反应对心脏持续损伤和功能恶化发挥着推动作用[56]。因此,针对炎症反应的干预方法可能成为改善急性心肌梗死后心脏重构的潜在治疗策略。
研究显示,miR-21、miR-146a在心脏重构、纤维化、炎症和肥大等进程中发挥重要作用[57-58]。
miR-21通过靶向磷酸酶和张力蛋白同源物/磷脂酰肌醇3-激酶/蛋白激酶B信号通路抑制炎症反应,从而减缓心脏重构[58-59]。
2.2.5 能量代谢与心脏重构的关系 能量代谢失衡是急性心肌梗死后心脏重构的一个重要因素。心脏是高能量消耗的器官,正常的能量代谢对于保持心脏功能至关重要。在急性心肌梗死发生后,由于供氧不足,心肌细胞的能量代谢受到影响,造成ATP合成降低,细胞功能受损,最终促进心脏重构的发生[60-62]。因此,维持正常的心脏能量代谢对于防止急性心肌梗死后心脏重构至关重要。研究者正在探索通过提升心脏能量代谢来预防或治疗急性心肌梗死后心脏重构的策略。
miR-29和miR-133等miRNA通过调控与能量代谢相关的基因,提升心肌细胞的代谢功能,减缓心脏重构进程[63-64]。AKKAYA等[65]研究了运动对CD4+ T细胞线粒体生物合成和活性氧产生的影响,为运动对细胞代谢的调节机制提供了见解。此外,研究还发现有氧运动能够显著提高心脏能量代谢水平,促进ATP合成,改善心脏功能[66]。
2.3 运动调控miRNA改善急性心肌梗死的病理机制
2.3.1 运动调控miRNA改善急性心肌梗死相关心脏纤维化的机制 急性心肌梗死引起的持续性心肌缺血能够造成心肌细胞凋亡以及病理性心脏重构,核心病理表现为心肌成纤维细胞异常增殖及细胞外基质过度沉积[9,67],通常伴随心肌收缩能力降低以及心室顺应性损伤,作用机制与转化生长因子β1信号通路的异常激活紧密关联[68]。研究发现,转化生长因子β1通过糖原合成酶激酶3β/β-Catenin/转化生长因子β1信号级联反应有效增强心肌纤维化进程[69]。然而,该通路的上游调控机制以及与其他信号网络的互动作用仍未完全清楚,特别是miRNA在此进程中的自适应调控特性有待进一步解析。
研究证实,适当的运动能够通过多方面调控miRNA表达来改善急性心肌梗死发生后心脏纤维化[70]。运动能够显著上调miR-17-5p、miR-125b-5p表达水平,miR-125b-5p不仅能够抑制白细胞活化,还能够通过正向调控机制抑制白细胞迁移,继而显著改善心肌纤维化相关的炎症微环境[71],这说明运动可能通过miRNA驱动的免疫调控机制影响纤维化进程。然而,miRNA在心肌细胞以及免疫细胞间的特定传递机制与时空表达特性尚需要确定。
在纤维化直接调控方面,运动促进miR-1192表达上调具有双向保护作用,一方面通过抑制心肌细胞凋亡保持组织结构完整性,另一方面通过靶向降低转化生长因子β1蛋白表达水平抑制促纤维化信号传递[72]。研究显示,为期8周的系统性运动能够同期上调miR-29a 以及miR-101a 的表达,通过抑制转化生长因子β1/Smad2/3信号轴及下游胶原合成关键基因表达,显著抑制心肌胶原沉积[51]。此外,研究发现,游泳训练能够显著提升miR-29家族(miR-29a/b/c)的表达,miR-29表达水平与心肌组织胶原含量呈负相关[52],这说明miR-29可能是运动的重要作用分子。然而,各类的运动模式对miRNA表达谱的差异性影响及剂量关系尚有待进一步研究。
综上,运动通过调控miR-125b-5p/miR-1192/ miR-29a-101a形成多通路的心肌纤维化调控策略,但尚存在以下局限性:①miRNA调控网络的时间和空间动态改变规律不清楚;②运动强度和miRNA表达剂量关系有待挖掘;③miRNA参与驱动的纤维化和心肌能量代谢的相互作用机制有待研究。因此,下一步文献将重点聚焦在:①建立miRNA-转化生长因子 β信号轴自适应性调控模型;②发现结合miRNA表达谱的运动处方个体化方案定制;③探索运动联合抗纤维化药物治疗的前景。
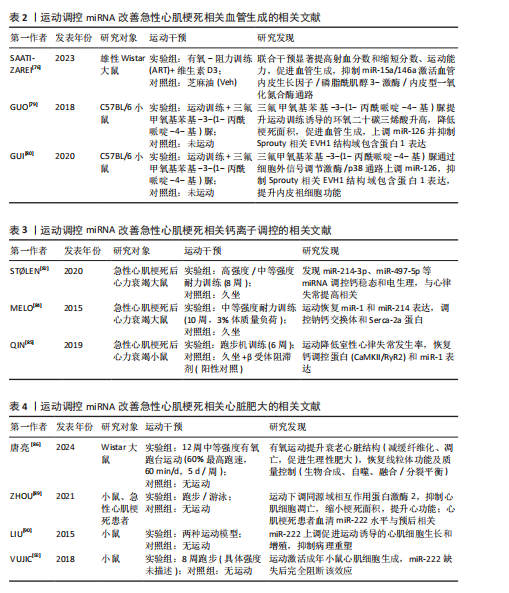
运动调控miRNA改善急性心肌梗死相关心脏纤维化的相关文献见表1[71-74]。
2.3.2 运动调控miRNA改善急性心肌梗死相关血管生成的机制 急性心肌梗死的核心病理表现是冠状动脉微循环系统的结构性破坏[75]。依据此,心肌组织的修复进程取决于有效激活血管生成反应[34]。血管内皮生长因子/磷脂酰肌醇3-激酶/内皮型一氧化氮合酶信号通路是调控急性心肌梗死后血管生成的关键节点[76]。当前研究已发现miR-15a、miR-126以及miR-146a通过靶向调控上述通路继而参与血管生成网络的动态平衡。
抗阻训练协同有氧运动能够有效加强急性心肌梗死后的心肌血管新生,表现为微血管密度提升以及心功能改善[76]。运动通过上调血管内皮生长因子/磷脂酰肌醇3-激酶/内皮型一氧化氮合酶通路的活性,抑制血管生成负调控因子miR-15a以及凋亡驱动因子miR-146a的表达,能够展现出加强血管生成的作用[76]。研究发现,运动以及维生素D3的协同使用通过抑制miR-15a以及miR-146a的表达,激活血管内皮生长因子/磷脂酰肌醇3-激酶/内皮型一氧化氮合酶通路来促进血管生成[54]。
miR-126在血管生成调控中展现出双向功能特性,一方面通过抑制血管内皮生长因子通路负调控因子Sprouty相关EVH1结构域包含蛋白1以及磷脂酰肌醇3-激酶调节亚基2促进内皮细胞增殖[77],另一方面通过激活磷脂酰肌醇3-激酶/蛋白激酶B通路调控血管网络形成[78]。此类双效调控机制使得miR-126变成急性心肌梗死发生后血管再生的重要调控枢纽。运动通过细胞外信号调节激酶/p38丝裂原活化蛋白激酶信号通路显著上调miR-126的表达,继而抑制靶蛋白Sprouty相关EVH1结构域包含蛋白1的功能,最终促进血管新生[79]。此外,三氟甲氧基苯基-3-(1-丙酰哌啶-4-基)脲是环氧二十碳三烯酸的强效抑制剂,能够通过增加运动驱动的环氧二十碳三烯酸累计进一步提升miR-126的表达水平[68]。GUI等[80]研究证实,三氟甲氧基苯基-3-(1-丙酰哌啶-4-基)脲联合运动干预通过细胞外信号调节激酶/p38通路调控 miR-126/Sprouty相关EVH1结构域包含蛋白1轴,形成“运动-三氟甲氧基苯基-3-(1-丙酰哌啶-4-基)脲-miR-126”的级联放大作用。
总之,运动靶向调控多种miRNA分子以及下游信号途径形成多维度血管生成调控网。然而,当前的研究还存在2个问题:其一是各类miRNA分子间的相互协同或拮抗作用;其二是运动的长期作用及其对心肌重构的影响还需要大样本队列研究验证。未来,有关运动的研究还需围绕:①miRNA分子调控相互作用网络的动态;②运动剂量及相关miRNA表达的剂量作用;③联合干预方案的临床转化应用前景,从而探索出明确解决此类重要科学问题,为制定精确化急性心肌梗死患者的运动康复干预策略提供依据。运动调控miRNA改善急性心肌梗死相关血管生成的相关文献见表2[76,79-80]。
2.3.3 运动调控miRNA改善急性心肌梗死相关钙离子调控的机制 钙离子是心肌细胞兴奋-收缩耦合的核心调控因子,钙离子稳态保持取决于钠离子跨膜转运、肌浆网钙库动态平衡以及兰尼碱受体以及心肌钠钙交换体的联合作
用[81]。急性心肌梗死引起的氧化应激以及能量
