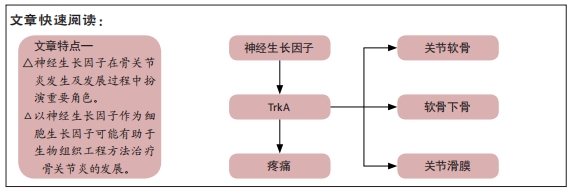
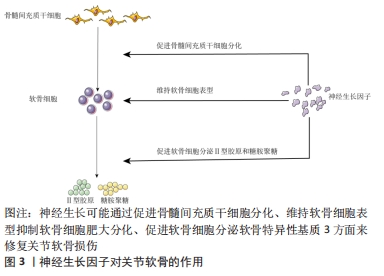
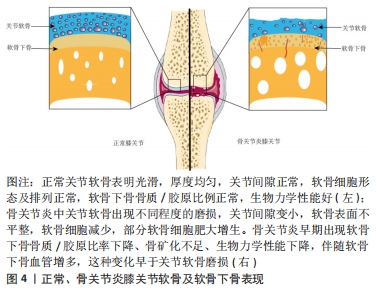
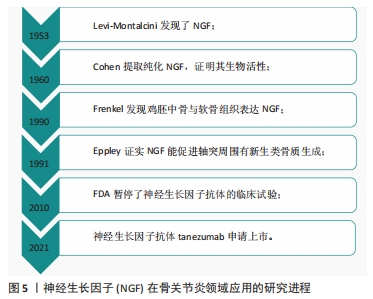
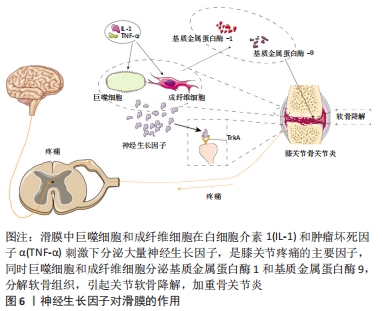
[1] ROCCO ML, SOLIGO M, MANNI L, et al. Nerve Growth Factor: Early Studies and Recent Clinical Trials. Curr Neuropharmacol. 2018;16(10): 1455-1465.
[2] DENK F, BENNETT DL, MCMAHON SB. Nerve Growth Factor and Pain Mechanisms. Annu Rev Neurosci. 2017;40:307-325.
[3] DO CARMO S, KANNEL B, CUELLO AC. Nerve Growth Factor Compromise in Down Syndrome. Front Aging Neurosci. 2021;13:719-507.
[4] PFLUGFELDER SC, MASSARO-GIORDANO M, PEREZ VL, et al. Topical Recombinant Human Nerve Growth Factor (Cenegermin) for Neurotrophic Keratopathy: A Multicenter Randomized Vehicle-Controlled Pivotal Trial. Ophthalmology. 2020;127(1):14-26.
[5] MONTAGNOLI C, TIRIBUZI R, CRISPOLTONI L, et al. β-NGF and β-NGF receptor upregulation in blood and synovial fluid in osteoarthritis. Biol Chem. 2017;398(9):1045-1054.
[6] JIANG Y, HU C, YU S, et al. Cartilage stem/progenitor cells are activated in osteoarthritis via interleukin-1β/nerve growth factor signaling. Arthritis Res Ther. 2015;17:327.
[7] 张滋彬,万千千,覃文聘,等. 神经-骨偶联机制促进骨关节退行性病发展的研究进展. 临床口腔医学杂志,2021,37(5):313-315.
[8] CHARTIER SR, MITCHELL SA, MAJUTA LA, et al. Immunohistochemical localization of nerve growth factor, tropomyosin receptor kinase A, and p75 in the bone and articular cartilage of the mouse femur. Mol Pain. 2017;13:1744806917745465.
[9] MANNI L, LUNDEBERG T, FIORITO S, et al. Nerve growth factor release by human synovial fibroblasts prior to and following exposure to tumor necrosis factor-alpha, interleukin-1 beta and cholecystokinin-8: the possible role of NGF in the inflammatory response. Clin Exp Rheumatol. 2003;21(5):617-624.
[10] KOONS GL, DIBA M, MIKOS AG. Materials design for bone-tissue engineering. Nat Rev Mater. 2020;5(8):584-603.
[11] ZHAN X, CAI P, LEI D, et al. Comparative profiling of chondrogenic differentiation of mesenchymal stem cells (MSCs) driven by two different growth factors. Cell Biochem Funct. 2019;37(5):359-367.
[12] YAN J, LI J, HU J, et al. Smad4 deficiency impairs chondrocyte hypertrophy via the Runx2 transcription factor in mouse skeletal development. J Biol Chem. 2018;293(24):9162-9175.
[13] HUANG H, SHANK G, MA L, et al. Nerve growth factor induced after temporomandibular joint inflammation decelerates chondrocyte differentiation. Oral Dis. 2013;19(6):604-610.
