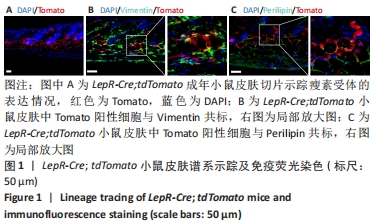
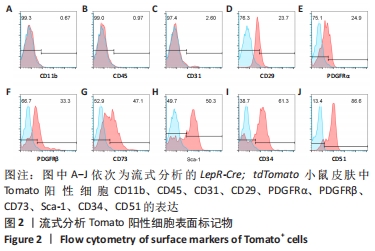
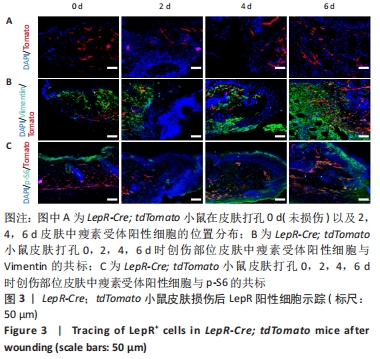
[1] PAN WW, MYERS MG JR. Leptin and the maintenance of elevated body weight. Nat Rev Neurosci. 2018;19(2):95-105.
[2] TRINH T, BROXMEYER HE. Role for Leptin and Leptin Receptors in Stem Cells During Health and Diseases. Stem Cell Rev Rep. 2021;17(2):511-522.
[3] FRIEDMAN JM. Leptin and the endocrine control of energy balance. Nat Metab. 2019;1(8):754-764.
[4] FU X, LIU G, HALIM A, et al. Mesenchymal Stem Cell Migration and Tissue Repair. Cells. 2019;8(8):784.
[5] KFOURY Y, SCADDEN DT. Mesenchymal cell contributions to the stem cell niche. Cell Stem Cell. 2015;16(3):239-253.
[6] TARTAGLIA LA. The leptin receptor. J Biol Chem. 1997;272(10):6093-6096.
[7] TRINH T, ROPA J, ALJOUFI A, et al. Leptin receptor, a surface marker for a subset of highly engrafting long-term functional hematopoietic stem cells. Leukemia. 2020 Nov 7. doi: 10.1038/s41375-020-01079-z.
[8] ZHOU BO, YUE R, MURPHY MM, et al. Leptin-receptor-expressing mesenchymal stromal cells represent the main source of bone formed by adult bone marrow. Cell Stem Cell. 2014;15(2):154-168.
[9] DING L, SAUNDERS TL, ENIKOLOPOV G, et al. Endothelial and perivascular cells maintain haematopoietic stem cells. Nature. 2012;481(7382):457-462.
[10] COMAZZETTO S, MURPHY MM, BERTO S, et al. Restricted Hematopoietic Progenitors and Erythropoiesis Require SCF from Leptin Receptor+ Niche Cells in the Bone Marrow. Cell Stem Cell. 2019;24(3):477-486.
[11] DECKER M, MARTINEZ-MORENTIN L, WANG G, et al. Leptin-receptor-expressing bone marrow stromal cells are myofibroblasts in primary myelofibrosis. Nat Cell Biol. 2017;19(6):677-688.
[12] ZHANG D, ZHANG S, WANG J, et al. LepR-Expressing Stem Cells Are Essential for Alveolar Bone Regeneration. J Dent Res. 2020;99(11):1279-1286.
[13] ROGNONI E, WATT FM. Skin Cell Heterogeneity in Development, Wound Healing, and Cancer. Trends Cell Biol. 2018;28(9):709-722.
[14] FUCHS E. Skin Stem Cells in Silence, Action, and Cancer. Stem Cell Reports. 2018;10(5):1432-1438.
[15] HORSLEY V. Skin in the Game: Stem Cells in Repair, Cancer, and Homeostasis. Cell. 2020;181(3):492-494.
[16] DEKONINCK S, BLANPAIN C. Stem cell dynamics, migration and plasticity during wound healing. Nat Cell Biol. 2019;21(1):18-24.
[17] GUERRERO-JUAREZ CF, PLIKUS MV. Emerging nonmetabolic functions of skin fat. Nat Rev Endocrinol. 2018;14(3):163-173.
[18] HU MS, BORRELLI MR, LORENZ HP, et al. Mesenchymal Stromal Cells and Cutaneous Wound Healing: A Comprehensive Review of the Background, Role, and Therapeutic Potential. Stem Cells Int. 2018;2018:6901983.
[19] RODRIGUES M, KOSARIC N, BONHAM CA, et al. Wound Healing: A Cellular Perspective. Physiol Rev. 2019;99(1):665-706.
[20] BOOTHBY IC, COHEN JN, ROSENBLUM MD. Regulatory T cells in skin injury: At the crossroads of tolerance and tissue repair. Sci Immunol. 2020;5(47): eaaz9631.
[21] SAXTON RA, SABATINI DM. mTOR Signaling in Growth, Metabolism, and Disease. Cell. 2017;168(6):960-976.
[22] LAPLANTE M, SABATINI DM. mTOR signaling in growth control and disease. Cell. 2012;149(2):274-293.
[23] KIM J, GUAN KL. mTOR as a central hub of nutrient signalling and cell growth. Nat Cell Biol. 2019;21(1):63-71.