代谢失衡能够显著降低钙离子瞬变振幅以及胞浆游离钙浓度,造成钠钙交换体活性损伤及钙超载现象[81-83]。在此病理进程中,肌浆/内质网钙ATP酶2通过驱动钙离子转运回肌浆网,成为保持钙稳态的核心调控节点[83]。
研究显示,高强度间歇训练能够通过上调miR-214-3p的表达和抑制miR-497-5p及miR-31a-5p的异常增加而双向调控钠钙交换体/内质网钙ATP酶2a轴来逆转急性心肌梗死发作后Ca2+调控异常[82]。其中miR-214-3p通过靶向调控钠钙交换体和内质网钙ATP酶2a的表达在重整Ca2+稳态进程中发挥重要作用;miR-31a-5p下调亦可有效降低内质网钙ATP酶2a异常所致的心律失常发生率[82]。高强度间歇训练的调控效果呈现剂量效应,说明运动强度可能会通过动态调控miRNA表达谱来提高治疗效果。
MELO等[84]的研究进一步发现了运动的分子机制:跑台间歇训练通过恢复miR-214基础水平,显著下调内质网钙ATP酶2蛋白表达,同时促进miR-1驱动的钠钙交换体上调,最终结果是改善梗死心脏的钙离子稳态及左心室顺应性。该研究与β受体阻滞剂治疗的钙调控作用具备相似性,均表现为内质网钙ATP酶2a表达降低,然而,运动未引起miR-133a的显著改变[85],这说明运动可能通过非β受体依赖途径实现钙稳态调控,详细分子机制尚有待进一步研究。
综上,运动通过调节miR-1/miR-214-3p/miR-31a-5p/miR-497-5p网络重塑钠钙交换体/内质网钙ATP酶2a信号轴形成多重钙稳态调控系统。然而,目前此类研究的空白点在于:①miRNA调控网络存在时空动态性改变规律;②运动强度和miRNA表达的剂量-作用关联需要明确;③miRNA驱动钙调控和心肌纤维化的相互作用机制。进一步的研究应该聚焦在于:①建立miRNA-Ca2+调控网络的数学模型;②寻找依据miRNA表达谱的运动处方的个体化方案定制;③探讨运动联合钙通道阻滞剂的联合治疗潜能。解决好此类核心问题为急性心肌梗死后心脏康复提供新的分子靶点和理论依据。运动调控miRNA改善急性心肌梗死相关钙离子调控的相关文献见表3[82,84-85]。
2.3.4 运动调控miRNA改善急性心肌梗死相关心脏肥大的机制 心脏重构是急性心肌梗死后的适应性病理进程,核心特性是心肌细胞肥大以及间质纤维化的动态平衡,运动能够在该进程中发挥作用[86]。研究发现,miRNA通过响应运动调控心肌细胞增殖以及分化,驱动生理性心脏肥大改变,成为改善急性心肌梗死后心功能的重要治疗靶标[87]。miR-222通过靶向调控多条肥大相关信号通路,在保持心肌细胞活力以及提升心脏功能中展现出核心作用。
miR-222的双向调控机制表现为:一方面,通过抑制心肌细胞生长转录因子同源盒包含基因1通路抑制病理性肥大进程[43],另一方面,通过调控同源域相互作用蛋白激酶通路抑制细胞凋亡,继而加强适应性重构。
miR-222是当前已知极少数能同时响应运动并直接调控心脏肥大的miRNA,miR-222表达水平与运动强度呈显著正相关[88]。ZHOU等[89]通过基因敲除模型研究表明,抑制同源域相互作用蛋白激酶 2可显著缩小梗死面积并改善心功能,而抑制miR-222则会加重心功能障碍。机制研究发现,运动通过上调miR-222水平,抑制靶标基因同源域相互作用蛋白激酶2表达,继而减少细胞凋亡并加强心肌修复[90]。研究进一步证实,急性心肌梗死患者血清miR-222水平显著降低,这说明miR-222兼具生物标志物以及治疗靶标的双向作用[91]。
研究显示,游泳以及自主跑台训练能够通过上调miR-222表达来抑制靶标基因同源域相互作用蛋白激酶1、同源域相互作用蛋白激酶2以及同源盒包含基因1的转录,继而驱动心脏的生理性肥大[90]。此类适应性改变主要表现为肌球蛋白重链α/β比例上升以及病理性肥大标志物(如心房钠尿肽和B型脑钠肽)的信使RNA表达显著下调[90],说明miR-222在运动适应性调控中起到不可或缺的作用。VUJIC等[92]研究发现,8周自主跑台训练能够显著抑制同源域相互作用蛋白激酶1的表达,同时促进心肌细胞增殖,然而在miR-222抑制组,运动驱动的心肌生成作用被彻底遏制,这说明miR-222不仅调控肥大反应,还介入心肌细胞再生进程。
综上所述,运动通过miR-222/同源域相互作用蛋白激酶/同源盒包含基因1轴,构建多维心肌重构调控轴,然而目前的研究还是存在以下局限:①miR-222与其他肥大相关miRNA的联合作用机制尚不明确;②运动的剂量-效应关系缺乏临床验证;③miR-222
在心肌细胞代谢重编程方面的作用机制尚有待研究。未来研究应聚焦于:①构建miR-222调控网络的分子互作图谱;②开发基于miR-222表达的运动处方的个体化定制;③阐明miR-222激动剂及运动联合干预的协同作用。解决此类关键问题将有望为急性心肌梗死后心脏重构的精准治疗提供新的理论依据。运动调控miRNA改善急性心肌梗死相关心脏肥大的相关文献见表4[86,89,90,92]。
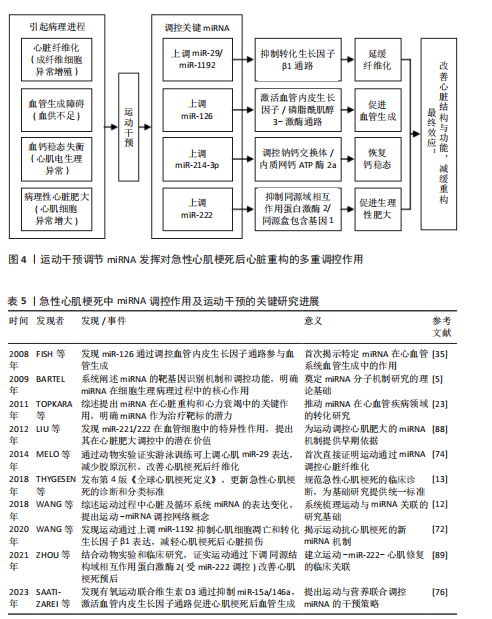
2.3.5 运动干预与miRNA在心脏重构中的协同作用 在急性心肌梗死后心脏重构进程中,运动通过调节miRNA的表达,显著影响多个病理机制,包括心脏纤维化、血管生成、钙稳态和心脏肥大。综合目前的研究表明,运动可通过多通路、多靶点的协同作用,改善心脏重构的治疗效果。
研究显示,运动通过调节miRNA(如miR-29、
miR-125b-5p和miR-1192等)有效抑制纤维化过程。miR-29家族在降低胶原沉积方面发挥重要作用[76]。此外,运动通过提高miR-125b-5p的表达抑制白细胞活化和迁移,继而缓解炎症反应,这对改善纤维化微环境具有积极作用[75]。
miRNA通过免疫调节和直接靶向转化生长因子β1通路的双重作用,使得运动成为治疗急性心肌梗死后心脏纤维化的重要策略[73]。血管生成是心脏重构中的另一个重要过程,运动能够通过调节miRNA(如miR-126、miR-15a、miR-146a等)促进血管新生。miR-126通过靶向血管内皮生长因子/磷脂酰肌醇3-激酶通路促进内皮细胞增殖,并通过磷脂酰肌醇3-激酶/蛋白激酶B通路促进血管网络形成[75]。此外,运动通过抑制miR-15a和miR-146a等负调控因子的表达,促进血管生成[76]。运动通过调节多种miRNA及其下游信号通路的协同作用,恢复急性心肌梗死后心脏的血供,从而促进心肌修复[77]。在急性心肌梗死引起的心脏重构过程中,钙稳态失衡会造成心肌细胞的电生理异常和心律失常。研究显示,运动通过调节miRNA(如miR-214-3p和miR-31a-5p)恢复心脏细胞内的钙稳态,特别是miR-214-3p通过调控钠钙交换体和肌浆网钙ATP酶的表达,缓解钙超载,提高心脏收缩功能[81-82]。心脏肥大是心脏重构的适应性反应,过度肥大可能造成心力衰竭。运动通过调节miRNA(如miR-222)在控制心脏肥大方面发挥重要作用。miR-222通过靶向多个信号通路(如同源域相互作用蛋白激酶 2和同源盒包含基因 1通路)抑制病理性肥大的发生[86]。运动通过上调miR-222的表达,抑制心肌细胞凋亡,促进心脏修复[87-88]。
综上所述,运动干预通过多种机制调节miRNA发挥对急性心肌梗死后心脏重构的多重调控作用。此外,运动干预通过多个信号通路的协同作用,展现出多维度的治疗潜力,见图4。然而,关于运动干预与miRNA之间的剂量效应、时空性改变以及长期效果等问题仍需进一步研究。因此,未来的研究应聚焦于运动干预与miRNA表达谱的个体化定制,结合更精确的运动方案,提供更加个性化的治疗策略。
