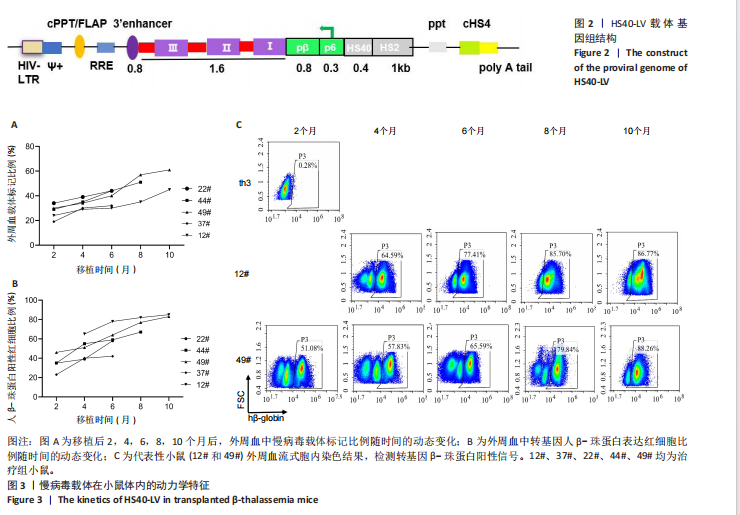
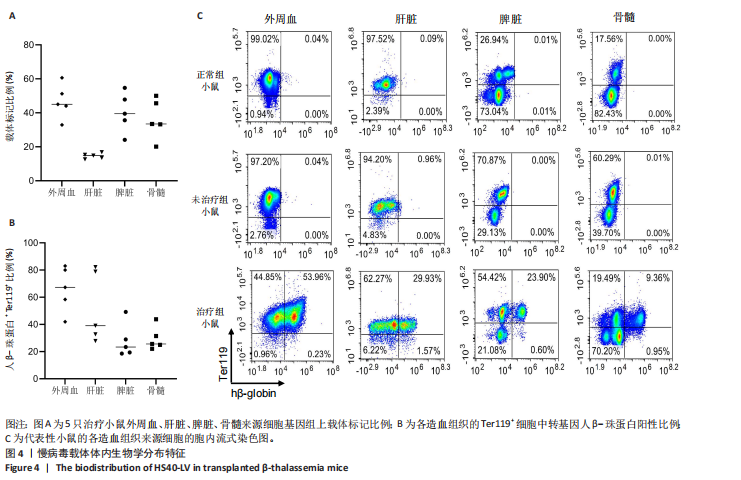
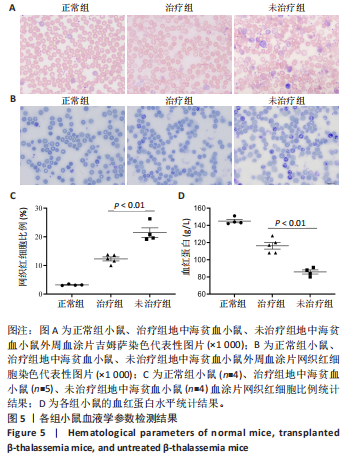
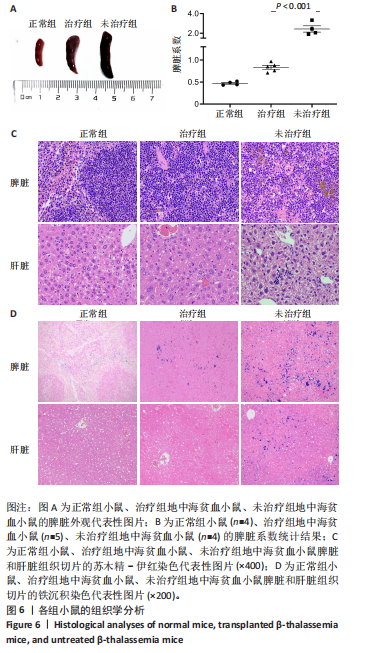
[1] KHANDROS E, THOM CS, D’SOUZA J, et al. Integrated protein quality-control pathways regulate free α-globin in murine β-thalassemia. Blood. 2012;119(22): 5265-5275.
[2] SANCHEZ-VILLALOBOS M, BLANQUER M, MORALEDA JM, et al. New Insights Into Pathophysiology of β-Thalassemia. Front Med (Lausanne). 2022;9:880752.
[3] FARMAKIS D, PORTER J, TAHER A, et al. 2021 Thalassaemia International Federation Guidelines for the Management of Transfusion-dependent Thalassemia. Hemasphere. 2022;6(8):e732.