[24] CRINO PB, NATHANSON KL, HENSKE EP. The tuberous sclerosis complex. N Engl J Med. 2006;355(13):1345-1356.
[25] KRETZSCHMAR K, WATT FM. Lineage tracing. Cell. 2012;148(1-2):33-45.
[26] HORIE T, PARK G, INABA Y, et al. MAPK Erk5 in Leptin Receptor‒Expressing Neurons Controls Body Weight and Systemic Energy Homeostasis in Female Mice. Endocrinology. 2019;160(12):2837-2848.
[27] DING X, BLOCH W, IDEN S, et al.mTORC1 and mTORC2 regulate skin morphogenesis and epidermal barrier formation. Nat Commun. 2016;7:13226.
[28] CIBRIAN D, DE LA FUENTE H, SÁNCHEZ-MADRID F. Metabolic Pathways That Control Skin Homeostasis and Inflammation. Trends Mol Med. 2020;26(11): 975-986.
[29] KRAMANN R, SCHNEIDER RK, DIROCCO DP, et al. Perivascular Gli1+ progenitors are key contributors to injury-induced organ fibrosis. Cell Stem Cell. 2015;16(1): 51-66.
[30] CURRIE JD, GROSSER L, MURAWALA P, et al. The Prrx1 limb enhancer marks an adult subpopulation of injury-responsive dermal fibroblasts. Biol Open. 2019; 8(7):bio043711.
[31] LEAVITT T, HU MS, BORRELLI MR, et al. Prrx1 Fibroblasts Represent a Pro-fibrotic Lineage in the Mouse Ventral Dermis. Cell Rep. 2020;33(6):108356.
[32] ABBASI S, SINHA S, LABIT E, et al. Distinct Regulatory Programs Control the Latent Regenerative Potential of Dermal Fibroblasts during Wound Healing. Cell Stem Cell. 2020;27(3):396-412.e6.
[33] TIKHONOVA AN, DOLGALEV I, HU H, et al. The bone marrow microenvironment at single-cell resolution. Nature. 2019;569(7755):222-228.
[34] BLANPAIN C, SIMONS BD. Unravelling stem cell dynamics by lineage tracing. Nat Rev Mol Cell Biol. 2013;14(8):489-502.
[35] EL AGHA E, KRAMANN R, SCHNEIDER RK, et al. Mesenchymal Stem Cells in Fibrotic Disease. Cell Stem Cell. 2017;21(2):166-177.
[36] SEROWOKY MA, ARATA CE, CRUMP JG, et al. Skeletal stem cells: insights into maintaining and regenerating the skeleton. Development. 2020;147(5): dev179325.
[37] RIFFAULT M, JOHNSON GP, OWEN MM, et al. Loss of Adenylyl Cyclase 6 in Leptin Receptor-Expressing Stromal Cells Attenuates Loading-Induced Endosteal Bone Formation. JBMR Plus. 2020;4(11):e10408.
[38] FUCHS E, BLAU HM. Tissue Stem Cells: Architects of Their Niches. Cell Stem Cell. 2020;27(4):532-556.
[39] MACHADO L, GEARA P, CAMPS J, et al. Tissue damage induces a conserved stress response that initiates quiescent muscle stem cell activation. Cell Stem Cell. 2021;28(6):1125-1135.e7.
[40] WELLS JM, WATT FM. Diverse mechanisms for endogenous regeneration and repair in mammalian organs. Nature. 2018;557(7705):322-328.
[41] LIU GY, SABATINI DM. mTOR at the nexus of nutrition, growth, ageing and disease. Nat Rev Mol Cell Biol. 2020;21(4):183-203.
[42] HENSKE EP, JÓŹWIAK S, KINGSWOOD JC, et al. Tuberous sclerosis complex. Nat Rev Dis Primers. 2016;2:16035.
[43] MENG D, FRANK AR, JEWELL JL. mTOR signaling in stem and progenitor cells. Development. 2018;145(1):dev152595.
[44] XIANG X, ZHAO J, XU G, et al. mTOR and the differentiation of mesenchymal stem cells. Acta Biochim Biophys Sin (Shanghai). 2011;43(7):501-510.
[45] HE D, WU H, XIANG J, et al. Gut stem cell aging is driven by mTORC1 via a p38 MAPK-p53 pathway. Nat Commun. 2020;11(1):37.
[46] ROBB KP, FITZGERALD JC, BARRY F, et al. Mesenchymal stromal cell therapy: progress in manufacturing and assessments of potency. Cytotherapy. 2019; 21(3):289-306.
[47] GOLCHIN A, FARAHANY TZ, KHOJASTEH A, et al. The Clinical Trials of Mesenchymal Stem Cell Therapy in Skin Diseases: An Update and Concise Review. Curr Stem Cell Res Ther. 2019;14(1):22-33.
[48] ZHANG R, MA J, HAN J, et al. Mesenchymal stem cell related therapies for cartilage lesions and osteoarthritis. Am J Transl Res. 2019;11(10):6275-6289.
[49] STAFF NP, JONES DT, SINGER W. Mesenchymal Stromal Cell Therapies for Neurodegenerative Diseases. Mayo Clin Proc. 2019;94(5):892-905.
[50] CAPLAN AI, CORREA D. The MSC: an injury drugstore. Cell Stem Cell. 2011;9(1): 11-15.
|