| [1] 颜流霞,厚磊,蔡小宁,等.2015-2019年中国居民急性心肌梗死发病和死亡特征分析[J].中国循环杂志,2024,39(10):968-975. [2] 王帅,朱建华.microRNA在急性心肌梗死诊治中的研究进展[J].心脏杂志,2013,25(5):592-594+602. [3] ZHU W, SUN L, ZHAO P, et al. Macrophage migration inhibitory factor facilitates the therapeutic efficacy of mesenchymal stem cells derived exosomes in acute myocardial infarction through upregulating miR-133a-3p. J Nanobiotechnology. 2021;19(1):61. [4] 马丽娟,周祁娜,张健,等.miRNA-208a-3p过表达致慢性心衰大鼠心肌细胞线粒体钙超载和功能障碍的机制研究[J].新疆医科大学学报, 2024,47(6):791-797. [5] BARTEL DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136(2):215-233. [6] 唐宇.血浆miR-1、miR-133a、理想心血管健康与急性心肌梗死的相关性研究[D].呼和浩特:内蒙古医科大学,2020. [7] 袁媛,韩大鹏,朱永新.miR-21通过激活PI3K/Akt信号通路对缺氧诱导的大鼠心肌细胞活性的作用机制[J].重庆医学,2021,50(13): 2165-2170. [8] 何梓峰,吕祥威,赵位昆,等.微小RNA-29b-3p对大鼠心肌细胞焦亡和心房纤维化的影响及其机制研究[J].中华老年心脑血管病杂志, 2023,25(2):179-182. [9] 高逸凡,胡荣,张瀛月,等.急性心肌梗死患者的院内早期心脏康复新进展[J].中国介入心脏病学杂志,2023,31(7):541-546. [10] MA Y, LIU H, WANG Y, et al. Roles of physical exercise-induced MiR-126 in cardiovascular health of type 2 diabetes. Diabetol Metab Syndr. 2022;14(1):169. [11] SILVA FCD, IOP RDR, ANDRADE A, et al. Effects of Physical Exercise on the Expression of MicroRNAs: A Systematic Review. J Strength Cond Res. 2020; 34(1):270-280. [12] WANG L, LV Y, LI G, et al. MicroRNAs in heart and circulation during physical exercise. J Sport Health Sci. 2018;7(4):433-441. [13] THYGESEN K, ALPERT JS, JAFFE AS, et al. Fourth Universal Definition of Myocardial Infarction (2018). Circulation. 2018;138(20):e618-e651. [14] LIBBY P, RIDKER PM, HANSSON GK. Progress and challenges in translating the biology of atherosclerosis. Nature. 2011;473(7347):317-325. [15] FRANGOGIANNIS NG. The inflammatory response in myocardial injury, repair, and remodelling. Nat Rev Cardiol. 2014;11(5):255-265. [16] IBANEZ B, JAMES S, AGEWALL S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: The Task Force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European Society of Cardiology (ESC). Eur Heart J. 2018;39(2):119-177. [17] ROTH GA, JOHNSON C, ABAJOBIR A, et al. Global, Regional, and National Burden of Cardiovascular Diseases for 10 Causes, 1990 to 2015. J Am Coll Cardiol. 2017;70(1):1-25. [18] BARTEL DP. Metazoan MicroRNAs. Cell. 2018; 173(1):20-51. [19] ZHANG B, WANG Q, PAN X. MicroRNAs and their regulatory roles in animals and plants. J Cell Physiol. 2007;210(2):279-289. [20] THUM T, CONDORELLI G. Long noncoding RNAs and microRNAs in cardiovascular pathophysiology. Circ Res. 2015;116(4):751-762. [21] 侯谦,马雅楠,刘德敏.微小RNA靶基因调控的信号通路在急性心肌梗死演进过程中作用的研究进展[J].中国心血管杂志,2024,29(5):486-491. [22] 范亮亮,马立宁,彭元亮,等.PI3K/AKT信号通路与心力衰竭[J].生命科学研究,2015,19(1):85-90. [23] TOPKARA VK, MANN DL. Role of microRNAs in cardiac remodeling and heart failure. Cardiovasc Drugs Ther. 2011;25(2):171-182. [24] 余丽霞,王键.miRNA调控心肌梗死后心肌细胞凋亡的研究进展[J].心脏杂志,2019,31(2):227-231. [25] XIAO Y, ZHAO J, TUAZON JP, et al. MicroRNA-133a and Myocardial Infarction. Cell Transplant. 2019;28(7):831-838. [26] WEI J, FENG L, LI Z, et al. MicroRNA-21 activates hepatic stellate cells via PTEN/Akt signaling. Biomed Pharmacother. 