[14] GIGANTE A, SENESI L, MANZOTTI S, et al. Effect of nerve growth factor on cultured human chondrocytes. J Biol Regul Homeost Agents. 2016;30(4 Suppl 1):1-6.
[15] LU Z, LEI D, JIANG T, et al. Nerve growth factor from Chinese cobra venom stimulates chondrogenic differentiation of mesenchymal stem cells. Cell Death Dis. 2017;8(5):e2801.
[16] MIAO Z, LU Z, LUO S, et al. Murine and Chinese cobra venom‑derived nerve growth factor stimulate chondrogenic differentiation of BMSCs in vitro: A comparative study. Molecular Medicine Reports. 2018;18(3): 3341-3349.
[17] TAN J, LU Z, MIAO Z, et al. Effect of NGF From Venom of Chinese Cobra (Naja Atra)on Chondrocytes Proliferation and Metabolism In Vitro. J Cell Biochem. 2017;118(12):4308-4316.
[18] SANG XG, WANG ZY, CHENG L, et al. Analysis of the mechanism by which nerve growth factor promotes callus formation in mice with tibial fracture. Exp Ther Med. 2017;13(4):1376-1380.
[19] DONELL S. Subchondral bone remodelling in osteoarthritis. EFORT Open Rev. 2019;4(6): 221-229.
[20] SALTZMAN BM, RIBOH JC. Subchondral Bone and the Osteochondral Unit: Basic Science and Clinical Implications in Sports Medicine. Sports Health. 2018;10(5):412-418.
[21] HU W, CHEN Y, DOU C, et al. Microenvironment in subchondral bone: predominant regulator for the treatment of osteoarthritis. Ann Rheum Dis. 2020;80(4):413-422.
[22] HOLZER LA, KRAIGER M, TALAKIC E, et al. Microstructural analysis of subchondral bone in knee osteoarthritis. Osteoporos Int. 2020;31(10): 2037-2045.
[23] HU Y, CHEN X, WANG S, et al. Subchondral bone microenvironment in osteoarthritis and pain. Bone Res. 2021;9(1):20.
[24] LU J, ZHANG H, CAI D, et al. Positive-Feedback Regulation of Subchondral H-Type Vessel Formation by Chondrocyte Promotes Osteoarthritis Development in Mice. J Bone Miner Res. 2018;33(5): 909-920.
[25] ZHU X, CHAN YT, YUNG PSH, et al. Subchondral Bone Remodeling: A Therapeutic Target for Osteoarthritis. Front Cell Dev Biol. 2020;8:607-764.
[26] 常琦,阮贞,王宪伟,等.骨关节炎模型大鼠血清骨保护素及软骨下骨降钙素基因相关肽、神经生长因子表达及Mankin评分变化[J].中国老年学杂志,2019,39(4):909-913.
[27] JIANG Y, TUAN RS. Role of NGF-TrkA signaling in calcification of articular chondrocytes. FASEB J. 2019;33(9):10231-10239.
[28] YU X, QI Y, ZHAO T, et al. NGF increases FGF2 expression and promotes endothelial cell migration and tube formation through PI3K/Akt and ERK/MAPK pathways in human chondrocytes. Osteoarthritis Cartilage. 2019;27(3):526-534.
[29] MILLER RE, BLOCK JA, MALFAIT AM. Nerve growth factor blockade for the management of osteoarthritis pain: what can we learn from clinical trials and preclinical models? Curr Opin Rheumatol. 2017;29(1):110-118.
[30] JIN P, YIN F, HUANG L, et al. Guangxi cobra venom-derived NGF promotes the osteogenic and therapeutic effects of porous BCP ceramic. Exp Mol Med. 2017;49(4):e312.
[31] LI Z, MEYERS CA, CHANG L, et al. Fracture repair requires TrkA signaling by skeletal sensory nerves. J Clin Invest. 2019;129(12):5137-5150.
[32] ZHANG R, LIANG Y, WEI S. The expressions of NGF and VEGF in the fracture tissues are closely associated with accelerated clavicle fracture healing in patients with traumatic brain injury. Ther Clin Risk Manag. 2018;14:2315-2322.
[33] RIVERA KO, RUSSO F, BOILEAU RM, et al. Local injections of beta-NGF accelerates endochondral fracture repair by promoting cartilage to bone conversion. Sci Rep. 2020;10(1):22241.
[34] YU F, LI M, YUAN Z, et al. Mechanism research on a bioactive resveratrol- PLA-gelatin porous nano-scaffold in promoting the repair of cartilage defect. Int J Nanomedicine. 2018;13:7845-7858.
[35] SCHLICHTING K, SCHELL H, KLEEMANN RU, et al. Influence of scaffold stiffness on subchondral bone and subsequent cartilage regeneration in an ovine model of osteochondral defect healing. Am J Sports Med. 2008;36(12):2379-2391.
