Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (32): 5191-5196.doi: 10.12307/2021.223
Previous Articles Next Articles
Effects of resolvin on vital pulp conservation and regeneration during the inflammatory environment of dental pulp, periapical and periodontal tissues
Qiu Wandi, Cui Caiyun, Li Yanjun#br#
- Department of Oral Medicine, Affiliated Hospital of Binzhou Medical University, Binzhou 256600, Shandong Province, China
-
Received:2020-08-05Revised:2020-08-08Accepted:2020-09-19Online:2021-11-18Published:2021-07-26 -
Contact:Cui Caiyun, MD, Attending physician, Department of Oral Medicine, Affiliated Hospital of Binzhou Medical University, Binzhou 256600, Shandong Province, China -
About author:Qiu Wandi, Master candidate, Department of Oral Medicine, Affiliated Hospital of Binzhou Medical University, Binzhou 256600, Shandong Province, China -
Supported by:the Natural Science Foundation of Shandong Province, No. ZR2019PH083 (to CCY); the Medical, Health Science and Technology Development Plan of Shandong Province, No. 2017WS552 (to CCY)
CLC Number:
Cite this article
Qiu Wandi, Cui Caiyun, Li Yanjun. Effects of resolvin on vital pulp conservation and regeneration during the inflammatory environment of dental pulp, periapical and periodontal tissues[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(32): 5191-5196.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 消退素 消退素是由ω-3多不饱和脂肪酸衍生而来的,可分为源自ω3-二十碳五烯酸的E类消退素(RvE) 和源自ω3-二十碳六烯酸的D类消退素(RvD)类型。 RvE包括RvE1(5S,12R,18R-三羟-6Z,8E,10E,14Z,16E-二十碳五烯酸) 、RvE2(5S,18R-三羟-6E,8Z,11Z,14Z,16E-二十碳五烯酸) 、RvE3(17S,18R-三羟-5Z,8Z,11Z,13E,15E-二十碳五烯酸) [2],RvE通过趋化素受体(ChemR23)和白三烯B4受体1作用于靶细胞发挥炎症消退作用[3]。RvE1与ChemR23结合,可以调节巨噬细胞和树突状细胞的迁移和细胞因子的产生,增强巨噬细胞对凋亡中性粒细胞的吞噬作用,从而抑制炎症反应[4]。RvE1和RvE2是白三烯B4受体1受体上的拮抗剂,具有反调节作用,可抑制中性粒细胞趋化性、钙动员和核因子κB活化[5]。 D类消退素共12种,分别为RvD1-RvD6、AT-RvD1-AT-RvD6。RvD1的受体有2种,包括脂氧素A4受体(ALX/FPR2) 及G蛋白偶联受体32,G蛋白偶联受体32也是RvD3和RvD5的受体[5]。RvD1作用于ALX/FPR2,能调节中性粒细胞的趋化性,提高其吞噬和杀灭细菌的能力[6]。RvD1与ALX/FPR2和G蛋白偶联受体32结合,调节特定的miRNA分子,下调白细胞介素8、白细胞介素10、白细胞介素12受体β2、趋化因子CC基序受体3、干扰素1α和干扰素β1等细胞因子和趋化因子,发挥促炎消退作用[7]。RvD2与其受体G蛋白偶联受体18(现称DRV2) 结合并通过其发出信号,以限制中性粒细胞的浸润,增强细菌的吞噬细胞清除率,并在小鼠急性炎症模型中促进组织分解[5]。 消退素在不同组织中都发挥着促炎症消退作用,主要作用机制包括抑制中性粒细胞浸润[8]、促进巨噬细胞极化为非经典型(M2)型、加强对细胞碎片和凋亡中性粒细胞的吞噬作用[1,9]、调节活性氧的生成[10]、调节骨代谢、抑制骨吸收、诱导骨重建[11-12]、镇痛[13-16],具体作用见表1。此外,消退素在2型糖尿病、动脉粥样硬化、眼部感染性疾病、下颌下腺炎、传染性疾病中发挥良好的促炎症消退作用[17-26]。 "

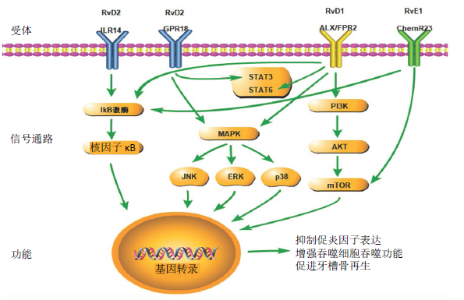
2.2 消退素在牙髓、根尖周及牙周组织炎症性疾病中的应用 2.2.1 消退素在牙髓炎中的应用 RvE1可抑制牙髓组织早期炎症阶段炎症细胞浸润和促炎细胞因子表达。20 μL皮质类固醇/ 抗生素混合物和20 μL RvE1 (1 mg/L) 分别作用于暴露24 h的大鼠炎症牙髓,皮质类固醇/抗生素混合物在术后24和72 h内不能改善暴露牙髓的炎症反应,20 μL RvE1(1 mg/L) 可减少以单核细胞浸润为主的牙髓组织细胞浸润,且局限于冠部和根管口处[27]。50 ng RvE1(1 mg/L)作用于暴露0,9,24,48 h的大鼠炎症牙髓24 h,0,9和24 h组RvE1能显著减少炎症牙髓细胞浸润(CD11b、髓过氧化物酶活性下降) ,降低促炎细胞因子白细胞介素6、肿瘤坏死因子α和白细胞介素1β mRNA表达,但48 h组RvE1无明显作用;100 nmol(35 ng) RVE1可降低体外炎症牙髓与牙髓细胞中的ChemR23表达,抑制脂多糖诱导的IKB-a磷酸化和核因子κB p65核转位增加,ChemR23基因敲除后,RvE1的抗炎作用消失[8]。RvE1在牙髓组织炎症的早期阶段(牙髓暴露24 h内) 通过激活ChemR23受体途径促进炎性细胞数量及降低促炎因子表达从而抑制牙髓炎症,其通过抑制核因子κB信号通路发挥作用。