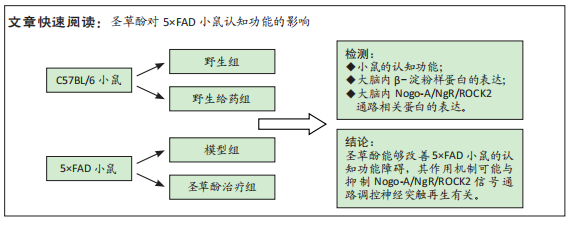
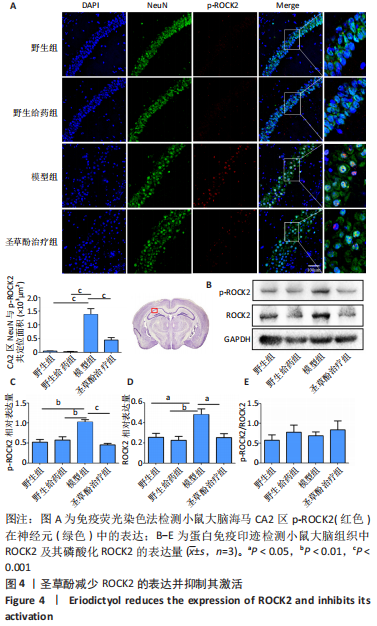
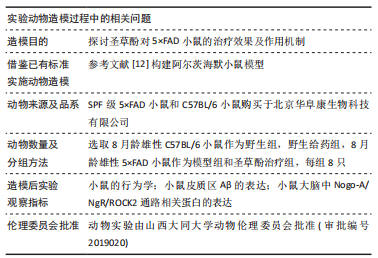
[1] LANE CA, HARDY J, SCHOTT JM. Alzheimer’s disease. Eur J Neurol. 2018; 25(1):59-70.
[2] BREIJYEH Z, KARAMAN R. Comprehensive Review on Alzheimer’s Disease: Causes and Treatment. Molecules. 2020;25(24):5789.
[3] JULIAN L, OLSON MF. Rho-associated coiled-coil containing kinases (ROCK): structure, regulation, and functions. Small GTPases. 2014;5:e29846.
[4] CAI R, WANG Y, HUANG Z, et al. Role of RhoA/ROCK signaling in Alzheimer’s disease. Behav Brain Res. 2021;414:113481.
[5] KEMPF A, SCHWAB ME. Nogo-A represses anatomical and synaptic plasticity in the central nervous system. Physiology (Bethesda). 2013;28(3):151-163.
[6] 刘颖,刘敬. Rho-ROCK信号通路研究进展[J].中国儿童保健杂志,2012, 20(9):822-825.
[7] MUELLER BK, MACK H, TEUSCH N. Rho kinase, a promising drug target for neurological disorders. Nat Rev Drug Discov. 2005;4(5):387-398.
[8] KOCH JC, TATENHORST L, ROSER AE, et al. ROCK inhibition in models of neurodegeneration and its potential for clinical translation. Pharmacol Ther. 2018;189:1-21.
[9] HE P, YAN S, ZHENG J, et al. Eriodictyol Attenuates LPS-Induced Neuroinflammation, Amyloidogenesis, and Cognitive Impairments via the Inhibition of NF-κB in Male C57BL/6J Mice and BV2 Microglial Cells. J Agric Food Chem. 2018;66(39):10205-10214.
[10] JING X, SHI H, ZHU X, et al. Eriodictyol Attenuates β-Amyloid 25-35 Peptide-Induced Oxidative Cell Death in Primary Cultured Neurons by Activation of Nrf2. Neurochem Res. 2015;40(7):1463-1471.
[11] 李苏垚,郭敏芳,于婧文,等.圣草酚对过氧化氢所致SH-SY5Y细胞线粒体动力学失衡及凋亡的影响[J].中国组织工程研究,2022,26(31): 4975-4981.
[12] 张婧.圣草酚通过促进小胶质细胞增殖改善阿尔茨海默病认知功能[D].太原:山西中医药大学,2020.