2013;67(5):387-392. [27] PU Y, LI C, QI X, et al. Extracellular Vesicles from NMN Preconditioned Mesenchymal Stem Cells Ameliorated Myocardial Infarction via miR-210-3p Promoted Angiogenesis. Stem Cell Rev Rep. 2023;19(4):1051-1066. [28] 张诺琪,于国慧,王亚玲.基于TGF-β1/Smads信号通路的miRNA在心肌纤维化的研究进展[J].河北医科大学学报,2024,45(1):89-93. [29] 肖一佳,沈祥春,李杰平,等.心肌纤维化与TGF-β-Smad信号系统[J].中国新药杂志,2016, 25(2):182-186+192. [30] 陈昱竹,乔宇,刘娅红,等.心房颤动心肌纤维化的分子机制研究进展[J].中国循环杂志, 2024,39(12):1229-1233. [31] WANG Q, TAO Y, XIE H, et al. MicroRNA101 inhibits renal tubular epithelialtomesenchymal transition by targeting TGFβ1 type I receptor. Int J Mol Med. 2021;47(6):119. [32] CHOW KVC, O’LEARY C, PAXTON-HALL F, et al. Pembrolizumab-induced toxic epidermal necrolysis: case report. Oxf Med Case Reports. 2022;2022(3):omac025. [33] ZHANG J, GUO Y, BAI Y, et al. Application of biomedical materials in the diagnosis and treatment of myocardial infarction. J Nanobiotechnology. 2023;21(1):298. [34] WU X, REBOLL MR, KORF-KLINGEBIEL M, et al. Angiogenesis after acute myocardial infarction. Cardiovasc Res. 2021;117(5):1257-1273. [35] FISH JE, SANTORO MM, MORTON SU, et al. miR-126 regulates angiogenic signaling and vascular integrity. Dev Cell. 2008;15(2):272-284. [36] GOMEZ-CABRERA MC, DOMENECH E, VIÑA J. Moderate exercise is an antioxidant: upregulation of antioxidant genes by training. Free Radic Biol Med. 2008;44(2):126-131. [37] YANG L, LI RC, XIANG B, et al. Transcriptional regulation of intermolecular Ca2+ signaling in hibernating ground squirrel cardiomyocytes: The myocardin-junctophilin axis. Proc Natl Acad Sci U S A. 2021;118(14):e2025333118. [38] 王非多,李腾龙,唐克强.老年急性ST段抬高型心肌梗死血清高敏肌钙蛋白Ⅰ与心电图Ⅲ级缺血的相关性[J].中国老年学杂志,2019, 39(1):12-13. [39] HU T, LIU L. Effects of MiR-214-3p Regulation of SERCA2a Expression on Contractility of Cardiomyocytes in Heart Failure Model. Cell Mol Biol (Noisy-le-grand). 2022;68(4):208-216. [40] 刘冠楠,陈钢.运动性与病理性心脏肥大[J].沈阳体育学院学报,2014,33(2):90-96. [41] RYSÄ J, TOKOLA H, RUSKOAHO H. Mechanical stretch induced transcriptomic profiles in cardiac myocytes. Sci Rep. 2018;8(1):4733. [42] KESIDOU D, BEQQALI A, BAKER AH. The dual effects of miR-222 in cardiac hypertrophy: bridging pathological and physiological paradigms. Cardiovasc Res. 2024;120(3):217-219. [43] DENG J, ZHONG Q. Advanced research on the microRNA mechanism in heart failure. Int J Cardiol. 2016;220:61-64. [44] LIU YQ, XU YC, SHUAI ZW. Mir-142-3P regulates MAPK protein family by inhibiting 14-3-3η to enhance bone marrow mesenchymal stem cells osteogenesis. Sci Rep. 2023;13(1):22862. [45] MURTHA LA, SCHULIGA MJ, MABOTUWANA NS, et al. The Processes and Mechanisms of Cardiac and Pulmonary Fibrosis. Front Physiol. 2017;8:777. [46] DING WX, YIN XM. Mitophagy: mechanisms, pathophysiological roles, and analysis. Biol Chem. 2012;393(7):547-564. [47] AZEVEDO PS, POLEGATO BF, MINICUCCI MF, et al. Cardiac Remodeling: Concepts, Clinical Impact, Pathophysiological Mechanisms and Pharmacologic Treatment. Arq Bras Cardiol. 