RvE1可能成为改善牙髓炎症状态一种新的治疗策略,但是研究仅集中于大鼠炎症牙髓组织及人牙髓细胞,RvE1药物作用浓度以1 mg/L为主,作用浓度单一,对牙髓炎晚期作用效果和作用机制仍未明确,可能成为今后研究的重要方向。 2.2.2 消退素在根尖周炎中的应用 消退素作为根管内封药可促进根尖周炎症的消退,尤其是促进根尖未闭合的年轻恒牙根尖周炎症消退及根尖闭合。大鼠第一磨牙开髓行牙髓切断术后开放3周构建根尖周炎模型,将20 μL RvE1 (1 mg/L)和三联抗生素糊剂(50 g/L) 处理牙髓组织3周后,组织学染色观察仅RvE1组根尖周病变范围减少,术后6 周 RvE1组和三联抗生素糊剂组均观察到根尖周病变减少和牙根发育,且RvE1组炎症消退效果明显,钙化组织沉积较规则[28]。 20 μL RvD2(1 mg/L) 作为根管内药物治疗大鼠根尖周病变,封药4周后RvD2(1 mg/L) 可降低根尖周吞噬细胞的髓过氧化物酶活性,并上调其受体G蛋白偶联受体18和牙本质基质蛋白1表达;体外100 nmol/L RvD2(约376 μg/L) 能显著促进牙髓细胞矿化结节的形成,促进牙本质基质蛋白1的mRNA、蛋白表达水平及磷酸化STAT3蛋白表达[29]。体内RvD2和RvE1均能有效促进根尖周炎症消退和牙根发育;体外RvD2-G蛋白偶联受体18受体相互作用可使STAT3磷酸化,从而促进牙髓细胞分化为成牙本质细胞并上调牙本质基质蛋白1表达。消退素有望作为一种新型根管内用药应用于根尖周炎症的治疗,消退素对根尖周炎症的研究主要集中于大鼠根尖周炎症及人牙髓细胞,RvE1和RvD2主要作用浓度为1 mg/L,但是消退素促进根尖周病变愈合的作用机制仍需进一步研究。 2.2.3 消退素在牙周炎中的应用 消退素用于牙周炎症治疗的相关研究相较于牙髓和根尖周炎症方面增多,消退素通过抑制炎症细胞的过度活化、促进牙周膜细胞活性等措施预防和恢复牙槽骨丧失,对结扎诱导及牙龈卟啉单胞菌诱导的实验性牙周炎都有促进愈合的作用。 RvE1可通过抑制炎症细胞浸润、降低破骨细胞浓度、抑制炎症因子表达、抑制骨吸收相关基因表达等促进牙周炎症愈合。0.1 g/L和0.5 g/L RvE1均可显著预防牙槽骨高度的丧失,降低破骨细胞浓度和减少炎性细胞浸润,同时能降低组织表达骨吸收及炎症相关基因(趋化因子配体3、CXCL、Acp5、趋化因子配体9等)[30]。局部注射5 mg/L RvE1,10 d后牙周组织炎症细胞浸润减轻,体外1 nmol/L RvE1(约0.35 μg/L) 可显著抑制牙周膜细胞中炎症因子白细胞介素1β、白细胞介素6、白细胞介素8在mRNA和蛋白水平的表达[31]。RvE1预防结扎诱导的实验性牙周炎中牙槽骨丧失的作用机制及临床作用效果仍不明确。 RvD1以依赖Del-1的方式抑制小鼠牙周炎症,Del-1是一种内皮细胞分泌的抗炎蛋白,在黏膜部位和中枢神经系统在内的各种器官和组织中均具有抑制炎症的作用。体外白细胞介素17以糖原合成酶激酶3β和C/EBPβ依赖的方式下调人脐静脉内皮细胞Del-1的表达,100 nmol/L RvD1(约376 μg/L)和RvD2(约376 μg/L)可逆转白细胞介素17对Del-1的抑制作用,而敲除G蛋白偶联受体32和/或ALX/FPR2后,该作用受到抑制;体外0.2 ng RvD1可降低牙周炎小鼠牙龈组织中髓过氧化物酶水平和干扰素相关分子(粒细胞集落刺激因子、趋化因子配体1、趋化因子配体2、趋化因子配体3和趋化因子配体5)的mRNA表达,同时RvD1不能抑制Del-1缺陷小鼠的骨丢失[32]。此外,1,10,50和100 μg/L RvD1能降低牙周袋深度、松动度、牙槽骨丧失量及龈沟液中炎症因子白细胞介素1β、肿瘤坏死因子α、白细胞介素6水平[33],而且白细胞介素6的表达随着RvD1浓度的升高而降低,具有一定的RvD1浓度依赖性[34],组织化学染色结果与体外实验结果一致[35],但其作用机制仍未明确。 消退素亦能防止牙龈卟啉单胞菌(Prophyromonas gingivalis,P.g)引起的实验性牙周炎的牙槽骨丧失。0.5 μg RvD2可通过减少牙龈RANKL/骨保护素比率,抑制中性粒细胞聚集,促进M2型巨噬细胞极化,调节促炎和抗炎细胞因子的表达,抑制全身和牙龈的TH1型适应性反应,防止P.g诱导的牙槽骨丢失[36]。马飞等[37]综述了RvE对牙龈卟啉单胞菌注入鼠爪诱发炎症的抑制作用,但未见RvE对牙龈卟啉单胞菌诱导牙周炎模型作用的报道。 此外,消退素能促进不同来源成纤维细胞的增殖、迁移,促进牙周组织愈合和再生,但其本身并不促进细胞成骨方向分化。10,100 nmol/L LXA4(约3.52,35.2 μg/L)或RvD2(约3.76,37.6 μg/L)能抑制转化生长因子β1诱导的小鼠成纤维细胞的增殖和迁移,但α-平滑肌动蛋白和Ⅰ、Ⅲ型胶原的表达没有明显改变[38],LXA4或RvD2可在早期限制纤维化,但不干扰肌成纤维细胞的分化,从而使细胞外基质中的胶原沉积,促进伤口愈合。 综上所述,主要通过结扎和P.g诱导实验性牙周炎动物模型来模拟人牙周炎环境,RvE1的作用浓度有0.1 g/L和0.5 g/L, RvD的作用浓度范围为1-100 μg/L,而且作用方式不同,浓度差别较大,可能是实验设计方式、模型构建方法不同;学者大多采用纳摩尔浓度(0.35 μg/L)的RvE1或(3.8-376 μg/L)RvD进行体外研究,可显著降低炎性因子的表达,减少牙槽骨丧失,其浓度差异可能跟不同来源、种属的细胞有关。此外,近6年仅见2篇RvE1应用于牙周组织疾病的研究,未见对P.g诱导实验性牙周炎动物模型和促进牙周再生的研究。 2.3 消退素参与信号通路 消退素在全身性疾病、牙髓及牙周的炎症控制上都观察到了促进炎症消退的作用,但其作用机制仍不明确,因此进一步回顾总结了消退素涉及参与的信号通路,主要包括核因子κB、丝裂原活化蛋白激酶(mitogen activated protein kinases,MAPK)、磷脂酰肌醇3-激酶(phosphatidylinositol 3-kinase,PI3K)、JAK-STAT信号通路,如图2所示。 "
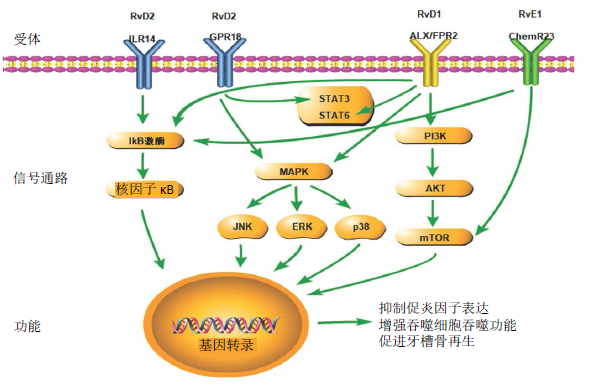