[4] TAHER AT, MUSALLAM KM, CAPPELLINI MD. β-Thalassemias. N Engl J Med. 2021; 384(8):727-743.
[5] ALGERI M, LODI M, LOCATELLI F. Hematopoietic Stem Cell Transplantation in Thalassemia. Hematol Oncol Clin North Am. 2023;37(2):413-432.
[6] LUCARELLI G, GAZIEV J. Advances in the allogeneic transplantation for thalassemia. Blood Rev. 2008;22(2):53-63.
[7] BARONCIANI D, ANGELUCCI E, POTSCHGER U, et al. Hemopoietic stem cell transplantation in thalassemia: a report from the European Society for Blood and Bone Marrow Transplantation Hemoglobinopathy Registry, 2000-2010. Bone Marrow Transplant. 2016;51(4):536-541.
[8] PSATHA N, PAPAYANNI PG, YANNAKI E. A New Era for Hemoglobinopathies: More Than One Curative Option. Curr Gene Ther. 2017;17(5):364-378.
[9] LEONARD A, TISDALE JF, BONNER M. Gene Therapy for Hemoglobinopathies: Beta-Thalassemia, Sickle Cell Disease. Hematol Oncol Clin North Am. 2022;36(4): 769-795.
[10] FERRARI G, CAVAZZANA M, MAVILIO F. Gene Therapy Approaches to Hemoglobinopathies. Hematol Oncol Clin North Am. 2017;31(5):835-852.
[11] MAGRIN E, SEMERARO M, HEBERT N, et al. Long-term outcomes of lentiviral gene therapy for the β-hemoglobinopathies: the HGB-205 trial. Nat Med. 2022; 28(1):81-88.
[12] KWIATKOWSKI JL, WALTERS MC, HONGENG S, et al. Betibeglogene autotemcel gene therapy in patients with transfusion-dependent, severe genotype β-thalassaemia (HGB-212): a non-randomised, multicentre, single-arm, open-label, single-dose, phase 3 trial. Lancet. 2024;404(10468):2175-2186.
[13] MIRZA A, RITSERT ML, TAO G, et al. Gene therapy in transfusion-dependent non-β0/β0 genotype β-thalassemia: first real-world experience of beti-cel. Blood Adv. 2025;9(1):29-38.
[14] THOMPSON AA, WALTERS MC, KWIATKOWSKI J, et al. Gene Therapy in Patients with Transfusion-Dependent β-Thalassemia. N Engl J Med. 2018;378(16):1479-1493.
[15] MARKTEL S, SCARAMUZZA S, CICALESE MP, et al. Intrabone hematopoietic stem cell gene therapy for adult and pediatric patients affected by transfusion-dependent ß-thalassemia. Nat Med. 2019;25(2):234-241.
[16] ROSELLI EA, MEZZADRA R, FRITTOLI MC, et al. Correction of beta-thalassemia major by gene transfer in haematopoietic progenitors of pediatric patients. EMBO Mol Med. 2010;2(8):315-328.
[17] BOULAD F, MAGGIO A, WANG X, et al. Lentiviral globin gene therapy with reduced-intensity conditioning in adults with β-thalassemia: a phase 1 trial. Nat Med. 2022;28(1):63-70.
[18] OUYANG W, DONG G, ZHAO W, et al. Restoration of β-Globin Expression with Optimally Designed Lentiviral Vector for β-Thalassemia Treatment in Chinese Patients. Hum Gene Ther. 2021;32(9-10):481-494.
[19] LI S, LING S, WANG D, et al. Modified lentiviral globin gene therapy for pediatric β(0)/β(0) transfusion-dependent β-thalassemia: A single-center, single-arm pilot trial. Cell Stem Cell. 2024;31(7):961-973.
[20] HARRISON C. First gene therapy for β-thalassemia approved. Nature Biotechnology. 2019;37(10):1102-1103.
[21] JONES RJ, DEBAUN MR. Leukemia after gene therapy for sickle cell disease: insertional mutagenesis, busulfan, both, or neither. Blood. 2021;138(11):942-947.
[22] DUNCAN CN, BLEDSOE JR, GRZYWACZ B, et al. Hematologic Cancer after Gene Therapy for Cerebral Adrenoleukodystrophy. N Engl J Med. 2024;391(14):1287-1301.
[23] ROMERO Z, URBINATI F, GEIGER S, et al. β-globin gene transfer to human bone marrow for sickle cell disease. J Clin Invest. 2013;123(8):3317-3330.