2016; 106(1):62-69. [48] ZHANG X, SHAO S, LI Q, et al. Roles of Autophagy, Mitophagy, and Mitochondria in Left Ventricular Remodeling after Myocardial Infarction. Rev Cardiovasc Med. 2025;26(3):28195. [49] SCHEFFER DDL, GARCIA AA, LEE L, et al. Mitochondrial Fusion, Fission, and Mitophagy in Cardiac Diseases: Challenges and Therapeutic Opportunities. Antioxid Redox Signal. 2022; 36(13-15):844-863. [50] AI L, DE FREITAS GERMANO J, HUANG C, et al. Enhanced Parkin-mediated mitophagy mitigates adverse left ventricular remodelling after myocardial infarction: role of PR-364. Eur Heart J. 2025;46(4):380-393. [51] KIM J, FIESEL FC, BELMONTE KC, et al. miR-27a and miR-27b regulate autophagic clearance of damaged mitochondria by targeting PTEN-induced putative kinase 1 (PINK1). Mol Neurodegener. 2016;11(1):55. [52] TAI Y, PU M, YUAN L, et al. miR-34a-5p regulates PINK1-mediated mitophagy via multiple modes. Life Sci. 2021;276:119415. [53] MARKHAM BN, RAMNARINE C, KIM S, et al. miRNA family miR-29 inhibits PINK1-PRKN dependent mitophagy via ATG9A. bioRxiv [Preprint]. 2024: 2024.01.17.576122. [54] DUAN D, FAN T, ZHANG L, et al. The Correlation Between Cardiac Oxidative Stress and Inflammatory Cytokine Response Following Myocardial Infarction. Clin Appl Thromb Hemost. 2023;29:10760296231211907. [55] HAN A, LU Y, ZHENG Q, et al. Qiliqiangxin Attenuates Cardiac Remodeling via Inhibition of TGF-β1/Smad3 and NF-κB Signaling Pathways in a Rat Model of Myocardial Infarction. Cell Physiol Biochem. 2018;45(5):1797-1806. [56] SONG Y, ZHANG C, ZHANG J, et al. An injectable silk sericin hydrogel promotes cardiac functional recovery after ischemic myocardial infarction. Acta Biomater. 2016;41:210-223. [57] HE F, GUAN W. The role of miR-21 as a biomarker and therapeutic target in cardiovascular disease. Clin Chim Acta. 2025;574:120304. [58] PALOMER X, CAPDEVILA-BUSQUETS E, BOTTERI G, et al. miR-146a targets Fos expression in human cardiac cells. Dis Model Mech. 2015;8(9):1081-1091. [59] WANG J, LIU Y, MA C, et al. Ameliorative Impact of Liraglutide on Chronic Intermittent Hypoxia-Induced Atrial Remodeling. J Immunol Res. 2022; 2022:8181474. [60] HU Q, WANG X, LEE J, et al. Profound bioenergetic abnormalities in peri-infarct myocardial regions. Am J Physiol Heart Circ Physiol. 2006;291(2): H648-657. [61] OMEROVIC E, BOLLANO E, BASETTI M, et al. Bioenergetic, functional and morphological consequences of postinfarct cardiac remodeling in the rat. J Mol Cell Cardiol. 1999;31(9):1685-1695. [62] OMEROVIC E, BOLLANO E, SOUSSI B, et al. Selective beta1-blockade attenuates post-infarct remodelling without improvement in myocardial energy metabolism and function in rats with heart failure. Eur J Heart Fail. 2003;5(6):725-732. [63] LI Q, LIN X, YANG X, et al. NFATc4 is negatively regulated in miR-133a-mediated cardiomyocyte hypertrophic repression. Am J Physiol Heart Circ Physiol. 2010;298(5):H1340-H1347. [64] FELICIANO RDS, MANCHINI MT, ATUM ALB, et al. Photobiomodulation therapy’s effects on cardiac fibrosis activation after experimental myocardial infarction. Lasers Surg Med. 2022;54(6):883-894. [65] AKKAYA B, ROESLER AS, MIOZZO P, et al. Increased Mitochondrial Biogenesis and Reactive Oxygen Species Production Accompany Prolonged CD4+ T Cell Activation. J Immunol. 