核因子κB是细胞内重要的核转录因子,它参与机体的炎症反应、免疫应答,调节细胞凋亡、应激反应,包括RelA(p65)、RelB、c-Rel、核因子κB1(p50)和核因子κB2(p52)蛋白。RvE1与ChemR23结合显著降低了IKB-a磷酸化和核因子κB p65蛋白向细胞核的转运,抑制促炎因子白细胞介素6、肿瘤坏死因子α和白细胞介素1β的表达[8,39]。在脂多糖诱导的急性肾损伤模型中,RvD1可通过下调核因子κB炎症信号,抑制半胱氨酸蛋白酶3蛋白上调,降低肿瘤坏死因子α水平,减轻肾脏病理损伤,保护肾功能,提高动物存活率[40]。RvD1可通过激活ALX/FPR2受体和核因子κB p50/p50-COX-2信号通路,促进急性呼吸窘迫综合征炎症的缓解[41]。RvD1通过抑制核因子κB信号传导抑制肿瘤坏死因子α诱导的结肠癌细胞中c-Myc蛋白的过度表达[42];RvD2通过Toll样受体4抑制核因子κB通路从而下调病毒性心肌炎小鼠体内肿瘤坏死因子α和白细胞介素1β的表达[43]。RvD5能抑制脂多糖刺激的细胞外调节蛋白激酶(extracelluar regulated protein kinase,ERK)磷酸化及p60和p50向细胞核内转运,从而抑制白细胞介素6和趋化因子配体5的产生[44]。RvDs可显著降低慢性阻塞性肺疾病患者巨噬细胞中的RelB磷酸化,抑制白细胞介素6、肿瘤坏死因子α等促炎因子的表达,促进M2巨噬细胞表型[45]。 MAPK是一组能被细胞因子、神经递质、激素、细胞应激及细胞黏附等不同细胞外刺激激活的丝氨酸-苏氨酸蛋白激酶,常见的有ERK、p38、JNK通路。RvD1可通过阻断ERK1/2、p38/MAPK、JNK和核因子κB/p65的磷酸化,降低银屑病面积、严重程度指数评分及银屑病标志性细胞因子(白细胞介素23、白细胞介素17、白细胞介素22、肿瘤坏死因子α)的表达,改善皮肤病理改变[46]。RvD1通过与ALX/FPR2的相互作用,抑制ERK1/2磷酸化,减弱c-Myc蛋白在人结肠癌细胞中过度表达的稳定性[42]。RvD1可通过降低ERK、p38、JNK磷酸化靶向MAPK通路抑制脂多糖诱导的肝癌细胞增殖和肿瘤坏死因子α、白细胞介素6、白细胞介素1β等炎性细胞因子的表达[47]。RvD1显著抑制了衣霉素诱导的c-Jun N末端激酶(JNK)的表达,但P38和ERK1/2磷酸化不受影响,此外,一种JNK激活剂茴香霉素增加了半胱氨酸蛋白酶3活性、细胞凋亡以及三酰甘油的积累和胆固醇调节元件结合蛋白1的表达,RvD1治疗逆转了这些变化[48]。AT-RvD1通过降低p38和ERK1/2磷酸化,抑制白细胞介素1β诱导的肺泡上皮细胞活化(细胞因子分泌减少、黏附分子表达下降)[49]。RvD2可逆转大鼠脑缺血再灌注模型的脑损伤和G蛋白偶联受体18水平降低,但G蛋白偶联受体18拮抗剂O-1918可通过G蛋白偶联受体18-ERK1/2-NOS信号通路阻断RvD2的逆转作用[50]。 PI3K蛋白家族参与细胞增殖、分化、凋亡和葡萄糖转运等多种细胞功能的调节。PI3K-AKt通路在RvE1处理白细胞介素6刺激的成骨细胞时出现显著干扰,显著下调破骨细胞分化[12]。在链脲佐菌素诱导的2型糖尿病小鼠模型中,RvD1的处理使AKt(Ser473)磷酸化,随后PI3K和雷帕霉素靶蛋白下游分子被激活,从而提高脑内神经元的存活率[51]。RvD1可通过激活PI3K-AKt通路抑制半胱氨酸蛋白酶3裂解,下调促凋亡Bax基因和上调抗凋亡Bcl-2基因的表达,经PI3K抑制剂预处理后,RvD1的抗凋亡作用被消除[52]。在突出髓核诱导大鼠神经根痛模型中,RvD2与G蛋白偶联受体18结合,激活Akt/糖原合成酶激酶3β信号通路,减轻机械性痛觉超敏反应及脊髓肿瘤坏死因子α、白细胞介素6的表达[16]。雷帕霉素靶蛋白是PI3K下游靶点,RvE1/ RvE2通过ChemR23激活雷帕霉素靶蛋白C1信号,改善脂多糖诱导的小鼠抑郁样行为[53]。RvD1、RvD2能逆转脂多糖诱导的小鼠抑郁样行为,雷帕霉素靶蛋白C1抑制剂雷帕霉素能显著阻断RvD1和RvD2的抗抑郁作用[54]。AT-RvD1通过抑制雷帕霉素靶蛋白途径,抑制丙酮酸激酶M2的表达,从而抑制转化生长因子β1诱导的上皮间质转化,降低肺癌细胞的迁移和侵袭[55]。 JAK-STAT信号通路是由细胞因子刺激的信号转导通路,参与细胞的增殖、分化、凋亡以及免疫调节等许多重要的生物学过程,它主要由酪氨酸激酶相关受体、酪氨酸激酶JAK和转录因子STAT组成。RvD1增强STAT6磷酸化及STAT6和DNA的结合活性,促进白细胞介素4诱导的小胶质细胞选择性激活,治疗神经炎性疾病[56]。单纯疱疹病毒诱导的基质角膜炎小鼠模型中,AT- RvD1以剂量依赖性方式减弱CD4+ T细胞中STAT1磷酸化,减少角膜新生血管和基质角膜炎的病变程度[57]。在牙髓细胞中,RvD2-G蛋白偶联受体18受体相互作用使STAT3磷酸化,进一步将牙髓细胞分化成类成牙本质样细胞[29]。 此外,RvD1还参与了miR-138-5p/FOXC1[58]、过氧化物酶体增殖剂激活受体γ[13,56,59]、Rac1/NOX2[60]、鞘氨醇-1-磷酸(S1P)[61]、FPR2/ROS/FOXM1[62]、SIRT1[63]、Nrf2[64-65]、环磷酸腺苷/蛋白激酶A(cAMP/PKA)[66]、CXCL2/CXCR4[67]、NLRP3炎症小体等信号通路[68-69]。 综上所述,RvE通过核因子κB、MAPK、PI3K信号通路下调促炎因子白细胞介素6、肿瘤坏死因子α和白细胞介素1β的表达,抑制破骨细胞分化;RvD主要通过核因子κB、MAPK、PI3K、JAK-STAT等信号通路减少细胞凋亡,下调白细胞介素6、肿瘤坏死因子α、白细胞介素1β、白细胞介素17、白细胞介素22等促炎因子的表达,促进M2型巨噬细胞表型,降低癌细胞迁移和侵袭,改善机体过敏、抑郁样行为、肺水肿和脑损伤。 "

| [1] 张武霞,庄志彬,郭淑珍,等.特异性促炎症消退介质结构与功能研究进展[J]. 免疫学杂志,2020,36(5):432-441. [2] CHIANG N, SERHAN CN. Structural elucidation and physiologic functions of specialized pro-resolving mediators and their receptors. Mol Aspects Med. 2017;58:114-129. [3] HAAS-STAPLETON EJ, YAN L, SONG H, et al. Candida albicans modulates host defense by biosynthesizing the pro-resolving mediator resolvin E1. PLoS ONE. 2017;2(12):e1316. [4] BALTA MG, LOOS BG, NICU EA. Emerging Concepts in the Resolution of Periodontal Inflammation: A Role for Resolvin E1. Front Immunol. 2017;8:1682. [5] DUVALL MG, LEVY BD. DHA-and EPA-derived resolvins, protectins, and maresins in airway inflammation. Eur J Pharmacol. 