[36] EPSTEIN ME. Anti-nerve growth factor monoclonal antibody: a prospective new therapy for canine and feline osteoarthritis. Vet Rec. 2019;184(1):20-22.
[37] SHANG X, WANG Z, TAO H. Mechanism and therapeutic effectiveness of nerve growth factor in osteoarthritis pain. Ther Clin Risk Manag. 2017;13:951-956.
[38] MONT MA, CARRINO JA, NEMETH MA, et al. Postoperative Outcome of Patients Who Underwent Total Joint Replacement During the Tanezumab Phase 3 Osteoarthritis Development Program: A 24-Week Observational Study. Surg Technol Int. 2021;38:467-477.
[39] SEAH KTM, RAMMANOHAR J, SUTTON J, et al. The Effectiveness of Anti-Nerve Growth Factor Monoclonal Antibodies in the Management of Pain in Osteoarthritis of the Hip and Knee: A PRISMA Systematic Review and Meta-Analysis. Pain Med. 2021;22(5):1185-1204.
[40] BERENBAUM F, BLANCO F J, GUERMAZI A, et al. Subcutaneous tanezumab for osteoarthritis of the hip or knee: efficacy and safety results from a 24-week randomised phase III study with a 24-week follow-up period. Ann Rheum Dis. 2020;79(6):800-810.
[41] FAN ZR, MA JX, WANG Y, et al. Efficacy and safety of tanezumab administered as a fixed dosing regimen in patients with knee or hip osteoarthritis: a meta-analysis of randomized controlled phase III trials. Clin Rheumatol. 2021;40(6):2155-2165.
[42] BANNWARTH B, KOSTINE M. Targeting nerve growth factor (NGF) for pain management: what does the future hold for NGF antagonists? Drugs. 2014;74(6):619-626.
[43] SCHNITZER T J, EASTON R, PANG S, et al. Effect of Tanezumab on Joint Pain, Physical Function, and Patient Global Assessment of Osteoarthritis Among Patients With Osteoarthritis of the Hip or Knee: A Randomized Clinical Trial. JAMA. 2019;322(1):37-48.
[44] BERENBAUM F, LANGFORD R, PERROT S, et al. Subcutaneous tanezumab for osteoarthritis: Is the early improvement in pain and function meaningful and sustained? Eur J Pain. 2021;25(7):1525-1539.
[45] CHEN S, FU P, WU H, et al. Meniscus, articular cartilage and nucleus pulposus: a comparative review of cartilage-like tissues in anatomy, development and function. Cell Tissue Res. 2017;370(1):53-70.
[46] WISE BL, SEIDEL MF, LANE NE. The evolution of nerve growth factor inhibition in clinical medicine. Nat Rev Rheumatol. 2021;17(1):34-46.
[47] XU L, NWOSU L N, BURSTON J J, et al. The anti-NGF antibody muMab 911 both prevents and reverses pain behaviour and subchondral osteoclast numbers in a rat model of osteoarthritis pain. Osteoarthritis Cartilage. 2016;24(9):1587-1595.
[48] INGALE D, KULKARNI P, ELECTRICWALA A, et al. Synovium-Synovial Fluid Axis in Osteoarthritis Pathology: A Key Regulator of the Cartilage Degradation Process. Genes (Basel). 2021;12(7):989.
[49] PESSLER F, DAI L, DIAZ-TORNE C, et al. The synovitis of “non-inflammatory” orthopaedic arthropathies: a quantitative histological and immunohistochemical analysis. Ann Rheum Dis. 2008;67(8):1184-1187.
[50] UDOMSINPRASERT W, JINAWATH A, TEERAWATTANAPONG N, et al. Interleukin-34 overexpression mediated through tumor necrosis factor-alpha reflects severity of synovitis in knee osteoarthritis. Sci Rep. 2020; 10(1):7987.
[51] BENITO MJ, VEALE DJ, FITZGERALDO, et al. Synovial tissue inflammation in early and late osteoarthritis. Ann Rheum Dis. 2005;64(9):1263-1267.
[52] WAN QQ, QIN WP, MA YX, et al. Crosstalk between Bone and Nerves within Bone. Adv Sci (Weinh). 2021;8(7):2003390.
[53] FOERTSCH S, HAFFNER-LUNTZER M, KRONER J, et al. Chronic psychosocial stress disturbs long-bone growth in adolescent mice. Dis Model Mech. 2017;10(12):1399-1409.
[54] HAFFNER-LUNTZER M, FOERTSCH S, FISCHER V, et al. Chronic psychosocial stress compromises the immune response and endochondral ossification during bone fracture healing via beta-AR signaling. Proc Natl Acad Sci U S A. 2019;116(17):8615-8622.
|