2018;201(11):3294-3306. [66] BASSETT DR JR, HOWLEY ET. Limiting factors for maximum oxygen uptake and determinants of endurance performance. Med Sci Sports Exerc. 2000;32(1):70-84. [67] GIBB AA, LAZAROPOULOS MP, ELROD JW. Myofibroblasts and Fibrosis: Mitochondrial and Metabolic Control of Cellular Differentiation. Circ Res. 2020;127(3):427-447. [68] BIERNACKA A, DOBACZEWSKI M, FRANGOGIANNIS NG. TGF-β signaling in fibrosis. Growth Factors. 2011;29(5):196-202. [69] GUO Y, GUPTE M, UMBARKAR P, et al. Entanglement of GSK-3β, β-catenin and TGF-β1 signaling network to regulate myocardial fibrosis. J Mol Cell Cardiol. 2017;110:109-120. [70] 毋江波,鲍蒙蒙,方李,等.外泌体微小RNAs调控心肌线粒体稳态:运动改善心力衰竭的新视角[J].生理科学进展,2025,56(2):154-161. [71] LIAO Z, LI D, CHEN Y, et al. Early moderate exercise benefits myocardial infarction healing via improvement of inflammation and ventricular remodelling in rats. J Cell Mol Med. 2019;23(12):8328-8342. [72] WANG Y, TIAN MM, MI CJ, et al. Exercise protects the heart against myocardial infarction through upregulation of miR-1192. Biochem Biophys Res Commun. 2020;521(4):1061-1069. [73] XIAO L, HE H, MA L, et al. Effects of miR-29a and miR-101a Expression on Myocardial Interstitial Collagen Generation After Aerobic Exercise in Myocardial-infarcted Rats. Arch Med Res. 2017; 48(1):27-34. [74] MELO SF, FERNANDES T, BARAÚNA VG, et al. Expression of MicroRNA-29 and Collagen in Cardiac Muscle after Swimming Training in Myocardial-Infarcted Rats. Cell Physiol Biochem. 2014;33(3):657-669. [75] 王旭兰,张炜,李喆,等.冠状动脉缺血再灌注对循环微粒的影响[J].中国循证心血管医学杂志,2021,13(7):879-881. [76] SAATI-ZAREI A, DAMIRCHI A, TOUSI SMTR, et al. Myocardial angiogenesis induced by concurrent vitamin D supplementation and aerobic-resistance training is mediated by inhibiting miRNA-15a, and miRNA-146a and upregulating VEGF/PI3K/eNOS signaling pathway. Pflugers Arch. 2023;475(4): 541-555. [77] HE S, SINGH D, YUSEFI H, et al. Stable Cavitation-Mediated Delivery of miR-126 to Endothelial Cells. Pharmaceutics. 2022;14(12):2656. [78] ARDERIU G, PEÑA E, CIVIT-URGELL A, et al. Endothelium-Released Microvesicles Transport miR-126 That Induces Proangiogenic Reprogramming in Monocytes. Front Immunol. 2022;13:836662. [79] GUO Y, LUO F, ZHANG X, et al. TPPU enhanced exercise-induced epoxyeicosatrienoic acid concentrations to exert cardioprotection in mice after myocardial infarction. J Cell Mol Med. 2018; 22(3):1489-1500. [80] GUI Y, CHEN J, HU J, et al. Soluble epoxide hydrolase inhibitors improve angiogenic function of endothelial progenitor cells via ERK/p38-mediated miR-126 upregulation in myocardial infarction mice after exercise. Exp Cell Res. 2020; 397(2):112360. [81] BERS DM, DESPA S, BOSSUYT J. Regulation of Ca2+ and Na+ in normal and failing cardiac myocytes. Ann N Y Acad Sci. 2006;1080:165-177. [82] STØLEN TO, HØYDAL MA, AHMED MS, et al. Exercise training reveals micro-RNAs associated with improved cardiac function and electrophysiology in rats with heart failure after myocardial infarction. J Mol Cell Cardiol. 2020;148: 106-119. [83] GONNOT F, BOULOGNE L, BRUN C, et al. SERCA2 phosphorylation at serine 663 is a key regulator of Ca2+ homeostasis in heart diseases. Nat Commun. 