2016;785:144-155. [6] CIANCI E, RECCHIUTI A, TRUBIANI O, et al. Human Periodontal Stem Cells Release Specialized Proresolving Mediators and Carry Immunomodulatory and Prohealing Properties Regulated by Lipoxins. Stem Cells Transl Med. 2016;5(1):20-32. [7] FREDMAN G, SERHAN CN. Specialized proresolving mediator targets for RvE1 and RvD1 in peripheral blood and mechanisms of resolution. Biochem J. 2011;437(2):185-197. [8] XU H, CHEN J, GE J, et al. Resolvin E1 Ameliorates Pulpitis by Suppressing Dental Pulp Fibroblast Activation in a Chemerin Receptor 23-dependent Manner. J Endod. 2019;45(9):1126-1134.e1. [9] 徐华兴,张琪. 消退素在炎症消退中的研究进展及其在牙髓炎症调控中的应用[J]. 口腔医学,2018,38(8):743-746. [10] CHATTOPADHYAY R, RAGHAVAN S, RAO GN. Resolvin D1 via prevention of ROS-mediated SHP2 inactivation protects endothelial adherens junction integrity and barrier function. Redox Biol. 2017;12:438-455. [11] GYURKO R, VAN DYKE TE. The role of polyunsaturated omega-3 fatty acid eicosapentaenoic acid-derived resolvin E1 (RvE1) in bone preservation. Crit Rev Immunol. 2014;34(4):347-357. [12] EL KHOLY K, FREIRE M, CHEN T, et al. Resolvin E1 Promotes Bone Preservation Under Inflammatory Conditions. Front Immunol. 2018;9:1300. [13] SAITO T, HASEGAWA-MORIYAMA M, KURIMOTO T, et al. Resolution of Inflammation by Resolvin D1 Is Essential for Peroxisome Proliferator-activated Receptor-gamma-mediated Analgesia during Postincisional Pain Development in Type 2 Diabetes. Anesthesiology. 2015;123(6):1420-1434. [14] O YY, LEE JY, PARK CK. Resolvin E1 Inhibits Substance P-Induced Potentiation of TRPV1 in Primary Sensory Neurons. Mediators Inflamm. 2016;2016: 5259321. [15] MEESAWATSOM P, BURSTON J, HATHWAY G, et al. Inhibitory effects of aspirin-triggered resolvin D1 on spinal nociceptive processing in rat pain models. J Neuroinflammation. 2016;13(1):233. [16] ZHANG LY, LIU ZH, ZHU Q, et al. Resolvin D2 Relieving Radicular Pain is Associated with Regulation of Inflammatory Mediators, Akt/GSK-3beta Signal Pathway and GPR18. Neurochem Res. 2018;43(12):2384-2392. [17] FREIRE MO, DALLI J, SERHAN CN, et al. Neutrophil Resolvin E1 Receptor Expression and Function in Type 2 Diabetes. J Immunol. 2017;198(2): 718-728. [18] SIMA C, MONTERO E, NGUYEN D, et al. ERV1 Overexpression in Myeloid Cells Protects against High Fat Diet Induced Obesity and Glucose Intolerance. Sci Rep. 2017;7(1):12848. [19] SIMA C, PASTER B, VAN DYKE TE. Function of Pro-Resolving Lipid Mediator Resolvin E1 in Type 2 Diabetes. Crit Rev Immunol. 2018;38(5):343-365. [20] HASTURK H, ABDALLAH R, KANTARCI A, et al. Resolvin E1 (RvE1) Attenuates Atherosclerotic Plaque Formation in Diet and Inflammation-Induced Atherogenesis. Arterioscler Thromb Vasc Biol. 2015;35(5):1123-1133. [21] ROSSI S, DI FILIPPO C, GESUALDO C, et al. Protection from endotoxic uveitis by intravitreal Resolvin D1: involvement of lymphocytes, miRNAs, ubiquitin-proteasome, and M1/M2 macrophages. Mediators Inflamm. 2015:149381. [22] MAISTO R, TROTTA MC, PETRILLO F, et al. Resolvin D1 Modulates the Intracellular VEGF-Related miRNAs of Retinal Photoreceptors Challenged With High Glucose. Front Pharmacol. 