[13] WEBSTER SJ, BACHSTETTER AD, NELSON PT, et al. Using mice to model Alzheimer’s dementia: an overview of the clinical disease and the preclinical behavioral changes in 10 mouse models. Front Genet. 2014;5:88.
[14] FERRARI C, SORBI S. The complexity of Alzheimer’s disease: an evolving puzzle. Physiol Rev. 2021;101(3):1047-1081.
[15] BRIGGS R, KENNELLY SP, O’NEILL D. Drug treatments in Alzheimer’s disease. Clin Med (Lond). 2016;16(3):247-253.
[16] SPIRES-JONES TL, HYMAN BT. The intersection of amyloid beta and tau at synapses in Alzheimer’s disease. Neuron. 2014;82(4):756-771.
[17] XU YQ, SUN ZQ, WANG YT, et al. Function of Nogo-A/Nogo-A receptor in Alzheimer’s disease. CNS Neurosci Ther. 2015;21(6):479-485.
[18] OERTLE T, VAN DER HAAR ME, BANDTLOW CE, et al. Nogo-A inhibits neurite outgrowth and cell spreading with three discrete regions. J Neurosci. 2003; 23(13):5393-5406.
[19] XIAO F, LIN LF, CHENG X, et al. Nogo-66 receptor activation inhibits neurite outgrowth and increases β-amyloid protein secretion of cortical neurons. Mol Med Rep. 2012;5(3):619-624.
[20] VENKATESH K, CHIVATAKARN O, LEE H, et al. The Nogo-66 receptor homolog NgR2 is a sialic acid-dependent receptor selective for myelin-associated glycoprotein. J Neurosci. 2005;25(4):808-822.
[21] ZHOU XF, LI HY. Roles of glial p75NTR in axonal regeneration. J Neurosci Res. 2007;85(8):1601-1605.
[22] ANDREWS JL, FERNANDEZ-ENRIGHT F. A decade from discovery to therapy: Lingo-1, the dark horse in neurological and psychiatric disorders. Neurosci Biobehav Rev. 2015;56:97-114.
[23] THEOTOKIS P, GRIGORIADIS N. p75NTR and TROY: Uncharted Roles of Nogo Receptor Complex in Experimental Autoimmune Encephalomyelitis. Mol Neurobiol. 2018;55(8):6329-6336.
[24] SAHA N, KOLEV M, NIKOLOV DB. Structural features of the Nogo receptor signaling complexes at the neuron/myelin interface. Neurosci Res. 2014; 87:1-7.
[25] SCHMANDKE A, SCHMANDKE A, STRITTMATTER SM. ROCK and Rho: biochemistry and neuronal functions of Rho-associated protein kinases. Neuroscientist. 2007;13(5):454-469.
[26] TAN HB, ZHONG YS, CHENG Y, et al. Rho/ROCK pathway and neural regeneration: a potential therapeutic target for central nervous system and optic nerve damage. Int J Ophthalmol. 2011;4(6):652-657.
[27] LIU J, GAO HY, WANG XF. The role of the Rho/ROCK signaling pathway in inhibiting axonal regeneration in the central nervous system. Neural Regen Res. 2015;10(11):1892-1896.
[28] LU W, WEN J, CHEN Z. Distinct Roles of ROCK1 and ROCK2 on the Cerebral Ischemia Injury and Subsequently Neurodegenerative Changes. Pharmacology. 2020;105(1-2):3-8.
[29] MASLIAH E, XIE F, DAYAN S, et al. Genetic deletion of Nogo/Rtn4 ameliorates behavioral and neuropathological outcomes in amyloid precursor protein transgenic mice. Neuroscience. 2010;169(1):488-494.
[30] GUO MF, ZHANG HY, ZHANG PJ, et al. Fasudil reduces β-amyloid levels and neuronal apoptosis in APP/PS1 transgenic mice via inhibition of the Nogo-A/NgR/RhoA signaling axis. J Integr Neurosci. 2020;19(4):651-662.
|