2023;14(1):3346. [84] MELO SF, BARAUNA VG, NEVES VJ, et al. Exercise training restores the cardiac microRNA-1 and -214 levels regulating Ca2+ handling after myocardial infarction. BMC Cardiovasc Disord. 2015;15:166. [85] QIN R, MURAKOSHI N, XU D, et al. Exercise training reduces ventricular arrhythmias through restoring calcium handling and sympathetic tone in myocardial infarction mice. Physiol Rep. 2019;7(4):e13972. [86] 唐亮,王合霞,王庆博,等.有氧运动调控线粒体质量控制系统逆转衰老大鼠心脏的病理性重塑[J].中国组织工程研究,2024,28(16):2534-2541. [87] WANG J, YANG X. The function of miRNA in cardiac hypertrophy. Cell Mol Life Sci. 2012;69(21):3561-3570. [88] LIU X, CHENG Y, YANG J, et al. Cell-specific effects of miR-221/222 in vessels: molecular mechanism and therapeutic application. J Mol Cell Cardiol. 2012;52(1):245-255. [89] ZHOU Q, DENG J, YAO J, et al. Exercise downregulates HIPK2 and HIPK2 inhibition protects against myocardial infarction. EBioMedicine. 2021;74:103713. [90] LIU X, XIAO J, ZHU H, et al. miR-222 is necessary for exercise-induced cardiac growth and protects against pathological cardiac remodeling. Cell Metab. 2015;21(4):584-595. [91] WANG Y, MENG D, SHI X, et al. Injectable hydrogel with miR-222-engineered extracellular vesicles ameliorates myocardial ischemic reperfusion injury via mechanotransduction. Cell Rep Med. 2025;6(3):101987. [92] VUJIC A, LERCHENMÜLLER C, WU TD, et al. Exercise induces new cardiomyocyte generation in the adult mammalian heart. Nat Commun. 2018;9(1):1659. |
| [1] | 张庆彤, 陈乐琴, 刘昶, 陈昱廷, 郭睿武. 内源性大麻素系统调控运动动机的神经机制[J]. 中国组织工程研究, 2026, 30(在线): 1-11. |
| [2] | 刘金龙, 阿卜杜吾普尔•海比尔, 白 臻, 苏丹阳, 苗 鑫, 李 菲, 杨晓鹏. 不同非手术方法治疗青少年特发性脊柱侧凸效果的系统综述与网状Meta分析[J]. 中国组织工程研究, 2026, 30(9): 2370-2379. |
| [3] | 李智斐, 韩 斌, 柳秋丽, 张展鸣, 韦浩凯, 左匡时, 张翼升. 基于动作捕捉技术分析神经根型颈椎病患者的颈椎运动特征[J]. 中国组织工程研究, 2026, 30(9): 2286-2293. |
| [4] | 潘 冬, 杨加玲, 田 卫, 王东济, 朱 政, 马文超, 刘 娜, 付常喜. 抗阻运动激活衰老大鼠骨骼肌卫星细胞:脂联素受体1途径的作用[J]. 中国组织工程研究, 2026, 30(7): 1736-1746. |
| [5] | 曹 涌, 滕虹良, 邰鹏飞, 李骏达, 朱腾旗, 李兆进. 细胞因子和卫星细胞在肌肉再生中的相互作用[J]. 中国组织工程研究, 2026, 30(7): 1808-1817. |
| [6] | 钟彩红, 肖晓歌, 李 明, 林剑虹, 洪 靖. 运动相关髌腱炎发病的生物力学机制[J]. 中国组织工程研究, 2026, 30(6): 1417-1423. |
| [7] | 侯超文, 李兆进, 孔健达, 张树立. 骨骼肌衰老主要生理变化及运动的多机制调控作用[J]. 中国组织工程研究, 2026, 30(6): 1464-1475. |
| [8] | 孙尧天, 徐 凯, 王沛云. 运动影响铁代谢对免疫性炎症疾病调控的潜在机制[J]. 中国组织工程研究, 2026, 30(6): 1486-1498. |
| [9] | 曹新燕, 于子夫, 冷晓轩, 高世爱, 陈金慧, 刘西花. 重复经颅磁刺激和经颅直流电刺激对脑瘫患儿运动功能及步态影响的网状Meta分析[J]. 中国组织工程研究, 2026, 30(6): 1539-1548. |
| [10] | 李郝静, 王 新, 宋成林, 张胜男, 陈云昕. 上斜方肌处体外冲击波与运动控制训练治疗慢性非特异性颈痛[J]. 中国组织工程研究, 2026, 30(5): 1162-1170. |
| [11] | 刘 煜, 雷森林, 周锦涛, 刘 辉, 李先辉. 有氧和抗阻运动改善肥胖相关认知障碍的作用机制[J]. 中国组织工程研究, 2026, 30(5): 1171-1183. |
| [12] | 王正业, 刘万林, 赵振群. miRNA在激素诱导股骨头坏死机制中的研究进展[J]. 中国组织工程研究, 2026, 30(5): 1207-1214. |
| [13] | 文 凡, 向 阳, 朱 欢, 庹艳芳, 李 锋. 运动干预改善2型糖尿病患者的微血管功能[J]. 中国组织工程研究, 2026, 30(5): 1225-1235. |
| [14] | 陈 强, 伍文娟, 江舒华, 黄 达. 体育锻炼改善烧伤患者身体功能的系统评价与Meta分析[J]. 中国组织工程研究, 2026, 30(5): 1269-1281. |
| [15] | 金智勇, 汪宇峰, 赵滨杰, 熊敏全, 严 力. 基于双侧肢体控制策略视角分析肢体间不对称对运动能力的影响[J]. 中国组织工程研究, 2026, 30(4): 949-963. |