2020;11:235. [23] TROTTA MC, PIERETTI G, PETRILLO F, et al. Resolvin D1 reduces mitochondrial damage to photoreceptors of primary retinal cells exposed to high glucose. J Cell Physiol. 2020;235(5):4256-4267. [24] NELSON JW, LEIGH NJ, MELLAS RE, et al. ALX/FPR2 receptor for RvD1 is expressed and functional in salivary glands. Am J Physiol Cell Physiol. 2014; 306(2):C178-C185. [25] WANG CS, WEE Y, YANG CH, et al. ALX/FPR2 Modulates Anti-Inflammatory Responses in Mouse Submandibular Gland. Sci Rep. 2016;6:24244. [26] RUSSELL CD, SCHWARZE J. The role of pro-resolution lipid mediators in infectious disease. Immunology. 2014;141(2):166-173. [27] DONDONI L, SCARPARO RK, KANTARCI A, et al. Effect of the pro-resolution lipid mediator Resolvin E1 (RvE1) on pulp tissues exposed to the oral environment. Int Endod J. 2014;47(9):827-834. [28] SSCARPARO R K, DONDONI L, BOTTCHER DE, et al. Intracanal delivery of Resolvin E1 controls inflammation in necrotic immature rat teeth. J Endod. 2014;40(5):678-682. [29] SIDDIQUI YD, OMORI K, ITO T, et al. Resolvin D2 Induces Resolution of Periapical Inflammation and Promotes Healing of Periapical Lesions in Rat Periapical Periodontitis. Front Immunol. 2019;10:307. [30] LEE CT, TELES R, KANTARCI A, et al. Resolvin E1 Reverses Experimental Periodontitis and Dysbiosis. J Immunol. 2016;197(7):2796-2806. [31] 程抒华,徐华兴,陈杰,等. 消退素E1调控牙周膜成纤维细胞改善牙周组织炎症的研究[J]. 口腔生物医学,2019,10(2):68-72. [32] MAEKAWA T, HOSUR K, ABE T, et al. Antagonistic effects of IL-17 and D-resolvins on endothelial Del-1 expression through a GSK-3beta-C/EBPbeta pathway. Nat Commun. 2015;6:8272. [33] 唐彩金,曾启新,方梅飞.消退素D1治疗实验性大鼠牙周炎的研究[J]. 口腔医学研究,2017,33(12):1270-1273. [34] 马飞,唐彩金,方梅飞,等. 消退素D1对牙周炎大鼠龈沟液中炎症因子影响的研究[J]. 实用口腔医学杂志,2018,34(5):610-613. [35] 马飞,唐彩金,方梅飞,等. 消退素D1对牙周炎大鼠牙龈组织中IL-1β、IL-6表达的影响[J]. 牙体牙髓牙周病学杂志,2018,28(10):571-574+604. [36] MIZRAJI G, HEYMAN O, VAN DYKE TE, et al. Resolvin D2 Restrains Th1 Immunity and Prevents Alveolar Bone Loss in Murine Periodontitis. Front Immunol. 2018;9:785. [37] 马飞,唐彩金,曾启新. 消退素与牙周病关系的研究进展[J]. 牙体牙髓牙周病学杂志,2017,27(12):733-735+720. [38] HERRERA BS, KANTARCI A, ZARROUGH A, et al. LXA4 actions direct fibroblast function and wound closure. Biochem Biophys Res Commun. 2015;464(4): 1072-1077. [39] RREY C, NADJAR A, BUAUD B, et al. Resolvin D1 and E1 promote resolution of inflammation in microglial cells in vitro. Brain Behav Immun. 2016;55:249-259. [40] ZHAO YL, ZHANG L, YANG YY, et al. Resolvin D1 Protects Lipopolysaccharide-induced Acute Kidney Injury by Down-regulating Nuclear Factor-kappa B Signal and Inhibiting Apoptosis. Chin Med J (Engl). 2016;129(9):1100-1107. [41] GAO Y, ZHANG H, LUO L, et al. Resolvin D1 Improves the Resolution of Inflammation via Activating NF-kappaB p50/p50-Mediated Cyclooxygenase-2 Expression in Acute Respiratory Distress Syndrome. J Immunol. 