[24] NEGRE O, EGGIMANN AV, BEUZARD Y, et al. Gene Therapy of the β-Hemoglobinopathies by Lentiviral Transfer of the β(A(T87Q))-Globin Gene. Hum Gene Ther. 2016;27(2):148-165.
[25] MORGAN RA, UNTI MJ, ALESHE B, et al. Improved Titer and Gene Transfer by Lentiviral Vectors Using Novel, Small β-Globin Locus Control Region Elements. Mol Ther. 2020;28(1):328-340.
[26] HANAWA H, HARGROVE PW, KEPES S, et al. Extended beta-globin locus control region elements promote consistent therapeutic expression of a gamma-globin lentiviral vector in murine beta-thalassemia. Blood. 2004;104(8):2281-2290.
[27] LISOWSKI L, SADELAIN M. Locus control region elements HS1 and HS4 enhance the therapeutic efficacy of globin gene transfer in beta-thalassemic mice. Blood. 2007;110(13):4175-4178.
[28] MICCIO A, CESARI R, LOTTI F, et al. In vivo selection of genetically modified erythroblastic progenitors leads to long-term correction of beta-thalassemia. Proc Natl Acad Sci U S A. 2008;105(30):10547-10552.
[29] RIVELLA S, SADELAIN M. Genetic treatment of severe hemoglobinopathies: the combat against transgene variegation and transgene silencing. Semin Hematol. 1998;35(2):112-125.
[30] YANNAKI E, TUBB J, AKER M, et al. Topological constraints governing the use of the chicken HS4 chromatin insulator in oncoretrovirus vectors. Mol Ther. 2002; 5(5 Pt 1):589-598.
[31] KAISER J. Gene therapy. Seeking the cause of induced leukemias in X-SCID trial. Science. 2003;299(5606):495.
[32] EMERY DW, YANNAKI E, TUBB J, et al. Development of virus vectors for gene therapy of beta chain hemoglobinopathies: flanking with a chromatin insulator reduces gamma-globin gene silencing in vivo. Blood. 2002;100(6):2012-2019.
[33] BROWNING DL, TROBRIDGE GD. Insulators to Improve the Safety of Retroviral Vectors for HIV Gene Therapy. Biomedicines. 2016;4(1):4.
[34] GOODMAN MA, ARUMUGAM P, PILLIS DM, et al. Foamy Virus Vector Carries a Strong Insulator in Its Long Terminal Repeat Which Reduces Its Genotoxic Potential. J Virol. 2018;92(1):e01639-17.
[35] IMREN S, PAYEN E, WESTERMAN KA, et al. Permanent and panerythroid correction of murine beta thalassemia by multiple lentiviral integration in hematopoietic stem cells. Proc Natl Acad Sci U S A. 2002;99(22):14380-14385.
[36] MAY C, RIVELLA S, CHADBURN A, et al. Successful treatment of murine beta-thalassemia intermedia by transfer of the human beta-globin gene. Blood. 2002;99(6):1902-1908.
[37] NEGRE O, FUSIL F, COLOMB C, et al. Correction of murine β-thalassemia after minimal lentiviral gene transfer and homeostatic in vivo erythroid expansion. Blood. 2011;117(20):5321-5331.
[38] PERSONS DA, ALLAY ER, SABATINO DE, et al. Functional requirements for phenotypic correction of murine beta-thalassemia: implications for human gene therapy. Blood. 2001;97(10):3275-3282.
[39] ANDREANI M, NESCI S, LUCARELLI G, et al. Long-term survival of ex-thalassemic patients with persistent mixed chimerism after bone marrow transplantation. Bone Marrow Transplant. 2000;25(4):401-404.
[40] FITZHUGH CD, CORDES S, TAYLOR T, et al. At least 20% donor myeloid chimerism is necessary to reverse the sickle phenotype after allogeneic HSCT. Blood. 2017; 130(17):1946-1948.
[41] AIUTI A, SLAVIN S, AKER M, et al. Correction of ADA-SCID by stem cell gene therapy combined with nonmyeloablative conditioning. Science. 2002; 296(5577):2410-2413.
[42] RAFTOPOULOS H, WARD M, LEBOULCH P, et al. Long-term transfer and expression of the human beta-globin gene in a mouse transplant model. Blood. 1997;90(9):3414-3422.
[43] NEGRE O, BARTHOLOMAE C, BEUZARD Y, et al. Preclinical evaluation of efficacy and safety of an improved lentiviral vector for the treatment of β-thalassemia and sickle cell disease. Curr Gene Ther. 2015;15(1):64-81.