运动已被广泛运用于急性心肌梗死的康复治疗。适量的运动能够改善急性心肌梗死患者的心脏功能,提高生活质量[9]。目前已知运动能够通过上调miR-126影响血管生成[10];运动能够下调miR-15a和miR-146a抑制炎症反应[11-12]。然而,不同运动类型、强度及持续时长对miRNA表达的影响仍不统一,此外,miRNA作为急性心肌梗死的生物标志物,在临床上的应用潜力亦需要更多的研究支撑。虽然运动改善心脏功能与miRNA调控存在密切关联,但运动调节miRNA的机制尚不完全明确。鉴于此,该综述探究miRNA在急性心肌梗死病理进程中的作用及运动调控机制。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1 资料来源
1.1.1 检索人及检索时间 第一作者于2025年2月进行文献检索。
1.1.2 检索文献时限 从各数据库建库至2025年2月发表的相关文献。
1.1.3 检索数据库 Web of Science、PubMed、Embase、中国知网、万方和维普等数据库。
1.1.4 检索路径 通过主题词、关键词相结合的方式进行检索,使用布尔逻辑运算符“AND”和“OR”进行连接。
1.1.5 检索研究类型 原始研究、综述、Meta分析等。
1.1.6 检索词 中文检索词:急性心肌梗死,心脏重构,微小RNA,心肌细胞凋亡,心脏纤维化,血管生成,钙稳态,心脏肥大,运动;英文检索词:Acute Myocardial Infarction,Cardiac remodeling,MicroRNAs,Myocardial cell apoptosis,Myocardial fibrosis,Angiogenesis,Calcium homeostasis,Cardiac hypertrophy,Exercise。
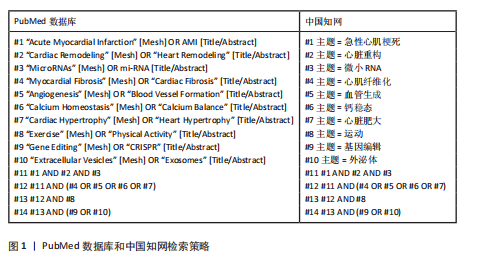
1.1.7 检索策略 以PubMed数据库和中国知网为例,检索策略见图1。
1.1.8 检索文献量 共检索到相关文献3 014篇,包括中文1 230篇、英文1 784篇。
1.2 入组标准
1.2.1 纳入标准 ①文献内容涉及miRNA在急性心肌梗死病理机制中的作用,如心肌纤维化、血管生成、钙稳态、心脏肥大等;②文献内容涉及运动对急性心肌梗死后心脏重构的调控机制,特别是通过miRNA驱动的分子机制;③研究类型包括实验文献(动物模型或细胞实验)、临床文献、Meta分析或综述;④文献语言为中文或英文。
1.2.2 排除标准 ①与急性心肌梗死、miRNA或运动调控机制无关的文献;②重复发表的文献;③文献设计不严谨、数据不完整或存在方法学缺陷的文献(原始文献由Cochrane量表评估,Meta分析由AMSTAR2量表评估);④无法获取全文的文献(如会议摘要、未发表的文献);⑤非原创性文献,如评论、社论等。
1.3 文献检索结果 共检索到相关文献3 014篇,中文1 230篇,英文1 784篇,最终符合上述入选标准的文献92篇,纳入文献流程见图2。
然而,现有研究也发现了一些仍不能解决的难题。miRNA具有多靶标作用,导致作用机制复杂多变。不同miRNA可能在不同细胞类型和病理条件下发挥不同作用,这使得精准调控特定miRNA的表达以实现治疗效果成为一大挑战。此外,miRNA在血液中的稳定性和作为生物标志物的应用潜力仍然存在不确定性,亟需更多临床和实验数据支持。
表5展示了miRNA调控及运动干预急性心肌梗死的关键研究进展时间线。
3.2 作者综述区别于他人之篇的特点 此综述在于将miRNA与运动调控机制结合,为运动治疗提供了新的理论依据,尤其是在个性化治疗方案的设计上,探索了miRNA作为靶向干预的可行性。此综述通过整合近几年的研究成果,深入讨论了miRNA在急性心肌梗死治疗中的多重作用,尤其是在纤维化、血管生成、钙稳态等方面的调节作用。与现有文献相比,此综述不仅仅关注miRNA在急性心肌梗死发生中的作用,还探讨了运动如何通过调节miRNA表达改善急性心肌梗死后的心脏重构,为心脏修复和功能恢复提供新的思路。
3.3 综述的局限性 此综述的内容主要基于实验动物模型和临床初步研究,缺乏大规模人群研究的验证。因此,未来的研究需要更广泛的人群样本和长时间的随访数据,才能确认运动干预与miRNA调控在临床应用中的有效性和可行性。此外,虽然miRNA作为心脏疾病的潜在靶点受到广泛关注,但在心脏修复中的具体机制仍然复杂。因此,未来的研究应更加注重miRNA在心脏修复不同阶段的功能,以及与其他分子和信号通路的相互作用。
3.4 综述的核心意义 此综述不仅为miRNA作为治疗靶点提供了理论依据,还为未来个性化运动干预方案的设计提供了新的视角。此外,还强调了运动与miRNA调控之间的相互作用,进一步验证了运动作为一种非药物干预手段提升心脏健康的潜力。通过结合运动和miRNA的作用机制,提出了多靶点、多途径的干预模式,为急性心肌梗死的精准治疗和康复提供了新的方案 。
3.5 对未来的建议 基于现有的研究成果,提出以下几点建议,以促进未来在该领域的研究进展:①miRNA时空表达的动态研究:各类miRNA在心脏重构的不同阶段可能表现出不同的作用,因此未来应更加关注miRNA在一定时间点的时空表达规律,并探讨在各类病理状态下的功能;②运动干预与个性化治疗方案:运动干预的类型、强度和持续时间对miRNA表达的影响存在差异,未来研究应探讨如何根据个体的具体情况(如年龄、性别、心脏功能等)制定个性化的运动干预方案,以最大限度地提高治疗效果;③miRNA作为生物标志物的稳定性研究:尽管miRNA在心血管疾病中的潜力已被初步证实,但在临床中的应用仍面临稳定性等问题,因此未来应加大对miRNA作为生物标志物的验证力度,尤其是在循环系统中的稳定性和临床转化的可行性;④运动与基因编辑技术的结合:未来的研究可以探索将运动干预与基因编辑技术结合,如CRISPR-Cas9技术,以精确调控miRNA的表达,继而促进心脏修复和功能恢复。通过深入探讨不同研究方向,有望为急性心肌梗死患者提供更加精准和个性化的治疗方案,推动心脏病治疗领域的创新发展。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
在急性心肌梗死研究领域,心脏重构的机制及调控已经成为当前研究的热点。近年来,越来越多的研究聚焦于miRNA在心脏重构中的作用,尤其是在调控心肌细胞凋亡、纤维化、血管生成和钙稳态等方面。例如,miRNA-1、miRNA-133a、miRNA-21等miRNA在急性心肌梗死后心脏修复中展现出重要作用。与传统的药物治疗方案相比,运动干预逐渐被认为是通过调节miRNA表达来提升心功能的一种有效方法。现有的实验数据显示,运动不仅能够通过上调某些miRNA(如miRNA-29、miRNA-126、miRNA-222等)来促进心脏修复,还能够与药物和营养物质联合调节miRNA的表达,提高治疗效果。本综述探讨了运动如何通过调节miRNA来改善急性心肌梗死后的心脏重构进程,与目前领域内的研究成果相契合。现阶段,运动干预调控miRNA在心脏康复中的应用尚处于探索阶段,尚需更多实验数据来进一步验证运动与miRNA联合治疗的效果。未来的研究方向包括结合人工智能和精准运动干预方案,进一步提升治疗效果,同时探索miRNA作为心脏疾病生物标志物的临床应用潜力,为运动与miRNA结合的干预策略提供了新的理论依据,并为未来的个性化治疗方案设计奠定了基础。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||