2017;199(6):2043-2054. [42] ZHONG X, LEE HN, SURH YJ. RvD1 inhibits TNFalpha-induced c-Myc expression in normal intestinal epithelial cells and destabilizes hyper-expressed c-Myc in colon cancer cells. Biochem Biophys Res Commun. 2018;496(2):316-323. [43] 石哲玮,刘胜新,张银宇,等. Resolvin D2通过抑制NF-κB通路减轻病毒性心肌炎小鼠炎症反应的研究[J]. 心电与循环,2020,39(1):35-40. [44] CHUN HW, LEE J, PHAM TH, et al. Resolvin D5, a Lipid Mediator, Inhibits Production of Interleukin-6 and CCL5 Via the ERK-NF-kappaB Signaling Pathway in Lipopolysaccharide-Stimulated THP-1 Cells. J Microbiol Biotechnol. 2020;30(1):85-92. [45] CROASDELL A, THATCHER TH, KOTTMANN RM, et al. Resolvins attenuate inflammation and promote resolution in cigarette smoke-exposed human macrophages. Am J Physiol Lung Cell Mol Physiol. 2015;309(8):L888-L901. [46] XU J, DUAN X, HU F, et al. Resolvin D1 attenuates imiquimod-induced mice psoriasiform dermatitis through MAPKs and NF-kappaB pathways. J Dermatol Sci. 2018;89(2):127-135. [47] LU Y, XU Q, YIN G, et al. Resolvin D1 inhibits the proliferation of lipopolysaccharide- treated HepG2 hepatoblastoma and PLC/PRF/5 hepatocellular carcinoma cells by targeting the MAPK pathway. Exp Ther Med. 2018;16(4):3603-3610. [48] JUNG TW, HWANG HJ, HONG HC, et al. Resolvin D1 reduces ER stress-induced apoptosis and triglyceride accumulation through JNK pathway in HepG2 cells. Mol Cell Endocrinol. 2014;391(1-2):30-40. [49] COX R JR, PHILLIPS O, FUKUMOTO J, et al. Resolvins Decrease Oxidative Stress Mediated Macrophage and Epithelial Cell Interaction through Decreased Cytokine Secretion. PLoS One. 2015;10(8):e0136755. [50] ZUO G, ZHANG D, MU R, et al. Resolvin D2 protects against cerebral ischemia/reperfusion injury in rats. Mol Brain. 2018;11(1):9. [51] BATHINA S, GUNDALA NKV, RHENGHACHAR P, et al. Resolvin D1 Ameliorates Nicotinamide-streptozotocin-induced Type 2 Diabetes Mellitus by its Anti-inflammatory Action and Modulating PI3K/Akt/mTOR Pathway in the Brain. Arch Med Res. 2020;51(6):492-503. [52] XIE W, WANG H, LIU Q, et al. ResolvinD1 reduces apoptosis and inflammation in primary human alveolar epithelial type 2 cells. Lab Invest. 2016;96(5):526-536. [53] DEYAMA S, SHIMODA K, SUZUKI H, et al. Resolvin E1/E2 ameliorate lipopolysaccharide-induced depression-like behaviors via ChemR23. Psychopharmacology (Berl). 2018;235(1):329-336. [54] Deyama S, Ishikawa Y, Yoshikawa K, et al. Resolvin D1 and D2 Reverse Lipopolysaccharide-Induced Depression-Like Behaviors Through the mTORC1 Signaling Pathway. Int J Neuropsychopharmacol. 2017;20(7):575-584. [55] LIU Y, YUAN X, LI W, et al. Aspirin-triggered resolvin D1 inhibits TGF-beta1-induced EMT through the inhibition of the mTOR pathway by reducing the expression of PKM2 and is closely linked to oxidative stress. Int J Mol Med. 2016;38(4):1235-1242. [56] LI L, WU Y, WANG Y, et al. Resolvin D1 promotes the interleukin-4-induced alternative activation in BV-2 microglial cells. Neuroinflammation. 