[44] FINKELSHTEIN D, WERMAN A, NOVICK D, et al. LDL receptor and its family members serve as the cellular receptors for vesicular stomatitis virus. Proc Natl Acad Sci U S A. 2013;110(18):7306-7311.
[45] LEBOULCH P, HUANG GM, HUMPHRIES RK, et al. Mutagenesis of retroviral vectors transducing human beta-globin gene and beta-globin locus control region derivatives results in stable transmission of an active transcriptional structure. EMBO J. 1994;13(13):3065-3076.
[46] WANG CX, SATHER BD, WANG X, et al. Rapamycin relieves lentiviral vector transduction resistance in human and mouse hematopoietic stem cells. Blood. 2014;124(6):913-923.
[47] DELVILLE M, SOHEILI T, BELLIER F, et al. A Nontoxic Transduction Enhancer Enables Highly Efficient Lentiviral Transduction of Primary Murine T Cells and Hematopoietic Stem Cells. Mol Ther Methods Clin Dev. 2018;10:341-347.
[48] PETRILLO C, CALABRIA A, PIRAS F, et al. Assessing the Impact of Cyclosporin A on Lentiviral Transduction and Preservation of Human Hematopoietic Stem Cells in Clinically Relevant Ex Vivo Gene Therapy Settings. Hum Gene Ther. 2019;30(9):1133-1146.
[49] MASIUK KE, ZHANG R, OSBORNE K, et al. PGE2 and Poloxamer Synperonic F108 Enhance Transduction of Human HSPCs with a β-Globin Lentiviral Vector. Mol Ther Methods Clin Dev. 2019;13:390-398.
[50] UCHIDA N, NASSEHI T, DRYSDALE CM, et al. High-Efficiency Lentiviral Transduction of Human CD34(+) Cells in High-Density Culture with Poloxamer and Prostaglandin E2. Mol Ther Methods Clin Dev. 2019;13:187-196.
[51] LEVASSEUR DN, RYAN TM, PAWLIK KM, et al. Correction of a mouse model of sickle cell disease: lentiviral/antisickling beta-globin gene transduction of unmobilized, purified hematopoietic stem cells. Blood. 2003;102(13):4312-4319.
[52] RIVELLA S, MAY C, CHADBURN A, et al. A novel murine model of Cooley anemia and its rescue by lentiviral-mediated human beta-globin gene transfer. Blood. 2003;101(8):2932-2939.
[53] GOTT KM, POTTER CA, DOYLE-EISELE M, et al. A Comparison of Cs-137 γ Rays and 320-kV X-Rays in a Mouse Bone Marrow Transplantation Model. Dose Response. 2020;18(2):1559325820916572.
[54] LI Z, DÜLLMANN J, SCHIEDLMEIER B, et al. Murine leukemia induced by retroviral gene marking. Science. 2002;296(5567):497.
[55] UCHIDA N, WEITZEL RP, EVANS ME, et al. Evaluation of engraftment and immunological tolerance after reduced intensity conditioning in a rhesus hematopoietic stem cell gene therapy model. Gene Ther. 2014;21(2):148-157.
[56] WYSS BK, MEYERS JL, SINN AL, et al. A novel competitive repopulation strategy to quantitate engraftment of ex vivo manipulated murine marrow cells in submyeloablated hosts. Exp Hematol. 2008;36(4):513-521.
[57] MARDINEY M 3RD, JACKSON SH, SPRATT SK, et al. Enhanced host defense after gene transfer in the murine p47phox-deficient model of chronic granulomatous disease. Blood. 1997;89(7):2268-2275.
[58] COWEN D, RICHAUD P, MARIT G, et al. Regimen-related toxicity in patients undergoing BMT with total body irradiation using a sweeping beam technique. Bone Marrow Transplant. 1992;10(6):515-519.
[59] BEARMAN SI, APPELBAUM FR, BACK A, et al. Regimen-related toxicity and early posttransplant survival in patients undergoing marrow transplantation for lymphoma. J Clin Oncol. 1989;7(9):1288-1294.
[60] ANURATHAPAN U, PAKAKASAMA S, MEKJARUSKUL P, et al. Outcomes of thalassemia patients undergoing hematopoietic stem cell transplantation by using a standard myeloablative versus a novel reduced-toxicity conditioning regimen according to a new risk stratification. Biol Blood Marrow Transplant. 2014;20(12):2066-2071.
[61] SAITO AM, ZAHRIEH D, CUTLER C, et al. Lower costs associated with hematopoietic cell transplantation using reduced intensity vs high-dose regimens for hematological malignancy. Bone Marrow Transplant. 2007;40(3):209-217. |