2014;11:72. [57] RAJASAGI NK, BHELA S, VARANASI SK, et al. Frontline Science: Aspirin-triggered resolvin D1 controls herpes simplex virus-induced corneal immunopathology. J Leukoc Biol. 2017;102(5):1159-1171. [58] BAI X, SHAO J, ZHOU S, et al. Inhibition of lung cancer growth and metastasis by DHA and its metabolite, RvD1, through miR-138-5p/FOXC1 pathway. J Exp Clin Cancer Res. 2019;38(1):479. [59] XIA H, WANG J, SUN S, et al. Resolvin D1 Alleviates Ventilator-Induced Lung Injury in Mice by Activating PPARgamma/NF-kappaB Signaling Pathway. Biomed Res Int. 2019;2019:6254587. [60] LIU W, HUANG J, DOYCHEVA D, et al. RvD1 binding with FPR2 attenuates inflammation via Rac1/NOX2 pathway after neonatal hypoxic-ischemic injury in rats. Exp Neurol. 2019;320:112982. [61] KANG JW, CHOI HS, SHIN JK, et al. Resolvin D1 activates the sphingosine-1-phosphate signaling pathway in murine livers with ischemia/reperfusion injury. Biochem Biophys Res Commun. 2019;514(4):1058-1065. [62] SUN L, WANG Y, WANG L, et al. Resolvin D1 prevents epithelial-mesenchymal transition and reduces the stemness features of hepatocellular carcinoma by inhibiting paracrine of cancer-associated fibroblast-derived COMP. J Exp Clin Cancer Res. 2019;38(1):170. [63] ZHUO Y, ZHANG S, LI C, et al. Resolvin D1 Promotes SIRT1 Expression to Counteract the Activation of STAT3 and NF-kappaB in Mice with Septic-Associated Lung Injury. Inflammation. 2018;41(5):1762-1771. [64] ZHANG Z, HU X, QI X, et al. Resolvin D1 promotes corneal epithelial wound healing and restoration of mechanical sensation in diabetic mice. Mol Vis. 2018;24:274-285. [65] POSSO SV, QUESNOT N, MORAES JA, et al. AT-RVD1 repairs mouse lung after cigarette smoke-induced emphysema via downregulation of oxidative stress by NRF2/KEAP1 pathway. Int Immunopharmacol. 2018;56:330-338. [66] MOTTOLA G, CHATTERJEE A, WU B, et al. Aspirin-triggered resolvin D1 attenuates PDGF-induced vascular smooth muscle cell migration via the cyclic adenosine monophosphate/protein kinase A (cAMP/PKA) pathway. PLoS One. 2017;12(3):e0174936. [67] YAXIN W, SHANGLONG Y, HUAQING S, et al. Resolvin D1 attenuates lipopolysaccharide induced acute lung injury through CXCL-12/CXCR4 pathway. J Surg Res. 2014;188(1):213-221. [68] YIN Y, CHEN F, WANG W, et al. Resolvin D1 inhibits inflammatory response in STZ-induced diabetic retinopathy rats: Possible involvement of NLRP3 inflammasome and NF-κB signaling pathway. Mol Vis. 2017;23:242-250. [69] 方晨,梅咏玉,王晶,等. 消退素D1抑制NLRP3信号通路对DSS结肠炎小鼠的影响[J]. 中国药理学通报,2019,35(4):580-585. |
| [1] | Li Mengfei, Zhang Hong, Zhao Shaojian, Yin Guanghao, Wang Qibao. Expression of forkhead box protein 3 in refractory periapical periodontitis in rats with Enterococcus faecalis infection [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1187-1192. |
| [2] | Zhao Lu, Zhao Yifei, Gao Da, Liu Yanfang, Fu Tingting, Xu Jiangyan. Expression of suppressor of Zeste 12 in kidney tissues of rats with diabetic nephropathy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1179-1186. |
| [3] | Li Xinyue, Li Xiheng, Mao Tianjiao, Tang Liang, Li Jiang. Three-dimensional culture affects morphology, activity and osteogenic differentiation of human periodontal ligament stem cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 846-852. |
| [4] | Yuan Wei, Liu Jingdong, Xu Guanghui, Kang Jian, Li Fuping, Wang Yingjie, Zhi Zhongzheng, Li Guanwu. Osteogenic differentiation of human perivascular stem cells and its regulation based on Wnt/beta-catenin signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 866-871. |
| [5] | Yuan Bo, Xie Lide, Fu Xiumei. Schwann cell-derived exosomes promote the repair and regeneration of injured peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 935-940. |
| [6] | Wang Jinling, Huang Xiarong, Qu Mengjian, Huang Fujin, Yin Lingwei, Zhong Peirui, Liu Jin, Sun Guanghua, Liao Yang, Zhou Jun. Effects of exercise training on bone mass and bone microstructure in aged osteoporotic rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 676-682. |
| [7] | Wu Yujie, Wan Xiaofang, Wei Mianxing, Peng Shiyuan, Xu Xiaomei. Correlation between autophagy and the Hippo-YAP protein pathway in periodental ligament cells on the pressure side of a mouse model of orthodontic tooth movement [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 683-689. |
| [8] | Tao Xin, Xu Yi, Song Zhiwen, Liu Jinbo. Hippo signaling pathway in the regulation of spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(4): 619-625. |
| [9] | Zong Mingrui, Liu Haiyan, Li Bing, Wu Xiuping. Application of carboxymethyl chitosan in tissue engineering of stomatology [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(3): 447-452. |
| [10] | Zhang Jie, Tian Ai. Advances in the signaling pathway of M2 macrophages involved in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 314-321. |
| [11] | Luo Di, Liang Xuezhen, Yan Bozhao, Li Jiacheng, Xu Bo, Li Gang. Mechanism of Bushen Huoxue Capsule in repair of bone defects due to steroid-induced osteonecrosis of the femoral head in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 184-191. |
| [12] | Zhao Feng, Fan Shaoqing, Cheng Xiaoyan, Li Xiaona, Li Changsheng, Ma Haojie. miR-132-3p targets and modulates Ptch1 to reduce neuropathic pain in mice with chronic constriction injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 230-236. |
| [13] | Zhong Jinglin, Cao Huimin, Pan Yaru, Jian Wenxuan, Wang Qi. Ginsenoside Rg3 protects PC12 cells against oxygen-glucose deprivation/reoxygenation-induced damage [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(2): 177-183. |
| [14] | Li Zhe, Yuan Changshen, Guan Yanbing, Xu Wenfei, Liao Shuning, Rong Weiming, Mei Qijie, Duan Kan. Bioinformatic analysis and experimental validation of ferroptosis in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(17): 2637-2643. |
| [15] | Fan Qinghua, Qi Jie, Zhang Jun. The regulatory role of Hippo-YAP signaling pathway in exercise-induced cardiac hypertrophy [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(17): 2708-2715. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||